Abstract
Hypoglycemia is a rare complication of diffuse large B-cell lymphoma. We are presenting a case of 67-year-old woman presented to her primary care physician with fatigue and hyperhidrosis. Laboratory evaluation revealed a glucose level of 1.9 mmol/L. Computed tomographic scan of the abdomen and subsequent positron emission tomographic scan revealed extensive lymphadenopathy. The patient was then diagnosed with CD5-positive-diffuse large B-cell lymphoma and developed recurrent hypoglycemia despite continuous infusion of glucose. Following immunochemotherapy, hypoglycemia was resolved. Several explanations have been postulated but the exact pathophysiology is not well understood. Further investigation is warranted to more clearly define the pathophysiology of persistent hypoglycemia in patients with diffuse large B-cell lymphoma.
Introduction
Hypoglycemia is a rare complication in malignancies, especially in lymphoproliferative tumors such as diffuse large B-cell lymphoma. It has been described in few case reports and is especially more pronounced in cachectic patients. Several explanations have been postulated but the pathophysiology is not well understood. Our case is a 67-year-old female who had newly diagnosed MCD-diffuse large B-cell lymphoma (MCD-DBLCL). MCD-DLBCL is a genetic subset of DLBCL with gain-of-function mutations in both MYD88 and CD79B. 1 The patient had refractory hypoglycemia despite continuous intravenous fluid administration with glucose. C-peptide levels are normal. Patient was suspected of having hypoglycemia related to the lymphoma and was given immunochemotherapy. She never had any further episode of hypoglycemia.
Case Presentation
Clinical findings on examination and laboratory investigations at the time of admission
This was a 67-year-old woman, who presented to the department of endocrinology with a history of generalized weakness, fatigue and hyperhidrosis for 1 week. On arrival, the patient was found conscious, alert, looking ill, and cachexic. She was hemodynamically stable and lung sounds are normal. On auscultation, S1 and S2 was audible with no added third sound or murmur. Her initial blood pressure was 120/81 mmHg, her heart rate was 84 beats per minute with an oxygen saturation of 99% on room air, and she was breathing spontaneously. The initial blood tests showed spanemia, with hemoglobin of 100 g/l, red blood cell count of 3.39*1012/l, and platelet count of 57*109/l. Plasma glucose level was 1.9 mmol/l. The hospital specialists’ team advised getting her serum insulin level, C-peptide, proinsulin, cortisol, and thyroid profile including TSH and free T4 at the time of hypoglycemia. The C-peptide level was low at 0.6 ng/mL which suggested nonpancreatic insulin source as the cause of hypoglycemia. Table 1 showed the laboratory values of the patient at the time of admission.
Clinical examinations performed on the patient.

Diagnosis
During the hospital course, the patient was given intravenous glucose and adequate nutritional support. Computed tomographic (CT) scan of the abdomen revealed soft tissue shadows in the right renal sinus, multiple nodules appear in the right ureteral, and splenomegaly (Figure 1). Subsequent PET/CT scan revealed extensive lymphadenopathy. The patient was then evaluated by specialists from department of hematology. Bone marrow biopsy confirmed the presence of diffuse large B-cell lymphoma. On immunostaining, the tumor cells are positive for CD20, CD79a, CD79b, CD5, CD10, MUM1, BCL-2, C-myc (10%+) and Ki-67 (95%) and negative for CD3, CD34, CD117, BCL-6, CyclinD1, SOX-11 and CD23. Furthermore, flow cytometry analysis of marrow cells showed 51% mature CD5+ B lymphocytes. The current DLBCL case was rare with mutations positive for MYD88L265P and CD79B, named MCD genetic subtype.
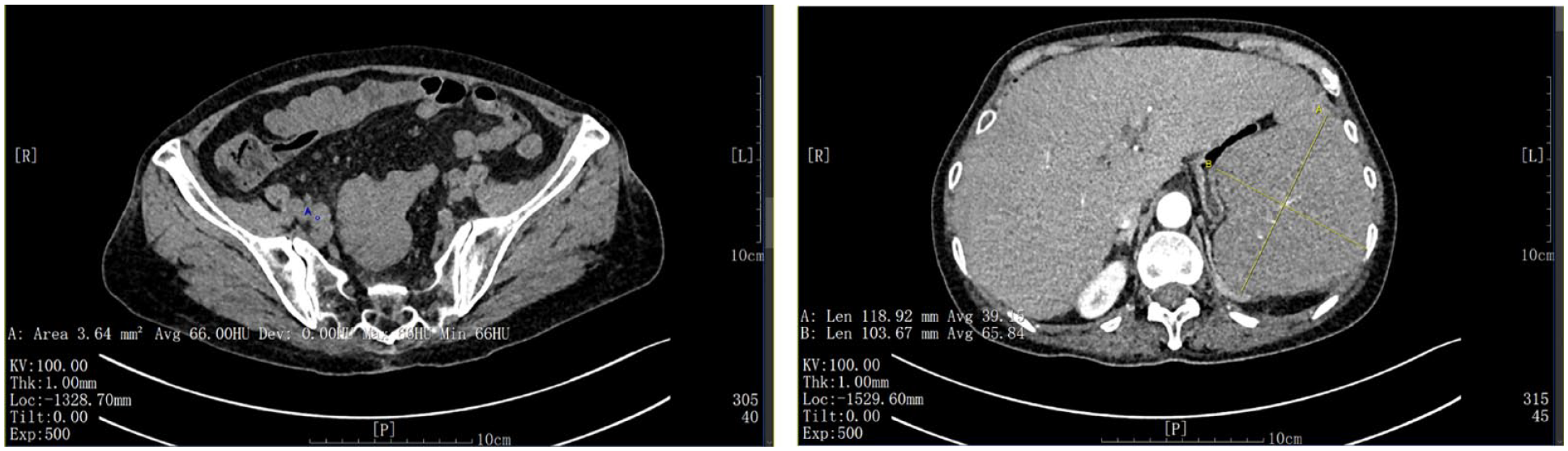
Computed tomographic scan of the abdomen of the patient at admission.
Treatment and follow-up
As mentioned above, during hospitalization in the department of Endocrinology, the patient was given intravenous glucose and adequate nutritional support. However, it’s hard to raise glucose levels. At the same time, initial findings suggested that the patient might have lymphoma. Then based on the suggestion by specialists from department of hematology, glucocorticoid treatment was started before chemotherapy. Fasting blood glucose was 1.9 mmol/L before chemotherapy. After diagnosed with diffuse large B-cell lymphoma, the patient was given an immunochemotherapy using the R-VLDP protocol (rituximab, cyclophosphamide, mitoxantrone hydrochloride liposome, vincristine and hexadecadrol).2-4 The patient’s labs were repeated after 2 weeks, and hypoglycemia was resolved, the fasting glucose level was 4.2 mmol/L. After 2 months, the patient then underwent one course of R-CHOP (rituximab, cyclophosphamide, doxorubicin hydrochloride, vincristine sulfate and prednisone) therapy and was evaluated again. Table 1 shows the laboratory values of the patient after two courses of chemotherapy. Besides, CT scan of the abdomen showed spleen shrinkage (Figure 2). The patient then underwent another four courses of R-CHOP therapy. The patient had another PET/CT scan after 6 months, and achieved a complete response.

Computed tomographic scan of the abdomen of the patient after two courses of chemotherapy.
Discussion
Hypoglycemia is often related to diabetes treatment with insulin or sulfonylureas, and drugs are the most common cause of hypoglycemia. 5 But a variety of conditions can cause hypoglycemia in people who do not have diabetes: alcohol drinking, critical illness, starvation, hormone deficiencies, and tumors. 6 A rare tumor of the pancreas (insulinoma) is associated with increased insulin secretion. 7 Through increased secretion of insulin-like growth factor 2 (IGF-2), 8 non-islet cell tumors may also cause hypoglycemia. IGF-2 increases glucose utilization, which can lead to hypoglycemia. 8
There have been several hypotheses proposed for the reasons of hypoglycemia as a complication of lymphoma, but the mechanism has been poorly defined (Table 2). In a case reported by Aldahash, 9 it was noted that, in a patient with ALK-1-negative anaplastic large-cell lymphoma, laboratory investigations showed reduced plasma insulin and C-peptide levels. The patient’s glucose storage was depleted most likely due to liver involvement and poor general condition. Another case reported by Lim et al 10 speculated that hypoglycemia associated with Hodgkin’s disease was probably related to a substance that mimicked the effects of insulin, an insulin-like growth factor causing hypoglycemia. Elhomsy et al 11 reported that in a patient with diffuse large B-cell lymphoma, excessive glucose consumption is an oncogenic switch that shifts metabolism toward a glycolytic state. Kulkarni et al 12 reported that patient with Hodgkin’s disease with severe hypoglycemia was probably due to the production of poorly characterized insulin-like substances by malignant cells especially in patients who are cachectic with extensive liver infiltration. Al Maqrashi et al 13 speculated that the development of hypoglycemia in association with the onset of multiorgan failure, hepatic dysfunction would have ensued and worsened the hypoglycemia eventually resulting in circulatory collapse. Tanios et al 14 presented a case of diffuse large B cell lymphoma with persistent hypoglycemia, cancer cells have higher rates of replication and glycolysis, they cannot rely on normal oxidative phosphorylation that is the main source of energy in normal cells. Therefore, they shift to a glycolytic pathway at a higher rate (>200-fold) in aerobic conditions. A case by Soleja et al 15 speculated that reprograming of glucose metabolism through p53, IDH2 mutations, and MYC overexpression may have contributed to the fatal outcome in the patient characterized by refractory hypoglycemia. Recently, Rosales-Castillo and Bustos-Merlo reported a case on refractory hypoglycemia as a form of presentation of diffuse B lymphoma. The proposed mechanisms may be related to increased IGF-2, normal-low or decreased levels of IGF binding protein (IGFBP) 3 or elevated IGF-2/IGF-1 ratio. 16 As IGF-2 exerts insulin-like activity and promotes hypoglycemia, IGF-2 could suppress both insulin and growth hormone, which in turn decreases the production of IGFBP3. 17
Review of the case reports of hypoglycemia in patients with lymphoma.

A case of MCD-DLBCL is reported that encroached multiple vital organs including the kidney and spleen. The MCD genotype is a genetic subset of DLBCL with the co-occurrence mutations in both MYD88 and CD79B, 18 and MCD genotype is characterized by significantly inferior outcomes. The mechanism of the pathophysiology of hypoglycemia has been poorly defined. The molecular mechanism of DLBCL with the concurrence of MYD88 and CD79B mutations are summarized as follows. Previous study found chronic active B-cell receptor (BCR) signaling and oncogenic MYD88 mutations in MCD-DLBCL could drive pathological nuclear factor kappa B (NF-κB) activation. 18 NF-κB could inhibit glucose production induced by glucocorticoid and glucagon. 19 Besides, activation of NF-κB can also inhibit glucose production by stimulating the production of nitric oxide. 20 Furthermore, it was suggested that the phosphatidylinositol 3-kinases (PI3K) pathway activation is an essential driver of the development of MCD-DLBCL. 21 One hypothesis stated that activation of hepatic PI3K is related to inappropriate inhibition of hepatic glycogenolysis and gluconeogenesis. Pharmacological suppression of the PI3K pathway can increase hepatic glucose output and induce hyperglycemia. 22 Another possibility is that the PI3K/AKT pathway could also reflect the trafficking and activity of GLUT-1 transporter. 23 In cancer cells, it was found that the hyperactivation of the PI3K/AKT pathway may promote GLUT-1 overexpression and increase glucose utilization. 24 It has also been recognized that the PI3K/AKT/mTOR signaling pathway could participate in β cell proliferation and survival. 25 Excessive secretion of IGF-2 is a common cause of non-islet cell tumor hypoglycemia. 26 IGF-2 has been found to activate the insulin receptor, and then inhibit gluconeogenesis, glycogenolysis, and ketogenesis. Besides, IGF-2 could increase the uptake of glucose by muscles and inhibit lipolysis by activating insulin receptor and decreasing glucagon production.27,28 In the current case, the availability of the tests is limited in our hospital, we didn’t get IGF-2 levels before chemotherapy. After one course of chemotherapy, IGF-2 level was 365.8 ng/mL (ref: 267-616 ng/mL). If we ever get a case like this in the future, IGF-2 levels are needed to further study the reason for hypoglycemia.
In this case, we described a patient with hypoglycemia associated with diffuse large B-cell lymphoma. Hypoglycemia corrected rapidly after chemotherapy. Chemotherapy can disrupt the metabolic pathways of malignant cells and correct these abnormalities, and should be initiated without delay. We recommend that, for patients with recurrent hypoglycemia refractory to continuous enteral feeds and other treatment modalities, comprehensive examination should be instituted. Diffuse large B cell lymphomas causing hypoglycemia is a rare manifestation. We should suspect a patient with profound or resistant hypoglycemia diagnosed with hematologic malignancy or nonsolid tumors.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Yangfan Project, Science and Technology Commission of Shanghai Municipality (23YF1411000) and Qingmiao project of Huadong Hospital (QMRC2204).
Contributors
All authors made individual contributions to authorship. All authors were involved in the diagnosis and management of patients and manuscript submission. All authors reviewed and approved the final draft.
Data Availability Statement
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Informed Patient Consent for Publication
Signed informed consent was obtained directly from the patients’ guardians.
