Abstract
Introduction:
Pulmonary artery aneurysms encompass a wide range of presentations and forms. Mycotic aneurysms represent a particular subset of focal dilatation of the vessel wall with high morbidity and mortality rates. Herein, we report the case of a 32 year old patient, with a prior history of ventricular septal defect presenting with a mycotic pulmonary artery aneurysm associated with infective endocarditis and septic emboli.
Case presentation:
We present the case of a 32 year old male with known history of congenital ventricular septal defect presented to the emergency department with signs of sepsis and dyspnea. Blood cultures were positive for methicillin-sensitive Staphylococcus aureus. An echocardiogram found evidence of endocarditis with multiples intra cavitary vegetations. A CT angiogram demonstrated major right ventricular dilatation, multiple nodules and peripheral opacities, scattered throughout the lungs, indicative of septic emboli. Segmental saccular dilatation of the left lateral basal pulmonary artery consistent with a mycotic aneurysm formation was found. The patient was started on intravenous antibiotics and given the overall satisfactory evolution a conservative approach was pursued. The patient was discharged with antibiotics and scheduled for surgical repair of the ventricular septal defect.
Conclusion:
To our knowledge, mycotic aneurysms associated to congenital heart malformation like ventricular septal wall defect remains a rare condition with few reported cases in the literature. Being aware of this entity is important for every practicing radiologist to allow for early diagnosis and treatment.
Introduction
Mycotic pulmonary artery aneurysms associated with ventricular septal defect are uncommon, 1 but remain important to recognize due to very high morbidity and mortality associated with the risk of rupture.
Infection either bacterial or fungal can lead to deficiency of the vessel wall resulting in focal dilatation of the pulmonary artery or one of its branches. 2
Pyogenic bacteria are usually associated with infective endocarditis and septic emboli.
CT angiogram provides valuable information regarding location, number and size of the focal aneurysmal dilatation of the pulmonary arterial wall. 3
Both clinicians and radiologists should be aware of this entity to ensure an accurate diagnosis and early treatment in order to reduce morbidity and mortality.
We report the case of a 32 year old patient, with a prior history of ventricular septal defect presenting with a mycotic pulmonary artery aneurysm associated with infective endocarditis and septic emboli.
Case Presentation
We present the case of a 32 year old male presented to the emergency department with signs of sepsis, fever of 39°, cough and chest discomfort.
At physical examination, the patient presented a shortness of breath with polypnea.
Laboratory tests found a mildly elevated white blood cell count of 10 230/mm3 (normal range 4000-10 000/mm3) and elevated C reactive protein at 125 mg/L (normal below 3 mg/L).
Blood cultures were positive for methicillin-sensitive Staphylococcus aureus, thought to have originated from a dental disorder. An echocardiogram found severe tricuspid valve regurgitation and evidence of elevated right ventricular pressures with a right ventricle just above the upper size limit and a RV/LV ratio at 0.8.
The right atrium was dilated measured at 38 cm2. The left ventricle and atrium were normal with no ventricular hypertrophy, along with a normal pulmonary, aortic and mitral valve.
An infra valvular septal defect, inferior to the septal leaflet of the tricuspid valve was found consistent with an indirect, type I of Gerbode’s classification of septal defects. Also, multiple hyperechoic sessile and oscillating vegetations on both the septal defect and on the septal, posterior and anterior leaflets of the tricuspid valve.
A routine plain chest radiography was performed in the emergency department showing pulmonary consolidations and cardiomegaly. However, given the severity of respiratory symptoms, signs of sepsis and the presence of vegetations on the echocardiography the decision to perform a CT angiogram to better assess for the parenchymal lesions and potential vascular lesions was made.
An initial CT angiogram showed cardiomegaly with major right ventricular dilatation (Figure 1). Multiple nodules and nodular ground glass peripheral opacities, with some cavitation, scattered throughout the lungs were indicative of septic emboli (Figure 2). Segmental saccular dilatation of the left lateral basal pulmonary artery consistent with a mycotic aneurysm formation was found (Figures 3 and 4).
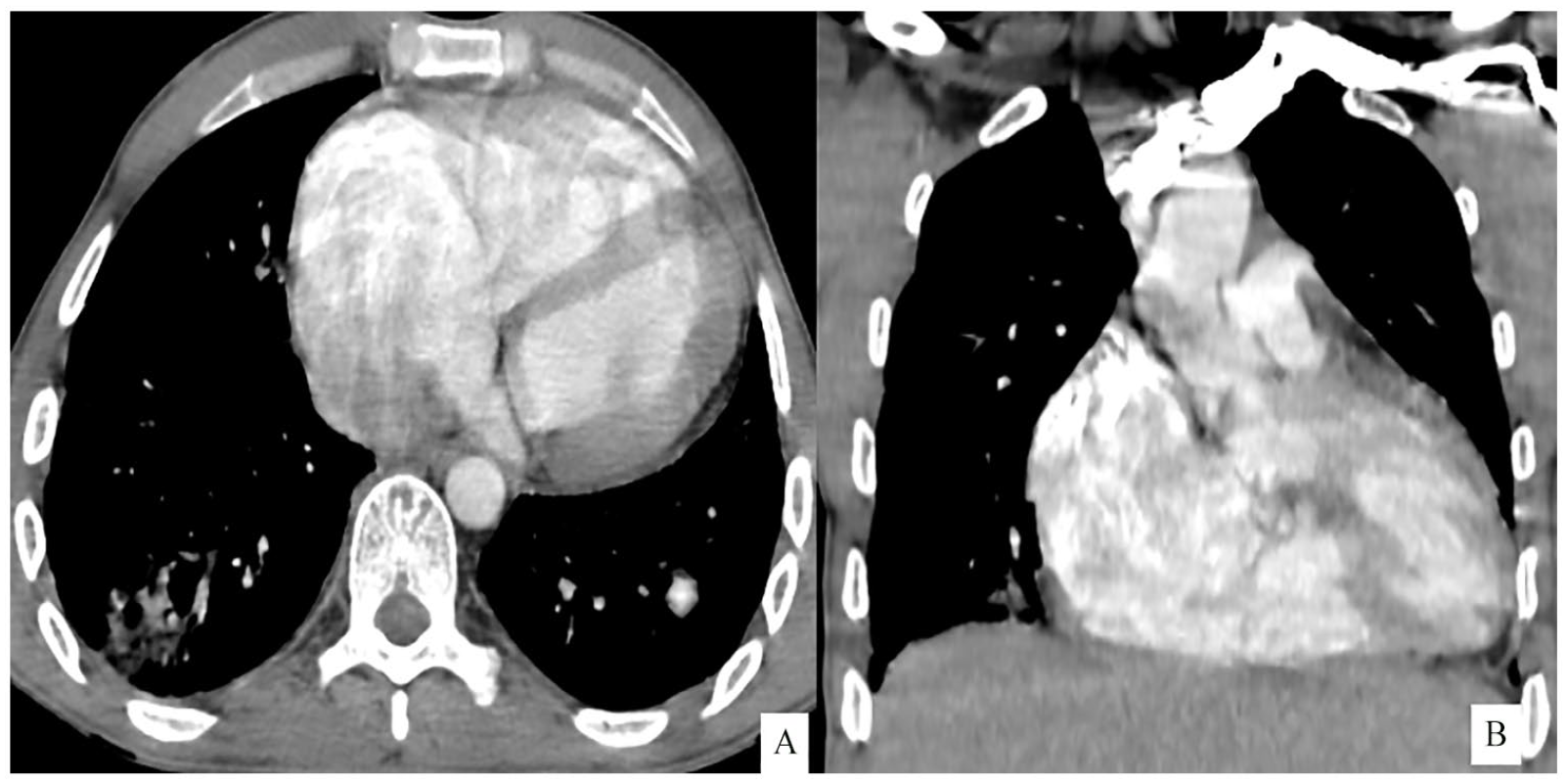
Axial (A) and coronal (B) CT angiogram showing a cardiomegaly with major right ventricular dilatation.

Axial (A), axial MIP reconstruction (B) and Sagittal (C) CT in lung window showing multiple peripheral nodules and wedge shaped subpleural consolidation (arrows) especially in the right lower lobe and cavitary Fowler nodule (arrow head).
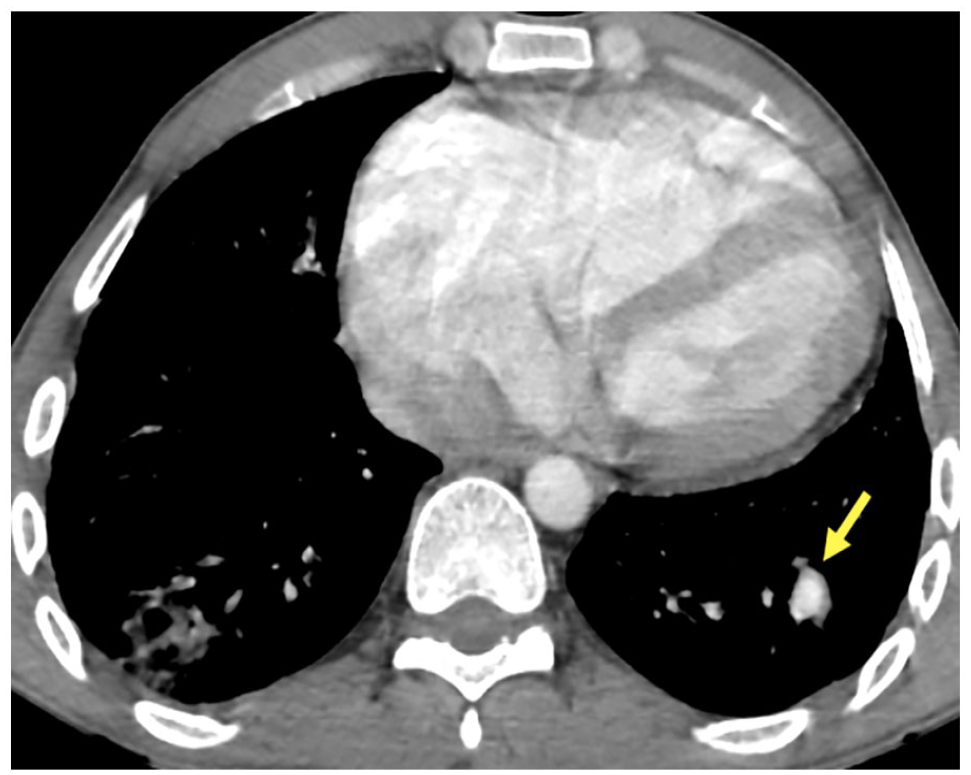
Axial CT angiogram showing a segmental saccular dilatation of the left lateral basal pulmonary artery (yellow arrow).

Coronal (A) and sagittal (B) MIP reconstruction demonstrating a segmental saccular dilatation of the left lateral basal pulmonary artery.
The patient was started on intravenous antibiotics and given the overall satisfactory evolution a conservative approach was pursued.
A follow up CT angiogram 9 weeks later demonstrated relative regression of the septic emboli with stability of the previously described mycotic aneurysm.
The patient was later discharged with antibiotics and scheduled for surgical repair of the ventricular septal defect. In fact, besides the infective endocarditis related to the complicated ventricular septal defect, he presented major tricuspid regurgitation. After careful multi-disciplinary discussion the decision was made to opt for a surgical treatment to proceed with both closure of the ventricular septal defect and valve annuloplasty to improve the surface of coaptation and reshape the tricuspid annulus to restore function with overall satisfactory outcome and evolution.
Discussion
Infective endocarditis refers to the infection of the endocardium. It usually affects the valves and chordae tendineae as well as prosthetic valves and implanted devices. 4
Among the risk factors, intravenous drug use, hematogenous spread from an oral, gastro intestinal or genital source of infection and valvular or congenital heart disease play a critical role in the pathogenesis of an endocardial lesion. Once the endocardial lesion is initiated, the clotting pathway enables vegetation formation. Vegetation fragments can than detach and undergo embolization through the bloodstream, either the pulmonary circulation causing pneumonia or to the systemic circulation particularly the brain causing strokes.
Diagnosis of infective endocarditis is based on modified Duke criteria. 5 Echocardiography is the first line modality to assess infective endocarditis, vegetation representing the hallmark of this affection. They appear as irregular, iso or hyper echoic lesions, oscillating or non-oscillating that preferentially congregate on the leading, low pressure side of the valves.
Pulmonary artery aneurysms represent focal dilatation involving all layers of the vessel wall. The normally expected diameter of the main pulmonary artery should not exceed 29 mm, and 17 mm for inter lobar arteries, a focal dilatation beyond their maximal caliber is defined as an aneurysm. 1
Pulmonary artery aneurysms can be due to a variety of causes ranging from congenital, to acquired secondary to pulmonary arterial hypertension, vasculitis, infections, neoplasms, connective tissue abnormalities and iatrogenic. 3
Mycotic aneurysms represent a specific subset of aneurysms resulting from infection of the vessel wall. Only a few cases of infectious aneurysms involving intra pulmonary arteries have been reported in the literature. 1
Furthermore, this type of aneurysms is associated to a high morbidity and mortality rate when not diagnosed and treated promptly.
Pulmonary artery aneurysms are found to be associated to congenital heart diseases in around 50% of the cases. However, among these affections very few cases of ventricular septal defects are reported in the literature. 6
The main risk factors for bacterial mycotic aneurysm on the other hand, include intra venous drug use, immunodeficiency and bacterial endocarditis.
It can develop from hematogenous spread of septic embolization and septicemia, or direct extension from an adjacent source of infection. 7
After septic embolization either bacterial or fungal, it can lead to progressive wall damage, arterial wall weakening and aneurysmal sac development. Mild injuries can develop into a true focal dilatation involving all wall layers, whereas severe destruction of the arterial wall tend to be associated with pseudo aneurysms. 3
In our case, the patient had history of a ventricular septal defect, although this congenital heart malformation could be in itself responsible for aneurysm formation it would have to involve the main pulmonary artery.
In fact, ventricular septal defect causes left to right shunts resulting in increased hemodynamic shear stresses with pulmonary artery volume and pressure overload resulting in major pulmonary artery dilatation. 2
Also, congenital cardiac abnormalities are at higher risk to develop bacteremia, subsequent endocarditis and consecutive septic emboli.8,9
The etiology was therefore certainly infectious since the patient presented with fever and signs of infective endocarditis.
Parts of the vegetations found on the tricuspid valve are thought to have dislodged and traveled through the pulmonary bloodstream allowing for the septic emboli insult to target the lungs as well as the distal pulmonary vessels. The location of vegetations determines the pathogenesis of mycotic aneurysms, involvement of the right cardiac chambers and valves, especially the tricuspid valve is more likely to cause pulmonary septic thrombo-emboli and subsequent pulmonary artery vessel walls lesions. 10
Staphylococcus aureus and Streptococcus are the 2 main pathogens encountered.
Mycobacterium Tuberculosis is also associated with aneurysms and pseudoaneurysms of the pulmonary artery, known as Rasmussen aneurysms. The diagnosis is readily made on the association of characteristic focal pulmonary dilatation located in the upper lobes and findings of post primary tuberculosis. 11
In the setting of a mycotic aneurysms related to infective endocarditis, trans-thoracic echocardiography is the first-line imaging modality especially to look for vegetations. Transesophageal echocardiography on the other hand seems to have a superior temporal and spatial resolution. 12
Computed tomography chest angiogram is the gold standard modality to identify focal dilatation of the pulmonary artery or one of its branches and remains the most effective imaging modality for treatment planning.13,14
The main imaging findings include parenchymal consolidation, well or ill-defined pulmonary nodules and/ or hilar enlargement. Three dimensional reconstruction further delineates the arterial configuration of the aneurysm arising from the pulmonary artery.
The feeding peripheral pulmonary artery can be easily identified supplying the aneurysmal sac.
It appears as a hyper enhancing nodular lesion with similar attenuation and equal enhancement to the adjacent vessel. Like in our case, we found a saccular dilatation of a peripheral pulmonary artery in the left lower lobe.
Adjacent consolidation, centrilobular nodules or pulmonary abscesses are indicative of local pulmonary suppuration.
Other associated features help narrow the diagnosis like in our case with pulmonary septic emboli. They appear as bilateral peripheral nodules, with possible cavitation and a characteristic feeding vessel sign.
Subpleural wedge shaped consolidations can be found related to pulmonary infarcts.
Pulmonary artery pseudo aneurysms secondary to pyogenic bacteria remains a rare condition. 11
Our patient had history of septal wall defect with infective endocarditis and subsequent hematogenous spread with septic emboli making him at high risk of developing such a condition. 15
Imaging presentation however, can sometimes be misleading for several reasons. The margins of the mycotic aneurysm can be difficult to distinguish from the adjacent parenchyma hence it can mimic a neoplasm. Some cases have even been reported to be mistaken for a lung mass. 14
Also, the aneurysm wall can be calcified making it challenging to detect the mycotic aneurysm especially if the lumen is partially or completely thrombosed. 16
We should therefore consider the medical history, clinical presentation and key imaging features with both septic emboli and saccular peripheral arterial dilatation, like in our patient, to make a confident diagnosis.
Treatment is based on infection control with antibiotic therapy adapted to the result of blood cultures.
Mycotic aneurysms treatment protocol however, remains relatively controversial. No consensus nor treatment guidelines currently exist. Determining the most appropriate treatment should be tailored to achieve durable results with the least invasive procedure.
A conservative approach can be discussed for clinically stable patients with no hemoptysis or significant symptoms. 17
In that matter, follow up CT angiogram is a valuable tool to monitor any change in size or status of the aneurysmal sac. No recommended time frame interval is clearly established although we suggest it would be interesting to perform follow up examinations, after treatment completion and clinical stabilization around 8 to 12 weeks. 2
More aggressive interventions have been advocated depending on aneurysm size and risk of rupture, with lobectomy, pneumonectomy or emergency surgical ligation in case of rupture. Although, given the higher risk of morbidity and mortality associated with this kind of surgeries, endovascular treatment is preferred when feasible. 18
A minimally invasive approach in interventional radiology using coils or detachable balloons have been reported to be a very promising technique. Bronchial embolization in particular represents a good alternative. Endovascular treatment is overall thought to best serve saccular aneurysms, both in the central and peripheral pulmonary arteries. Fusiform aneurysms of the peripheral pulmonary arteries may be treated endovascularly, central fusiform aneurysms however, require surgical management. 19
Conclusion
Congenital heart disease is a primary predisposing factor to endocarditis. Ventricular septal defect with infective endocarditis and left to right shunts can lead to pulmonary septic embolization and subsequent mycotic pulmonary aneurysm formation. Although mycotic aneurysms secondary to ventricular septal defect are uncommon, knowledge of their presentation and radiologic manifestations is important to recognize.
Association with infective endocarditis and secondary septic emboli are valuable findings to make the correct diagnosis. Our patient had history of septal wall defect with infective endocarditis and subsequent hematogenous spread with septic emboli making him at high risk of developing such a condition. A multimodality imaging approach is pivotal : first to diagnose infective endocarditis with echocardiography, then to allow accurate evaluation of the pulmonary artery aneurysmal dilatation and treatment planning with CT angiograms.
CT also gives valuable information to monitor any change in size or status of the aneurysm to help guide the best treatment strategy.
Hereby, computed tomography angiogram plays a key role, allowing for accurate and timely diagnosis to prevent life threatening complications such as rupture.
Footnotes
Author Contributions
SK collected the data, contributed to the to the selection of images, creation of the report and writing the manuscript. All authors read and approved the final manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was approved by Ethics Committee. The patients/participants provided their written informed consent to participate in this study. No potentially identifiable human images or data is presented in this study.
