Abstract
Background:
Fahr’s syndrome a rare neurological condition characterized by an abnormal basal ganglia calcification. The condition has both genetic and metabolic causes. Here, we describe a patient who had Fahr’s syndrome and basal secondary to hypoparathyroidism, and her calcium level raised after the administration of steroid therapy.
Case report:
We presented a case of a 23-year-old female with seizures. Associated symptoms included headache, vertigo, disturbed sleep, and reduced appetite. Her laboratory workup revealed hypocalcemia and low parathyroid hormone level, computed tomographic (CT) scan of the brain showed diffuse calcification in the brain parenchyma. The patient was diagnosed as a case of Fahr’s syndrome secondary to hypoparathyroidism. The patient was started on calcium and calcium supplementations along with anti-seizure therapy. Her calcium level raised after the initiation of oral prednisolone and she remained asymptomatic.
Conclusion:
Steroid could be considered as an adjunct therapy with calcium and vitamin D supplementation in patient whose Fahr’s syndrome is secondary to primary hypoparathyroidism.
Introduction
Fahr’s Syndrome also known as idiopathic basal ganglia calcification or Bilateral Striato Pallido Dentate Calcinosis (BSPDC) is an uncommon neurodegenerative disorder characterized by abnormal calcified deposits in the basal ganglia and other parts of the central nervous system. The calcification in Fahr’s syndrome is bilateral and symmetrical most commonly in the globus pallidus of the basal ganglia but may also occur in the thalamus, dentate nuclei, centrum semiovale, and rarely other intracerebral structures. 1
Fahr’s Syndrome was first reported and described by a German neurologist, Karl Theodor Fahr in 1930. Fahr’s Syndrome has an autosomal dominant inheritance pattern with both familial and non-familial cases and has an age predilection between 40 and 50 years with a prevalence of less than 1/1 000 000.2,3 The clinical presentation includes varying paresthesia, neuropsychiatric, parkinsonism, convulsive seizures, and cerebellar symptoms. 4 The pathogenesis and etiologies of Fahr’s syndrome are not clearly understood but endocrine abnormalities of calcium metabolism (Parathyroid), toxins, infections, and genetic causes may be involved.1,5
The diagnosis of Fahr’s Syndrome is based on the clinical presentation of progressive neurological dysfunction combined with the radiographic finding of calcified deposits and the exclusion of other etiologies. 3 Fahr’s Syndrome is as yet considered to be incurable and management strategies are mainly supportive to relieve symptoms and treatment of the causative etiology. Evidence suggests that early diagnosis and treatment of the causative etiology may reverse the calcification process and hence improve neurological dysfunction. 6 Here, we reported a unique case of Fahr’s syndrome secondary to primary hypoparathyroidism in which the patient calcium level raised after the addition of steroids as an adjunct to the replacement therapy.
Case Report
A 23-year-old female presented to the emergency department with a 1-day history of fits. It was described as a generalized tonic-clonic seizure with loss of consciousness for about 30 minutes. There was no aura or focal neurological deficit. She reported having headache, vertigo, disturbed sleep, and reduced appetite. The patient had a history of similar seizures that progressively worsened over the last 1 month. The patient was taking sodium valproate for the last 6 months but the seizures were uncontrolled despite of strict medication compliance. In addition, she also had generalized body weakness, intermittent twitching and tightness in the hands for the past 7 years. She did not have a history of seizures, head trauma, central nervous system infection, stroke, hypertension, diabetes, thyroid disease, or autoimmune disease. Family history was not significant for seizures disorder.
Her vital signs were as follows: blood pressure was 120/70 mm Hg; pulse rate was 74 beats per minute, respiratory rate of 20 breaths per minute, and body temperature of 37.3°C. Neurological examination revealed clear consciousness with no cognitive impairment or cranial nerve dysfunction. Her systemic and general physical examinations were unremarkable except for a positive Chvostek sign.
Initially workup was done for the metabolic causes of seizures and the laboratory evaluation revealed hypocalcemia as shown in Table 1
Initial laboratory investigations.

Abbreviations: g/dL, gram per deciliter; fL, femtoliter; mg/dL, milligram per deciliter; µL, microliter; U/L, units per liter.
Computed tomographic (CT) scan of the brain was ordered that diffuse brain parenchymal calcification involving globus pallidus, putamen, caudate, internal capsule, thalamus, dentate nucleus, and subcortical white matter in scattered areas of fronto parietal regions as shown in Figure 1.

A Computed tomographic scan of the brain showing diffuse calcification in the parenchyma of the brain involving bilateral globus pallidus, putamen, caudate, thalamic, cerebellar dentate nuclei, and subcortical white matter with subsequent atrophy in scattered areas of bilateral frontoparietal regions.
Based on the patient laboratory investigation and radiological finding a differential diagnosis of fahr’s disease and fahr’s syndrome were made. Initially the patient received injection calcium gluconate 20 mL of calcium gluconate 10% diluted in 50 mL of dextrose infused over 10 minutes by intravenous route, injection midazolam and an injection diavalproex sodium 500 mg. Patient was admitted to the medical unit and further workup for hypocalcemia was initiated that revealed hypoparathyroidism as shown in the Table 2.
Hypocalcemia workup.
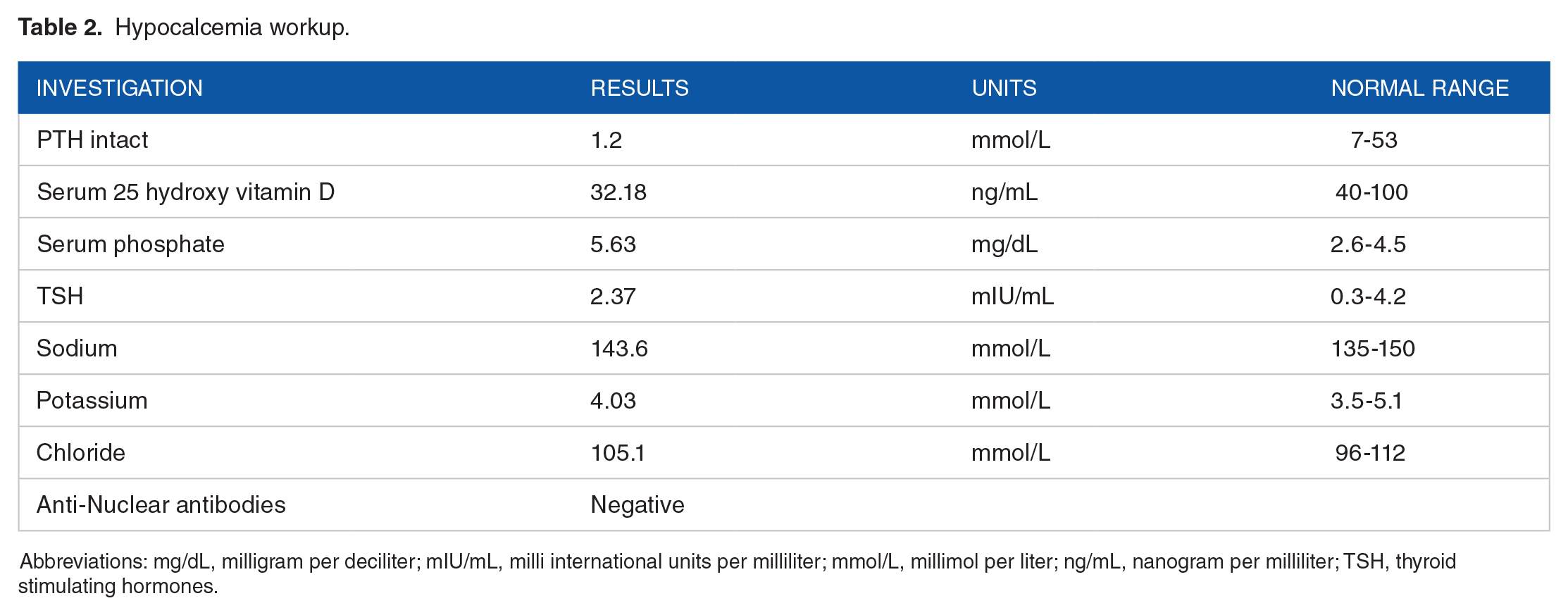
Abbreviations: mg/dL, milligram per deciliter; mIU/mL, milli international units per milliliter; mmol/L, millimol per liter; ng/mL, nanogram per milliliter; TSH, thyroid stimulating hormones.
Due to the absence of secondary causes that is, congenital defects, thyroid surgery, neck radiation in the patient history, negative anti-nuclear antibodies a diagnosis of Fahr’s syndrome secondary to primary hypoparathyroidism was made. The patient was placed on oral sodium valproate 500 mg twice daily, calcium citrate 600 mg twice daily, vitamin D3 50 000 once weekly for 6 weeks. At sixth week follow up, the patient remained symptomatic having 2 episodes of seizure in the last 15 days despite of strict compliance to anti-seizures and calcium supplements. Her calcium remained low (5.2 mg/dL). She was prescribed oral prednisolone 2 mg/kg for 4 weeks along with the anti-seizures and calcium and vitamin D supplement. After 1 month of starting the steroid therapy her calcium level raised to 7.2 mg/dL, she remained asymptomatic and her steroid therapy was tapered.
Discussion
Fahr’s syndrome or Fahr’s disease is suspected to be a calcification of the basal ganglia of the brain based on brain imaging studies of patients with neurological or psychiatric symptoms such as seizure, dyskinesia, dementia, and depression. Related diseases include hyperparathyroidism, hypoparathyroidism, hypoparathyroidism, hypervitaminosis D, tuberculosis, cytomegalovirus infection, toxoplasmosis, and astrocytoma. Fahr’s disease is diagnosed when the most prevalent parathyroid abnormalities are present along with other neurological or metabolic problems and basal ganglia calcification. 7 Although the exact pathophysiology of Fahr’s syndrome remains unclear, there is an established link to imbalances in calcium and phosphorus levels regulated by the parathyroid gland. Parathyroid hormone (PTH) is important in maintaining calcium levels in the cerebrospinal fluid and preventing phosphorus deposition in the periventricular regions, which may partially explain why hypoparathyroidism coincides with the occurrence of Fahr’s syndrome in some patients. 8 Although there are many specific hypotheses, the causes of abnormal calcification in the brain are related to parathyroid dysfunction or other causes of calcium metabolism overall 9
Brain calcification in Fahr’s syndrome is widespread at various sites; therefore, there is a high frequency of neurological symptoms. Some of the major complications found in patients are depression, dementia, psychosis, speech difficulty, epilepsy, recurrent falls and orthopedic consequences. 10 Clinical symptoms of Fahr’s syndrome are slow and differ into 3 subset of groups: movement disorders, neuropsychiatric features, and other central nervous system features. In the literature, several possible symptoms have been reported in patients such as dystonia, gait dysfunctions, seizures, and symptoms resembling parkinsonism. 11 López-Villegas et al reported that 18 patients with Fahr’s syndrome had different neurological symptoms according to calcified areas of the basal ganglia. 12 Hempel et al analyzed positron emission tomography (PET) images of patients with Fahr’s disease and found that glucose uptake was reduced in the temporal lobe, parietal lobe, and the basal ganglia 13 ; these functional changes in the brain cause a variety of mental neurological symptoms.
The major setback while making a differential diagnosis is the lack of precise criteria. Therefore a major criteria is to detect calcification foci greater than 800 mm in surface area. 14 Electroencephalography is not particularly useful and has a low diagnostic value, due to all kinds of alterations in the central electrophysiological activity are possible. The innovation and availability of computed tomography (CT) has greatly increased the detection of intracranial calcifications and the number of diagnosed Fahr’s syndrome cases. Currently, CT has become the gold standard mode of diagnosis and has even surpassed the use of magnetic resonance imaging (MRI). Calcified areas can be visualized as hyperdense lesions on CT, and that is considered decisive for accurate diagnosis. 14
Thus, neurological and psychological examinations as well as CT imaging remain the basic techniques for the diagnosis of Fahr’s syndrome, other diagnostic criteria used for diagnosing Fahrs syndrome in our patient include Bilateral calcifications of the basal ganglia of the brain on neuroimaging (may not apply to patients from families with Fahr’s syndrome); Progressive neurological dysfunction and/or psychiatric symptoms; Absence of biochemical abnormalities and somatic states suggestive of metabolic or mitochondrial disease; Absence of toxic, infectious or traumatic causes of intracranial calcifications; Positive family history of Fahr’ syndrome. 14
In our patient, we had considered metabolic/endocrine disorders such as hypoparathyroidism or pseudohypoparathyroidism, Hemochromatosis/Wilson’s Disease, or other Idiopathic causes. These were ruled out one by one after we met the diagnostic criterion for Fahr syndrome. Our patient’s blood tests showed hypocalcemia, hyperphosphatemia, low PTH, and low vitamin D. As our patient did not have a history of surgery, radiation therapy, or autoimmune disorders to suggest secondary hypoparathyroidism, these findings indicated primary hypoparathyroidism. Moreover, the imaging findings of the symmetric and extensive calcification are usually typical features as our patient showed bilateral basal ganglia calcification on a head CT, which suggested the diagnosis of Fahr’s syndrome secondary to hypoparathyroidism.
Unfortunately at present, there is neither a specific cure for Fahr’s syndrome nor a standard course of treatment. The main treatment modality of Fahr’s syndrome focuses on symptomatic management and correction of calcium and phosphate levels. 15 Any etiologies of hypoparathyroidism are treated with the standard therapy of active vitamin D such as calcitriol and calcium supplements. If calcitriol is not available cholecalciferol can also be used. In addition, calcium supplements are given with a dose of 1 to 3 g in divided doses. The desired therapeutic result is to maintain the serum calcium close to the lower limit of the normal range. 16 Evidence from prior case reports has suggested that correcting underlying abnormalities may improve the presenting symptoms in cases where metabolic disturbances are found. Since hypoparathyroidism is a common cause, we can set dietary regimens for the patient. Antipsychotic, antidepressant, and/or antiepileptic medications with appropriate rehydration and electrolyte and hemodynamic maintenance are given in combination for the symptomatic treatment of the neurological and psychiatric symptoms. If the patient calcium level is not responding to the replacement therapy in case of primary hypoparathyroidism, systemic steroid should be considered as adjunct therapy. Meanwhile, the patient urinary calcium should be monitored. 17
Conclusion
Further workup should be done in case of abnormal brain calcification to find out the secondary causes. If Fahr’s syndrome is secondary to primary hypoparathyrodism, and the patient calcium level does not respond to replacement therapy a trial of systemic steroid could be considered.
Footnotes
Author Contributions
S.Y.S and M.I coceived the idea and were the treating physicians of the patient. Q.A.K collected the data. F.A.H, Y.L.C, H.P, M.D.O and A.N wrote the original manuscript. Q.A.K critically revised the manuscript and did final editing. All the authors reviewed and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent
Written informed consent was taken from the patient for publication of this case report and any accompanying images.
