Abstract
It is already known that Coronavirus disease 2019 (COVID-19) may lead to various degrees and forms of lung parenchyma damage, but some cases take a strikingly severe course that is difficult to manage. We report the case of a 62-year old male, non-obese, non-smoker, and non-diabetic, who presented with fever, chills, and shortness of breath. The infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was diagnosed by real-time Polymerase Chain Reaction. Although the patient had been vaccinated with 2 doses of Pfizer-BioNTech COVID-19 vaccine 7 months earlier and had no risk factors for a severe outcome, serial computed tomography (CT) scan revealed lung involvement progressively extending from an initial 30% to 40% to almost 100% 2.5 months later. The spectrum of lung lesions included at first only ground-glass opacities and some tiny emphysema bullae, but later also bronchiectasis, pulmonary fibrosis, and large emphysema bullae as post-COVID-19 pulmonary sequelae. For fear of severe evolution of superimposed bacterial infection (Clostridoides difficile enterocolits and possibly bacterial pneumonia) the administration of corticosteroids was intermittent. Massive right pneumothorax secondary to bulla rupture, possibly favored by the indispensable high flow oxygen therapy, led to respiratory failure compounded by hemodynamic instability, and ultimately to the patient’s final demise. COVID-19 pneumonia may cause severe lung parenchyma damage which requires long-term supplemental oxygen therapy. Beneficial or even lifesaving as it might be, high flow oxygen therapy may nonetheless have deleterious effects too, including the development of bullae that may rupture engendering pneumothorax. Corticosteroid treatment should probably be pursued despite superimposed bacterial infection to limit the viral induced damage to lung parenchyma.
Background
More than 2 years have passed since the COVID-19 pandemic started, during which time the diagnosis, evolution, and treatment have been intensively studied. The observance of the current treatment guidelines increases the chance of a favorable outcome for patients with COVID-19, but some cases have an unpredictable course that is difficult to manage. We present the case of a patient diagnosed with COVID-19 pneumonia, who had a progressively unfavorable course, leading to patient’s death due to severe pulmonary sequelae.
Case presentation
A 62-year old caucasian non-smoker male, presented to the emergency department with an 8 days history of fever and chills, followed by shortness of breath 2 days prior to his presentation. He denied cough or sputum production. The patient had a history of benign prostatic hyperplasia and dyslipidemia and was on daily treatment with Tamsulosin and Rosuvastatin. The patient had received 2 doses of Pfizer-BioNTech COVID-19 vaccine, the second 7 months earlier, the first about 3 weeks prior to the second. The physical examination revealed a normal weight male, with a slight fever (37.8°C), normal breath sounds with bilateral crackles in the middle, and lower lung fields, a respiratory rate of 25 min, a peripheral oxygen saturation (SpO2) of 89% on room air that climbed to 98% when the patient was given oxygen at a rate of 4 L/min on simple face mask, normal blood pressure (125/65 mmHg), regular heart rhythm at a rate of 90 bpm, soft abdomen with no tenderness, normal urine output, no edema. Arterial blood gas analysis on admission revealed type 1 respiratory failure (pH: 7.48, pCO2: 29.9 mm Hg, paO2: 56.8 mm Hg, arterial oxygen saturation: 92%). Blood tests revealed normal hemoglobin (12.6 g/dL), thrombocytes (158 000/μL) and leukocytes (5750 μL) but lymphopenia (800 μL), high inflammatory markers: C-reactive protein (CRP = 149.21 mg/L), serum ferritin (>500 ng/dL), erythrocyte sedimentation rate (ESR = 41 mm/h), and interleukin-6 (IL-6 = 267.98 pg/mL) with normal fibrinogen (486 mg/dL). D-dimers were not significantly increased (0.833 µg/mL). Renal and hepatic function tests were normal. High-sensitivity cardiac troponin was normal (12.8 ng/L). The only abnormality on the 12-lead ECG was sinus tachycardia. At admission, the real-time Polymerase Chain Reaction for SARS-CoV-2 was positive. Anteroposterior chest radiograph showed bilateral ground-glass opacities in the mid and lower zones (Figure 1). Nonenhanced thorax CT scan revealed isolated consolidation areas in the upper (left > right) and lower lobes, diffuse, predominantly subpleural, consolidation in the lower lobes associated with bronchiectasis, and mild, diffuse, bilateral centrilobular, and subpleural emphysema, as well as nonspecific enlargement of upper and middle mediastinal lymph nodes (Figure 2A). The percentage of lung involvement was estimated to 30% to 40% by visual assessment. According to the COVID-19 treatment guidelines1,2 (including local ones 3 ), the antiviral drug Remdesivir was given in combination with systemic corticosteroids (Dexamethasone 8 mg intravenous) and antithrombotic therapy (Dalteparin 5000 units subcutaneous once daily). CRP decreased in the next 2 days (61.98 mg/L respectively 30.70 mg/L), and therefore the patient was not started on an immunomodulatory agent as, according to the local COVID-19 treatment protocol, such an agent is not warranted if inflammatory markers decrease following corticosteroid combined with antiviral treatment. Blood glucose level increased significantly, probably because of corticosteroid therapy.
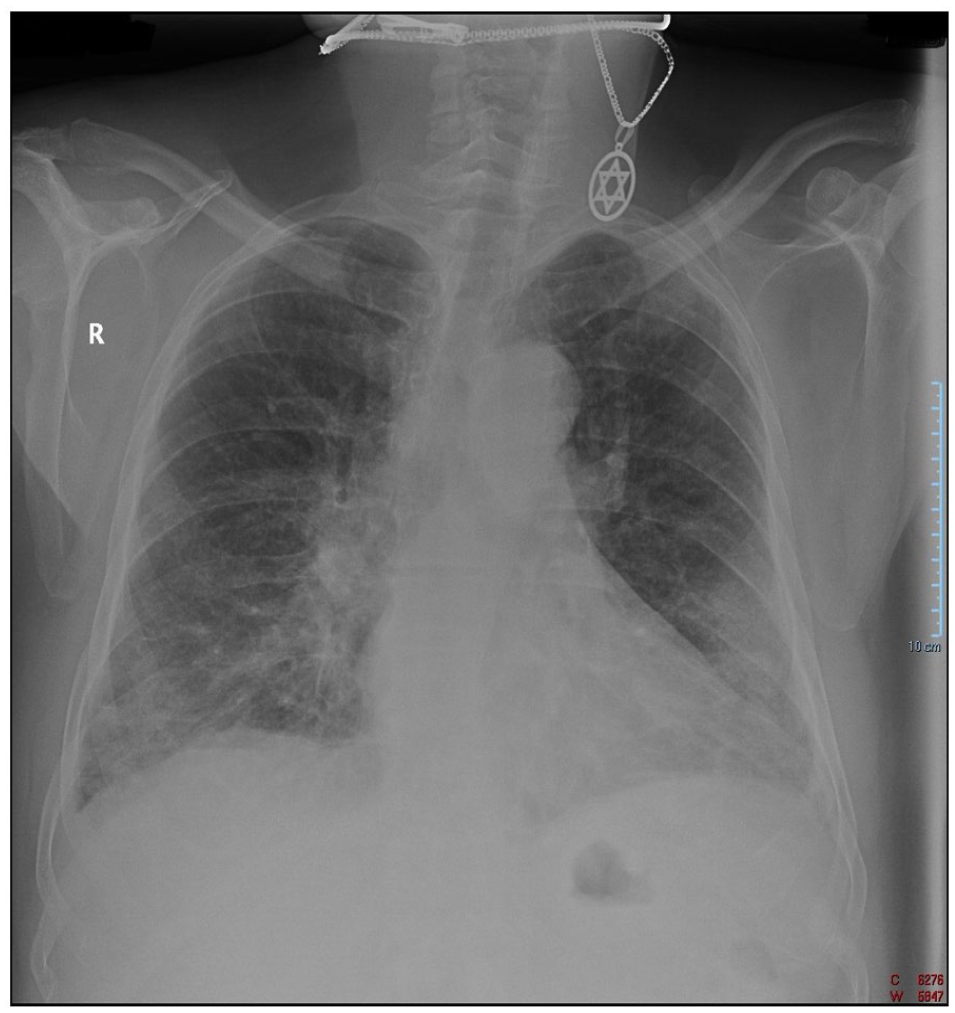
Anteroposterior chest radiograph: bilateral, ground-glass opacities in the mid, and lower zone.

Thorax CT scan. (A) (at admission): diffuse consolidation in the subpleural areas of the upper, middle and lower lobes with ground glass opacities associated with bronchiectasis and mild emphysema, (B) (2 weeks later): widespread ground-glass opacities and consolidation areas in both lungs, (C) (2 months later): ground-glass opacities progression leading to 100% lung involvement, subpleural reticular pattern, cavitary lesions, thin-walled with several septum-like structures inside in right upper, and lower lobe.
On the third in-hospital day (22.10) the patient developed diarrhea, which proved to be Clostridoides enterocolitis, paralleled by a surge in CRP (256.03 mg/l), fibrinogen (621 mg/dl), ESR (67 mm/h), and leukocyte count (13 400 /μL) (the latter could have also been the result of corticosteroid therapy). The only identifiable risk factors were proton pump inhibitors 4 and corticosteroids, 5 therefore these agents were discontinued and oral Vancomycin (250 mg every 6 hours) and intravenous Metronidazole were started with consequent diarrhea resolution and a drop in CRP (79.51 mg/L) and fibrinogen (482 mg/dL), but with persistent leukocytosis (14 000 /μL) after 6 days of treatment (28.10). The treatment for Clostridoides enterocolitis was continued for 15 days (25.10-10.11).
On the seventh day (26.10), shortness of breath worsened, fever recurred, and SpO2 decreased. In the next days, progressively higher oxygen flow was needed to maintain a satisfactory SpO2 (up to 20 L/min on the 14th day) (02.11). Blood and a urine culture were negative. Blood tests revealed an increase in fibrinogen (707 mg/dL), ESR (72 mm/h), and leukocyte (18 600 /μL) and neutrophil count (15 900 /μL) with a lymphocyte count of 1300 /μL and a slightly elevated procalcitonin (0.5 ng/ml). A new contrast enhanced thorax CT scan was performed on the 15th day, which revealed widespread ground-glass opacities and consolidation areas in both lungs, involving 70% of lung parenchyma (Figure 2B), as well as nonocclusive blood clots in various segmental arteries in both lungs and enlarged mediastinal lymph nodes. As the increase in neutrophil count could not be attributed to corticosteroid treatment (as this had been already stopped), the infectious disease specialist suspected a bacterial lung infection (superimposed on COVID-19 pneumonia) as the main culprit for the progression of the lung involvement and recommended Meropenem in addition to oral Vancomycin (03.11); therefore, the patient was started on Meropenem (1000 mg thrice a day intravenously). However, corticosteroid treatment was also resumed (intravenous Dexamethasone at a dose 16 mg/day) for fear that COVID-19 pneumonia could also have contributed to the increased lung involvement. Starting the patient on an immunomodulatory drug (Baricitinib) was also considered appropriate and the drug was asked to the hospital’s pharmacy department, which unfortunately could not provide it due to the limited availability. Given the blood clots in the segmental pulmonary arteries, unfractionated heparin as a continuous infusion was given for 2 days, followed by low-molecular-weight heparin (Dalteparin 7500 units twice daily).
As SpO2 declined to 81% on 20 L/min oxygen, continuous positive airway pressure (CPAP) noninvasive ventilation was started. SpO2 remained satisfactory under CPAP noninvasive ventilation and after 10 days the patient was switched to high flow oxygen therapy with an oxygen flux of 35 L/min and a FiO2 of 75% (on the 28th day) (15.11).
Dexamethasone dose was reduced to 8 mg/day on the 25th day then stopped on the 35th day (21.11) as the patient developed again diarrhea (on the 34th day). Repeated tests for Clostridioides difficile toxins were at first negative, but finally became positive (on the 40th day). Therefore, the patient was once again started on oral Vancomycin (250 mg every 6 hours), which was continued for another 14 days. The inflammatory markers had a surge, which was attributed to the Clostridioides difficile infection, and then fluctuated and finally dropped to almost normal values (CRP = 3.00 mg/L, fibrinogen = 287 mg/dL, ferritin = 373.1 ng/dl, IL-6 = 7.48 pg/mL, while leukocytes and lymphocytes reached normal levels (9200 μL and 1900 μL respectively).
Although the patient continued to need high flow oxygen therapy, the oxygen flow could be reduced progressively down to 10 L/min and FiO2 95%. Repeated trials of switching the patient back to oxygen by facial mask were unsuccessful. As it is well known that barotrauma might be due to the wrong mechanical ventilator setting, 6 the ventilator parameters were checked daily and set to the lowest oxygen flow to provide a convenient SpO2 with the lowest risk of barotrauma. The patient began a rehabilitation program including breathing and mobilization exercises supervised by a physiotherapist. Unfortunately, contrast enhanced thorax CT scan performed about 2 months after admission (22.12) showed ground-glass opacities progression encompassing the entire lung parenchyma and thin-walled cavitary lesions with septum-like structures inside in the right upper and lower lobes (the largest 2 having transaxial diameters of 86/58 mm and 37/34 mm), besides emphysematous spaces, bronchiectasis, pulmonary fibrosis, and persistent nonspecific enlarged mediastinal lymph nodes (Figure 2C). Post-COVID-19 organizing pneumonia was suspected and consequently pulse methylprednisolone therapy (500 mg for 3 days) was given (24-26.12), followed by Prednisone 40 mg/zi. On the 78th day (12 days into the corticosteroid therapy course), the patient developed shortness of breath, stabbing chest pain, and dry cough with no audible breath sounds on right hemithorax auscultation. The contrast enhanced thorax CT scan (04.01) demonstrated massive right pneumothorax inducing complete right lung collapse, left displacement of the mediastinum, and left lung compression (Figure 3). A chest tube drain was inserted into the right pleural cavity, but endotracheal intubation and mechanical ventilation were needed due to severe respiratory failure. Chest radiograph performed 2 days later (06.01) demonstrated almost complete right lung reexpansion with only a small residual apical pneumothorax (Figure 4). Despite advanced medical life support the patient had an unfavorable course, continuing to need mechanical ventilator support. A few days later, he became hemodynamically unstable requiring norepinephrine to maintain arterial pressure and finally died after a 3 months in-hospital stay.

Thorax CT scan: massive right pneumothorax associated with complete right lung collapse, displacement the mediastinum and compression the left lung.

Anteroposterior chest radiograph: lung reexpansion with small right-sided apical pneumothorax and diffuse ground-glass opacity.
Figure 5 represents a summary of the patient’s course in terms of various complications, most relevant laboratory tests, imaging studies, and treatment, while Table 1 is a summary of lab results at the time of each main clinical event

The summary of the patient’s course.
Summary of lab results at the time of each main clinical event.
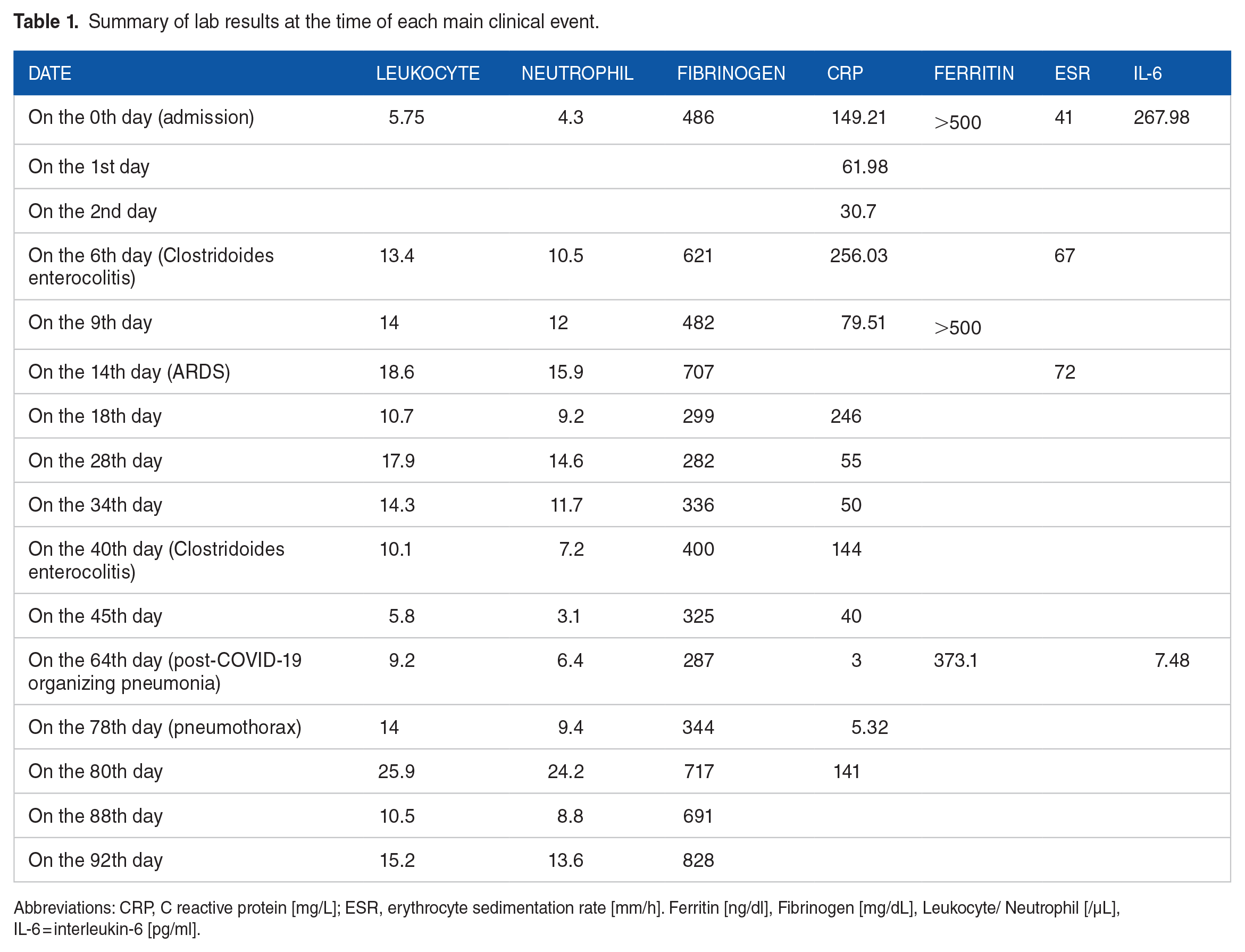
Abbreviations: CRP, C reactive protein [mg/L]; ESR, erythrocyte sedimentation rate [mm/h]. Ferritin [ng/dl], Fibrinogen [mg/dL], Leukocyte/ Neutrophil [/µL], IL-6 = interleukin-6 [pg/ml].
Discussion
COVID-19 is caused by SARS-CoV-2 and may lead to various degrees of respiratory illness, from mild, even asymptomatic, to severe requiring hospitalization, and supplemental oxygen. 7 Unfavorable outcomes in COVID-19 are linked to many risk factors, including obesity, smoking current or former, chronic lung diseases, chronic liver diseases, diabetes mellitus, heart disease.8-10 The risk is higher in unvaccinated individuals, but also in vaccinated ones aged 75 years or older and in those aged 65 years or older and with additional risk factors.1,2 Our patient was a relatively young, vaccinated individual with no risk factors.
Inflammatory markers, including CRP, ferritin, procalcitonin, ESR, IL-6, are used to assess the severity and prognosis of COVID-19. 11 A meta-analysis showed that inflammatory markers are much higher in severe forms compared to mild ones and can predict the progression of the COVID-19 to a severe form. 11 In our case, inflammatory parameters, especially IL-6, predicted a severe outcome. The local protocol 3 recommends the administration of immunomodulatory agents only if the inflammatory and respiratory status of the patients worsens despite antiviral treatment combined with corticosteroids. Indeed, initially the level of inflammatory markers declined, therefore immunomodulatory treatment was not considered appropriate. Later, inflammation intensified, but that seemed to be the result of a bacterial infection (enteral with Clostridoides difficile and/or respiratory) as it was accompanied by a rise in leukocyte count and therefore immunomodulatory treatment was not considered adequate. Later however, when CT scan showed the progression of the pulmonary lesions, an immunomodulatory agent was considered necessary, and prescribed but, on shortage grounds, the pharmacy department did not supply it. It should be noted that a bacterial infection was also suspected at that time, but this was not considered a contraindication for the immunomodulatory treatment. 12
Radiological sequelae after COVID-19 may vary depending on the infection severity including ground-glass opacities, fibrosis-like changes, bronchiectasis, and bullae, the latter occurring as a result of a resorption process following the pulmonary lesions inflicted by COVID-19. 13 Based on several studies, which showed that ground-glass opacities and consolidation14,15 may be reversible under steroid therapy15-18 methylprednisolone pulse therapy followed by prednisone was started in the hope of improving radiological changes and implicitly patient’s outcome.
The formation of pulmonary emphysematous bullae (whose rupture may lead to pneumothorax) has already been described in COVID-19,13,19 and may be ascribed to the severe damage to the alveolar walls and the consecutive fibrous changes. 20 However, it is thought that non-invasive or invasive forms of ventilation may also be involved in the incidence of these complications.20-22 It is known that mechanical ventilation can lead to barotrauma and pneumothorax risk is high in some circumstances especially adult respiratory distress syndrome, pneumonia, chronic obstructive lung disease, positive pressure ventilation. 23 Lung diseases affect lung structure and compliance increasing alveolar pressure and predisposing to barotrauma. 24 Although no single strategy prevents barotrauma, we typically use lung protective ventilatory approaches by limiting plateau pressure (Pplat) ⩽30 cm H2O and using low tidal volume ventilation (6-8 mL/kg ideal body weight [IBW]). Additional prevention principles include avoiding overventilation, adopting measures to avoid or treat dynamic hyperinflation (auto-positive end-expiratory pressure [PEEP]), and the cautious use of high levels of PEEP. The risk of barotrauma is not eliminated until the underlying predisposing condition is resolved and the patient is weaned from mechanical ventilation. It is debatable whether the corticosteroid treatment given for organizing pneumonia may have also contributed to the development of pneumothorax.25,26
Conclusion
Distinctive in the present case was the uncertainty regarding the most appropriate treatment because of the coexistence of the viral infection with a bacterial infection (a certain one, Clostridoides enterocolitis, and a presumed one, lung infection superimposed on the viral pneumonia) which led to the intermittent administration of corticosteroids which may have contributed to the progression of the lung injuries. The lack of immunomodulatory treatment (due to scarcity) may have also had a hand in this. COVID-19 pneumonia may cause severe lung parenchymal damage, which requires long-term supplemental oxygen therapy. Beneficial or even lifesaving as it might be, high flow oxygen therapy and corticosteroid treatment may nonetheless have deleterious effects, including the development of bullae that may rupture, engendering pneumothorax. Corticosteroid treatment should probably be pursued despite superimposed bacterial infection to limit the viral induced damage to lung parenchyma.
Footnotes
Acknowledgements
Not applicable.
Author Contributions
MIG, MMM, DT, AEBS, DI, RT contributed equally to this work and share first authorship.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Note
Name of Department and Institution where the work was done: 1st Internal Medicine Clinic of University Emergency Hospital Bucharest.
Consent for Publication
Written informed consent was obtained from the patient (when he was alive) for publication of the case report and any accompanying images. After patient’s death, a family member (the patient’s sister) has also provided written informed consent for publication of the data and images pertaining to this case.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
