Abstract
Hydralazine is an antihypertensive medication that has been associated with drug-associated autoimmune syndromes like interstitial lung disease, pauci-immune glomerulonephritis, and hypocomplementemia. Hydralazine-induced ANCA-associated vasculitis and hydralazine induced positive antinuclear antibodies are rare. Clinical manifestations range from arthralgia, petechiae, or rash to multiorgan involvement. When presented as pulmonary-renal syndrome, it can be rapidly progressive and fatal. Here, we describe a case of hydralazine-associated vasculitis with rapidly progressive glomerulonephritis and highlight the importance of early diagnosis and prompt treatment.
Key Clinical Message
The most life-threatening complication of hydralazine use in patients is the development of pulmonary-renal syndrome or ANCA-associated vasculitis. Primary care physicians should be aware of this potential complication as timely diagnosis with urinalysis, and kidney biopsy, and prompt treatment with withdrawal of hydralazine along with corticosteroids and immunosuppressive agents are essential to stop the multi-organ damage.
Background
Hydralazine is a widely prescribed vasodilator that is used in the clinical setting—both inpatient and outpatient since 1951. 1 Hydralazine-induced ANCA-associated vasculitis and lupus was first described in 1953. 2 It is a rare entity which can be missed due to myriad clinical presentation and when lupus and ANCA-associated vasculitis occur together.
Hydralazine-induced antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis is a necrotizing vasculitis that affects small vessels in a multisystem organ involvement fashion including lungs, kidney, skin, heart, blood, and joints. In the literature, only 4 studies have described kidney-limited presentation of hydralazine-induced ANCA-associated vasculitis.3 -7
We review this case to discuss and highlight the importance of a high degree of clinical acumen, and early diagnosis for better clinical outcomes.
Case Presentation
A 71-year-old Caucasian female with a history of stage 3 chronic kidney disease, hypertension, and congestive heart failure with reduced ejection fraction (45%) presented to the hospital with shortness of breath, productive cough, and hypertensive urgency. On presentation, she was afebrile with a blood pressure of 190/92 mm Hg, heart rate of 86 beats per minute, pulse oxygenation of 95% on 2 L of oxygen. On physical examination, the patient had tachypnea, fine rales, and pitting pedal edema. Initial laboratory workup showed a serum creatinine of 2.6 mg/dL, blood urea nitrogen of 88 mg/dL with an estimated glomerular filtration rate of 18 mL/min/1.73 m2 (eGFR; baseline is 40-45 mL/min/1.73 m2), potassium of 4.5 mmol/L, and hemoglobin of 7.8 g/dL. A urinalysis showed 3+ protein, large blood, and >100 red blood cells (RBC)/high power field with spot urine protein creatinine ratio of 2.7 g. Chest X-ray was notable for multifocal pneumonia and mild central vascular congestion. A kidney ultrasound showed smaller sized kidneys (9.3 cm on the right and 9.6 cm on the left) with no hydronephrosis, calculi, but mild to moderate diffuse parenchymal thinning. Her home medications included furosemide 40 mg daily, metoprolol 25 mg twice daily, and lisinopril 40 mg daily in addition to multivitamins and acetaminophen. Despite this, patient’s blood pressure was uncontrolled in the outpatient setting and she was started on hydralazine 50 mg 3 times a day 1 month prior to this admission. The patient otherwise had no family history of kidney disease.
She was admitted to the intensive care unit for worsening hypoxic respiratory failure, and was initiated treatment with meropenem, linezolid, and levofloxacin for possible sepsis and clevidipine drip for a brief period for hypertensive urgency. CT chest was done which showed bilateral diffuse infiltrates and bronchoscopy revealed occasional blood which raised suspicion for a possible diffuse alveolar hemorrhage. The bronchoscopy results, in conjunction with her proteinuria and rapidly declining renal function (creatinine increased from 2.6 mg/dL to 6.5 mg/within the first 7 days of hospitalization), led us to believe that the patient’s acute kidney injury was possibly due to a nephritic syndrome. Further investigation revealed hypocomplementemia with low C3 (83 mg/dL; reference range 88-201) and normal C4 (20 mg/dL; reference range 10-40) levels. Anti-nuclear antibodies (ANA) along with anti-histone antibody was positive. Anti-dsDNA was positive at 10 IU/mL (negative is <10 IU/mL), anti-proteinase 3 (Anti-PR3) was positive at 11.3 EU/mL (negative is <0.4 EU/mL), and both anti-myeloperoxidase (anti-MPO) and anti-glomerular basement membrane (Anti-GBM) were negative. With the raising concern for ANCA vasculitis and drug-induced positive anti-histone antibodies, hydralazine was stopped. Pulse intravenous steroids were administered while awaiting kidney biopsy. In the meantime, patient was initiated on hemodialysis for concern of uremia.
The kidney biopsy (Figure 1) specimen contained 32 glomeruli, 2 of which were globally sclerosed and about 5% to 10% interstitial fibrosis. Three small cellular crescents (Figure 2) were identified. There was no significant endocapillary proliferation, no evidence of significant immune complex disease. Immunofluorescence showed trace mesangial staining for IgM and C3 of doubtful significance. Electron microscopy showed moderate foot process effacement of podocytes with corrugation of glomerular basement membranes (Figure 3) without significant subepithelial or subendothelial immune complex deposits. Endothelial cells were swollen without reticular aggregates. There were no significant mesangial immune complex or tubular basement membrane deposits.
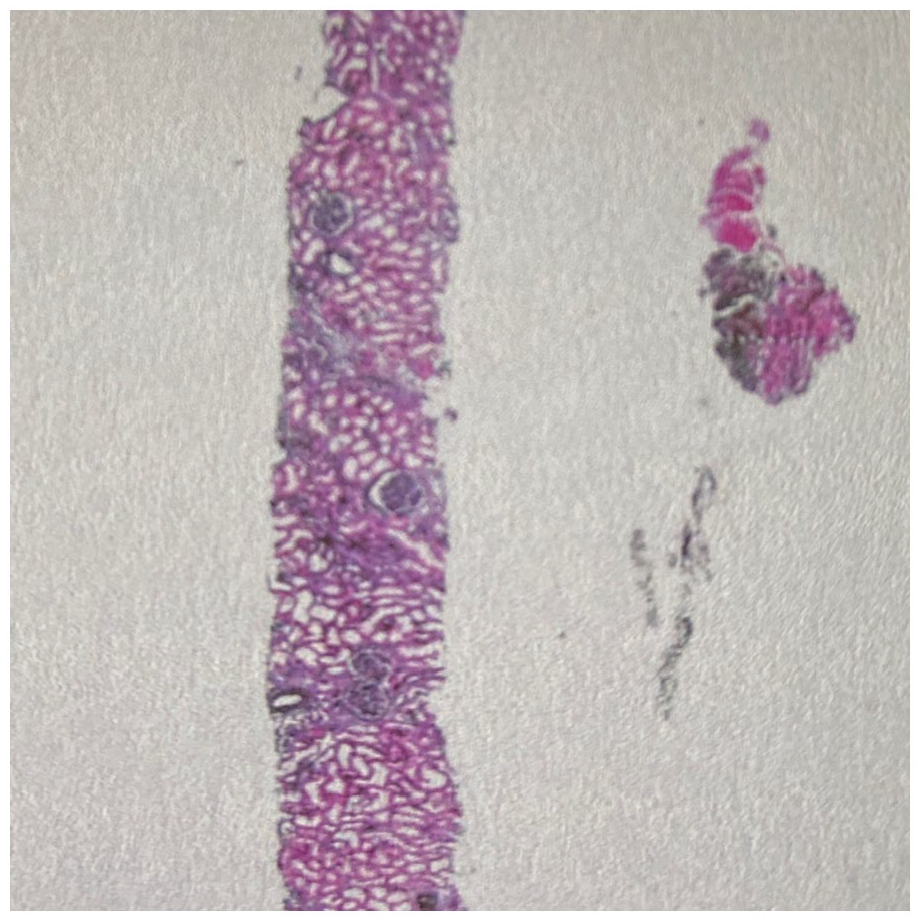
The kidney biopsy specimen contained 32 glomeruli, 2 of which were globally sclerosed and about 5% to 10% interstitial fibrosis.
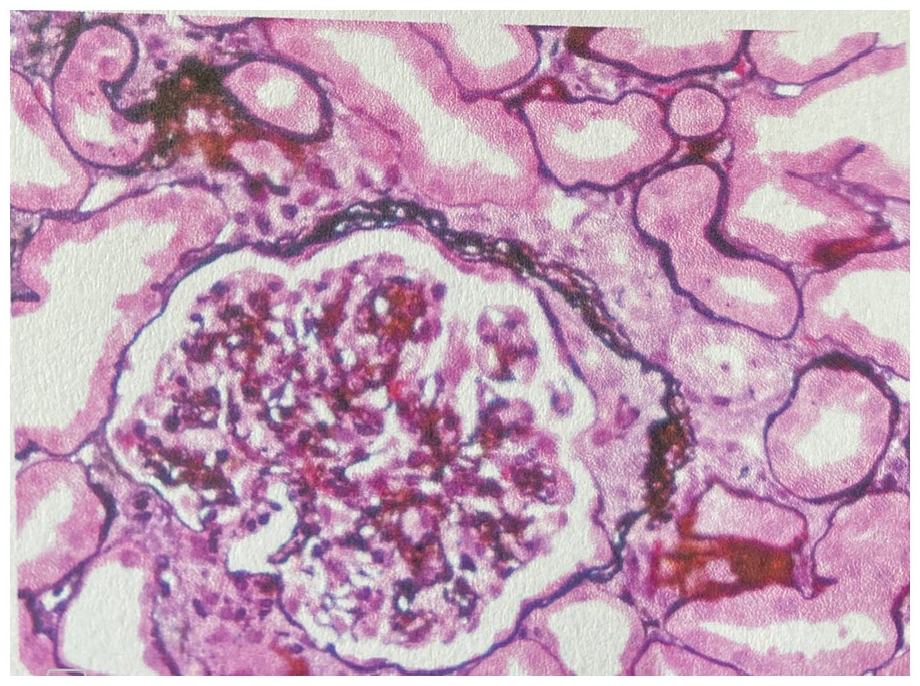
Cellular crescent identified.

Electron microscopy showed moderated foot process effacement of podocytes with corrugation of glomerular basement membranes without significant subepithelial or subendothelial immune complex deposits.
The patient was established on hemodialysis and treatment with intravenous rituximab 375 mg/m² once a week for 4 weeks for immunosuppression in addition to a taper of methylprednisolone 70 mg daily over a 3 to 4-month period. She received atovaquone 1500 mg daily for pneumocystis pneumonia prophylaxis and she received entecavir weekly for prophylaxis against hepatitis B since her hepatitis total core antibody was positive. The patient remained clinically stable with improvement in oxygen requirements to being on room air, blood pressure controlled on furosemide 40 mg on non-dialysis days, metoprolol 25 mg twice a day, lisinopril 40 mg daily, and clonidine 0.1 mg 3 times a day at the time of discharge. Over the next 3 months, the patient was taken off hemodialysis and she remain on prednisone 20 mg daily.
Discussion
Hydralazine was first discovered as a treatment for malaria but given its direct smooth muscle vasodilator effect, it has been used as an adjunctive treatment for hypertension, heart failure with reduced ejection fraction, and in hypertensive emergencies in pregnancy. 8 Hydralazine-induced ANCA-associated vasculitis and hydralazine induced lupus (HIL) are rare presentations, has an incidence of 5.4% in patients on 100 mg/day to 10.4% with 200 mg/day for >3-years duration 9 and incidence of 7% to 13% for HIL. 10
In addition to AAV, hydralazine is known to cause or result in other positive serologies such as ANA, anti-double stranded DNA, anti-histone antibodies, etc. A study performed by Kumar et al 8 revealed that 323 patients with ANCA-associated vasculitis (12 had exposure to hydralazine) had serologic features overlapping with SLE, as all 12 were found to be positive for ANA (titers 1:160 to 1:2560; 10 diffuse pattern and 2 nucleolar), ANCA (titers 1:320 to 1:2560; 11 perinuclear ANCA pattern, 1 cytoplasmic ANCA), and anti-histone. Eleven of the 12 patients also were positive for anti-MPO and anti-double stranded DNA (anti-dsDNA), 4 had positive anti-cardiolipin IgG or IgM, and 9 had hypocomplementemia. A literature review performed by Battisha et al 11 showed that of 35 patients with lung-kidney syndrome secondary to hydralazine-induced, ANCA-associated vasculitis, 29 of 33 had positive ANA, 33 of 34 had positive anti-MPO, 6 of 19 had positive anti-PR3, 20 of 21 had positive anti-histone, and 12 of 30 had positive anti-dsDNA antibodies; 9 of 22 had low C3 and C4; none of 13 had anti-GBS antibodies.
Pathogenesis of hydralazine-induced ANCA associated vasculitis include binding to myeloperoxidase (MPO) leading to neutrophil apoptosis resulting in the production of multiple antibodies, reversal of epigenic silencing of MPO and proteinase3 (PR3) leading to increased expression of neutrophil autoantigens and a break in tolerance in slow versus fast acetylators of hydralazine. 12
Hydralazine induced lupus usually present with positive antinuclear and anti-histone antibodies. It usually presents with systemic symptoms but standalone kidney involvement is uncommon (<5%). Pathogenesis of hydralazine induced vasculitis and lupus is unknown but could be related to its auto-immunogenic potential-decrease in DNA methyltransferase expression which inhibits extracellular signal-regulated kinase (ERK) pathway signaling resulting in disruption of proteinase 3 (PR3) and MPO, 13 metabolism of hydralazine by MPO released from activated neutrophils to form reactive intermediates that result in the formation of anti-MPO antibody. It was also reported that hydralazine-induced vasculitis is more common in slow acetylators since hydralazine acetylation will be slower and that gives more chance for a break in tolerance. 14 Antibodies associated with Hydralazine induced vasculitis include MPO-ANCA, ANA, Anti-histone, anti-elastase, and antiphospholipid antibody. The combination of anti-histone antibody, MPO, and/or PR3 ANCA and absence of anti-dsDNA antibody could be used to support the diagnosis of hydralazine-induced vasculitis in the appropriate clinical setting with evidence of pauci-immune glomerulonephritis.
Risk factors for ANCA associated vasculitis-include cumulative dose of more than 100 g, female gender, and thyroid disease. 15 Other risk factors include HLA-DR4 genotype, slow hepatic acetylation, and the null gene for C4. 16
Several risk factors for drug-induced lupus have been identified: the drug dose (more than 200 mg/day and/or a cumulative dose of more than 100 g), female sex, slow hepatic acetylation, HLA-DR4 genotype, and the null gene for the fourth component of the complement, C4. 16
Even though the patient had positive ANA, anti-histone antibody, and ANCA, the presence of pauci-immune glomerulonephritis instead of immune complex deposits on kidney biopsy helped us to make a definitive diagnosis. While lupus nephritis usually presents with immune complex deposits when kidney function is severely impaired (class III, IV, V, and VI), biopsy would show an immunofluorescence profile with staining for IgG, IgA, C3, and C1q. However, the renal biopsy in our patient only showed weak, nonspecific IgM. Active lupus nephritis would also be expected to show endocapillary proliferation and wire loops and/or hyaline thrombi, which were mentioned as pertinent negatives in our patient’s kidney biopsy.
Currently, there are no guidelines or randomized control trials for the treatment of hydralazine induced vasculitis. Treatment should be individualized to the patient, based on age, disease severity, and co-morbidities. In mild cases, discontinuation of hydralazine is sufficient. In severe cases like pulmonary-renal syndrome, aggressive management with prompt withdrawal, and immunosuppressive regimens are necessary. Corticosteroids with cyclophosphamide or rituximab for rapidly progressive glomerulonephritis should be considered. In a very similar case by Agarwal et al 17 on a 62-year-Hispanic male with pulmonary-renal syndrome from hydralazine, the drug was discontinued and the patient was treated with pulse intravenous methylprednisolone and cyclophosphamide with taper over a period of 3 months. The MEPEX trial showed that the risk of progression to end-stage kidney disease was reduced by 24% when plasma exchange was used compared to IV methylprednisolone in patients with newly diagnosed AAV with severe kidney failure over a period of 12 months. 18 Conversely, the PEXIVAS trial showed that plasma exchange did not reduce the incidence of death or end-stage kidney disease in patients with severe AAV with renal or lung involvement. In our patients, plasma exchange was not required as the patient did not have hemoptysis.19,20
It is highly important to educate the patients about future drug exposure, with hydralazine added to the allergies list in the medical record.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Lakshmi Kannan-Conception, design of the study, review of literature and analysis of data. UmmeRubab Syeda-Drafting of the manuscript and review of the final manuscript. All authors have given final approval of the version for publication.
Consent
Written informed consent was obtained from the patient to publish this report in accordance with the journal’s patient consent policy.
Data Availability Statement
All data generated or analyzed during this study are included in this article.
