Abstract
Tumor lysis syndrome (TLS) is an oncologic emergency that is usually associated with hematologic malignancies either spontaneously or following early chemotherapy and is caused by massive tumor cell lysis. However, it has been rarely reported in solid tumors. We report a case of 25-year-old lady recently diagnosed with metastatic gastric adenocarcinoma who developed TLS after the fourth cycle of chemoimmunotherapy (FOLFOX plus Nivolumab). She presented with abdominal pain, decrease in oral intake and decreased urine output. Laboratory studies showed acute kidney injury with electrolyte disturbances and was diagnosed initially with autoimmune nephritis secondary to Nivolumab but was later found to have TLS and recovered after appropriate treatment. Soon after this complication, our patient was found to have disease progression on imaging which makes the incidence of TLS an indicator of disease progression.
Introduction
Tumor lysis syndrome (TLS) is a life-threatening complication of chemotherapy treatment. It is usually reported in hematologic malignancies with rapid cellular turnover and is rarely seen in solid tumors. The syndrome can occur spontaneously but primarily occurs in patients undergoing induction chemotherapy and is often evident within 12 to 72 hours after the initiation of cytotoxic therapy due to rapid destruction of large number of tumor cells. Few case reports have reported TLS associated with solid tumors after the first cycle of chemotherapy but extremely rare cases have been reported after several cycles of FOLFOX chemotherapy.
Case Presentation
A 25-year-old lady recently diagnosed with gastric adenocarcinoma metastatic to peritoneum and lymph nodes receiving first-line treatment with FOLFOX and Nivolumab presented 3 days after receiving the fourth cycle of treatment with abdominal pain and decrease in oral intake and urine output. Vitals signs on presentation were within normal limits, with temperature of 36.3°C, heart rate of 89 beats/minute, blood pressure of 129/98 mm Hg, and a pulse oximetry of 100% on room air. On physical examination, she was alert and oriented to person, place, and time. Neurologic and cardiopulmonary examinations were normal and abdominal examination showed soft, distended abdomen with diffuse tenderness.
Computed tomography scan of chest, abdomen, and pelvis showed large pericardial effusion, new left and right moderate pleural effusions, rectal wall thickening suggestive of proctitis, and diffuse severe body wall edema with large volume ascites and increase in the innumerable sclerotic and lytic metastatic osseous lesions (Figure 1). Laboratory findings (Table 1) were remarkable for elevated serum creatinine of 1.4 mg/dL (normal range: 0.6-1.2 mg/dL), BUN of 69 mg/dL (normal range: 8-25 mg/dL), potassium 7.3 mmol/L (normal range: 3.5-5.1), phosphorus of 5.7 mg/dL (normal range: 2.7-4.8 mg/dL), calcium 8.6 mg/dL (normal range: 8.5-10.5 mg/dL), uric acid of 13.8 mg/dL (normal range: 3.5-8 mg/dL), bicarbonate 15 mmol/L (normal range: 24-30 mmol/L), LDH 607 IU/L (normal range: 110-265 IU/L). EKG showed sinus regular rhythm. It was thought that her presentation is likely related to an underlying autoimmune nephritis secondary to Nivolumab thus she was started empirically on prednisone 1 mg/kg daily. However, because her electrolyte derangements were highly suggestive of TLS, she was started on allopurinol and aggressive intravenous hydration. Over the next 5 days, the electrolyte abnormalities improved (Table 1), with serum creatinine of 0.8 mg/dL, potassium of 4.5 mmol/L, phosphorus of 1.8 mg/dL, calcium of 8.4 mg/dL, and uric acid of 4.1 mg/dL. To note, prednisone was stopped only after 3 days.

Pericardial and bilateral pleural effusions.
Laboratory findings.

Day 1: Day of admission to the hospital.
Discussion
Tumor lysis syndrome is an oncologic emergency that occurs when tumor cells are lysed, either spontaneously or in response to cancer-directed therapy, and subsequently release their contents into the bloodstream. This leads to the typical TLS electrolyte disturbances of hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia and this in turn may cause life-threatening complications such as arrhythmias, seizures, renal failure, and death secondary to multi-organ failure.
Only few case reports have been published on TLS in solid tumors, either due to its rarity or due to underreporting or under diagnosis. We did a review of literature, and we summarized the 9 reported cases of TLS in gastric adenocarcinoma in Table 2.1 -9 Of these, 4 cases were spontaneous TLS, and the other 5 were chemotherapy-induced, with only 1 of the 5 cases being FOLFOX-induced. The mean age at presentation was 55 years and all 9 patients were males. All patients with chemotherapy-induced TLS (including our patient) received regimens combining fluoropyrimidine therapy (capecitabine, floxuridine, 5-fluorouracil, and tegafur) with platinum-based therapy (cisplatin and oxaliplatin). TLS secondary to FOLFOX regimen has been frequently described with colon cancer but only 1 case of TLS secondary to FOLFOX regimen in gastric cancer was reported. Not only we are describing the second case of FOLFOX induced TLS in gastric cancer but the first after the fourth cycle.
Literature review. 9
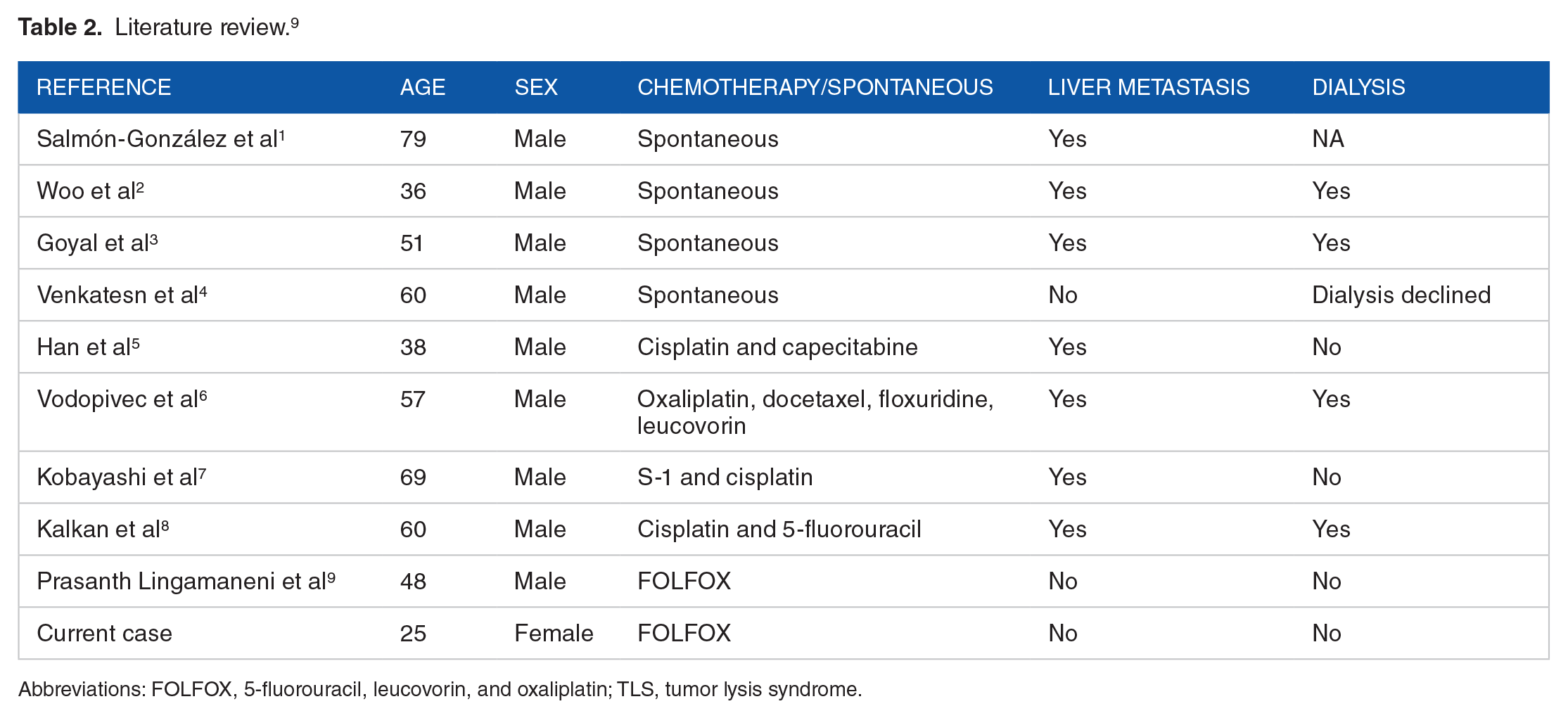
Abbreviations: FOLFOX, 5-fluorouracil, leucovorin, and oxaliplatin; TLS, tumor lysis syndrome.
There are several factors that increase the risk of developing TLS including renal insufficiency, dehydration, highly proliferating malignancies, sensitivity of the tumor cells to chemotherapy, baseline elevations in uric acid and phosphorus, 10 and exposure to nephrotoxic substances. Liver metastasis is known to be a strong risk factor for the development of TLS in solid tumors 11 .Our patient, however, did not have liver metastasis.
The risk of TLS in solid tumor patients is higher in advanced stages and metastatic disease and usually occurs after initiation of cancer targeted therapy (radiation, chemotherapy, or hormonal therapy). 12
In our case, TLS occurred after the fourth cycle of chemotherapy not immediately after initiating treatment, which could be explained by disease progression that was evident on imaging despite receiving appropriate treatment regimen. Where our patient developed bilateral malignant pleural effusions and malignant pericardial effusion (Figure 1) confirmed by cytology, in addition to the increase in the number of sclerotic and lytic osseous lesions with still no apparent bulky disease or liver metastasis.
Thus, because there was no new obvious bulky lesion that could explain this TLS, instead there were new metastatic lesions at different sites of the body, we could attribute the TLS to disease progression (cumulative effects of these new small metastatic lesions). Thus, our assumption would be that TLS does not always occur secondary to the effect of early chemotherapy to bulky lesions, instead it can occur secondary to new multiple metastatic lesions and be a sign of disease progression.
Conclusion
In this report, we described the first case of TLS in a young gastric cancer female patient occurring after fourth cycle of FOLFOX chemotherapy indicating disease progression. Despite the rarity of this complication in solid tumors and after multiple cancer targeted therapy sessions in the absence of new bulky lesion, physicians should be aware of this life-threatening complication and should expect it in patients who are not responding to their cancer targeted therapy as it can still occur indicating disease progression.
Should TLS occur in solid tumor patients on cancer targeted therapy, we believe physicians should investigate not only for the therapy side effect but rather for disease progression, especially if it occurs after several cycles of therapy.
Footnotes
Acknowledgements
The authors would like to thank the patient and her family for consenting to publish the case and contribute to medical knowledge.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Informed consent
Oral informed consent was obtained from the patient.
