Abstract
Budd Chiari syndrome (BCS) is defined as obstruction of hepatic venous outflow that can be located anywhere from small hepatic venules up to the entrance of inferior vena cava (IVC) into right atrium. Etiologies of BCS include myeloproliferative disorders, congenital, and acquired hypercoagulable states. Anticoagulation is the mainstay of treatment for all cases of BCS with a demonstrable hypercoagulable state. Interventional radiology procedures such as transjugular intrahepatic portosystemic shunting (TIPS) can be utilized to reduce portal hypertension and to improve complications related to portal hypertension. We present a patient with systemic lupus erythematosus who first presented with fever, weight loss, malar rash, alopecia, livedo reticularis, symmetric polyarthritis, pancytopenia, and class IV lupus nephritis when she was 23 years old. After receiving an induction treatment of cyclophosphamide and glucocorticoids, she received a maintenance treatment of azathioprine. She presented with ascites and abdominal pain when she was 36 and the abdominal imaging revealed reduced calibration of hepatic venules and intrahepatic segment of inferior vena cava. Lupus anticoagulant was positive and anti cardiolipin IgM and IgG were positive. Work up for hereditary hypercoagulable states was negative. Thus, the diagnosis was secondary antiphospholipid syndrome where BCS was the first clinical manifestation of the antiphospholipid syndrome. Patient was anticoagulated with warfarin and received diuretics for ascites. After the ascites became refractory to diuretics and the patient had multiple vertebral compression fractures due to volume overload secondary to ascites, she was successfully treated with TIPS. When control imaging was performed, 50% of stenosis was observed in the stent. Balloon dilation of the stent was performed. Interventional radiology techniques like TIPS can be used in BCS patients secondary to APS, in cases when medical treatment is insufficient to control complications of portal hypertension. In BCS patients secondary to APS, TIPS enables clinical improvement but due to the presence of endothelial dysfunction in APS patients, there is a risk of shunt dysfunction secondary to thrombosis or stenosis secondary to intimal hyperplasia. Therefore, strict anticoagulation and regular follow up of patients after TIPS is recommended. In cases with stent stenosis, reintervention may be necessary.
Introduction
Budd Chiari syndrome (BCS) is a rare vascular disorder of the liver, which is defined as obstruction of hepatic venous outflow that can be located anywhere from the small hepatic venules up to the entrance of inferior vena cava (IVC) into right atrium. 1
BCS is characterized by abdominal pain, hepatomegaly, and ascites, and the clinical presentation can range from nearly asymptomatic cases to fulminant liver failure. 2
Etiology of BCS is variable. While myeloproliferative disorders such as polycythemia vera and essential thrombocythemia have been responsible in some patients; congenital hypercoagulable states such as antithrombin III, protein C and protein S deficiency, factor V Leiden, prothrombin G20210A mutation, and acquired hypercoagulable states such as antiphospholipid syndrome (APS), paroxysmal nocturnal hemoglobinuria, Behçet’s disease, use of oral contraceptives, pregnancy and postpartum states have been responsible in other patients.2,3 Regarding BCS in APS, literature is limited with case reports and case series which show that BCS can be the first clinical manifestation of APS in some patients. 2
Anticoagulation is the mainstay of treatment for all cases of BCS with a demonstrable hypercoagulable state and should be initiated to all patients in the absence of contraindications.2,4 Interventional radiology procedures such as transjugular intrahepatic portosystemic shunting (TIPS) can be utilized to reduce portal hypertension and to improve complications related to portal hypertension. 4
We hereby present a patient with systemic lupus erythematosus and secondary antiphospholipid syndrome where BCS was the first clinical manifestation of the antiphospholipid syndrome. Patient was anticoagulated with warfarin and received diuretics for ascites. After the ascites became refractory to diuretics and the patient had multiple vertebral compression fractures due to volume overload secondary to ascites, she was successfully treated with TIPS. Written informed consent of the patient was obtained.
Case Report
In 1997, a 23 year-old female patient presented with fever, weight loss, malar rash, diffuse alopecia, livedo reticularis, symmetric polyarthritis, and pretibial edema. She had pancytopenia; the erythrocyte sedimentation rate (ESR) was 100 mm/hour, C-reactive protein (CRP) was 20 mg/L. Antinuclear antibody was positive (titer was 1/640 with a speckled pattern), anti-dsDNA was positive, C3 and C4 levels were low. Anticardiolipin IgM and IgG were both elevated (45 MPLU/mL and 100 GPLU/mL respectively) and lupus anticoagulant was positive. In urinalysis, there was nephrotic range proteinuria and active urinary sediment. Renal biopsy was performed, which revealed an immune complex nephritis consistent with proliferative lupus nephritis (Class IV lupus nephritis, activity index: 10/24, chronicity index: 0/12). Echocardiography was normal. She was diagnosed with systemic lupus erythematosus. She received 500 mg intravenous cyclophosphamide every 15 days for 6 doses (ELNT protocol), along with methylprednisolone and hydroxychloroquine followed by azathioprine as the maintenance treatment. Patient was in complete remission at the sixth month of treatment and her methylprednisolone dose was tapered to 4 mg/day.
Patient remained in remission until 2010. In June 2010 patient presented with fever, malaise, and acute abdominal pain. Physical examination revealed tenderness in the lower quadrants of the abdomen, hepatomegaly, and shifting dullness up to 3 cm below umbilicus (ascites). Blood count revealed leukopenia and lymphopenia, ESR was 89 mm/hour, CRP was 14 mg/L. Urinalysis revealed an active urine sediment and a proteinuria of 2 g/day. C3, C4 levels were low, anti-dsDNA and lupus anticoagulant were positive. Anti cardiolipin IgM and IgG were both positive in high titers (>100 MPLU/mL and >100 GPLU/mL respectively). Ascites analysis demonstrated a transudative ascites with a serum-ascites albumin gradient larger than 1.1 g/dL and total protein of ascites was 3 g/dL. Gastroscopy revealed reflux esophagitis and alkaline reflux gastritis. Abdominal ultrasound revealed abundant ascites, hepatomegaly, heterogeneous echogenicity of liver parenchyma. Doppler ultrasound of portal system demonstrated decreased hepatic vein diameter, normal portal vein diameter but a decreased flow rate in portal veins. CT angiography of portal venous system was performed, where portal vein diameter was normal but calibration of hepatic venules and intrahepatic segment of inferior vena cava were thin. Ascites and heterogeneous contrast enhancement of liver parenchyma were also noted. Patient underwent liver biopsy, which demonstrated mononuclear inflammation of portal vein, dilation of central and midzonal sinusoids, congestion and hepatocellular damage in the centrilobular area. Work up for hereditary hypercoagulable states such as Factor V Leiden and prothrombin 20210 mutation was negative. Patient was diagnosed with subacute Budd-Chiari syndrome related to active systemic lupus erythematosus and secondary antiphospholipid syndrome and received monthly pulses of 1 g cyclophosphamide for 6 months, followed by 3 g/day of mycophenolate mofetil, together with methylprednisolone, hydroxychloroquine, warfarin, spironolactone, and furosemide. She was regularly followed up by the outpatient clinics of our Rheumatology and Gastroenterohepatology departments.
Although the disease activity was under control, her ascites became refractory to diuretics and she had to undergo frequent therapeutic paracentesis. She also developed osteoporotic vertebral compression fractures, umbilical hernia, total uterine prolapse, and cystorectocele due to the presence of massive ascites. In 2014, a repeat CT angiography of portal venous system revealed decreased calibration of hepatic veins and intrahepatic segment of inferior vena cava. After consultation with gastroenterohepatology and interventional radiology departments, patient underwent transjugular intrahepatic portosystemic shunt (TIPS). Pre-treatment and post-treatment CT angiography images are demonstrated in Figure 1 and pre-treatment and post-treatment MR angiography images are demonstrated in Figure 2. These images demonstrate the success of TIPS in reducing ascites and restoring homogeneity of liver parenchyma.

Pre-treatment (A) and post-treatment (B) CT angiography images. In the pre-treatment image, there is no flow in hepatic veins which is consistent with Budd-Chiari syndrome. Also in figure A, liver parenchyma is non-homogeneous because of the perfusion defect. In the post-treatment image, with improved perfusion, liver parenchyma is relatively homogenous. TIPS can be noted (arrow). Also, extensive ascites which can be seen in pre-treatment image is relatively decreased in post-treatment images.
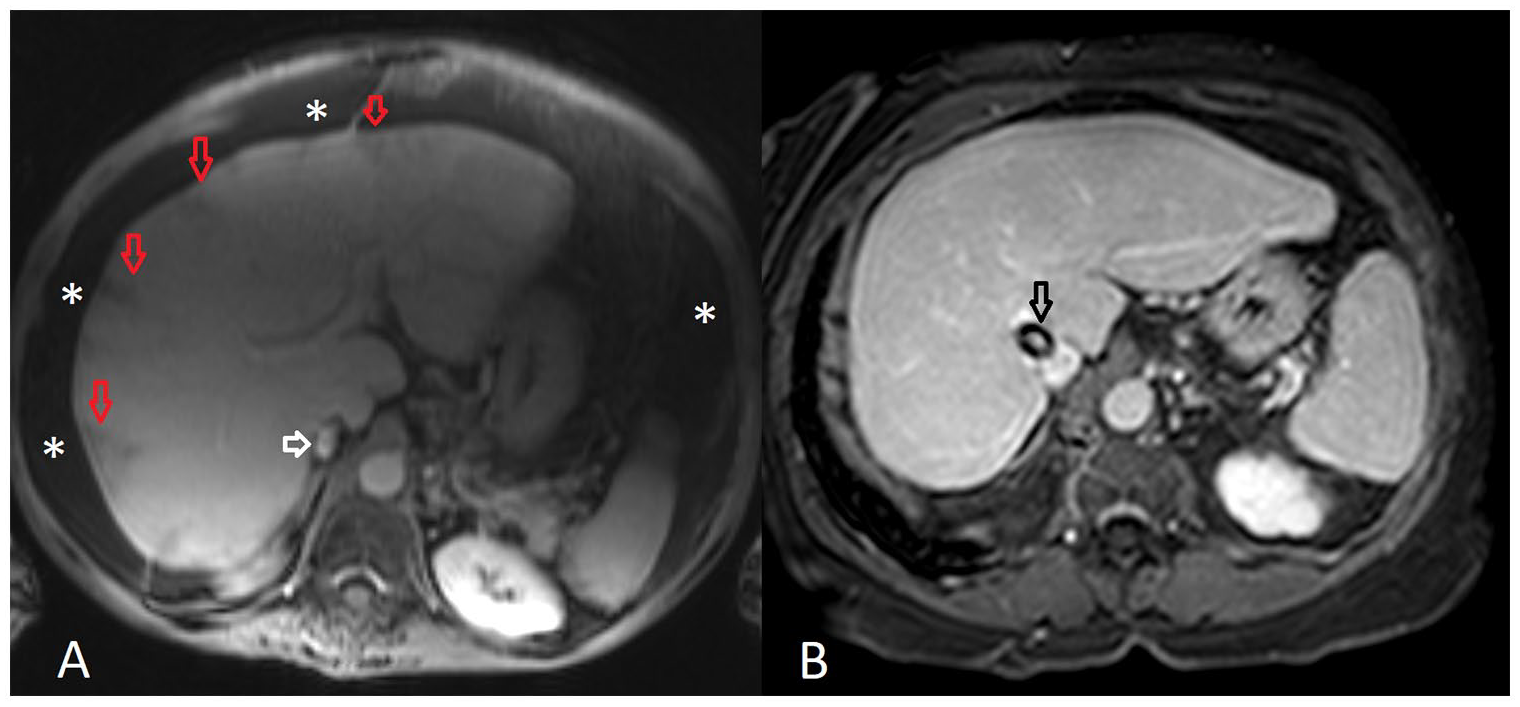
Pre-treatment (A) and post-treatment (B) MR angiography images. Liver parenchyma heterogeneity, peripherally hypointense triangular shaped regions (red arrows) caused by perfusion defect, decreased flow in hepatic veins, relatively low flow in intrahepatic segments of inferior vena cava (white arrow) and extensive ascites (white stars) can be seen in pre-treatment image (A). In post-treatment image, with improved perfusion, liver parenchyma is relatively more homogenous and ascites is relatively decreased compared to pre-treatment image. Also, TIPS (black arrow) and contrast material within the TIPS can be seen.
After this procedure, surgery was performed for uterine prolapse and cystorectocele. In 2016, control imaging demonstrated 50 % of stenosis in the stent, ascites was absent and liver parenchyma seemed homogeneous. Balloon dilation of the stent was performed. As of March 2020, patient is ascites-free and is receiving a maintenance treatment of 2 g/day of mycophenolate mofetil, 4 mg of methylprednisolone, 200 mg of hydroxychloroquine, warfarin, and teriparatide.
Discussion
BCS can be the presenting feature of APS, therefore antiphospholipid antibodies, namely lupus anticoagulant, anticardiolipin IgM, IgG, and antibeta-2-glycoprotein 1 IgM, IgG should be a part of the routine workup of patients with BCS. 3 In a review of 43 APS patients with BCS by Espinosa et al, 2 32 patients had primary APS, 8 patients had APS secondary to systemic lupus erythematosus (SLE), and 3 patients had APS secondary to other diseases. BCS was the first clinical manifestation of APS in 65% of these patients. In the same review, 1 patient initially had lupus anticoagulant positivity without any symptoms of APS and developed BCS 3 years later. Our patient initially had lupus anticoagulant and anticardiolipin IgM, IgG positivity when she was diagnosed with SLE. She did not have any arterial, venous thrombosis, or heart valve involvement. She developed BCS 13 years later when she experienced a disease flare.
In the same review, presentation of BCS was subacute in 70% of the patients, chronic in 23% of the patients, and fulminant in the remaining 7%. Most common symptoms were abdominal pain (56%) and abdominal distention (21%) and most common physical examination findings were hepatomegaly (58%) and ascites (53%). Among these 43 patients, lupus anticoagulant was positive in 77% of patients, anticardiolipin IgG was positive in 80%, and anticardiolipin IgM was positive in 59% of patients. 2
Imaging plays a crucial role in the diagnosis of BCS. Identification of obstructed hepatic venous outflow by the non-invasive techniques of Doppler ultrasound, triphasic multidetector computed tomography (CT), and magnetic resonance imaging (MRI) is usually sufficient for diagnosis. 3 Dilation of the veins upstream from an obstacle, solid endoluminal material inside the vein, transformation of the veins into a cord without flow signal, and presence of venous collaterals are findings that suggest obstruction of hepatic venous outflow. 3 Patchy enhancement of liver parenchyma may suggest a perfusion defect but this finding is non-specific and may be present in other vascular disorders of the liver. 3 Since the current imaging modalities are efficient, direct or retrograde venography are seldom used to diagnose BCS. 3 Liver biopsy is reserved for cases when large veins appear normal on imaging and thrombosis is present in only small intrahepatic veins. 1 Among 43 APS-BCS patients in the review of Espinosa et al, 2 20 APS-BCS patients had undergone liver biopsy and sinusoidal dilation was present in 50% of the cases, centrilobular necrosis was present in 35% of the cases and centrilobular congestion was present in 25% of the cases. In our case, Doppler ultrasound revealed decreased hepatic vein diameter, CT angiography revealed reduced calibration of hepatic vein and inferior vena cava, and liver biopsy demonstrated sinusoidal dilation, centrilobular congestion, and centrilobular necrosis.
Current guideline of American Association for the Study of Liver Diseases recommends a stepwise approach for the management of BCS. Anticoagulation should be initiated to all patients without contraindications. 4 Local thrombolytic therapy can be used for acute/recent thrombosis of hepatic vein. 4 Percutaneous transluminal angioplasty with or without stenting can restore hepatic blood flow in cases with segmental hepatic vein obstruction or inferior vena cava obstruction. 4 TIPS is indicated in cases where angioplasty/stenting is not technically feasible, in cases with complications of severe portal hypertension or in cases that have persistently deteriorating liver functions. 4 Liver transplantation is reserved for severe cases that are unresponsive to other treatment modalities. 4
TIPS is currently the most common interventional treatment modality in BCS patients that are unresponsive to medical treatment.5,6 TIPS can be used as a “bridging modality” for liver transplantation but in some cases it can alleviate the need for transplantation. 7 TIPS has a high technical success rate of 93% and patients have a 1- and 5-year transplant-free survival rate of 93% and 74% respectively. Clinical response has been demonstrated in 84% of patients. 5
In a review of literature of TIPS in BCS patients, Qi et al 4 reported that TIPS related complications ranged from 0% to 56% and that procedure related death was rare. They noted that shunt dysfunction was more frequent in BCS patients due to their procoagulant state and that the rate of shunt dysfunction ranged from 18% to 100% depending on the type of stents, number of patients, and the length of follow up period. 4 They also concluded that covered stents significantly reduced the rate of shunt dysfunction compared to bare metal stents. 4 In order to prevent shunt dysfunction secondary to thrombosis, strict anti-coagulation with an international normalized ratio (INR) goal greater than 2.5 is recommended to BCS patients that undergo TIPS. 2
APS is an acquired thrombotic disorder where endothelial dysfunction is a key pathological feature. 8 Therefore, one may assume that APS patients could be at a higher risk for shunt dysfunction due to thrombosis or stent stenosis after procedures like TIPS, because these procedures can result in additional endothelial injury. In the review of Espinosa et al, 2 4 APS-BCS patients were treated with TIPS: 3 patients for portal hypertension and 1 patient for progressive liver failure. Shunt dysfunction secondary to intimal hyperplasia was observed in 3 patients, which necessitated reintervention using coaxial stents or angioplasty. 2 In 2 of the 3 patients INR was below the therapeutic range at the time of stent dysfunction. 2 Intimal hyperplasia causing narrowing or stenosis of the lumen of the shunt is a common cause of delayed shunt dysfunction. 2 Thrombin has been shown to cause smooth muscle cell migration and intimal hyperplasia. 2 In APS patients, plasma levels of prothrombin fragments 1 + 2 are increased during thrombotic events, which results in increased thrombin generation. 3 This can explain occurrence of intimal hyperplasia in APS patients after TIPS. 2 Espinosa et al 2 concluded that TIPS placement may be effective in APS-BCS patients, if used together with long term high intensity anticoagulation to prevent recurrent thrombosis. In our patient, 2 years after TIPS, there was a stenosis in the stent. It did not cause ascites but the patient had to undergo balloon angioplasty to prevent shunt dysfunction. Her INR at the time of stenosis was under therapeutic range. This underlines the significance of effective and sustained anticoagulation in maintaining stent patency in APS-BCS patients.
In conclusion, interventional radiology techniques like TIPS can be used in BCS patients secondary to APS, in cases when medical treatment is insufficient to control complications of portal hypertension. In BCS patients secondary to APS, TIPS enables clinical improvement but due to the presence of endothelial dysfunction in APS patients, there is a risk of shunt dysfunction secondary to thrombosis or stenosis secondary to intimal hyperplasia. Therefore, strict anticoagulation and regular follow up of the patients after TIPS is recommended. In cases with stent stenosis, reintervention may be necessary.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Ege Sinan Torun and Mert Erciyestepe drafted the paper, Bahar Artım-Esen critically revised and made suggestions. All authors read and approved the manuscript.
