Abstract
Introduction:
ROS1 rearrangement has recently emerged as a new molecular subtype in non–small-cell lung cancer (NSCLC) and is predominantly found in lung adenocarcinoma compared with other oncogenes such as EGFR, KRAS, or ALK. It has been identified in only 1% to 2% of NSCLC cases.
Case Report:
We report a case of 52-year-old man (nonsmoker) with a medical history of allergic rhinitis and bronchial asthma. Histopathologic examination of bronchoscopic-guided biopsy showed adenocarcinoma histology on September 2015. After 2 months, he developed left-sided pneumonia for which he was treated with multiple intravenous antibiotics. In the meantime, fiberoptic bronchoscopy was done which revealed purulent secretion from right upper lobe and narrowed opening of right middle lobe. His cancer symptoms got worsened and bronchial biopsy showed EGFR mutation negative. For further diagnosis, fluorescent in situ hybridization test was done which showed ROS1 mutation positive. By then, the patient was started with crizotinib 250 mg twice daily for ROS1 mutation in July 2016. Later, patient appears to benefit from treatment with crizotinib. X-ray report and positron emission tomographic-computed tomographic scan revealed that the patient was overall better with clear chest and well tolerated with the therapy. Crizotinib was approved on March 11, 2016 by Food and Drug Administration for the treatment of patients with ROS1-positive NSCLC.
Conclusions:
In this report, crizotinib showed marked antitumor activity in patients with advanced ROS1 rearrangement, a third molecular subgroup of NSCLC.
Introduction
Lung cancer is the most commonly occurring cancer worldwide and nearly 80% to 85% of all cases accounts for non–small-cell lung cancer (NSCLC).1,2 Targeted molecular therapy is efficient for patients with advanced NSCLC with related gene mutations. 3 Despite driver genes, including epidermal growth factor receptor (EGFR) and Kirsten rat sarcoma viral oncogene (KRAS), commonly found in lung adenocarcinomas, the c-ros oncogene 1 receptor tyrosine kinase (ROS1) rearrangement is found only in 1% to 2% of NSCLC cases and emerged as a new molecular subtype. 4
ROS1 is a transmembrane tyrosine kinase receptor with similar ALK protein kinase domain. ROS1 rearrangement is observed more often in patients who never smoked and have histologic features of adenocarcinoma. Fluorescent in situ hybridization (FISH) analysis is believed as the best technique to identify ROS1 rearrangement. 5 Crizotinib, a highly effective inhibitor of ROS1 kinase activity, was approved by Food and Drug Administration (FDA) in 2016 for the patients with ROS1-positive NSCLC. 2
A study conducted by Padma 2 found that the sensitive method for the detection and evaluation of pulmonary nodules is fluorodeoxyglucose positron emission tomographic-computed tomographic (FDG PET-CT) scan.
Case Report
A 52-year-old man, nonsmoker (body mass index: 28 kg/m2), with the medical history of allergic rhinitis and bronchial asthma, was admitted to our institution in September 2015 due to the complaints of cough and recurrent shortness of breath of 3 months duration. He had been evaluated elsewhere with chest CT, bronchoscopy, and CT-guided biopsy. Chest CT found a nonenhancing, homogeneous mass lesion on the right upper lobe.
Social history was significant for a several year history of electrical work and negative for tobacco or alcohol use. On examination, dyspnea score was 4, pain score was 1 to 3, temperature was 38°C, blood pressure was 140/86 mm Hg, and pulse was 75 beats per minute.
Bronchial biopsy (right middle lobe) from our institution revealed lung adenocarcinoma with signet-ring cell morphology. Chest X-ray was ordered revealing an ill-defined enhancing right hilar mass suggestive of malignancy (Figure 1A). The PET-CT scan suggested abnormal increased FDG uptake in right lung (Figure 2A) and stage IIIB (T4N2M0) disease. The patient received first-line chemotherapy with gemcitabine 2000 mg and carboplatin 600 mg. Gemcitabine and carboplatin were selected as the patient’s bronchial biopsy was showing signet-ring cells and his electrocardiography was within the normal limits.
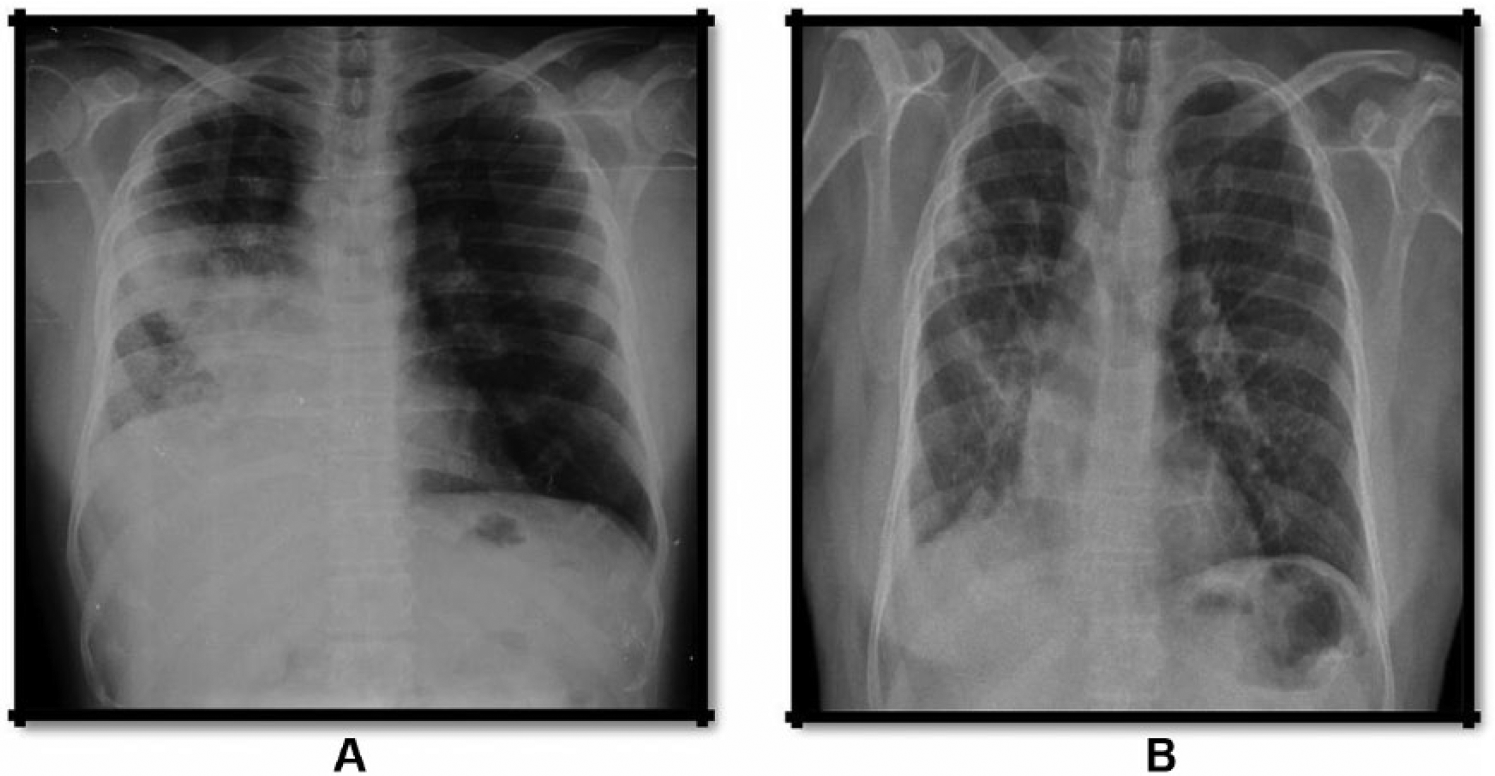
Chest X-ray showing the primary lung adenocarcinoma lesion: (A) before crizotinib treatment and (B) after crizotinib treatment.
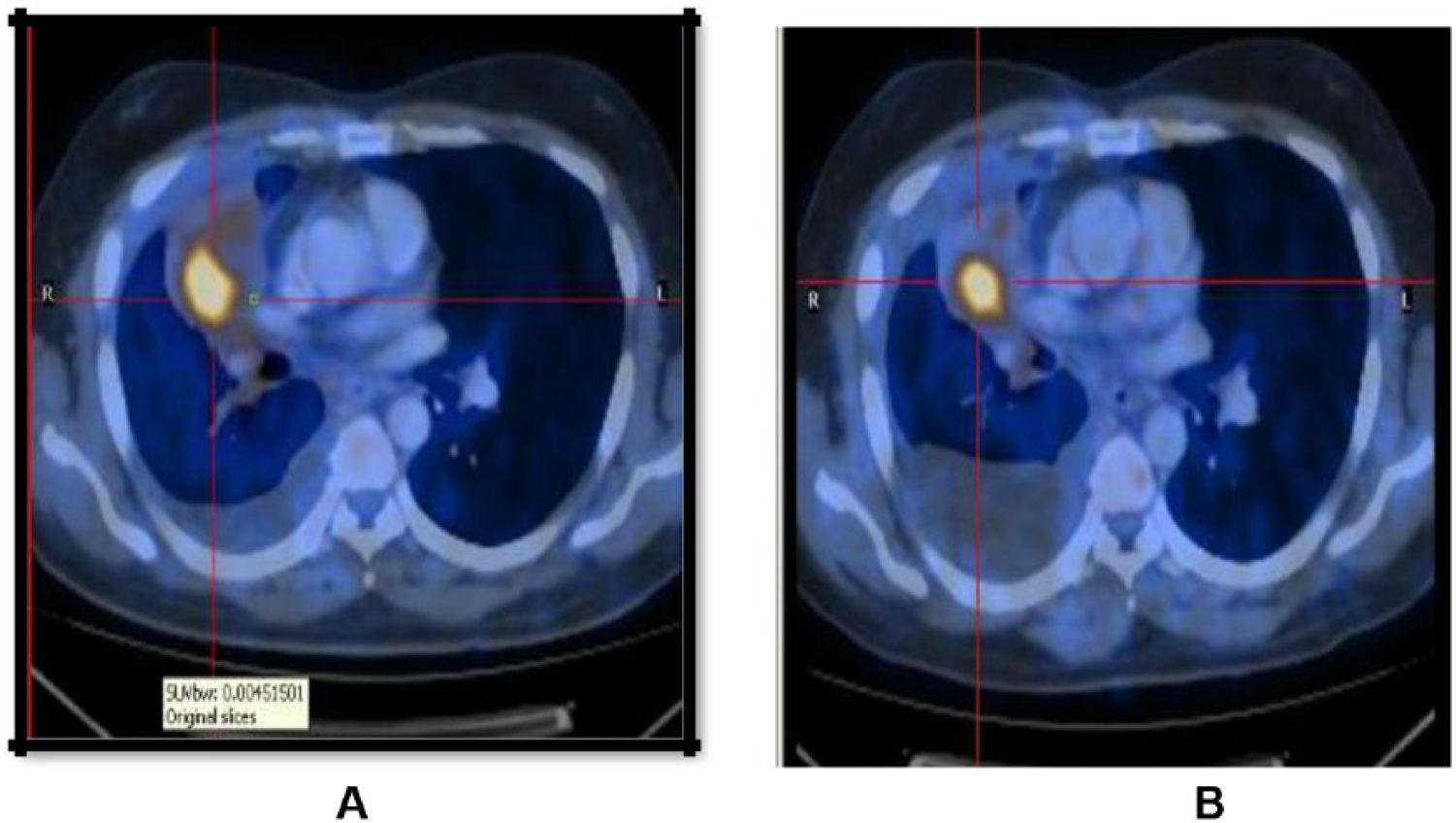
Computed tomographic and positron emission tomographic images showing FDG uptake: (A) before crizotinib treatment and (B) after crizotinib treatment.
In November 2015, following 2 months of first-line treatment, the patient again complained of fever and cough with scanty mucoid expectoration. A CT scan revealed obstructive pneumonia. He was treated with multiple intravenous antibiotics (amoxicillin:clavulanate 875 mg/125 mg, clarithromycin 500 mg). But he again experienced shortness of breath and cough, so the antibiotics were changed to piperacillin-tazobactam 4.5 mg, clindamycin 600 mg, and amikacin 1000 mg for 14 days. However, the chemotherapeutic treatment was discontinued after 4 cycles because of severe bone marrow suppression (Table 1). In the meantime, fiberoptic bronchoscopy was done which revealed purulent secretion from right upper lobe and narrowed opening of right middle lobe. Bronchoalveolar lavage was taken from right upper lobe and culture showed no growth. So he was further referred to oncology department and the biomarker testing showed EGFR mutation negative. Due to the lack of EGFR mutation, FISH analysis was performed which showed ROS1 rearrangement positive. In July 2016, the patient began to receive crizotinib (250 mg, twice per day), a ROS1 kinase inhibitor, and was continued for 2 months.
Time line of selected laboratory values.

Abbreviations: Hb, hemoglobin; Hct, hematocrit.
After 2 months of crizotinib treatment, chest CT scan images demonstrated a decrease in tumor size (Figure 1B) and decreased FDG uptake (Figure 2B). According to Response Evaluation Criteria in Solid Tumors guidelines (version 1.1), this patient was considered as having a partial response to crizotinib and improved quality of life (dyspnea score and pain score lowered). During crizotinib treatment, abnormal hepatic and renal functions were not found. There were no treatment-related adverse events, including gastrointestinal reaction and flickering vision. The patient remained asymptomatic and has no recurrence and progression of tumor at the last follow-up in September 2017 and, at present, he continues to receive crizotinib with the same dose.
Discussion
Non–small-cell lung cancer is becoming a life-threatening condition. 6 The identification of NSCLC is poor, even with the most current treatment regimens, which include surgery, chemotherapy, and radiation.7,8 Non–small-cell lung cancer with adenocarcinoma histology has been known to be genetically diverse. The understanding of its genetic diversity has led to the discovery of new molecular-targeted approaches. A moderately new player in NSCLC is the ROS1 fusion gene announced in around 1% patients with NSCLC.9,10 On March 11, 2016, the FDA formally approved crizotinib (Pfizer, New York City, NY, USA) for the treatment of patients with advanced (metastatic) NSCLC harboring ROS1 gene fusion. 11 The researchers shows that crizotinib also inhibits the tumors caused by the oncogene called ALK and depicted as remarkable response of patient to crizotinib treatment. 12
In the phase 1 clinical trial of crizotinib, 50 patients with NSCLC with ROS1 fusion were included, including 49 cases detected by FISH and 1 by reverse transcription-polymerase chain reaction. The overall response rate was 72%, with a median duration of response of 17.6 months and a median progression-free survival of 19.2 months.13,14 The current guidelines from the National Comprehensive Cancer Network (NCCN) suggested for EGFR testing as well as ROS1 rearrangement in all patients with advanced NSCLC as well as crizotinib as an option for first-line therapy. 15
Crizotinib is a targeted therapy that targets and binds to tyrosine kinase receptor and inhibits ALK. By binding to these receptors, crizotinib blocks an important pathway that promotes cell division. The common side effects of crizotinib are vision problems, nausea, vomiting, and diarrhea (30%). The half-life of crizotinib is 45 hours.
The patient we presented here is benefitted from crizotinib therapy, a report that is consistent with the ROS1-mutated lung adenocarcinoma. Apart from as an oral drug which is effective in patients with ROS1 rearrangement–positive NSCLC, its safety profile makes crizotinib a successful and a suitable therapeutic option for patients with NSCLC.16,17
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
JMJ wrote the first draft of the manuscript. JMJ, SK, PK and MV participated in manuscript development. All authors reviewed and approved the final manuscript.
