Abstract
Background:
Hypertension affects over 1.3 billion globally, many remain uncontrolled, and aldosterone synthase inhibitors (ASIs) are emerging as a treatment. This network meta-analysis compared the effectiveness and safety of ASI regimens in hypertensive patients.
Methods:
PubMed, Cochrane Central, and ScienceDirect were searched till December 2025. We conducted a frequentist network meta-analysis using RStudio version 4.3.3 with the “meta” and “netmeta” packages. P-scores were used for treatment ranking.
Results:
Ten randomized controlled trials were included in the analysis. Baxdrostat 1 mg, 2 mg, lorundrostat 100 mg once daily, 12.5 mg twice daily, 25 mg twice daily, 50 mg once daily, 50 mg and escalation to 100 mg, osilodrostat 0.25, 0.5, and 1.0 mg once daily, vicadrostat 10, 20, and 3 mg plus empagliflozin, and 20 mg significantly reduced the systolic blood pressure (SBP), where the baxdrostat 2 mg (P-score = 0.89) was ranked best. Lorundrostat 50 mg once daily, Baxdrostat 1 and 2 mg significantly reduced the diastolic blood pressure (DBP) with Lorundrostat 50 mg once daily ranked best (P-score = 0.84). Adverse events were increased with lorundrostat regimens, but serious adverse events were similar across regimens. Hyperkalemia was more frequent with Baxdrostat 1 and 2 mg and all lorundrostat regimens. Symptomatic hypotension occurred mainly with lorundrostat 50 mg daily.
Conclusion:
The SBP and DBP were significantly reduced with the baxdrostat and lorundrostat regimens, although the risk of hyperkalemia increased. Adverse events were also higher in the lorundrostat regimens, whereas serious adverse events were comparable.
Keywords
Introduction
Hypertension remains a leading global health challenge, affecting over 1.3 billion individuals and contributing significantly to cardiovascular morbidity and mortality.1,2 Approximately 1 in 10 individuals with hypertension is affected by this condition, which contributes disproportionately to the global cardiovascular disease burden.3,4 Despite the availability of multiple antihypertensive drug classes, including angiotensin-converting enzyme (ACE) inhibitors, Angiotensin Receptor Blockers (ARBs), calcium channel blockers, and diuretics, optimal blood pressure control remains elusive for a substantial proportion of patients.5 -7 While mineralocorticoid receptor antagonists (MRAs) like spironolactone and eplerenone have been used to counteract aldosterone’s downstream effects, their efficacy is limited by off-target hormonal side effects and incomplete suppression of aldosterone synthesis. 8 This therapeutic gap has prompted the exploration of novel pharmacologic agents, among which aldosterone synthase inhibitors (ASIs) have emerged as a promising class.
ASIs such as Lorundrostat and Baxdrostat directly target CYP11B2, the enzyme responsible for aldosterone synthesis, offering a mechanistically distinct and potentially more effective strategy for blood pressure control and cardiovascular risk reduction in this high-burden subgroup. 9 Recent randomized controlled trials (RCTs) and meta-analyses have evaluated the blood pressure–lowering effects and safety profiles of ASIs in diverse hypertensive populations.10 -13 A recent large-scale analysis of individual patient data published in August 2025 has reinforced the therapeutic potential of aldosterone synthase inhibitors in managing hypertension, demonstrating consistent benefits across varied clinical environments. 14 However, findings across studies remain heterogeneous, particularly regarding the magnitude of systolic and diastolic blood pressure reduction, incidence of hyperkalemia, and overall tolerability. Moreover, direct head-to-head comparisons between ASIs and other antihypertensive agents are scarce, limiting the ability to contextualize their relative efficacy and safety.
To address these gaps, we conducted a network meta-analysis (NMA) of RCTs evaluating ASIs for hypertension. This approach enables indirect comparisons across multiple interventions. This NMA quantifies the blood pressure-lowering efficacy of ASIs relative to placebo, assesses their safety profile with a focus on adverse events and the risk of hyperkalemia, and ranks available treatment regimens based on their benefit-risk profiles. Despite the growing interest in ASIs as a novel therapeutic class for hypertension, direct comparative evidence between ASIs and established antihypertensive agents remains limited. By leveraging NMA’s strengths, this study aims to provide a nuanced, comparative assessment of ASIs within the broader antihypertensive landscape, addressing critical gaps in evidence synthesis and enhancing the precision of therapeutic recommendations.
Methods
This systematic review and network meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 15 and followed the Cochrane Handbook for Systematic Reviews and Interventions. 16 The protocol for this review was registered on PROSPERO under the ID: CRD420251147042.
Literature Search
Electronic databases, including PubMed, Cochrane Central, and ScienceDirect, were searched up to December 2025 to identify potential articles. Gray literature, including conference proceedings and abstracts, was also explored to ensure comprehensive coverage of the topic, using search engines such as Google Scholar. The bibliographies of included articles were also searched for relevant articles. The MeSH terms and keywords used were “Hypertension,” “resistant hypertension,” “uncontrolled hypertension,” “aldosterone synthase inhibitor,” “aldosterone synthase inhibition,” “osilodrostat,” “lorundrostat,” “baxdrostat,” and “vicadrostat.” A detailed breakdown of the search strategies employed in each database is provided in Supplemental Table 1.
Study Selection and Eligibility Criteria
Two authors (A.S. and H.A.A.R.) independently searched the databases to retrieve potential articles. After removing duplicates, the remaining articles were screened based on their titles and abstracts. The articles that passed primary screening underwent a full-text secondary review to determine which would be included in the meta-analysis. Any discrepancy during the study selection process is resolved by a third author (M.H.W.). The entire study selection process is outlined in the PRISMA flowchart (Figure 1).

PRISMA flowchart of the study selection process.
Patients had to be at least 18 years old and have a confirmed diagnosis of hypertension to be included in the study. The intervention involved various ASI regimens, while the placebo group served as a comparison. Only RCTs were considered for the study design; studies involving patients under 18 years of age, as well as reviews, editorials, and observational studies, were excluded.
Data Extraction
Two authors (Z.U.A. and A.S.) independently extracted data on baseline variables and outcomes using a pre-defined Excel sheet. Baseline variables extracted included study ID, location, sample size, mean age, sex distribution, ASI treatment regimens, study duration, diagnostic criteria for high blood pressure, and follow-up. The efficacy outcomes extracted were changes in systolic blood pressure (SBP) and diastolic blood pressure (DBP). The safety endpoints extracted were incidence rates of adverse events, serious adverse events, hyperkalemia, and symptomatic hypotension. Any discrepancies during the extraction process were resolved by a third author (M.H.W.).
Risk of Bias
Two authors (H.A.A.R. and Z.U.A.) independently assessed the risk of bias in the included RCTs using the Cochrane Risk of Bias (RoB) 2.0 tool. 17 The RoB 2.0 tool evaluates bias across the following domains: bias in the randomization process, bias due to deviations from the intended interventions, bias due to missing outcome data, bias in the measurement of the outcome, and bias in the selection of the reported result. Any discrepancies during the bias assessment were resolved by a third author (A.H.C.). The details of the risk of bias are presented in the traffic light plot (Supplemental Figure 1).
Statistical Analysis
We conducted a network meta-analysis using the frequentist approach with the “meta” and “netmeta” packages in RStudio version 4.3.3. The analysis pooled mean differences (MDs) and risk ratios (RRs) with 95% confidence intervals (CIs) to estimate network effects for continuous and dichotomous outcomes. We assessed heterogeneity using the Cochrane Q test and Higgins I2 statistics, considering P-values less than .05 to be statistically significant. We created network plots using NMAstudio, where each node represents an intervention, and line thickness indicates the number of studies comparing these interventions. We also generated forest plots and league tables to illustrate the network’s structure. We used P-scores to rank treatments, with a P-score of 1 indicating the most effective or safe intervention and 0 indicating the least effective or safe. Rankograms illustrated treatment rankings by P-score. To identify inconsistencies between direct and indirect evidence, we performed a split node analysis. The CiNEMA GRADE assessment was employed to evaluate the certainty of the evidence. We assessed publication bias visually with funnel plots and statistically using Egger’s regression test.
Results
Search Results
Our initial search of databases, including PubMed, Cochrane Central, and ScienceDirect, yielded 1629 potential articles. After removing duplicates (n = 396), we narrowed it down to 151 articles that passed our primary title and abstract screening. These were then reviewed in full text, and 10 articles10,11,14,18 -24 were selected for inclusion in the network meta-analysis. The entire selection process is outlined in the PRISMA flowchart (Figure 1).
Study Characteristics
Ten RCTs10,11,14,18 -24 pooling a total of 3826 patients were included in this network meta-analysis. The mean age ranged from 54 to 69 years. The ASI regimens included were baxdrostat 0.5 mg, baxdrostat 1 mg, baxdrostat 2 mg, lorundrostat 100 mg once daily, lorundrostat 12.5 mg once daily, lorundrostat 12.5 mg twice daily, lorundrostat 25 mg twice daily, lorundrostat 50 mg once daily, lorundrostat 50 mg and then escalation to 100 mg, osilodrostat 0.25 mg once daily, osilodrostat 0.25 mg twice daily, osilodrostat 0.5/1mg twice daily, osilodrostat 0.5 mg once daily, osilodrostat 0.5 mg twice daily, osilodrostat 1.0 mg once daily, osilodrostat 1.0 mg twice daily, osilodrostat 2 mg once daily, vicadrostat 10 mg, vicadrostat 10 mg plus empagliflozin, vicadrostat 20 mg, vicadrostat 20 mg plus empagliflozin, vicadrostat 3 mg, vicadrostat 3 mg plus empagliflozin, vicadrostat 40 mg. The follow-up duration ranged from 1 week to 12 weeks. The detailed characteristics of the included studies are provided in Table 1.
Baseline Characteristics of the Included Studies.

Abbreviations: ACEi, angiotensin converting enzyme inhibitor; ARB, Angiotensin receptor blocker; ASIs, aldosterone synthase inhibitors; BD, twice daily; BP, blood pressure; C, comparator; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HTN, hypertension; mg: milligram; QD, once daily; SBP, systolic blood pressure.
Risk of Bias
For the included RCTs, we assessed the risk of bias using the Cochrane RoB 2.0 tool. All the studies had a low risk of bias across all 5 domains of the RoB 2.0 tool. The results are illustrated in Supplemental Figure 1 as a traffic light plot.
Network Meta-Analysis
Change in SBP
Ten studies, including 22 ASI regimens and 69 pairwise comparisons, were included in the network meta-analysis. Compared with placebo, baxdrostat 1 mg and 2 mg, lorundrostat (all regimens except 12.5 mg once daily), osilodrostat 0.25, 0.5, and 1.0 mg once daily, vicadrostat 10, 20, and 3 mg (all combined with empagliflozin) and 20 mg alone significantly decreased SBP. Other regimens were comparable to placebo (Figure 2A and Supplemental Table 2). The network plot for the change in SBP is shown in Figure 3A. Based on P-scores, baxdrostat 2 mg was ranked best (P-score = 0.89) in decreasing SBP, whereas the vicadrostat 10 mg was ranked worst (P-score = 0.20). The details regarding the P-score ranking are provided in Table 2 and Figure 4. No heterogeneity was detected (tau2 < 0.0001; I2 = 0% [0.0%; 62.4%]).
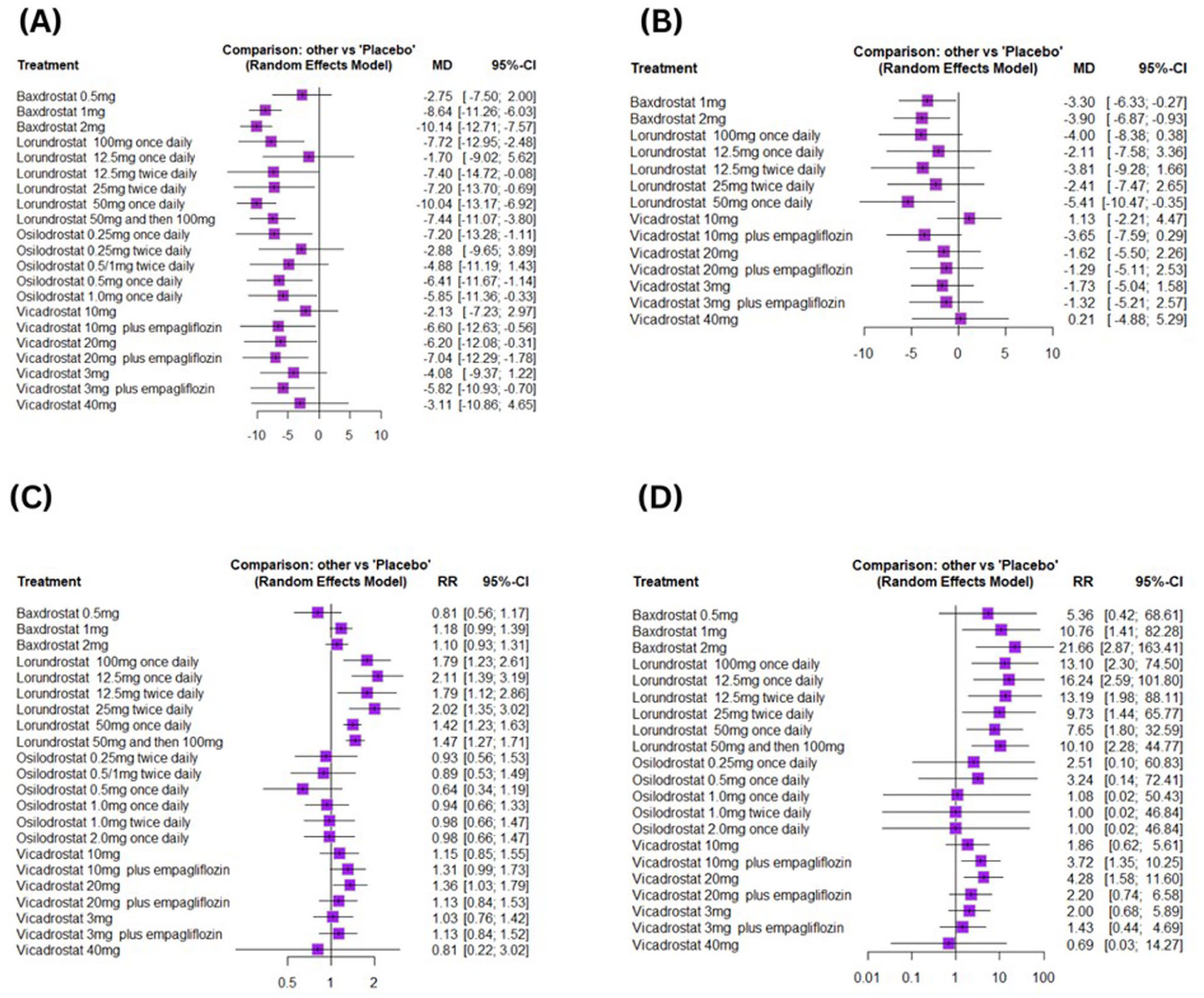
Forest plots: (A) change in systolic blood pressure, (B) change in diastolic blood pressure, (C) adverse events, and (D) hyperkalemia.

Network plot: (A) change in systolic blood pressure, (B) change in diastolic blood pressure, (C) adverse events, and (D) hyperkalemia.
Treatment ranking based on P-scores.


Rankogram for change in systolic blood pressure.
Change in DBP
Four studies, including 15 treatment regimens and 45 pairwise comparisons, were included in the network meta-analysis. Compared with placebo, baxdrostat 1 and 2 mg, and lorundrostat 50 mg once daily significantly decreased DBP, whereas the others showed effects comparable to placebo (Figure 2B and Supplemental Table 3). The network plot of DBP is shown in Figure 3B. Based on the P-scores, lorundrostat 50 mg once daily was ranked best (P-score = 0.84) in reducing DBP, whereas vicadrostat 10 mg was ranked worst (P-score = 0.10). The details regarding the P-score ranking are given in Table 2 and Supplemental Figure 2. Low heterogeneity was observed (tau2 = 1.4110; I2 = 24.8% [0.0%; 92.2%]).
Adverse Events
Nine studies, including 23 treatment regimens and 73 pairwise comparisons, were included in the network meta-analysis. Compared to placebo, lorundrostat all regimens and vicadrostat 20 mg significantly increased the risk of adverse events, whereas other regimens showed results comparable to placebo (Figure 2C and Supplemental Table 4). The network plot of the adverse events is shown in Figure 3C. Based on the P-scores, osilodrostat 0.5 mg once daily was ranked best (P-score = 0.92) in reducing the adverse events, whereas the lorundrostat 12.5 mg once daily was ranked worst (P-score = 0.05). The details regarding the P-score ranking of the treatments are provided in Table 2 and Supplemental Figure 3. No heterogeneity was detected (tau2 < 0.0001; I2 = 0% [0.0%; 64.8%]).
Hyperkalemia
Nine studies, including 22 treatment regimens and 68 pairwise comparisons, are included in the network meta-analysis. Compared with placebo, baxdrostat 1 and 2 mg, lorundrostat all regimens, vicadrostat 10 mg plus empagliflozin, and 20 mg significantly increased the risk of hyperkalemia, whereas other regimens showed comparable results (Figure 2D and Supplemental Table 5). The network plot of hyperkalemia is shown in Figure 3D. Based on P-score ranking, vicadrostat 40 mg (P-score = 0.80) was ranked best in decreasing hyperkalemia, whereas baxdrostat 2 mg was ranked worst (P-score = 0.13). The details regarding P-score ranking are given in Table 2 and Supplemental Figure 4. No heterogeneity was detected (tau2 < 0.0001; I2 = 0% [0.0%; 67.6%]).
Serious Adverse Events
Ten studies, including 25 treatment regimens and 83 pairwise comparisons, were included in the network meta-analysis. Compared to placebo, none of the treatment regimens related to baxdrostat, lorundrostat, osilodrostat, and vicadrostat showed a significant change in the risk of serious adverse events (Figure 5A and Supplemental Table 6). The network plot for serious adverse events is shown in Figure 5B. Based on P-scores, baxdrostat 0.5 mg is ranked best (P-score = 0.85) in reducing serious adverse events, whereas the lorundrostat 12.5 mg once daily is ranked worst (P-score = 0.11). The details regarding P-score ranking are provided in Table 2 and Supplemental Figure 5. No heterogeneity was detected (tau2 = 0.075; I2 = 0% [0.0%; 60.2%]).

(A) Forest plot for serious adverse events and (B) network plot for serious adverse events.
Symptomatic Hypotension
Six studies, including 16 treatment regimens and 51 pairwise comparisons, were included in the network meta-analysis. Compared to placebo, only lorundrostat 50 mg once daily significantly increased the risk of symptomatic hypotension, whereas other regimens showed results comparable to placebo (Figure 6A and Supplemental Table 7). The network plot for symptomatic hypotension is shown in Figure 6B. Based on P-score ranking, vicadrostat 3 mg plus empagliflozin was ranked best (P-score = 0.68) in reducing symptomatic hypotension, whereas vicadrostat 10 mg plus empagliflozin was ranked worst (P-score = 0.21). The details regarding the P-score ranking of the interventions are provided in Table 2 and Supplemental Figure 6. No heterogeneity was detected (tau2 < 0.0001; I2 = 0% [0.0%; 74.6%]).
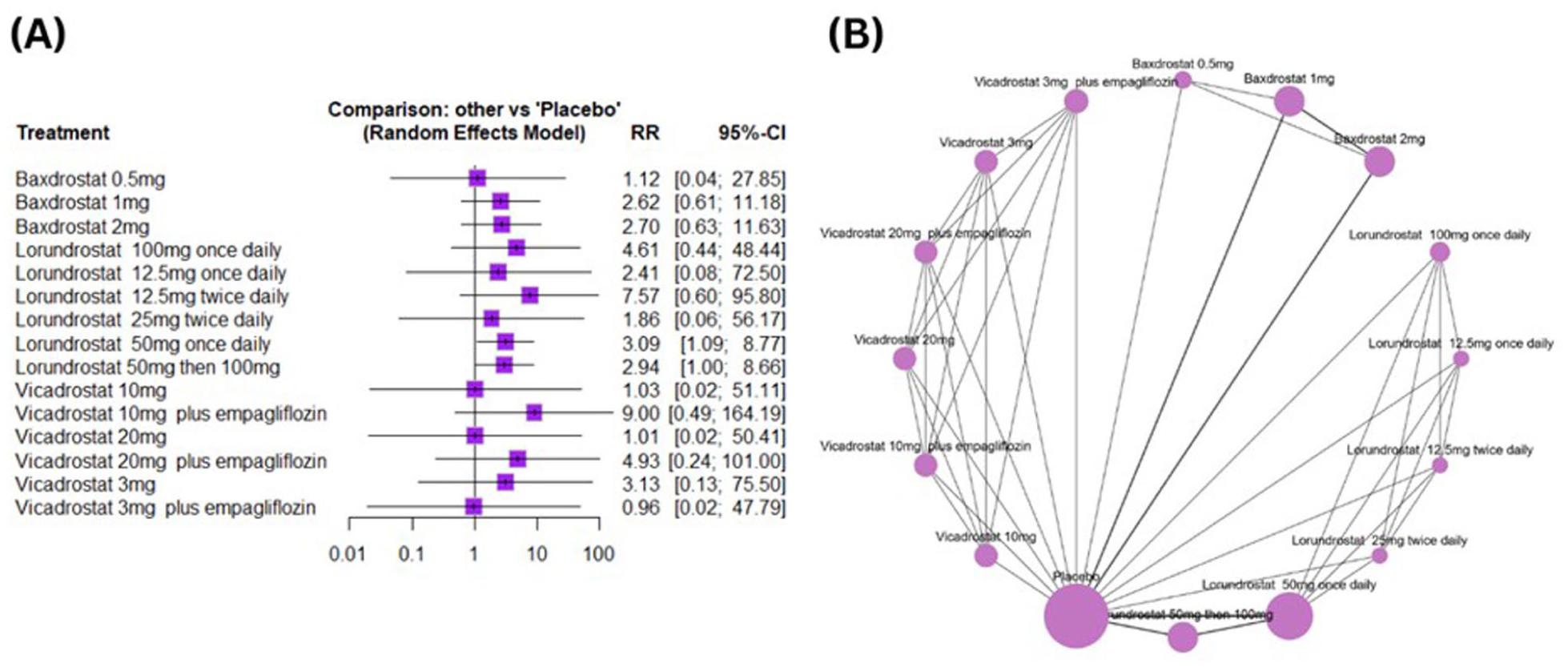
(A) Forest plot for symptomatic hypotension and (B) network plot for symptomatic hypotension.
Inconsistency and Publication Bias
To assess the inconsistency between direct and indirect evidence, we performed a nodal split analysis. No significant inconsistencies were found via the nodal split analysis. The details of this analysis are presented in Supplemental Tables 8–13. We evaluated publication bias by visually examining funnel plot asymmetry and statistically using Egger’s regression test. Our visual analysis of the funnel plots showed no publication bias, which was confirmed by the results of Egger’s regression test (Supplemental Figures 7–12). Prediction intervals indicate the range within which the effect of a future study is likely to fall, providing a means to assess the influence of heterogeneity in the results if present. The prediction intervals for all the outcomes are provided in the Supplemental Figures 13–18.
GRADE Assessment
The certainty of the evidence was evaluated using CiNEMA, which examines 6 domains: (1) Within-study bias, (2) Reporting bias, (3) Indirectness, (4) Imprecision, (5) Heterogeneity, and (6) Incoherence. Overall, the certainty of the evidence ranges from moderate to low. Details of the GRADE assessment are available in Supplemental Tables 14–19.
Discussion
This network meta-analysis pooled 10 RCTs to compare the efficacy and safety profile of ASI regimens among patients with uncontrolled or resistant hypertension. Efficacy outcomes were evaluated based on changes in SBP and DBP. Baxdrostat 1 and 2 mg; lorundrostat 100 mg once daily, 12.5 mg twice daily, 25 mg twice daily, 50 mg once daily, and 50 mg escalating to 100 mg; osilodrostat 0.25 mg once daily, 0.5 mg once daily, and 1 mg once daily; and vicadrostat 10, 20, and 3 mg plus empagliflozin, and 20 mg caused significant reductions in SBP. Significant reductions in DBP were observed with baxdrostat 1 and 2 mg, and lorundrostat 50 mg once daily. Regarding safety, adverse events were frequently reported with all lorundrostat regimens, and vicadrostat 20 mg. Serious adverse events were comparable across all regimens. Increased incidence of hyperkalemia was demonstrated with baxdrostat 1 and 2 mg; all lorundrostat regimens; and vicadrostat 10 mg plus empagliflozin and 20 mg, while symptomatic hypotension was reported with lorundrostat 50 mg once daily only.
Among the regimens analyzed in this network meta-analysis, baxdrostat 2 mg showed the highest efficacy in reducing SBP, securing the highest P-score ranking. The significant reduction of SBP by the baxdrostat is supported by the previous meta-analysis by Gao et al, 25 Siddiqui et al, 26 Louat et al. 27 A recent meta-analysis by Dewi et al 28 also corroborates our findings, ranking baxdrostat 2 mg as the highest for SBP reduction. The Baxdrostat operates through potent and selective antagonism of aldosterone synthase, the enzyme responsible for catalyzing the rate-limiting step of aldosterone biosynthesis.29,30 Also, improved nitric oxide bioavailability precipitates further vascular relaxation.31,32 These effects may underlie baxdrostat’s superior ranking in our pooled analysis, particularly at 2 mg, suggesting a dose-dependent relationship. Notably, baxdrostat 2 mg ranked lowest in reducing the risk of hyperkalemia, and significant hyperkalemia was observed with both 1 and 2 mg doses. Similar instances of hyperkalemia have been documented in the landmark trials BrigHTN 2023 and BaxHTN 202510,14; nevertheless, the investigators determined that the events were unlikely to be directly attributable to the drug. Hyperkalemia remains a recognized consequence of aldosterone synthase inhibition. The affected patients in the baxdrostat group did not develop cardiac arrhythmias and were able to safely resume therapy without subsequent complications.10,14
In contrast to other ASIs, the structural and pharmacodynamic specificity of baxdrostat enables it to lower BP without eliciting the cortisol increases commonly associated with other ASIs.33,34 Baxdrostat’s ability to lower BP without instigating adrenocortical insufficiency diminishes undesirable systemic effects and prevents the incidence of various factors linked to higher mortality and morbidity, including weakened immune system, compromised cardiovascular response, and dysregulated renal function.33,35,36 The biochemical profile of baxdrostat translated into a superior P-ranking for baxdrostat 0.5 mg, as demonstrated by our pooled estimates evaluating serious adverse effects. Overall, the distinct dose-dependent effects of baxdrostat underscore its potential to be precisely calibrated to patient-specific needs, enabling clinicians to optimize therapeutic benefit while carefully managing safety considerations in the context of individual comorbidities and baseline characteristics.
Although baxdrostat 1 and 2 mg produced significant reductions in DBP, the lorundrostat 50 mg once-daily dosage emerged as the most effective regimen for lowering DBP, securing the highest P-score in the treatment ranking. We speculate that, compared with other doses of lorundrostat, the 50 mg once-daily dose provides an optimal overnight period of “aldosterone escape” with significant hemodynamic benefits. This is consistent with earlier placebo-controlled trials that similarly established lorundrostat’s efficacy to reduce BP, thereby corroborating our findings.11,22,23 Our findings regarding the higher ranking of baxdrostat 2 mg and lorundrostat 50 mg once daily are corroborated by a recent meta-analysis by Dewi et al. 28 Although the reduction in DBP is not statistically significant compared to our study, which showed a significant reduction in DBP.
While lorundrostat 50 mg once daily demonstrated the greatest efficacy in reducing DBP, P-score ranking identified the 12.5 mg dose as the least favorable with respect to averting the risk of adverse and serious adverse events. This finding may be attributable to the relatively short half-life of lorundrostat, 37 possibly restricting the 24-hour pharmacodynamic efficacy of the 12.5 mg once-daily regimen. Despite the lorundrostat demonstrating a high specificity for aldosterone synthase, 38 inadequate trough concentrations with 12.5 mg may have caused insufficient aldosterone suppression during the dosing interval, thereby reducing the cumulative efficacy and predisposing to an elevated incidence of adverse outcomes. In addition to the 12.5 mg once-daily regimen, other doses of lorundrostat also showed a significant association with adverse events. This reinforces the notion that lorundrostat’s short half-life may result in insufficient inhibition of aldosterone synthase, thereby promoting sodium retention and variations in intravascular volume. Such hemodynamic instabilities subsequently increase the predisposition to adverse clinical outcomes despite an overall reduction in mean BP. 39
Notably, our pooled estimates showed that osilodrostat 0.5 mg once daily had the lowest incidence of adverse effects, with the highest P-score. Although a low incidence of adverse events was observed with osilodrostat, it cannot be interpreted as evidence of superior drug safety due to non-significant results. These findings are corroborated by the previous meta-analyses by Siddiqui et al 26 and Goh et al, 40 which also showed a non-significant reduction in the adverse events. This suggests that the superior P-score of osilodrostat in lowering the risk of adverse effects predominantly stemmed from the cautiously planned trial characteristics, including intensive run-in and strict exclusion criteria, producing lower-risk cohorts.
Our results identified vicadrostat as an important therapeutic agent with noteworthy dose-dependent and combination-specific implications. The vicadrostat 40 mg dose achieved the highest P-score for reducing hyperkalemia; however, intermediate doses, including vicadrostat 20 and 10 mg in combination with empagliflozin, were associated with a higher incidence of hyperkalemia. The findings are consistent with a multinational phase 2 trial evaluating the efficacy of vicadrostat among hypertensive patients with chronic kidney disease. 24 These findings indicate a critical interplay between the extent of aldosterone synthase inhibition and compensatory neurohormonal feedback, wherein higher-dose suppression may achieve more consistent correction of mineralocorticoid-driven potassium retention, while intermediate inhibition may insufficiently offset homeostatic mechanisms and thereby predispose to hyperkalemia.19,24,41
The SGLT2 inhibitors, such as empagliflozin, exert complementary effects on natriuresis, intravascular volume, and blood pressure. 42 Evidence from large-scale cardiovascular outcome trials has consistently shown that SGLT2 inhibitors are associated with a reduced incidence of clinically significant hyperkalemia.42 -44 Our findings suggested that, when combined with vicadrostat, this interaction is dose-dependent: at certain doses, the combination mitigated the risk of hyperkalemia, whereas at others, it appeared to exacerbate it. The assessment of hemodynamic outcomes also revealed a dose-dependent trend, with the vicadrostat 3 mg plus empagliflozin regimen conferring the greatest protection against symptomatic hypotension, while the 10 mg combination consistently emerged as the least favorable. A plausible explanation may stem from the balance between aldosterone synthase inhibition and the osmotic diuretic effects of SGLT2 blockade. At lower vicadrostat doses, empagliflozin’s hemodynamic influence remains proportionate, supporting circulatory stability and minimizing hypotension while sustaining cardiorenal benefit. 24 In contrast, intermediate dosing such as 10 mg may intensify fluid shifts or instigate disruption in RAAS responses, hence increasing the risk of symptomatic hypotension. These observations underscore that the safety profile of vicadrostat in combination with SGLT2 inhibitors is dose-dependent, warranting tailored regimens. Patients with a predisposition to hyperkalemia may benefit from higher doses under strict BP monitoring, while those at risk of hypotension may require lower doses. Careful dose selection, in conjunction with electrolyte and hemodynamic assessment, is the cornerstone of optimizing outcomes.
This study highlights the clinical relevance of ASIs as a new approach for managing hypertension, especially in cases of uncontrolled and treatment-resistant hypertension. The reductions in SBP and DBP observed with baxdrostat and lorundrostat suggest they may serve as effective alternatives when traditional antihypertensive treatments fail to adequately control blood pressure. Additionally, the different safety profiles of the ASIs highlight the importance of carefully choosing the appropriate regimen based on each patient’s risk factors. Clinicians should carefully weigh the efficacy of different ASI regimens against side effects such as hyperkalemia, with close monitoring for patients with additional comorbidities.
Our study is not free from limitations. First, the included trials had small sample sizes and short follow-up durations, which may limit the generalizability of our findings. Second, discrepancies among the trial designs, dosing strategies, and patient selection may have introduced variability and affected the comparability of results. However, our study consistently reported low statistical heterogeneity across all endpoints. Third, some regimens were evaluated in only 1 or 2 trials, reducing the confidence in estimates for those arms despite robust modeling. Fourth, including patients from different countries and age groups, and those with various comorbidities, can introduce variability in the patient population, which could affect the generalizability of our findings. Finally, various included studies are phase 2 trials and reflect a preliminary nature of the existing evidence base, which may affect the precision of effect estimates, thus mandating cautious interpretation and validation through larger, long-term phase 3 trials.
Conclusion
This network meta-analysis of ASI regimens for hypertension showed dose-dependent effects. Multiple regimens, including baxdrostat 1 mg, 2 mg; most lorundrostat regimens; osilodrostat 0.25, 0.5, and 1.0 mg once daily; vicadrostat 10, 20, and 3 mg plus empagliflozin, significantly lowered SBP. Baxdrostat 1 mg, 2 mg, and lorundrostat 50 mg once daily significantly reduce DBP. Adverse events were increased with lorundrostat regimens and vicadrostat 20 mg, but serious adverse events were similar across regimens. Hyperkalemia was more frequent with baxdrostat 1 and 2 mg, and with all lorundrostat regimens. Symptomatic hypotension occurred mainly with lorundrostat 50 mg daily. These findings underscore the importance of individualized treatment strategies, in which clinicians must carefully weigh efficacy against safety and select regimens tailored to patients’ specific risk profiles. Robust clinical trials with longer follow-up periods are necessary to fully capture the nuances of ASIs before they are widely adopted in clinical practice.
Supplemental Material
sj-docx-1-cic-10.1177_11795468261436180 – Supplemental material for Comparing Efficacy and Safety of Different Aldosterone Synthase Inhibitors for Hypertension: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials
Supplemental material, sj-docx-1-cic-10.1177_11795468261436180 for Comparing Efficacy and Safety of Different Aldosterone Synthase Inhibitors for Hypertension: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials by Muhammad Hassan Waseem, Zain Ul Abideen, Areeba Shoaib, Hafsa Arshad Azam Raja, Pawan Kumar Thada and Ameer Haider Cheema in Clinical Medicine Insights: Cardiology
Supplemental Material
sj-docx-2-cic-10.1177_11795468261436180 – Supplemental material for Comparing Efficacy and Safety of Different Aldosterone Synthase Inhibitors for Hypertension: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials
Supplemental material, sj-docx-2-cic-10.1177_11795468261436180 for Comparing Efficacy and Safety of Different Aldosterone Synthase Inhibitors for Hypertension: A Systematic Review and Network Meta-analysis of Randomized Controlled Trials by Muhammad Hassan Waseem, Zain Ul Abideen, Areeba Shoaib, Hafsa Arshad Azam Raja, Pawan Kumar Thada and Ameer Haider Cheema in Clinical Medicine Insights: Cardiology
Footnotes
Acknowledgements
None.
Author Note
This manuscript has not been published previously and is not under consideration for publication elsewhere.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Author Contributions
Study concept and design: MHW and ZUA; acquisition of data: ZUA and AS; analysis and interpretation of data: HAAR and ZUA; drafting of the manuscript: AS, HAAR, PKT, and AHC; critical revision of the manuscript: MHW and AHC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be made available upon reasonable request to the authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
