Abstract
Background:
Coronary artery disease (CAD) is the leading cause of death worldwide. After percutaneous coronary intervention (PCI), dual antiplatelet therapy (DAPT) is recommended to reduce thrombotic events. This meta-analysis assesses the effectiveness of clopidogrel compared to aspirin monotherapy following DAPT post-PCI.
Methods:
From inception to April 2025, an exhaustive literature search was conducted across electronic databases, including PubMed, Cochrane Library, ScienceDirect, EMBASE, and Web of Science. Risk ratios (RRs) along with 95% confidence intervals (CIs) were pooled using the random-effects model in Review Manager. Leave-one-out sensitivity analysis and funnel plots were used to evaluate heterogeneity and publication bias, respectively.
Results:
Six studies, including 3 RCTs and 3 observational studies, spanning over 19 494 patients, were included in our analysis. Clopidogrel significantly reduced major adverse cardiovascular events (MACE) (RR = 0.78; 95% CI: [0.69, 0.89]; P = .0002; I2 = 0%) and myocardial infarction (MI) (RR = 0.73; 95% CI: [0.56, 0.94]; P = .02; I2 = 21%) compared to aspirin. Likewise, the clopidogrel group demonstrated a substantial advantage in reducing the incidence of any stroke (RR = 0.66; 95% CI: [0.49, 0.89]; P = .006; I2 = 14%), including ischemic stroke (RR = 0.69; 95% CI: [0.49, 0.97]; P = .04; I2 = 0%). All other endpoints, including hemorrhagic stroke, all-cause mortality, cardiac death, major bleeding, stent thrombosis, repeat, and target vessel revascularization, were comparable between the 2 arms.
Conclusion:
Clopidogrel significantly reduced the incidence of MACE, MI, and stroke after DAPT following PCI compared to aspirin, indicating greater effectiveness. However, the main conclusion of this meta-analysis depends primarily on the estimates from RCTs. Additional randomized studies are necessary to confirm these results and support clinical decision-making.
Introduction
Coronary artery disease (CAD) is characterized by the buildup of atherosclerotic plaques in the coronary arteries. It encompasses conditions such as stable angina, acute coronary syndrome (ACS), and silent myocardial ischemia. CAD is the leading cause of death worldwide. In 2021, CAD was responsible for 9.4 million deaths globally. 1 It is the most prevalent cardiovascular disease in the United States, affecting 18.2 million adults. 2 In 2017, 637 650 percutaneous coronary intervention (PCI) procedures were performed in CAD patients in the United States, increasing from 550 872 in 2013. 3 Current guidelines by the American Heart Association, the American College of Cardiology, and others recommend that dual antiplatelet therapy (DAPT) reduce the risk of early and late thrombotic events among patients with ACS regardless of whether they are managed with an invasive strategy. 4 Previous meta-analyses have shown the effectiveness of Aspirin for the secondary prevention of atherosclerotic cardiovascular disease (ASCVD), and it is recommended in patients who complete DAPT following PCI.5,6 According to the 2024 European Society of Cardiology guidelines, clopidogrel can be an alternative in case the patient has any contraindication or intolerance to aspirin. 7
According to the CAPRIE trial, published in 1996, clopidogrel was more effective than aspirin for the secondary prevention of ASCVD, reducing major adverse cardiovascular events (MACE), combined risk of ischemic stroke, and myocardial infarction (MI) with a safety profile comparable to that of aspirin. 8 However, the trial included patients who had stable coronary disease and was done before the availability of drug-eluting stents (DES).
Most recently, the HOST-EXAM trial showed promising results of clopidogrel as monotherapy in patients who completed appropriate DAPT following PCI. 9 A recent review by Galli et al 10 discussed how P2Y12 inhibitors, such as clopidogrel, are now increasingly viewed not just as alternatives to aspirin; they might even be preferred over it. Despite prior meta-analyses on this subject by Sibbing et al, Marques et al, and Gragnano et al,11 -13 further investigation is needed in the form of high-power randomized trials. Given the significant findings from the recently published SMART-CHOICE 3 and STOPDAPT-2 trials comparing clopidogrel and aspirin monotherapy in high-risk PCI patients, there is a pressing need to update the existing evidence.14,15 These large, randomized studies employed robust methodologies and had long follow-up periods, thereby improving the reliability of their results. Incorporating this recent data enhances our understanding of the optimal antiplatelet therapy after DAPT ends post-PCI. Therefore, an updated meta-analysis is needed to enhance clinical decision-making and provide evidence-based guidance.
Methods
The systematic review and meta-analysis adhered rigorously to the established guidelines formulated by the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) 16 and the Cochrane Handbook for Systematic Reviews of Interventions. 17 This review was registered on PROSPERO under the ID: CRD420251045921.
Data Sources and Search Strategy
Two authors (AHC and ZUA) independently searched PubMed, Cochrane Central, ScienceDirect, EMBASE, and Web of Science from inception to April 2025 for studies that compared aspirin with clopidogrel after DAPT following PCI. Additionally, the relevant articles were identified through manual bibliographic searches. The following MeSH terms and keywords were utilized in the search strategy: “Clopidogrel,” “Aspirin,” “Dual antiplatelet therapy,” and “PCI.” A description of the search strategies used in each database is given in Supplemental Table 1.
Study Selection and Eligibility Criteria
All the articles retrieved in our initial search were imported into EndNote Reference Library software, and duplicates were removed. Two authors (AHC and MHW) independently screened the titles and abstracts of all retrieved articles, excluding those that did not meet the inclusion criteria. Full texts of the remaining articles were reviewed against the established eligibility criteria. Disagreements regarding the selection were resolved through discussion with the third reviewer.
We included studies in our systematic review and meta-analysis if they met the following eligibility criteria: (1) Study design: Randomized Controlled Trials (RCTs) or observational studies; (2) Population: Adults (age> 18 years) with ACS and chronic coronary syndrome (CCS) or stable CAD undergoing PCI who received DAPT for >3 months; (3) Intervention: Clopidogrel administration following DAPT; (4) Comparison: Aspirin administration following DAPT; (5) Outcomes: MACE, major bleeding, all-cause mortality, cardiac death, stroke (ischemic, hemorrhagic, or any), stent thrombosis, MI, repeat and target vessel revascularization.
We excluded the following studies: (1) Study design: Case reports, case series, editorials, systematic reviews, narrative reviews, and abstracts without full text, as well as animal studies; (2) Population: children or adolescents (under 18 years); Intervention: administration of P2Y12 inhibitors other than clopidogrel. No language or data restrictions were applied. The study selection is depicted in the PRISMA flowchart Figure 1.
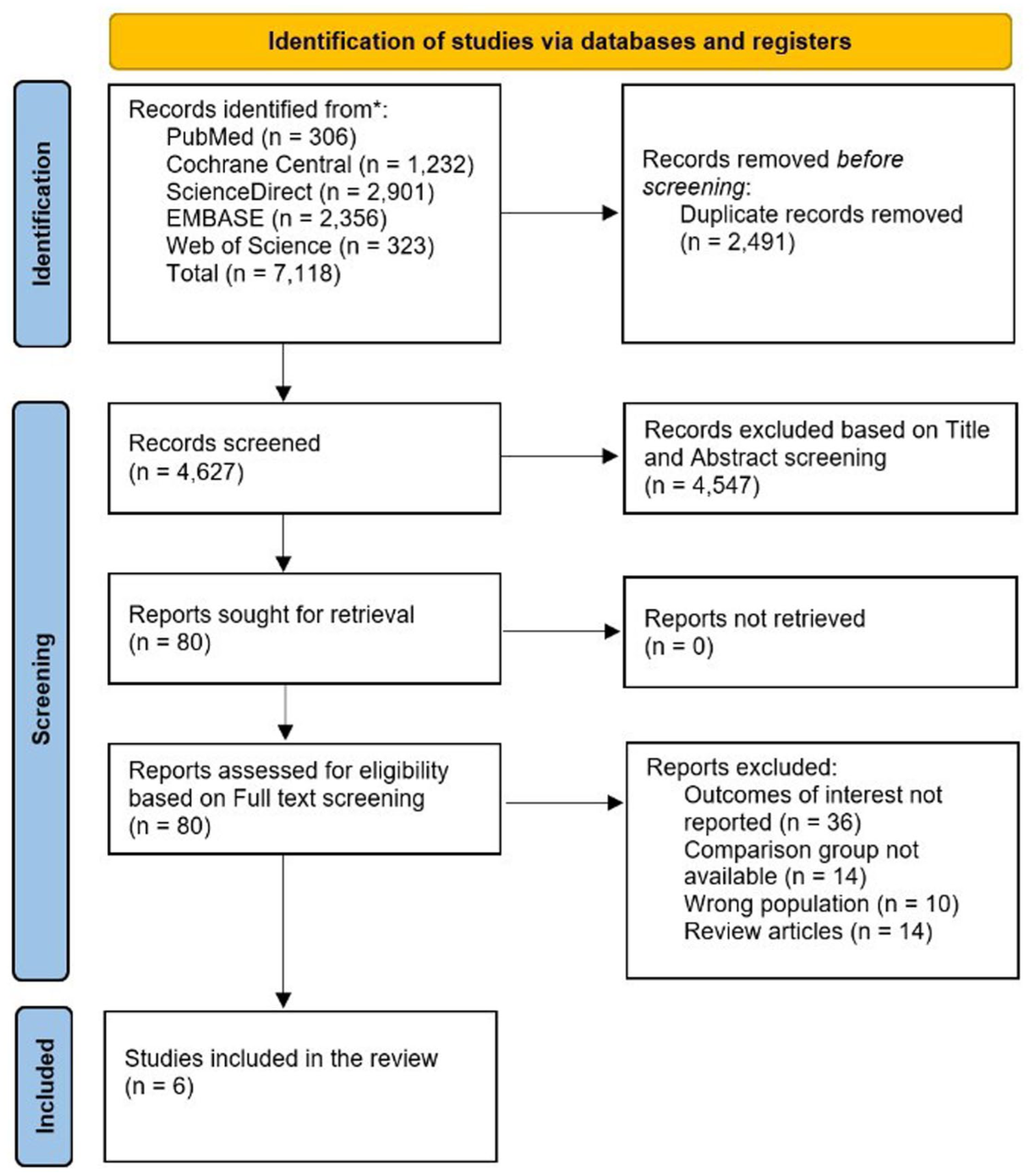
PRISMA flowchart of the study selection procedure.
Data Extraction
Data regarding the baseline characteristics and outcomes of interest were extracted on an Excel spreadsheet. The baseline data on study characteristics (study ID, location, study design, centers, inclusion criteria, DAPT regimen after PCI, clopidogrel/aspirin dosage, sample size of clopidogrel and aspirin arm, follow-up duration), patient characteristics (mean age, gender, body mass index [BMI], smoking status, hemoglobin levels, type of ACS at the time of presentation; comorbidities like diabetes, hypertension, dyslipidemia, chronic kidney disease [CKD], congestive heart failure [CHF]; medical history including history of previous MI, stroke, PCI, coronary artery bypass graft [CABG]; drug history including history of beta blocker, Angiotensin receptor blockers ARBs, Angiotensin converting enzyme [ACE] inhibitors or statin usage) and angiographic and procedural characteristics (percentage of 1, 2, or 3 vessel CAD, left main disease, first and second generation DES, mean stents implanted along with stents length) were extracted. Any discrepancies were resolved by group discussion. The primary and secondary endpoints of interest extracted were MACE, major bleeding, all-cause mortality, cardiac death, stroke (ischemic, hemorrhagic, or any), stent thrombosis, MI, repeat, and target vessel revascularization.
Risk of Bias
We evaluated the quality of the included RCTs and observational studies using the revised Cochrane risk of bias tool (RoB 2.0) and the ROBINS-I tool, respectively.18,19 RoB 2.0 consists of 5 domains: (1) bias from the randomization process, (2) bias from deviations in intended interventions, (3) bias from missing outcome data, (4) bias in outcome measurement, and (5) bias in selecting reported results. In contrast, the ROBINS-I tool evaluates 7 domains: (1) bias from confounding factors, (2) bias from participant selection, (3) bias in intervention classification, (4) bias from deviations in intended interventions, (5) bias due to missing data, (6) bias in outcome measurement, and (7) bias in the selection of reported outcomes results. Publication bias was evaluated visually using funnel plots.
Statistical Analysis
We conducted the statistical analysis on Review Manager software version 5.4.1. Clinical outcomes were evaluated using the Mantel-Haenszel random-effects model to pool risk ratios (RRs) with 95% confidence intervals (CIs). The pooled results were represented graphically as forest plots. The Chi-square test and the Higgins I2 statistics were used to evaluate the statistical heterogeneity. 20 A P-value of <.05 was considered statistically significant. With I2 values >50%, heterogeneity was assessed using a leave-one-out sensitivity analysis. Additionally, subgroup analyses were performed according to the included study designs and follow-up durations.
Results
Search Results
An exhaustive search across electronic databases, including PubMed, Cochrane Central, ScienceDirect, EMBASE, and Web of Science, yielded 7118 articles. After eliminating duplicates (2491), the remaining articles underwent title and abstract screening, yielding 80 selected for full-text review. Subsequently, only 6 studies9,14,15,21 -23 were included in the conclusive meta-analysis.
Characteristics of Included Studies and Patients
This systematic review included 3 RCTs9,14,15 and 3 observational studies21 -23 published between 2014 and 2025. There were 19 494 subjects from 6 studies. All studies were multicenter, except for Zhuang et al and Park et al.21,22 Most of the studies were conducted across South Korea9,15,22,23 except for Watanabe et al and Zhuang et al, which were conducted across Japan and China, respectively.14,21 The follow-up period ranged from 1 to 5 years. The most common dosages for clopidogrel and aspirin were 75 and 100 mg, respectively. The mean age ranged from 60.9 to 70.1 years, with the mean BMI ranging from 24.1 to 26.6 kg/m2.
Table 1 presents the baseline characteristics of the included studies, and Supplemental Table 2 displays those of the included patients. Supplemental Table 3 provides angiographic and procedural details. The definitions of MACE and major bleeding used across different studies are provided in Supplemental Table 4.
Baseline Characteristics of the Included Studies.

Abbreviations: ACS, acute coronary syndrome; CAD, coronary artery disease; CCS, chronic coronary syndrome; DAPT, dual antiplatelet therapy; MI, myocardial infarction; mo, month; NA, not available; PCI, percutaneous coronary intervention.
Risk of Bias
Two investigators (AN and FS) conducted the quality assessment. We assessed study quality using the ROBINS-I and Cochrane RoB 2.0 tools for eligible observational cohorts and randomized trials, respectively. Most cohort studies showed a moderate risk of bias, mainly due to confounding factors inherent to their observational study design. The details of the ROBINS-I tool for the quality assessment of observational studies are provided in Supplemental Table 5. All 3 RCTs showed some concerns due to deviations from intended interventions.9,14,15 The traffic plot for the RCTs quality assessment is provided in Supplemental Figure S1.
Outcomes
Major Adverse Cardiovascular Events (MACE)
All the included studies reported MACE. Our results showed that following DAPT after PCI, clopidogrel monotherapy significantly reduced MACE compared to aspirin (RR = 0.78; 95% CI: [0.69, 0.89]; P = .0002; I2 = 0%). Sub-group analysis based on the study design showed that the results became statistically insignificant in the case of observational studies (Figure 2). The Hartung-Knapp adjustment for MACE showed an RR of 0.78 with a 95% CI of 0.67-0.92, as shown in Supplemental Figure S2.
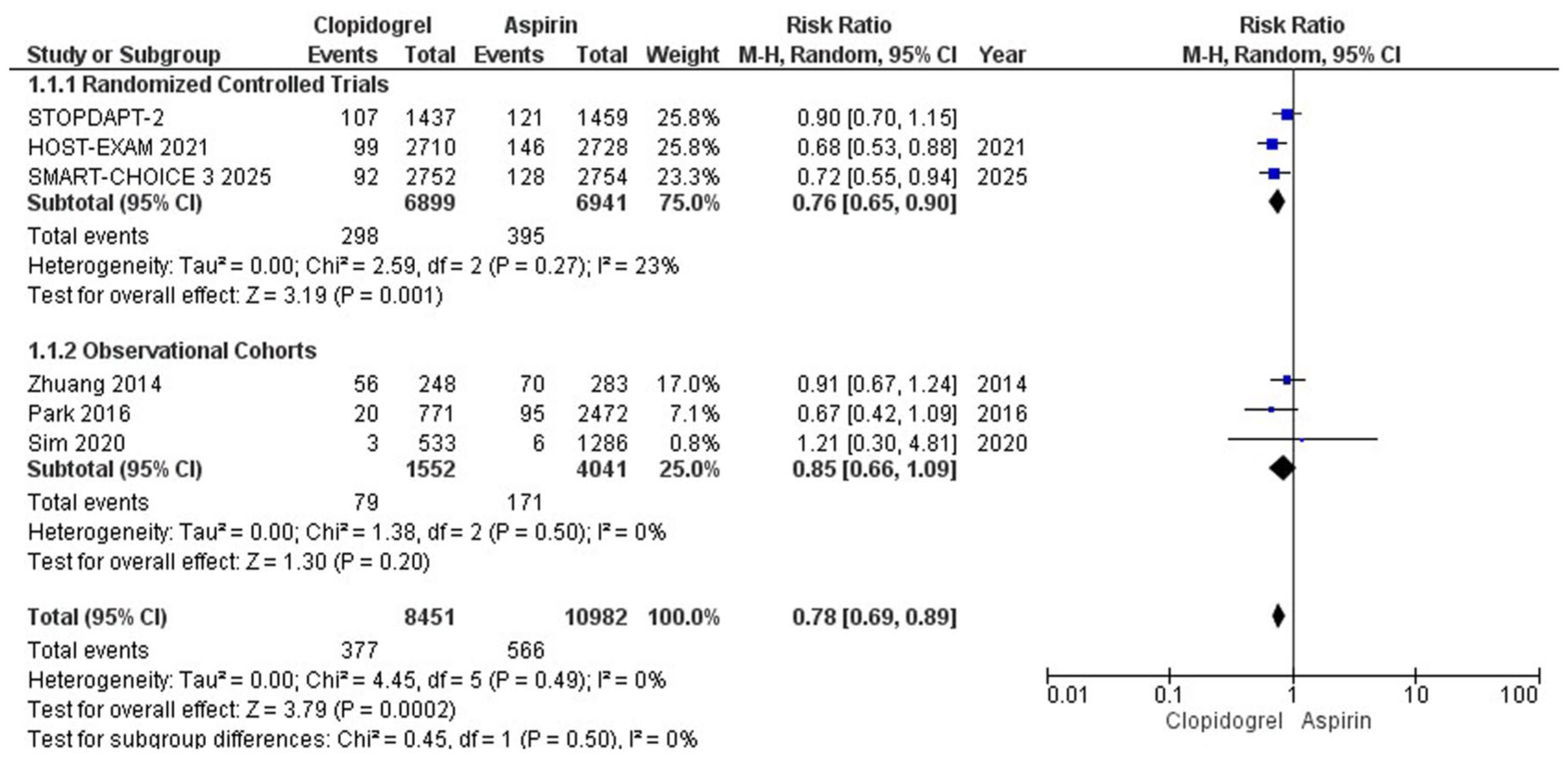
Major Adverse Cardiovascular Event (MACE) forest plot.
All-Cause Mortality
Five of the included studies reported all-cause mortality. Clopidogrel decreased mortality, but the results were statistically insignificant (RR = 0.94; 95% CI: [0.74, 1.19]; P = .59; I2 = 40%). On subgrouping based on study design, the results remained statistically insignificant in both subgroups (Figure 3). The Hartung-Knapp adjustment for all-cause mortality showed an RR of 0.94 with a 95% CI of 0.66-1.33, as shown in Supplemental Figure S3.
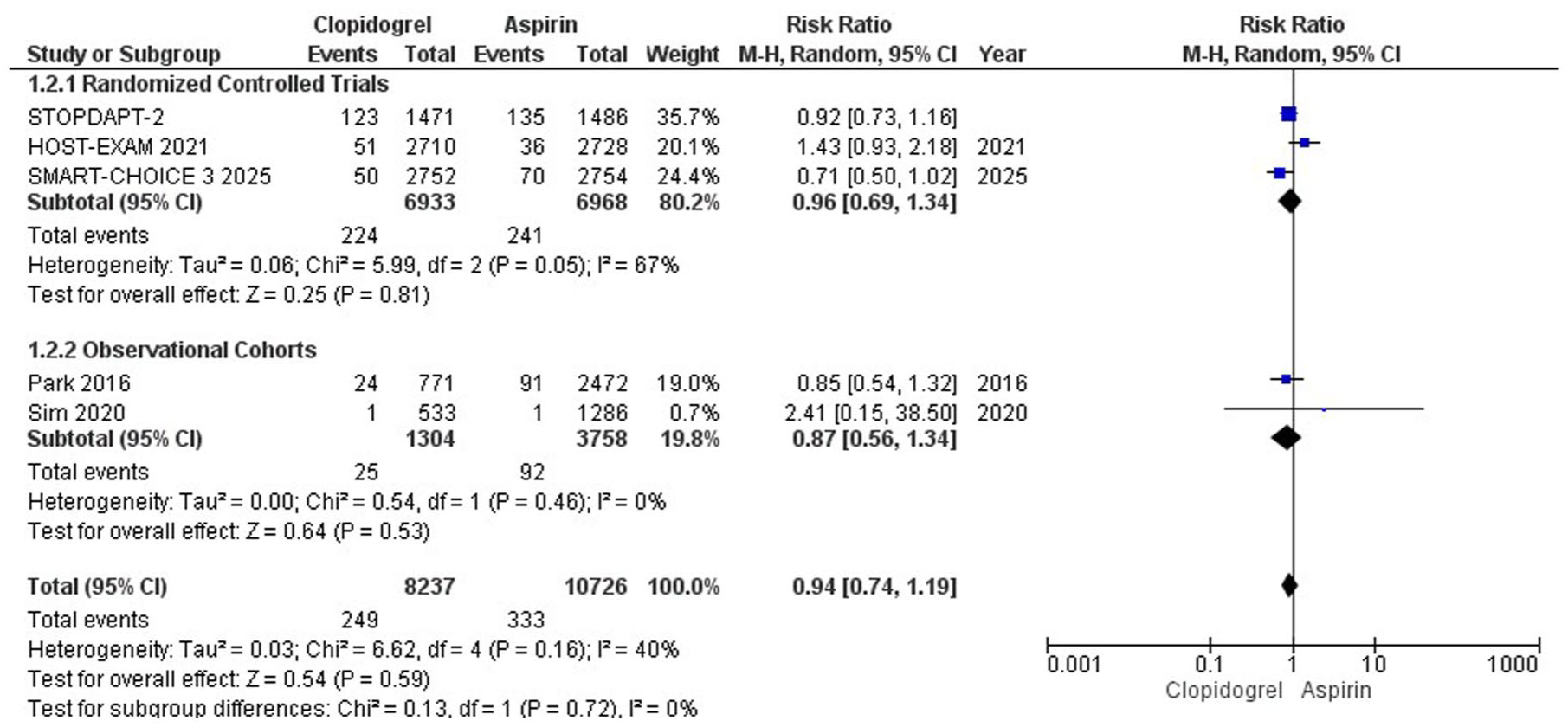
All-cause mortality forest plot.
Cardiac Death
Five studies reported cardiac death. Clopidogrel decreased cardiac death, but the results were statistically nonsignificant (RR = 0.82; 95% CI: [0.62, 1.08]; P = .15; I2 = 12%). The significance of the results from subgroup analysis based on study design showed no change in either subgroup (Figure 4). The Hartung-Knapp adjustment for cardiac death showed an RR of 0.82 with a 95% CI of 0.54-1.24, as shown in Supplemental Figure S4.

Cardiac death forest plot.
Major Bleeding
All the studies included reported this outcome. Our results showed no significant difference between clopidogrel and aspirin regarding major bleeding risk (RR = 0.95; 95% CI: [0.64, 1.40]; P = .79; I2 = 46%). When analyzed by study design subgroups, the results continued to be statistically insignificant in both categories (Figure 5). The Hartung-Knapp adjustment for major bleeding showed an RR of 0.95 with a 95% CI of 0.57-1.58, as shown in Supplemental Figure S5.

Major bleeding forest plot.
Stroke
Four studies reported the incidence of stroke. Compared to aspirin monotherapy, clopidogrel monotherapy after DAPT is associated with a lower risk of stroke, and the results are statistically significant (RR = 0.66; 95% CI: [0.49, 0.89]; P = .006; I2 = 14%). The results of subgroup analysis based on study design became insignificant for the observational studies subgroup (Figure 6). The Hartung-Knapp adjustment for stroke showed an RR of 0.66 with 95% CI of 0.41-1.06 as shown in Supplemental Figure S6.

Any stroke forest plot.
Ischemic Stroke
Four studies reported the incidence of ischemic stroke. Clopidogrel monotherapy is superior to aspirin and leads to a statistically significant reduction in the incidence of ischemic stroke (RR = 0.69; 95% CI: [0.49, 0.97]; P = .04; I2 = 0%) (Supplemental Figure S7). The Hartung-Knapp adjustment for ischemic stroke showed an RR of 0.69 with 95% CI of 0.43-1.12 as shown in Supplemental Figure S8.
Hemorrhagic Stroke
Three of the included studies reported this outcome. Compared to aspirin monotherapy, clopidogrel yielded no significant change in risk of hemorrhagic stroke in patients who underwent PCI and switched to clopidogrel after DAPT (RR = 0.54; 95% CI: [0.22, 1.33]; P = .18; I2 = 46%), (Supplemental Figure S9). The Hartung-Knapp adjustment for hemorrhagic stroke showed an RR of 0.55 with a 95% CI of 0.08-3.52, as shown in Supplemental Figure S10.
Myocardial Infarction
All studies reported MI. Our results showed a statistically significant decrease in the clopidogrel arm compared to aspirin regarding MI incidence (RR = 0.73; 95% CI: [0.56, 0.94]; P = .02; I2 = 21%). Upon subgrouping, the results became statistically insignificant for the observational studies subgroup (Supplemental Figure S11). The Hartung-Knapp adjustment for MI showed an RR of 0.73 with a 95% CI of 0.51-1.03, as shown in Supplemental Figure S12.
Repeat Revascularization
Four of the included studies reported repeat revascularization. The results are comparable between the 2 groups (RR = 0.93; 95% CI: [0.80, 1.09]; P = .39; I2 = 0%). The results of the subgroup analysis based on study design were insignificant for both groups (Supplemental Figure S13). The Hartung-Knapp adjustment for repeat revascularization showed an RR of 0.93 with 95% CI of 0.81-1.08 as shown in Supplemental Figure S14.
Target Vessel Revascularization
Target vessel revascularization was reported by 4 of the included studies. There is no statistically significant difference between clopidogrel monotherapy and aspirin monotherapy as shown by our results (RR = 0.80; 95% CI: [0.60, 1.07]; P = .13; I2 = 0%). The subgroup analysis results, categorized by study design, were insignificant for either group (Supplemental Figure S15). The Hartung-Knapp adjustment for target vessel revascularization showed an RR of 0.80 with a 95% CI of 0.68-0.95, as shown in Supplemental Figure S16.
Stent Thrombosis
Five of the included studies commented on the incidence of stent thrombosis. Our analysis shows that clopidogrel monotherapy reduces the incidence of stent thrombosis compared to aspirin alone, but the results are statistically nonsignificant (RR = 0.61; 95% CI: [0.34, 1.08]; P = .09; I2 = 0%). Results from the subgroup analysis, organized by study design, showed no significant differences for either group (Supplemental Figure S17). The Hartung-Knapp adjustment for stent thrombosis showed an RR of 0.61 with 95% CI of 0.29-1.28 as shown in Supplemental Figure S18.
Sensitivity Analysis, Subgroup Analysis, and Publication Bias
To explore the source of heterogeneity, we conducted a leave-one-out sensitivity analysis. Removing individual studies consecutively did not significantly affect the overall results of the meta-analysis. Removing studies that used the Thrombolysis In Myocardial Infarction (TIMI) criteria for major bleeding, namely Park et al Sim et al, and Watanabe et al,14,22,23 did not change the non-significant results, as shown in Supplemental Figure S19. Likewise, excluding studies that employed the Bleeding Academic Research Consortium (BARC) criteria for major bleeding, including Zhuang et al, SMART CHOICE 3, and HOST EXAM,9,15,21 also left the results statistically insignificant, as shown in Supplemental Figure S20.
The likelihood of publication bias was evaluated visually with funnel plots and statistically using Egger’s regression. The overall risk of publication bias is inconclusive since the funnel plots and Egger’s regression test have low sensitivity for detecting publication bias due to the small number of included articles in this meta-analysis (Supplemental Figures S21-S31).
In subgroup analyses by follow-up duration (12 months vs> 12 months), the results remained the same, except that MACE and MI became insignificant in the 12-month group, as shown in Supplemental Figures S32-S39.
Discussion
This meta-analysis evaluated the comparative efficacy and safety of clopidogrel versus aspirin monotherapy in patients who had undergone successful PCI and completed a standard DAPT duration. Our results showed that following DAPT after PCI, clopidogrel monotherapy significantly reduced MACE compared to aspirin. A previous meta-analysis study by Tan et al supported these findings. 24 However, subgroup analysis showed this effect was observed only in the RCT group. It is interesting to note that a similar trend was seen in the risk ratio for MI, a significant reduction in MI risk in the RCT group, but not in the observational studies group. Our analysis implies that MACE and MI were lower in the RCTs but not in observational studies.
Our findings of reduced MACE, MI, any stroke, and ischemic stroke are strongly supported by a recent individual-patient data meta-analyses by Giacoppo et al 25 and Valgimigli et al, 26 which also reported significant reductions in MACE, MI, any stroke, and ischemic stroke. Moreover, our meta-analysis includes both RCTs and observational cohort studies, thereby increasing statistical power, enhancing generalizability, and providing a more real-world perspective. Additionally, our analysis focuses exclusively on clopidogrel rather than the broader P2Y12 inhibitor class, with particular emphasis on its clinical profile.
Several key methodological and patient-level differences in the observational studies could explain these differences. Firstly, in the study by Park et al, despite using Inverse Probability of Treatment Weighting (IPTW) adjustment, patients in the clopidogrel group had significantly worse baseline profiles. 22 They were older and had a higher prevalence of diabetes, hypertension, CKD, prior stroke, and more complex coronary lesions. This suggests that physicians may have preferentially prescribed clopidogrel for patients with higher ischemic risk or aspirin intolerance. Sim et al analyzed acute MI patients from a nationwide Korean registry and found no difference in net adverse clinical events (NACE). 23 However, they lacked clarity on why patients were assigned to either drug, leaving room for significant treatment selection bias.
In the study by Zhuang et al, there were significant baseline differences in ischemic risk across the 3 treatment groups, which complicates direct comparisons of outcomes. 21 Patients in the standard-dose clopidogrel group had the highest baseline ischemic risk. They had a higher prevalence of multivessel coronary disease compared to the aspirin and low-dose clopidogrel groups. Additionally, the standard-dose clopidogrel group had a significantly higher proportion of patients with a history of MI, elevated LDL-C levels, and higher HbA1c, indicating poorer lipid and glycemic control. They also had a higher percentage of patients with chronic kidney disease and a higher BMI, all of which are well-established predictors of future cardiovascular events. This suggests that the patients in the standard-dose clopidogrel group had a higher cardiovascular risk profile compared to the aspirin group, which could have potentially made them more likely to have ischemic events regardless of treatment. Regarding other outcomes, our analysis indicates that clopidogrel is comparable to aspirin alone in terms of all-cause mortality, cardiac death, major bleeding, hemorrhagic stroke, repeat revascularization, target vessel revascularization, and stent thrombosis. These findings are supported by research from Tan et al 24 and by recent individual patient data meta-analyses by Giacoppo et al 25 and Valgimigli et al, 26 which also demonstrated comparable rates of all-cause mortality, cardiac death, major bleeding, and stent thrombosis.
Compared to aspirin monotherapy, clopidogrel monotherapy after DAPT is associated with a lower risk of stroke, and the results are statistically significant. These findings are similar to the landmark CAPRIE trial. 8 The trial involved 19 000 patients with recent ischemic stroke, MI, or peripheral arterial disease and compared the composite endpoint of stroke, MI, or vascular death between aspirin and clopidogrel. Notably, the greatest benefit was observed in the stroke subgroup, where clopidogrel reduced the relative risk of recurrent ischemic stroke. These findings laid the foundation for clopidogrel’s preferred use in patients with high cerebrovascular risk, which continues to this day and is reinforced by the findings in our analysis.
Importantly, major bleeding risk was comparable between the 2 groups, suggesting that clopidogrel does not confer an increased hemorrhagic burden relative to aspirin. 13 Aspirin irreversibly inhibits COX-1, reducing thromboxane A₂ and, ultimately, platelet aggregation. At the same time, it also suppresses protective gastric prostaglandins, theoretically increasing the risk of GI bleeding. 27 On the other hand, clopidogrel blocks the P2Y12 receptor without affecting prostaglandins, making it less ulcerogenic. 28 Confounding by indication may bias results as patients perceived to be at higher bleeding risk (eg, older age, GI history, anemia) are often selectively prescribed clopidogrel, which could obscure its safety advantage. Similar findings were seen in a recent meta-analysis by Wang et al, which evaluated RCTs comparing P2Y12 inhibitors and aspirin for secondary prevention. 29 They found that P2Y12 inhibitors, particularly clopidogrel, were associated with a modestly lower risk of gastrointestinal bleeding. However, the analysis also concluded that the overall risk of major bleeding was not significantly different between the 2 treatment strategies.
The CAPRIE trial also showed that while both drugs had similar safety profiles, clopidogrel was associated with fewer gastrointestinal side effects, including a lower rate of GI bleeding.8,27 The benefit of clopidogrel over aspirin might be slight, but it amplifies in some high-risk subgroups in the CAPRIE trial, such as those with previous cardiac surgery, diabetes, and at least one ischemic event.30 -32 Interestingly, in the HOST-EXAM study, clopidogrel monotherapy showed a significant reduction in both thrombotic and bleeding events compared to aspirin when used during the chronic maintenance phase after PCI. 33
In another study, patients who received a shorter duration of DAPT followed by P2Y12 inhibitor alone had significantly fewer major bleeding events compared to those who stayed on DAPT for a full 12 months. This supports the idea that stepping down to single therapy earlier may help lower bleeding risks without compromising protection. 34
Although the benefit of clopidogrel therapy over aspirin is significant in preventing major cardiovascular events, yet the cost-effectiveness of aspirin often takes precedence in many clinical settings. One relatively old yet significant study by Gaspoz et al revealed that expanding aspirin therapy to all eligible patients would cost approximately $11 000 per quality-adjusted life year (QALY) gained. 35 In contrast, prescribing clopidogrel to all patients resulted in an incremental cost exceeding $130 000 per QALY gained. The study concluded that, due to its higher cost, clopidogrel’s incremental cost-effectiveness is currently unattractive unless its use is limited to patients who are ineligible for aspirin. 35
The results of this meta-analysis are clinically meaningful. First, they suggest that clopidogrel can be considered a preferred long-term monotherapy. Second, this supports a personalized approach to antiplatelet therapy, where patient-specific factors such as tolerance to aspirin, comorbidities, and ischemic/bleeding risk profiles are used to guide post-DAPT treatment. In real-world settings, where aspirin intolerance and gastrointestinal side effects are common, clopidogrel offers an effective and safer alternative. However, the benefits of clopidogrel must be weighed against the increased costs, and shared decision-making between physician and patient is necessary. The burden of cardiovascular disease is growing globally and combined with longer life expectancy and the widespread use of DES, long-term antiplatelet strategy optimization remains a critical component of the management of ACS and CCS post-PCI and DAPT.
Limitations
Despite these promising findings, several limitations must be acknowledged: Most included studies were observational, only 3 being RCTs. Although multiple methods were used to minimize confounding, residual bias cannot be ruled out. Treatment decisions in observational studies may be influenced by unmeasured clinical factors, potentially skewing outcomes. This was especially prevalent in some of the studies in our analysis, where higher-risk patients were more likely to be prescribed clopidogrel. The comparable difference in major bleeding between clopidogrel and aspirin use might be explained by the inclusion of a population at particularly high risk of GI bleeding or by the frequent use of gastroprotective agents, such as proton pump inhibitors, by the population. The included studies varied in terms of patient populations, geographic locations, and clinical presentations (eg, STEMI, NSTEMI, unstable angina, and stable angina). For example, Zhuang et al focused on a Chinese population, while Park and Sim conducted their studies in Korean populations.21 -23 This may limit the generalizability of results to other ethnic groups, particularly Western populations with different genetic and metabolic profiles. Subgroup analysis based on ACS and CCS was not possible because most studies included both patient groups without separate data. Similarly, since most studies reported DAPT duration within a 6 to 18 month range, subgroup analysis according to standard (⩾6 months), short (<6 and >1 months), and ultrashort (⩽1 month) DAPT duration could not be conducted.
One of the most important unmeasured confounders is the CYP2C19 polymorphism, which affects clopidogrel metabolism. Individuals with poor metabolism may have reduced response to clopidogrel and be at higher risk for thrombotic events. The prevalence of these loss-of-function (LOF) alleles varies across populations, with a notably higher occurrence in Asian individuals. A meta-analysis by Xi et al focusing on Asian patients undergoing PCI found that carriers of at least one CYP2C19 LOF allele had a significantly increased risk of MACE compared to non-carriers. Additionally, the incidence of stent thrombosis was higher among LOF allele carriers. 36 Because Asians have a higher prevalence of the CYP2C19 LOF allele than non-Asians, the antiplatelet activity of clopidogrel may be reduced in Asians. Conversely, clopidogrel’s pharmacological activity may be more consistent in non-Asian populations. Therefore, this meta-analysis results should be interpreted carefully when applying them to populations with different ethnic and genetic backgrounds.
Given these limitations, further high-quality research is needed. Large-scale RCTs comparing aspirin and clopidogrel monotherapy post-DAPT are necessary, particularly in diverse, multi-ethnic populations. Future studies should include genetic testing for CYP2C19 polymorphisms to stratify patients based on clopidogrel responsiveness. This would enable precision medicine approaches in antiplatelet therapy. Studies with 5 to 10 year follow-up are needed to assess the durability of stroke prevention, bleeding safety, and survival benefit with clopidogrel monotherapy. Health-economic evaluations comparing the long-term costs of aspirin versus clopidogrel, especially in low-resource settings, would help guide policy-making.
Conclusion
Clopidogrel monotherapy after standard-duration DAPT seems to be a safe and effective alternative to aspirin in PCI patients. It provides better clinical outcomes concerning MACE, MI, and stroke prevention without raising major bleeding risk. However, the meta-analysis’s main conclusion relies solely on RCT data. These results endorse moving toward more personalized antiplatelet approaches, but additional long-term, randomized, and genetically informed research is essential to improve clinical decisions.
Supplemental Material
sj-docx-1-cic-10.1177_11795468251414841 – Supplemental material for Efficacy and Safety of Clopidogrel Versus Aspirin Monotherapy After Percutaneous Coronary Intervention: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-cic-10.1177_11795468251414841 for Efficacy and Safety of Clopidogrel Versus Aspirin Monotherapy After Percutaneous Coronary Intervention: A Systematic Review and Meta-analysis by Ameer Haider Cheema, Muhammad Hassan Waseem, Zain ul Abideen, Muhammad Zubair Tahir, Fahad Saleem, Amna Nadeem, Urvah Tauseef, Tahreem Qasim, Sania Aimen, Muhammad Bilal Zahid and Pawan Kumar Thada in Clinical Medicine Insights: Cardiology
Supplemental Material
sj-docx-2-cic-10.1177_11795468251414841 – Supplemental material for Efficacy and Safety of Clopidogrel Versus Aspirin Monotherapy After Percutaneous Coronary Intervention: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-2-cic-10.1177_11795468251414841 for Efficacy and Safety of Clopidogrel Versus Aspirin Monotherapy After Percutaneous Coronary Intervention: A Systematic Review and Meta-analysis by Ameer Haider Cheema, Muhammad Hassan Waseem, Zain ul Abideen, Muhammad Zubair Tahir, Fahad Saleem, Amna Nadeem, Urvah Tauseef, Tahreem Qasim, Sania Aimen, Muhammad Bilal Zahid and Pawan Kumar Thada in Clinical Medicine Insights: Cardiology
Footnotes
Author Contributions
Study concept and design: MHW and AHC; acquisition of data: ZUA and AHC; analysis and interpretation of data: MZT and FS; drafting of the manuscript: AN, UT, PKT and SA; critical revision of the manuscript: MHW.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
