Abstract
CAD and AF are 2 of the major cardiovascular challenges worldwide, often coexisting and complicating management strategies. PCI has revolutionized the treatment of CAD, reducing mortality and improving recovery. At the same time, AF increases the risk of stroke, thus requiring anticoagulation. The combination of antiplatelet and anticoagulant therapies in patients undergoing PCI is challenging because of increased bleeding risks. This literature review explores the controversies surrounding dual antithrombotic therapy (DAT) and triple antithrombotic therapy (TAT), evaluating their efficacy and safety in mitigating thrombotic and hemorrhagic risks. Key clinical trials and guidelines advocate the use of DAT, especially in patients at high bleeding risk, underlining its role in reducing complications without compromising ischemic protection. Furthermore, advances in stent technologies, personalized medicine tools, and pharmacogenomics have further refined treatment strategies toward tailored approaches. Despite these developments, the optimal duration of therapy remains debated, as do patient selection and the need to balance ischemic and bleeding risks, particularly in complex patients.This review underscores the need for an individualized, evidence-based approach to optimize outcomes and inform future guidelines in managing this high-risk population. Emerging therapies and real-world evidence—such as trials investigating Factor XI inhibitors and bioresorbable scaffolds—are promising, aiming to reduce bleeding risk and improve long-term outcomes in patients with CAD and AF. Additionally, newer DOACs are being evaluated for improved efficacy and safety profiles.
Keywords
Introduction
Coronary artery disease (CAD) remains the foremost global cause of mortality and disability, arising from progressive atherosclerotic plaque accumulation in the coronary arteries that precipitates myocardial ischemia, angina, and acute myocardial infarction. 1 Annually, CAD accounts for an estimated 7 million deaths and 129 million disability-adjusted life-years, with the burden disproportionately borne by low- and middle-income nations. 2 Percutaneous coronary intervention (PCI) is a cornerstone therapy: it alleviates angina, limits infarct size in acute coronary syndromes, and improves survival in selected high-risk presentations. 3 Its widespread adoption is reflected by more than 100,000 PCI procedures performed in the United Kingdom in 2020 alone, surpassing surgical revascularization volumes. 3 Successive technological and pharmacological advances continue to shorten recovery times and enhance long-term outcomes, underscoring the central role of PCI in contemporary CAD management.2,3
Atrial fibrillation (AF) is a common cardiac arrhythmia characterized by an irregular and frequently rapid heart rate, which substantially elevates the risk of stroke, heart failure, and other cardiovascular complications. 4 Globally, AF affects an estimated 33.5 million individuals, with prevalence increasing markedly with age, rising from approximately 2% in the general population to 10% to 12% among those aged 80 years and older.4,5 Importantly, AF is a significant risk factor for stroke, often doubling or even tripling its incidence, thereby representing a critical contributor to adverse cerebrovascular outcomes. 6
Beyond its clinical implications, AF necessitates continuous medical management, which can substantially reduce quality of life and impose significant economic burdens on patients.7,8 Effective prevention and mitigation strategies hinge on addressing modifiable risk factors, such as diabetes, hypertension, and physical inactivity. Early intervention targeting these determinants is critical, as it can decrease both the incidence of AF and the associated healthcare costs.4,5
Antiplatelet therapy is essential following percutaneous coronary intervention (PCI) to prevent thrombus formation at the stent site and reduce the risk of complications such as stent thrombosis. 3 In patients with AF, antithrombotic medications, including vitamin K antagonists like warfarin and direct oral anticoagulants (DOACs), are commonly prescribed to lower the risk of stroke and other thromboembolic events.3,4,8
Despite extensive research, substantial gaps remain in the optimal management of patients with concurrent CAD and AF, particularly those undergoing PCI. Existing clinical guidelines provide only limited guidance on the complex interplay between antiplatelet and anticoagulant therapies in this dual-disease context. This ambiguity complicates clinical decisions, as practitioners must carefully weigh the need to minimize thrombotic events against the increased risk of bleeding.
Moreover, the current evidence base lacks robust, long-term studies evaluating the safety and efficacy of combined antithrombotic regimens. This absence of conclusive data impedes the development of standardized treatment pathways and raises critical questions about emerging pharmacologic strategies.
Our research seeks to bridge these knowledge gaps by systematically evaluating the outcomes of various treatment protocols in patients with both CAD and AF. By doing so, we aim to establish a more definitive framework for clinical decision-making, enhance the understanding of dual antithrombotic therapy, and contribute to the refinement of future evidence-based guidelines.
Pathophysiology and Clinical Considerations
The development of atherosclerotic plaques begins with the subendothelial accumulation of cholesterol and the subsequent formation of foam cells—lipid-laden macrophages derived from circulating monocytes—within the arterial intima. These foam cells serve as the initial cellular foundation for plaque development. Simultaneously, smooth muscle cells (SMCs) from the tunica media migrate into the intima, where they proliferate and undergo phenotypic modulation. These transformed SMCs contribute to the formation of a fibrous cap over the lipid-rich core, a process essential for plaque stabilization. As the plaque enlarges, it can cause significant luminal narrowing, exceeding 50% of the arterial diameter or 75% of the cross-sectional area, potentially compromising perfusion and leading to clinical manifestations such as angina. 2 (Figure 1).

Progression of atherosclerosis and role of percutaneous coronary intervention (PCI). This figure illustrates the pathophysiological evolution of atherosclerosis, beginning with early-stage fatty streaks and progressing through the formation of atheromas and fibrous plaques, ultimately culminating in complicated lesions with potential for rupture. Advanced atherosclerotic disease is associated with significant clinical consequences, including myocardial infarction (MI), atrial fibrillation (AF), and left ventricular hypertrophy (LVH). The lower panel of the figure delineates the PCI procedure, encompassing balloon inflation, stent delivery, and deployment to restore coronary blood flow.
Percutaneous coronary intervention (PCI) is a revascularization procedure that typically involves the inflation of a balloon to reopen occluded coronary arteries, followed by the deployment of a stent to maintain vessel patency, particularly in patients with stable coronary artery disease (CAD) 3 (Figure 1). However, PCI introduces considerable thrombotic risk. The mechanical disruption of the vascular endothelium exposes subendothelial elements such as tissue factor and collagen, triggering platelet activation and initiating the extrinsic coagulation cascade.9,10 Balloon dilation and stent implantation further exacerbate these processes, often resulting in microvascular injury, distal plaque embolization, and potential obstruction of downstream vessels. These sequelae are closely associated with enhanced local thrombus formation and increased ischemic complications.10,11
Once deployed, the stent itself becomes a nidus for thrombosis. The exposed metal struts offer a thrombogenic surface for platelet adhesion and fibrin deposition, maintaining a prothrombotic milieu until complete endothelial coverage is achieved.10,11 However, endothelialization is a delayed process, particularly in drug-eluting stents (DES), which are coated with anti-proliferative agents to prevent restenosis. These agents inadvertently inhibit endothelial healing, thereby prolonging the thrombogenic period compared to bare-metal stents (BMS). 10
Beyond direct endothelial injury, PCI alters hemodynamic flow dynamics. The irregular geometry of stent struts disrupts laminar flow, creating areas of turbulence and low shear stress. Such conditions favor platelet aggregation and fibrin deposition. 11 These low-shear regions can trap circulating platelets and stabilize thrombi, heightening the risk of acute stent thrombosis. In cases where endothelialization remains incomplete, this risk extends over time, predisposing to late stent thrombosis—a rare but potentially fatal complication. 10
In AF, thrombus formation is driven primarily by blood stasis due to disorganized and ineffective atrial contractions. This stagnation, especially within the left atrial appendage (LAA), facilitates clot development. Additionally, AF induces endothelial dysfunction and promotes a pro-inflammatory, prothrombotic state. Together, these factors significantly elevate the risk of embolic stroke, reinforcing the pivotal role of anticoagulation in AF management. 10
In patients with AF undergoing PCI, a unique and complex dual thrombotic risk emerges. While the stented coronary artery presents a localized risk for thrombosis due to endothelial disruption and mechanical irritation, the hemodynamic abnormalities in AF predispose to atrial thrombus formation. This overlapping risk significantly amplifies the potential for stroke and systemic embolization, with thrombi potentially arising from either the coronary stent or the atrial chambers. Consequently, clinical management must navigate this dual pathway of thrombogenesis, requiring a carefully balanced approach to both stent thrombosis prevention and stroke risk reduction. 12
Dual antiplatelet therapy (DAPT)—typically combining aspirin with a P2Y12 inhibitor (eg, clopidogrel)—plays a crucial role in preventing thrombus formation at the stent site. DAT inhibits platelet aggregation and thromboxane A2 synthesis, thereby reducing the risk of both early and late thrombotic complications.13,14 In patients with AF, DAT is often used in conjunction with oral anticoagulants (OACs) during the highest-risk period, followed by de-escalation to monotherapy with anticoagulation as thrombotic risk decreases. This strategy reflects the need to address the dual thrombotic threats in individuals with coexisting CAD and AF. 12 Having established the dual thrombotic pathways in CAD with AF, we now turn to current guideline-based management strategies.
Current Guidelines for Management
Leading cardiovascular societies—including the American College of Cardiology (ACC), European Society of Cardiology (ESC), and American Heart Association (AHA)—advocate for personalized treatment strategies guided by validated risk stratification tools such as CHA2DS2-VASc (for stroke risk) and HAS-BLED (for bleeding risk).
Recommendations for Patients With AF, CAD, and/or PCI
For patients with AF undergoing PCI or presenting with acute coronary syndrome (ACS), current guidelines generally recommend dual antithrombotic therapy (DAT)—comprising a direct oral anticoagulant (DOAC) and a P2Y12 inhibitor (such as clopidogrel)—as the preferred strategy. This approach is favored over triple antithrombotic therapy (TAT), which includes aspirin, to mitigate bleeding risk without compromising ischemic protection.15 -18
The 2019 ESC guidelines recommend DAT over TAT for most patients with AF undergoing PCI. A short course of TAT—limited to 1 month—is advised only in patients with high thrombotic risk.18,19 This recommendation is supported by several large randomized controlled trials, including AUGUSTUS and PIONEER AF-PCI, which demonstrated that DAT incorporating DOACs significantly reduces bleeding compared with TAT, while maintaining comparable efficacy in preventing ischemic events.
However, nuances in trial design must be acknowledged. In PIONEER AF-PCI, rivaroxaban was administered at a reduced dose of 15 mg once daily in combination with clopidogrel—a deviation from the standard 20 mg dose approved for stroke prevention in AF. Importantly, the trial was not powered to assess thromboembolic outcomes as primary endpoints, limiting conclusions about comparative efficacy. Further contributing to clinical uncertainty, neither the ESC nor the AHA/ACC guidelines provide definitive recommendations on rivaroxaban dosing in this context.17 -20 (See Figure 2).

Comparative analysis of antithrombotic therapy strategies in clinical trials.
In patients with stable CAD and elevated CHA2DS2-VASc scores, anticoagulation alone may suffice, allowing clinicians to forgo antiplatelet therapy and thus minimize bleeding risk.15,16,19 This underscores the importance of integrating risk stratification tools such as CHA2DS2-VASc for stroke risk and HAS-BLED for bleeding risk to guide personalized treatment (See Table 1). PCI-specific bleeding risk tools that guide antiplatelet intensity and duration—PRECISE-DAPT, ARC-HBR, and CRUSADE—are summarized in Table 2.
Risk Stratification Tools Overview.

Bleeding Risk Assessment Tools for Patients Undergoing Antithrombotic Therapy.

Figure 2 presents a timeline overview of antithrombotic regimens assessed in major clinical trials, including WOEST, ISAR-TRIPLE, PIONEER AF-PCI, RE-DUAL PCI, and AUGUSTUS. It highlights key differences in therapeutic combinations, drug dosages, and treatment durations. These trials compared DOAC-based regimens (eg, rivaroxaban, dabigatran, apixaban) against traditional therapy with vitamin K antagonists (VKAs) and P2Y12 inhibitors, evaluating outcomes in terms of ischemic protection and bleeding risk in patients undergoing PCI or presenting with ACS.
Emerging evidence increasingly supports therapy simplification to reduce bleeding while maintaining antithrombotic efficacy. Recent trial data advocate for personalized treatment strategies over uniform protocols.18 -21
Patient-Specific Considerations
Antithrombotic management in patients with CAD and AF must be tailored to individual patient profiles. Decision-making should encompass not only clinical efficacy but also patient safety, quality of life, and treatment adherence. Simplified regimens that reduce bleeding risk are often associated with better long-term compliance and improved outcomes.
Age, Sex, and Comorbidities
Age is a crucial factor in therapy selection. Elderly patients are at elevated risk for both thrombotic and hemorrhagic complications, and DOACs have demonstrated a favorable safety profile, including lower rates of intracranial hemorrhage compared with warfarin.16,18 Sex-specific risk stratification is also essential; women with AF are at a higher risk of stroke and require careful assessment to ensure optimal anticoagulation. 18
Comorbidities such as diabetes mellitus, hypertension, and chronic heart failure further elevate thrombotic risk and complicate pharmacologic management due to potential drug-drug interactions and cumulative toxicity. 22 For instance, poorly controlled hypertension notably in-creases the risk of major bleeding during anticoagulant therapy. 18
Kidney and Hepatic Function Considerations
Renal and hepatic function critically influence the metabolism and safety profile of antithrombotic agents. In patients with chronic kidney disease (CKD), dose adjustments for direct oral anticoagulants (DOACs)—such as apixaban—are essential to prevent drug accumulation and mitigate bleeding risk.16,23 For individuals with severe renal impairment, warfarin may be a safer alternative, as it does not rely on renal excretion to the same extent as DOACs.
Similarly, hepatic dysfunction poses therapeutic challenges. Impaired liver function alters the synthesis of coagulation factors and affects the metabolism of various anticoagulants. Careful monitoring and therapeutic adjustments are crucial in these patients to maintain a delicate balance between thromboprophylaxis and bleeding prevention. 23
Patient Preference and Quality of Life
Antithrombotic therapy must be guided by shared decision-making that incorporates patient values, treatment preferences, and lifestyle factors.22,24 Patients reluctant to undergo frequent monitoring often favor DOACs over warfarin due to their fixed dosing and limited need for laboratory testing.
Moreover, simplified regimens that minimize side effects and preserve functional independence significantly improve quality of life. In high-risk patients, such as those with a history of bleeding, monotherapy with a single antiplatelet agent or reduced-dose anticoagulation may offer a better alignment with patient goals and therapeutic safety. 22
Type of Stent: BMS Versus DES Versus BVS
Bare-metal stents (BMS) and drug-eluting stents (DES) remain the 2 most common stent types in clinical practice. Among these, second-generation DES have demonstrated superior outcomes in reducing major adverse cardiovascular events (MACE), restenosis, and stent thrombosis, making them the preferred choice in most settings.
However, DES requires DAPT for 6 to 12 months, posing challenges for patients with AF who are also receiving oral anticoagulation. In such cases, BMS may be considered due to their shorter recommended DAT duration (typically 1 month). Despite this, the advanced design and safety profile of DES have led to their widespread use, even in complex patient populations.
A more recent innovation, the bioresorbable vascular scaffold (BVS), was developed to provide temporary vessel support before fully dissolving over time, with the goal of restoring natural vascular physiology and minimizing chronic inflammation and late stent thrombosis associated with permanent metallic implants. However, early clinical enthusiasm for BVS has been tempered by safety concerns.
Notably, the AIDA trial, one of the largest randomized studies comparing the Absorb BVS to everolimus-eluting stents, found a higher incidence of device thrombosis and target lesion failure with BVS over a 5-year follow-up period. These findings have been supported by subsequent meta-analyses and observational studies, leading to a substantial decline in the routine clinical use of first-generation BVS platforms. In response, newer-generation BVS devices are being redesigned to incorporate thinner struts and enhanced resorption kinetics, aimed at improving endothelialization and reducing thrombotic complications. Until robust long-term safety data become available, the use of BVS remains largely confined to clinical trials or highly selected patients with specific anatomical or clinical considerations.25,26
Figure 3 outlines key factors influencing antithrombotic therapy: patient demographics, comorbid conditions, pharmacologic interactions, and procedural characteristics. Treatment decisions aim to balance ischemic protection and bleeding risk. High ischemic risk may warrant extended DAT or P2Y12 inhibitor monotherapy, while patients at elevated bleeding risk may benefit from shortened DAT or early transition to single-agent therapy.25,26 With the pathophysiology and guidelines in view, the next key debate is whether dual therapy suffices, or triple therapy remains necessary

Patient-centered decision-making in antithrombotic therapy.
Triple Therapy Versus Dual Therapy: The Debate
Treating CAD patients undergoing PCI with AF presents a significant therapeutic challenge. The clinician must navigate the dual goals of stroke prevention in AF and coronary event management post-PCI. The controversy centers on whether TAT—typically combining a vitamin K antagonist (VKA) or DOAC with aspirin and a P2Y12 inhibitor—is superior to dual therapy (DAT) in achieving these goals.
Historically, warfarin (a VKA) was the standard anticoagulant in this setting, offering a 66% relative risk reduction in stroke. However, warfarin’s clinical utility is hampered by narrow therapeutic margins, extensive food-drug interactions, and frequent INR monitoring requirements. 1
Recent trials have established the superior safety profile and comparable efficacy of DOACs—including dabigatran, rivaroxaban, and apixaban—compared to warfarin, particularly in reducing major bleeding events. As such, current guidelines recommend DOACs as the first-line therapy in AF patients undergoing PCI. Yet, optimal drug selection and dosing remain critical, as over- or underdosing may result in thromboembolic events or major hemorrhage. 2
Real-World Underdosing of DOACs
Despite clear guideline recommendations, real-world data reveal a troubling trend of inappropriate underdosing of DOACs during combined antithrombotic therapy. In a comprehensive nationwide cohort study by Chan et al, elderly patients with AF and high bleeding risk were frequently prescribed subtherapeutic doses, such as apixaban 2.5 mg/day, or rivaroxaban 10 mg/day, in attempts to minimize bleeding. 27 However, this strategy was associated with poorer clinical outcomes, including increased venous thrombosis and all-cause mortality, underscoring the dangers of deviating from evidence-based dosing protocols.
Supporting these findings, the POL-AF Registry, a prospective observational study of nearly 4000 AF patients across 10 cardiology centers in Poland, revealed inappropriate underdosing in 71% of rivaroxaban users and 67% of apixaban users, often without clear clinical justification such as advanced age or renal impairment. By contrast, dabigatran was correctly dosed in 100% of cases, suggesting more consistent adherence to label recommendations for this agent. These patterns underscore clinicians’ cautious approach to bleeding risk, but simultaneously highlight the danger of inadequate thromboembolic protection. 28
The same registry reported that among 359 AF patients who underwent PCI, 56% continued to receive TAT, despite guideline preference for DAT. Furthermore, in the subgroup managed with DOAC-based combinations, only 29% of rivaroxaban and 33% of apixaban prescriptions followed recommended dose reduction guidelines, again contrasting with full compliance in dabigatran prescribing. This dual pattern—frequent use of intensified TAT regimens and widespread DOAC underdosing—likely reflects clinician concerns about hemorrhagic complications, but paradoxically may compromise stroke and stent thrombosis prevention. 28
One of the tools central to risk stratification in AF patients is the CHA2DS2-VASc score, which guides anticoagulation decisions by quantifying stroke risk relative to therapeutic benefit. 29 Traditionally, TAT has been the standard in AF patients undergoing PCI, aimed at reducing thromboembolic events such as stent thrombosis, myocardial infarction (MI), and ischemic stroke. 30 The combination of anticoagulants and antiplatelet agents provides comprehensive thromboprophylaxis, particularly in the early post-PCI phase. However, TAT is consistently associated with a substantially elevated bleeding risk, including major, nonfatal, and potentially fatal hemorrhagic events.
In response, DAT has emerged as a safer alternative, particularly in patients at elevated bleeding risk. While aspirin, a cyclooxygenase-1 (COX-1) inhibitor, offers partial platelet inhibition, its limitations are addressed by the addition of P2Y12 receptor antagonists, which block the adenosine diphosphate (ADP)-mediated pathway. New-generation agents such as ticagrelor and prasugrel have demonstrated superior platelet inhibition and clinical outcomes in large-scale trials compared to older agents like ticlopidine.
Although DAT improves compliance, simplifies regimens, and reduces hemorrhagic events, it may not provide sufficient protection in patients at very high risk for recurrent MI or stent thrombosis. Therefore, while dual therapy represents a pivotal advance in managing AF-PCI patients, its use must be carefully tailored, reserving TAT for select high-risk cases (Figure 4). 31

Balancing thrombosis and bleeding risks in antithrombotic therapy for PCI patients with atrial fibrillation. The interplay of Virchow’s triad components (A) in atrial fibrillation and PCI highlights the need for careful management with anticoagulants and antiplatelet agents (B). Triple therapy increases thrombosis risk but poses a high bleeding risk, while dual therapy may mitigate bleeding but insufficiently prevent thrombosis, requiring a delicate balance between these risks (C).
Reversal Agents and Future Anticoagulants
In clinical scenarios involving life-threatening bleeding or the need for urgent surgical intervention, the availability of reversal agents for DOACs has significantly improved treatment safety.
Idarucizumab, a humanized monoclonal antibody fragment, is a specific reversal agent for dabigatran. It binds dabigatran with high affinity and neutralizes its anticoagulant effect within minutes. Approval was granted following the RE-VERSE AD trial, which demonstrated its efficacy in emergencies. Idarucizumab has a short half-life of ~45 minutes, although this may be prolonged in patients with renal impairment.32,33
Andexanet alfa, approved for the reversal of rivaroxaban and apixaban, acts as a decoy recombinant factor Xa protein, sequestering factor Xa inhibitors and restoring coagulation. Despite its clinical utility, its use is often restricted due to limited availability and high cost, particularly in resource-constrained settings.34,35
In parallel, research into next-generation anticoagulants focuses on agents with reduced bleeding risk. Notably, factor XIa inhibitors such as asundexian and milvexian have shown promise in early-phase trials. 36 These agents target the intrinsic pathway of coagulation, minimally involved in hemostasis but relevant in pathological thrombosis, potentially offering effective antithrombotic activity with lower hemorrhagic risk. However, head-to-head trials against existing anticoagulants are necessary to validate efficacy and safety in broader populations. 37
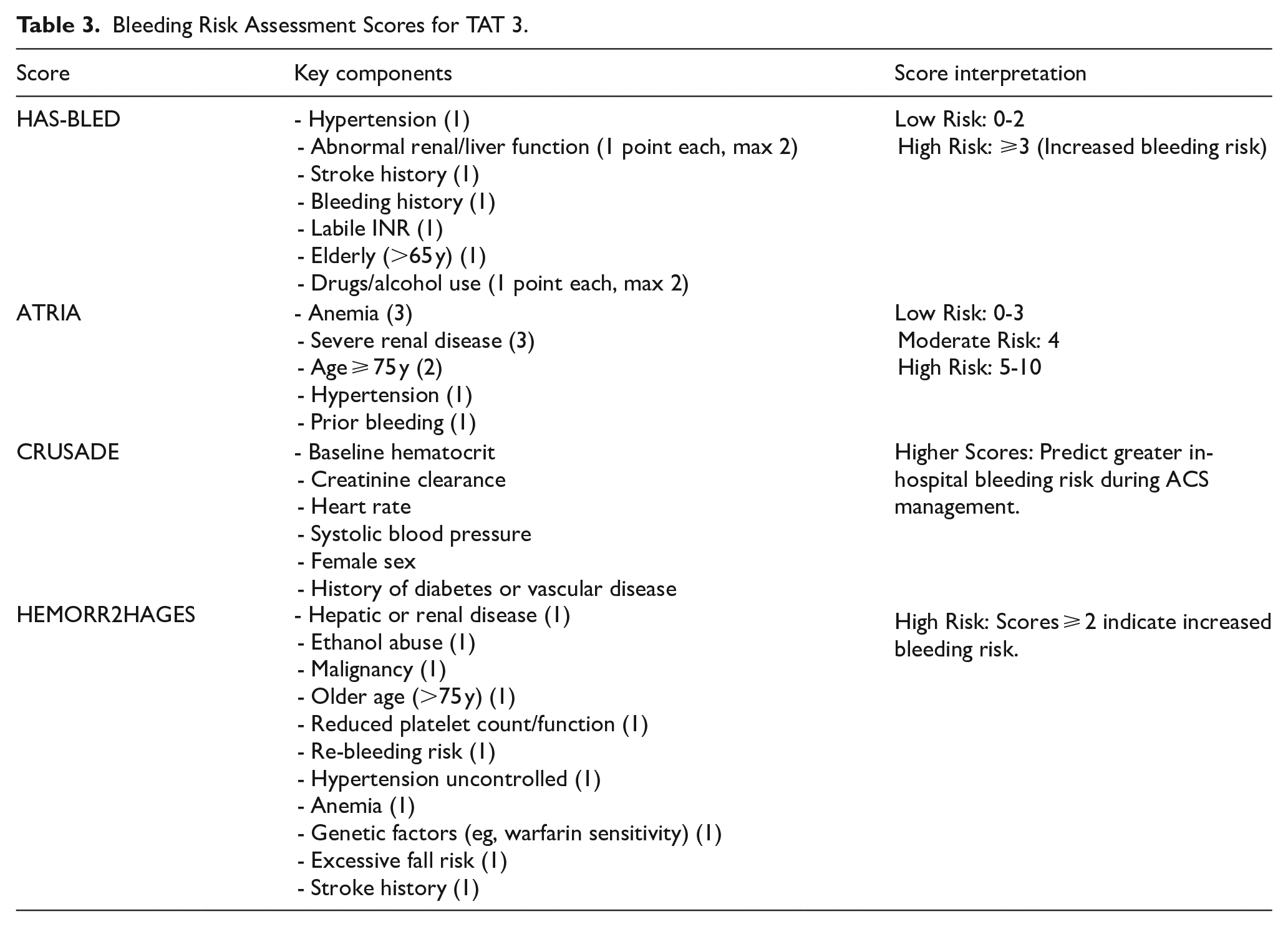
For patients with high bleeding risk or with comorbidities such as chronic kidney disease, advanced age, or concurrent antithrombotic use, DAT poses limitations. Tools such as the HAS-BLED, ATRIA, CRUSADE, and HEMORR2HAGES scores are critical for bleeding risk assessment. These tools provide a structured framework for adjusting therapy duration, intensity, and monitoring protocols in patients on TAT or DAT.29,38,39 (See Table 3). Common clinical scenarios with suggested strategies and monitoring are summarized in Table 4.
Bleeding Risk Assessment Scores for TAT 3.
Clinical Applications of Risk Stratification in Antithrombotic Therapy.

Major Clinical Trials, Controversies, and Unresolved Questions
The choice between DAT and TAT remains a significant therapeutic challenge in patients with AF undergoing PCI. This decision impacts both ischemic event prevention and bleeding risk, necessitating a careful, evidence-informed approach.
TAT Versus DAT: Safety and Efficacy in Trials
Several randomized controlled trials and subsequent meta-analyses have explored this balance17,40 -42:
Gargiulo et al concluded that DAT is associated with lower bleeding risks, including intracranial hemorrhage, though it may cause a non-significant rise in ischemic events. 43
Assaf et al supported DAT as it reduced major bleeding without increasing the risk of stent thrombosis (ST) or myocardial infarction (MI). 44
Ravi et al observed no significant differences in mortality, non-fatal MI, stroke, or ST between DAT and TAT, but emphasized that DAT had a lower bleeding risk, making it preferable in patients at high hemorrhagic risk. 45
Other meta-analyses46 -50 confirmed that DAT offers comparable protection against thrombotic outcomes and a significant reduction in major bleeding, bolstering its role as the default in many patients.
Yet, some studies urge caution:
Cappello et al found that DAT, while reducing bleeding, was linked to a higher risk of stent thrombosis and MI compared to TAT. 51
Golwala et al similarly noted that the benefit in bleeding reduction from DAT may be counterbalanced by increased thrombotic events. 47
Andò and Costa advocated for an initial short course of TAT followed by DAT, particularly in high ischemic-risk patients. 52
Figure 5. Antithrombotic Therapy Strategies for AF Patients Undergoing PCI. This timeline depicts antithrombotic therapy strategies in AF patients undergoing PCI, differentiated by risk profile:
TAT (OAC + aspirin + P2Y12 inhibitor) is used initially.
Transition to DAT or OAC monotherapy is based on patient-specific ischemic and bleeding risks, optimizing protection over 12 months.
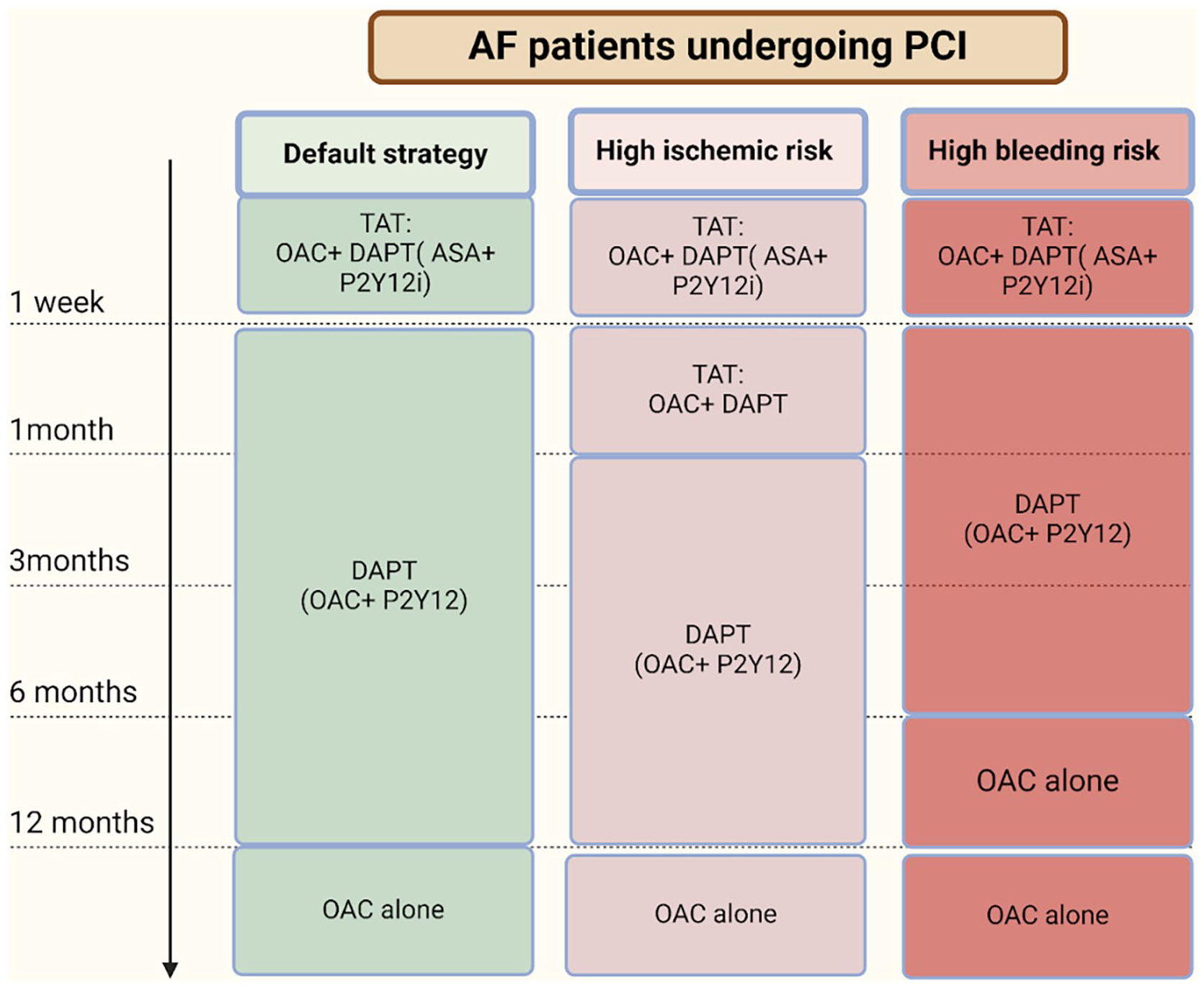
Antithrombotic therapy strategies for AF patients undergoing PCI.
Which DAT Regimen is Best?
The debate extends beyond the DAT versus TAT paradigm to the choice of specific DAT combinations:
In a network meta-analysis, Kuno et al compared 9 antithrombotic strategies, finding that the VKA + P2Y12 inhibitor regimen resulted in significantly higher bleeding than most DOAC-based DAT options. Among the latter, apixaban + P2Y12 inhibitor was associated with the lowest bleeding risk and no increase in ischemic events. 53
Liang et al found similar outcomes, ranking apixaban-based DAT regimens as safest while VKA + P2Y12 retained high efficacy, especially for preventing ischemic complications. 54
Golwala et al also favored DOAC + P2Y12 combinations over VKA-based approaches, underscoring both improved safety and comparable efficacy. 47
Altoukhi et al. further refined the assessment, ranking:
Apixaban highest for reducing ISTH major/clinically relevant bleeding and stroke,
Rivaroxaban as most effective in minimizing MI and ST, and
Dabigatran as optimal for reducing all-cause mortality. 55
Meta-analyses by Eyileten and Shen et al consistently showed that DOAC-based regimens—whether as part of DAT or TAT—offer superior bleeding profiles and maintain effective thromboembolic protection compared to VKA-based strategies.56,57 Observational data from Park et al in Korea corroborated these results, demonstrating favorable ischemic and bleeding outcomes for DOAC-based DAT and TAT over VKA-TAT at 3 months post-PCI. 58
Optimal DAT Strategy: Duration, De-escalation, and Customization
Kuno et al 59 conducted a systematic review and network meta-analysis exploring multiple DAT strategies:
Standard DAT (12 months),
Extended DAT (⩾18 months),
Short-term DAT (⩽6 months) followed by P2Y12 monotherapy or aspirin,
Unguided de-escalation (from potent to lower-dose or clopidogrel after 1 month),
Guided selection via genetic/platelet function testing.
They found:
Unguided de-escalation yielded the lowest MACE and bleeding rates,
Short DAT followed by P2Y12 inhibitor showed the lowest all-cause death and major bleeding rates.
Meanwhile, the optimal duration of TAT remains uncertain. In a network meta-analysis, Wang et al 60 evaluated 4 durations of TAT and found no significant differences in MACE, stroke, MI, or mortality across durations. However, short-term TAT was associated with a lower bleeding risk, supporting its use as a default, with longer TAT reserved for those with elevated ischemic risk.
Real-World Versus Clinical Trial Discrepancies
Despite robust evidence from RCTs, real-world outcomes remain variable:
ORBIT-AF and GARFIELD-AF registries offer insights into broader populations, reflecting real-life clinical diversity.61,62
Acconcia et al raised concerns that RCT results may not always mirror real-world outcomes, noting that observational studies have found higher stent thrombosis rates with DAT than predicted, challenging assumptions of universal DAT superiority. 63
This schematic illustrates recommended therapy strategies over a 12-month timeline, adapted to patient-specific ischemic and bleeding risks:
Initial TAT (OAC + aspirin + P2Y12 inhibitor) in high-risk cases,
Transition to DAT (OAC + P2Y12 inhibitor),
De-escalation to OAC monotherapy, guided by individual risk profiles.
Emerging Therapies and Future Directions
Advances in Stent Technology and Its Impact on Therapy Duration
Innovations in stent design have significantly influenced the management of patients with CAD and AF undergoing PCI. Second-generation DES, engineered with improved biocompatibility and polymer biodegradability, have demonstrated markedly lower rates of stent thrombosis and restenosis. 64 As a result, the recommended duration of DAT following PCI has been shortened, particularly beneficial in AF patients who require concurrent oral anticoagulation, thereby mitigating bleeding risks. 65
Emerging technologies such as BVS are designed to provide temporary mechanical support, eventually dissolving and restoring natural vessel function. Although initial results were promising, first-generation devices raised safety concerns, including increased thrombosis rates. Nevertheless, newer BVS platforms are being redesigned to improve strut thickness and resorption kinetics, offering potential for safer future use. 66
Personalized Medicine in Tailoring Antithrombotic Therapy
Given the heterogeneity of risk in patients with concurrent AF and CAD, the paradigm is shifting toward personalized medicine. The incorporation of clinical risk scores such as CHA2DS2-VASc and HAS-BLED enables stratification of thromboembolic and bleeding risks. 67 Moreover, pharmacogenomic testing, especially of CYP2C19 polymorphisms influencing clopidogrel metabolism, helps optimize antiplatelet selection and dosing. 67
Advanced artificial intelligence (AI) and machine learning (ML) algorithms now allow integration of complex patient-specific variables, including demographics, lab values, imaging, and comorbidities, to guide real-time therapeutic decisions and dynamically adjust treatment plans. 68
Biomarkers Predicting the Effectiveness of Treatment
Biomarkers offer additional layers of prognostic insight in the management of AF with CAD. Elevated cardiac troponin levels, beyond indicating myocardial injury, are independently associated with increased stroke and cardiovascular mortality in AF and enhance predictive accuracy when added to CHA2DS2-VASc scoring. 69
Similarly, B-type natriuretic peptide (BNP) and NT-proBNP, markers of atrial stress and wall tension, correlate with thromboembolic risk and inform anticoagulation strategies. 70 Soluble ST2, linked to cardiac fibrosis, has shown value in heart failure but adds little to AF-specific risk stratification. 71
Other promising biomarkers include:
D-dimer, elevated in thrombotic states and associated with adverse outcomes in trials like RE-LY and ARISTOTLE, 69
Platelet reactivity testing, aiding in customizing antiplatelet regimens,
Inflammatory markers, notably high-sensitivity C-reactive protein (CRP) and interleukin-6 (IL-6). Elevated IL-6 predicts stroke and cardiovascular death, while CRP correlates with reduced sinus rhythm maintenance and higher mortality.69,72
Future Directions in Research and Innovation
The therapeutic landscape for managing AF and CAD is advancing rapidly, driven by novel agents and technologies. Factor XIa inhibitors (eg, asundexian, milvexian) represent a promising class of anticoagulants with reduced bleeding potential due to their limited involvement in physiological hemostasis. 73
Nanotechnology-based drug delivery systems are being developed to localize antithrombotic effects directly to sites of vascular injury, minimizing systemic exposure and bleeding risk. 74 Additionally, wearable biosensors capable of monitoring arrhythmias and coagulation parameters in real-time are poised to revolutionize patient monitoring and therapy titration.
Ongoing clinical trials evaluating abbreviated DAT regimens are expected to further refine practice guidelines. 75 In parallel, shared decision-making—incorporating patient preferences, lifestyle considerations, and treatment burdens—remains a cornerstone for optimizing therapy adherence and overall outcomes.
Conclusion
The management of CAD in patients with AF undergoing PCI remains a complex and evolving clinical scenario. Balancing the prevention of thromboembolic events—including stent thrombosis and embolic stroke—against the risk of major bleeding requires an individualized, evidence-based approach.
In many cases, DAT—typically combining a DOAC with a P2Y12 inhibitor—has emerged as a safer and effective alternative to TAT, especially in patients at high bleeding risk. Concurrently, technological advancements in stent design, particularly with second-generation DES, have reduced restenosis and thrombosis rates, permitting shorter durations of antiplatelet therapy without compromising safety.
The integration of personalized medicine tools, including risk scores (CHA2DS2-VASc, HAS-BLED), pharmacogenomics, and biomarkers, continues to improve the precision and safety of therapy. Nevertheless, clinical controversies persist, especially around therapy duration, optimal DAT composition, and real-world versus trial outcomes. These knowledge gaps highlight the importance of ongoing research, real-world data collection, and emerging therapies to refine and personalize clinical practice further.
Ultimately, the key to success lies in harmonizing clinical evidence with patient-centered care, ensuring that treatment decisions reflect both scientific rigor and individual patient values to achieve the best possible outcomes.
Footnotes
Ethical Considerations
Ethical approval is not required for this review article.
Author Contributions
O.A. and B.E. conceptualized the study; F.M., Q.Z.A., A.M.D., and M.M.A. developed the review methodology and search strategy; F.M., Q.Z.A., H.N., A.O., O.H., S.S., A.Y., and S.H. conducted literature screening/selection and evidence synthesis; A.M.D., M.M.A., and M.B. drafted the initial manuscript; S.S., M.B., K.B.H., and A.I.M.A.Z. prepared tables/figures; A.Y. provided critical revision; H.N., A.S., A.O., S.S., O.H., A.M., K.B.H., A.I.M.A.Z., and S.H. reviewed and approved the final version; A.S. and M.B. supervised the work and revised the manuscript after peer review.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
This study did not involve the creation or analysis of any new data. Therefore, data sharing is not applicable.
Provenance and Peer Review
Not commissioned; externally peer reviewed.
