Abstract
A 50-year-old male had intermittent episodes of dyspnea for 7 days with persistent atrial fibrillation (AF) for 2 years. New York Heart Association (NYHA) functional class is IV status. The echocardiographic evaluation (Philps EPIQ 7C, S5-1 probe, 1.0-5.0 MHz) was performed showing a left ventricular ejection fraction (LVEF) of 48% (measured by the Simpson biplane method). His-Purkinje conduction system pacing (HPCSP) may enhance electrical resynchronization as well as the clinical and echocardiographic outcomes of patients with advanced heart failure (HF) who require resynchronization therapy (CRT). The use of left anterior fascicular areas pacing/left posterior fascicular areas pacing (LAFAP/LPFAP) may offer a significant advantage in patients for whom both biventricular pacing (BVP) and His bundle pacing (HBP) have not delivered substantial therapeutic benefits. In this study, we optimized electrical resynchronization by combining the concept of fused simultaneous pacing of LAFA/LPFA with an atrioventricular node ablation (AVNA) in a patient with HF and atrial fibrillation (AF). The maximal electrical resynchronization presented better hemodynamic and demonstrated comparable clinical and echocardiographic responses.
Keywords
Introduction
The most prevalent form of arrhythmia in individuals with heart failure (HF) is atrial fibrillation (AF). His-Purkinje conduction system pacing (HPCSP) is a viable substitute for biventricular pacing (BVP). 1 The feasibility and effectiveness of HPCSP combined with AVNA in patients with HF and AF over medium-term follow-up have been investigated in several recent studies. 2 Patients who received both HPSCP and AVNA showed significant improvements in left ventricular (LV) function and better outcomes. Patients receiving left bundle branch area pacing (LBBAP) also showed lower pacing thresholds and improved sensing relative to those treated with HBP. 3 As pacing at a more distal point within the conduction system may lessen the chance of a threshold rise during ablation, LBBAP may be preferable to HBP in AVNA. Simultaneous left anterior fascicular area pacing (LAFAP) and left posterior fascicular area pacing (LPFAP) may be a potential bail-out option when either HBP or LBBAP cannot be achieved. This study reported the performance of simultaneous pacing of LAFA/LPFA followed by AVNA on AF in an HF patient.
Case report
A 50-year-old male was admitted to hospital with intermittent episodes of dyspnea for 7 days and persistent AF for 2 years. The patient was classified as class IV according to the NYHA functional classification. A Holter ECG at admission showed 5394 premature ventricular beats, maximal heart ventricular rate of 209 bpm, mean heart ventricular rate of 119 bpm, AF with intraventricular conduction disorder, baseline QRS duration of 112 ms, and baseline blood pressure of 110/85 mmHg. Chest CT at admission showed bilateral pulmonary edema, bilateral pleural effusion, cardiac enlargement. Transthoracic echocardiography (Philps EPIQ 7C, S5-1 probe, 1.0-5.0 MHz) showed an LVEF (measured by the Simpson biplane method) of 48%. The LV end-diastolic, left atrial (LA), and right atrial (RA) diameters were 62.9 mm, 52.6 mm, and 52.8 mm, respectively. He was diagnosed with dilated cardiomyopathy, AF, and HF with mildly reduced ejection fraction (HFmrEF). The patient had received optimal medical therapy of HF, such as drugs to reduce cardiac preload: intravenous nitroprusside (25 mg), furosemide (20 mg), and oral hydrochlorothiazide (50 mg). Cardiotonic: intravenous milrinone (10 mg), cedilan (0.2 mg), and oral digoxin (0.125 mg). The ‘new quadruple’ treatment of HF: metoprolol (47.5 mg), benazepril (10 mg), dapagliflozin (10 mg), and spironolactone (20 mg). The patient was treated with rate controlling drugs and anticoagulant drugs, including metoprolol (47.5 mg), digoxin (0.125 mg) and rivaroxaban (20 mg), but the dyspnea symptoms did not resolve, ventricular rate was not controlled after treatment with β-blockers and digoxin, the maximal heart ventricular rate of 199 bpm, mean heart ventricular rate of 110 bpm, The patient had no history of cardiovascular risk factors (hypertension, diabetes and smoking) and infection associated myocarditis. The patient did not have clinical symptoms of persistent angina. Laboratory assessments revealed 1438 pg/mL N-terminal pro-brain natriuretic peptide (NT-proBNP) level at admission. Therefore, permanent pacemaker implantation with conduction system pacing and atrioventricular node ablation (AVNA) were planned.
A C315 His sheath (Medtronic, Minneapolis, USA) was inserted into the tricuspid annular area using a guidewire. Contrast was then injected through the sheath in the right anterior oblique view (RAO) at a 30° angle. A 12-lead electrocardiogram (ECG) and intracardiac electrocardiograms (EGMs) were recorded continuously using an electrophysiology (EP) system (Boston Scientific Corporation, USA). The 3830-69 electrode was successfully implanted with the aid of a C315-His sheath. The potential of the His bundle was first determined by positioning the tip of the pacing lead at the location of the His bundle. The His-ventricular (HV) interval at this site was 52 ms (Figure 1a). The sheath and lead were positioned 1 cm toward the right ventricular (RV) apex using the fluoroscopic HBP location as a guide. The 3830-69 electrode was implanted at the LAFA region deep within the interventricular septum (Figure 1b). A sharp LBB potential was recorded at the location with LBB-V interval of 30 ms. During pacing at an output of 5 V at 0.5 ms, unipolar pacing displayed Qr in lead V1, a peak left ventricular activation time (PLVAT) of 82 ms in lead V5, and a QRS duration of 144 ms. Unipolar pacing showed QS in lead V1, a PLVAT of 98 ms in lead V5, and a QRS duration of 170 ms at 2.5 V @ 0.5 ms (Figure 2c). At this site, the R-wave amplitude was 10.7 mV, the pacing impedance was 920 Ω, and the pacing threshold was 0.4 V @ 0.5 ms. The other Medtronic 3830-69 pacing lead was then implanted into the LPF region for left ventricular bifascicular pacing. The lead-tip EGM displayed a sharp fascicular potential preceding the local ventricular signal with the potential to surface QRS duration of 26 ms. During pacing at an output of 5 V at 0.5 ms, a PLVAT of 102 ms was in lead V5, and the QRS duration was 160 ms. Unipolar pacing displayed QS in lead V1, a PLVAT of 102 ms in lead V5, and a QRS duration of 156 ms at 2.5 V @ 0.5 ms. A left anterior hemiblock pattern in the surface ECG was recorded (Figure 2d). The sensed R-wave was 5.6 mV, the threshold of LPFA pacing was 0.4 V@ 0.5 ms, and the pacing impedance was 873 Ω. Simultaneously LAFA/LPFA unipolar pacing at the final sites revealed a paced QRS duration of 148 ms and a PLVAT of 90 ms in lead V5. Bipolar pacing revealed a paced QRS duration of 122 ms and a PLVAT of 82 ms in lead V5 (Figure 2e). Next, the Select Secure 3830-59 pacing lead’s right atrial electrode was implanted by C304-S59 sheath (Medtronic, Minneapolis, USA). The RA, LV, and RV ports were individually wired to the right atrial (RA), LAFA, and LPFA electrodes. Finally, the Medtronic C2TR01 was successfully inserted. During AVNA, a long sheath (SR0) (Abbott, Plymouth, MN, USA) was employed to ensure enough stability. An irrigated ablation catheter is typically utilized for AVNA during ablation. Complete AV block was achieved using AVNA without affecting LBBP parameters (Figure 2f).
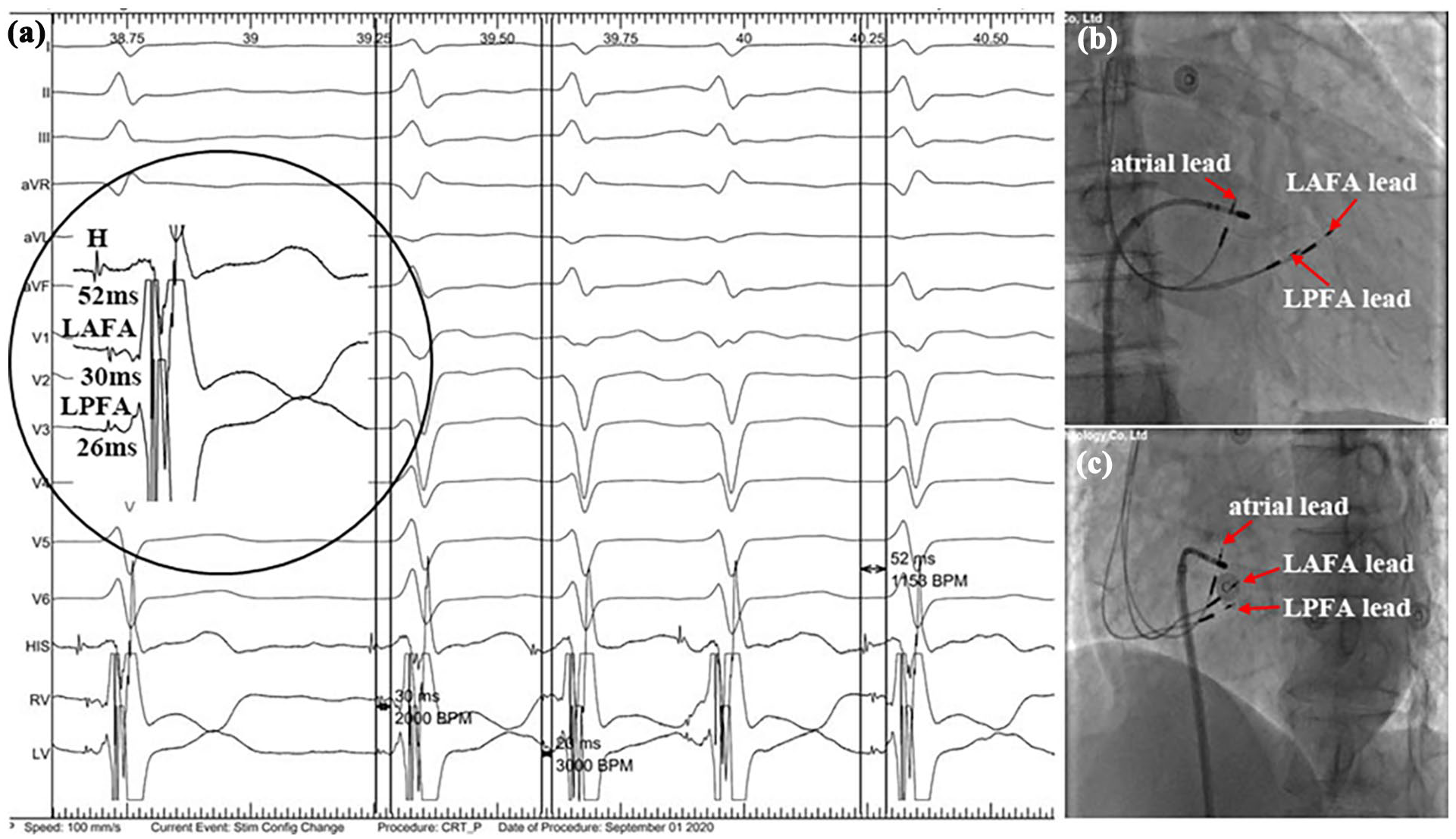
The 12-lead ECG, intracardiac electrogram, and fluoroscopic images of the patient (a) Baseline 12-lead ECG showed AF rhythm. The inset showed the His potential and sharp fascicular potentials. (b) RAO 30° fluoroscopic view showed the actual placements of the LAFA and LPFA leads in the anterior and posterior fascicle areas. (c) Fluoroscopic imaging of the final atrial pacing lead, LAFA and LPFA pacing leads (red arrows), and the ablation catheter positions in LAO 45° view.

Intracardiac electrogram of simultaneous pacing of LAFA/LPFA combined with AVNA (a) The native ECG. (b) His pacing with 5 V/0.5 ms showed QRS morphology, the SPLVAT of 106 ms in lead V5, and a QRS duration of 166 ms. (c) The SPLVAT of LAFA pacing was shortened from 98 ms to 82 ms with increased output from 2.5 V/0.5 ms to 5.0 V/0.5 ms. This demonstrated that septal was captured with 2.5 V/0.5 ms and septal and LAF were captured with 5 V/ 0.5 ms. (d) The SPLVAT of LPFA pacing did not differ between high and low voltages. The ECG morphology of paced QRS by LPFA pacing was left anterior branch fascicular block. LPF was captured and non-selective LBBP (NS-LBBP) and selective LBBP (S-LBBP) were observed at 2.5 V/0.5 ms and 5 V/0.5 ms in unipolar. The postoperative output voltage is 3.5 V, which would be LAFA pacing and LPFA pacing. (e) The ECG of simultaneous pacing of LAFA / LPFA with unipolar pacing. (f) The ECG after AVNA demonstrated complete AVB with simultaneous bipolar pacing of LAFA / LPFA.
The pacing parameters were steady during the follow-up appointment after 3 months. The measured R-wave was 6.6 mV, the RV pacing threshold was 0.5 V @ 0.5 ms, and the pacing impedance was 570 Ω. The LV pacing threshold was 0.25 V @ 0.5 ms, and the pacing impedance was 361 Ω. On follow-up, the NYHA functional class IV status has significantly improved to the class I position. The patient’s capacity for daily activity had increased noticeably. The patient’s subsequent echocardiographic results are shown in Figure 3. The transthoracic echocardiography (Philps EPIQ 7C, S5-1 probe, 1.0-5.0 MHz) supported the deep septal placements of the LBB pacing leads. Three months after the operation, the LVEF (measured by the Simpson biplane method) had increased to 59% (up from 48% before the treatment). The postoperative reassessment revealed 485 pg/mL NT-proBNP level, indicating a significant decrease. The follow-up LV end-diastolic dimensions had also significantly decreased relative to the baseline (62.9 mm to 54.1 mm) (Figure 3). The postoperative Holter ECG after 1 month showed 2 premature ventricular beats, maximal heart ventricular rate of 84 bpm, mean heart ventricular rate of 73 bpm, pacemaker rhythm and persistent AF of 24 h. We evaluated LV synchrony under simultaneous pacing of LAFA/LPFA using speckle-tracking echocardiography (STE) in the patient. During simultaneous pacing of LAFA/LPFA, STE showed no evident worsening of LV global longitudinal strain (Figure 3).

Postoperative speckle-tracking echocardiography of simultaneous pacing of LAFA/LPFA (a and b) Echocardiographic images revealed the deep septal location of the LAFA and LPFA pacing leads in the right ventricular (RV) cavity with long-axis and short-axis echocardiographic views. (c and d) Speckle-tracking echocardiography long-axis and short-axis echocardiographic views (bull’s eye plots display time to peak longitudinal strain) showed that simultaneous left anterior fascicular areas pacing/left posterior fascicular areas pacing (LAFAP/LPFAP) did not create left ventricular desynchronization.
Discussion and Conclusion
Patients with AF and HF on the best guideline-directed medical therapy (GDMT), Simultaneous LAFAP/LPFAP with AVNA may be a potential bail-out option when either HBP or LBBAP cannot be achieved. Here, we optimized electrical resynchronization by combining the concept of fused simultaneous pacing of LAFA/LPFA with an AVNA in the patient with AF and cardiac insufficiency (NYHA functional class IV). This optimal dual-site activation potentially lowers pacing thresholds and extends device longevity while maintaining LV systolic function, improving the symptoms of patients.
The 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure recommends that if a rhythm control strategy fails or is undesired, and ventricular rates remain rapid despite medical therapy after all other options are exhausted, atrioventricular nodal ablation with implantation of a CRT device can be considered as a treatment option. 4 The treatment of ventricular rate control is the vital cornerstone in dealing with AF. Ventricular rate control is inseparable from the treatment of regular rate controlling drugs and AV node ablation combined with physiological pacing. The strict ventricular rate control (a resting ventricular rate of less than 80 beats per minute and a moderate activity ventricular rate of less than 110 beats per minute) as the initial treatment goal has been shown to effectively prevent and treat tachycardia-induced cardiomyopathy, reduce the mortality of AF patients with HF. His Bundle Pacing (HBP) can reverse ventricular remodeling, restore ventricular electromechanical synchronization and cardiac function by utilizing the normal conduction system, and provides the most physiological pacing. It is not only suitable for traditional pacing indications but also can be used in the treatment of patients with AF complicated by cardiac dysfunction or complete LBBB requiring resynchronization therapy. However, compared with the extremely small target provided by the His bundle, the LBB offers a bigger target for physiologic pacing. LBBP significantly improved left ventricular ejection fraction, similar to HBP, but it had a higher implantation success rate, better pacing parameters, and fewer long-term lead-related complications. LBBP is technically easier to perform AVNA and usually does not require the implantation of an additional spare lead. 5 Additionally, pacing at a more distal site within the conduction system may theoretically lower the chance of a threshold rise during ablation, making LBBP preferable to HBP in the context of AVNA. Therefore, LBBP is feasible, safe, and effective in AF patients with HF following AVNA. Pacing at different sites of the LBB resulted in similar intraventricular and interventricular synchrony in AF patients with HF. Notably, LAFA/LPFA pacing demonstrated preserved left ventricular global longitudinal strain (LV-GLS) in recent speckle-tracking echocardiography studies (mean ΔGLS = −2.1 ± 1.8% vs −5.3 ± 2.3% in conventional pacing). 6 In conclusion, compared with LAFA or LPFA pacing, simultaneous LAFA/LPFA pacing are more similar to physiological pacing (similar to LBB trunk pacing) in patients who the His or LBB trunk cannot be captured. In addition, simultaneous LAFA/LPFA pacing may improve interventricular synchrony, have the lower difficulty and risk than CRT. Fusion optimization using LAFA/LPFA pacing with AVNA may achieve synchronous interventricular pacing theoretically due to concurrent capture of RV and LV myocardium during bipolar pacing. However, there is a lack of prospective evidence evaluating LBBP as a pacing technique in patients with AF and HF following AVNA.
In this case, simultaneous LAFA/LPFA pacing was selected as the therapeutic strategy due to the patient’s severe intraventricular conduction delay, AF, and refractory symptoms to medical therapy. Patients with severe cardiac insufficiency, significant intraventricular conduction delay, poor response to traditional treatment, combined with AF may be more suitable for simultaneous LAFA/LPFA pacing. Further validation through randomized trials (DUAL-PACE study, NCT05234567) is warranted to establish definitive indications. Further studies must be performed to determine optimal patients’ selection, suitable for simultaneous LAFA/LPFA pacing, effects and clinical outcomes, intended role in this group of patients specifically, we expect better and longer-term follow-up results.
Limitations
Firstly, the representativeness of patients was limited, and a single case may not represent the heterogeneous population of AF patients with HF. Different patients may have different responses to simultaneous LAFA/LPFA pacing. The results based on a single case may not be generalizable, limiting the statistical reliability and generalizability of the study results. Secondly, this case report lack an appropriate control group, making it difficult to determine whether the observed improvement in heart function was attributed to the treatment plan adopted, difficult to compare the effects of different treatment, or other factors such as natural course, medication adjustments. Finally, the follow-up time for patients was short, and detailed follow-up data and long-term prognosis information were not provided, making it difficult to evaluate the long-term effectiveness and safety of simultaneous LAFA/LPFA pacing, while simultaneous LAFA/LPFA pacing demonstrated preserved left ventricular global longitudinal strain, didnot have confirmed clinical endpoints like reduced HF hospitalization.
Footnotes
Acknowledgements
We would like to thank the patient and their family for consenting to the construction of this case report.
Ethics Considerations
Written informed consent was obtained from the individual (s) for the publication of any potentially identifiable images or data included in this article. The patient consent form was read and signed by the patient.
Consent to Participate
Not applicable.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Author Contributions
BG and WD were responsible for the clinical design and conceptualization. JX, YL, HX, XL and GD were involved in the acquisition of the clinical data. SL and WD conducted the clinical diagnosis. SL, BY, and WD analyzed and interpreted the data. BG and WD wrote the manuscript. All authors discussed, read, and approved the submission of this manuscript to the journal.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The raw data supporting the conclusions of this case report will be made available by the authors, without undue reservation. All relevant data supporting the conclusions of this article are included within the article.
Clinical Trial Number
Not applicable.
