Abstract
Introduction:
Tricuspid valve prolapse (TVP) is characterized by the tricuspid valve leaflets’ displacement beyond the annular plane during midsystole. Although less common than mitral valve prolapse, TVP often coexists with it, sharing similar pathophysiological mechanisms. This case highlights new-onset tricuspid regurgitation (TR) following cavotricuspid isthmus ablation for atrial flutter, stressing the need for vigilant monitoring after such procedures.
Case:
A 68-year-old male with atrial flutter, coronary artery disease, and type 2 diabetes underwent cavotricuspid isthmus ablation in August 2023 after failed rate control. By September 2023, he developed a grade 4/6 systolic murmur. Imaging confirmed TVP with mild to moderate TR, though he remained asymptomatic. Conservative management was adopted. Follow-up in October 2024 showed mild TR without prolapse, indicating improvement.
Conclusion:
This case underscores the importance of recognizing valvular complications after atrial flutter ablation and conducting thorough post-procedural imaging to enable timely intervention and improved outcomes.
Keywords
Introduction
Tricuspid valve prolapse (TVP) is defined as the systolic displacement of 1 or more tricuspid valve leaflets beyond the annular plane, with coaptation occurring above the annulus. 1 Although less common than mitral valve prolapse (MVP), TVP frequently coexists with it, and both conditions may share similar pathophysiological mechanisms.
We present the case of a 68-year-old man with a history of atrial flutter who developed a new-onset heart murmur following cavotricuspid isthmus ablation, subsequently diagnosed as TVP. A detailed description of imaging and laboratory studies performed in this case are included in the text, and a timeline can be observed in Figure 1.

Patient timeline from initial atrial flutter diagnosis to post-ablation follow-up. Major events include atrial flutter detection and management, cavotricuspid isthmus ablation, new onset of tricuspid valve prolapse (TVP) and moderate tricuspid regurgitation (TR) identified on transesophageal echocardiography, confirmation of right atrial dilation and annular fibrosis on cardiac magnetic resonance imaging, and clinical improvement at follow-up.
Clinical Case
This case involves a 68-year-old male with a history of severe monomorphic ventricular arrhythmia, for which he underwent catheter ablation in 2014, as well as coronary artery disease and recently diagnosed type 2 diabetes mellitus. Coronary angiography performed in 2014 revealed significant involvement of the anterior descending artery (ADA) and less severe disease in the right coronary artery and intermediate ramus, without indication for stenting. Since then, the patient has been under regular follow-up with electrophysiology and clinical cardiology services.
In May 2023, the patient’s medications included sitagliptin/metformin, candesartan, low-dose aspirin, metoprolol succinate, and rosuvastatin. During a routine cardiology follow-up, he reported dizziness, prompting an electrocardiogram (ECG), which revealed atrial flutter with a 4–6:1 conduction ratio; cardiopulmonary auscultation at that time was unremarkable. Given his CHA₂DS₂-VA score of 4 (indicating a moderate to high risk of stroke) therapy with a non-vitamin K antagonist oral anticoagulant (NOAC) was initiated, specifically rivaroxaban, and aspirin was discontinued. Transesophageal echocardiography (TEE) was performed to assess candidacy for electrical cardioversion and demonstrated no intracavitary thrombus or valvular abnormalities. Laboratory results showed mildly elevated fasting glucose (114 mg/dL), a glycated hemoglobin of 6.3%, and a brain natriuretic peptide (BNP) level of 46 pg/mL, suggesting no significant cardiac strain.
On May 15, 2023, the patient underwent successful electrical cardioversion with 50 J, which resulted in sinus bradycardia, a first-degree atrioventricular (AV) block, and findings consistent with Bayés syndrome type I. A 24-hour Holter monitor was subsequently performed, revealing persistent atrial flutter with variable AV conduction and nocturnal RR intervals lasting up to 8 seconds. Metoprolol succinate was discontinued. Given the time elapsed since his last coronary assessment in 2014, a stress echocardiogram was scheduled to reassess coronary artery disease.
During a cardiology follow-up in June 2023, the patient demonstrated progression to second-degree atrioventricular (AV) block Mobitz type II and continued atrial flutter. He was referred to an electrophysiology specialist for further evaluation and ablation. In August 2023, successful cavotricuspid isthmus (CTI) ablation was performed, although electrical cardioversion was required intraoperatively due to severe hypotension during anesthesia induction. No additional procedural complications occurred.
At a follow-up visit in September 2023, a grade 4/6 systolic murmur was auscultated at the tricuspid area. Subsequent TEE revealed tricuspid valve prolapse with moderate regurgitation. Given the patient’s stable clinical status and absence of symptoms such as dyspnea or angina, conservative management with clinical observation and additional imaging was elected.
Cardiac magnetic resonance imaging (CMR) performed on December 19, 2023, showed a normal-sized left ventricle with a preserved ejection fraction of 64%. The right atrium was dilated, with an indexed volume of 71 mL/m². The right ventricle displayed normal morphology and an ejection fraction of 59%. Significant prolapse of the tricuspid valve was observed (9.6 mm), along with mild regurgitation (regurgitant fraction 11%). Late gadolinium enhancement (LGE) imaging revealed fibrosis of the inferolateral tricuspid annulus. Cine sequences and native T1 mapping are shown in Figure 2.

Patient images: (a) cine-MRI sequence of the right ventricular outflow tract showing tricuspid regurgitation (white arrow), (b) static four-chamber image showing the tricuspid valve leaflets exceeding the tricuspid annulus, indicating a prolapse (white arrow), (c) late gadolinium enhancement (LGE) in the plane of the mitral and tricuspid rings, with gadolinium retention visible at the 7 o’clock position (white arrow) on the tricuspid ring, and (d) native T1-mapping sequence with a normal value according to our lab for this time in the muscular portion of the interventricular septum.
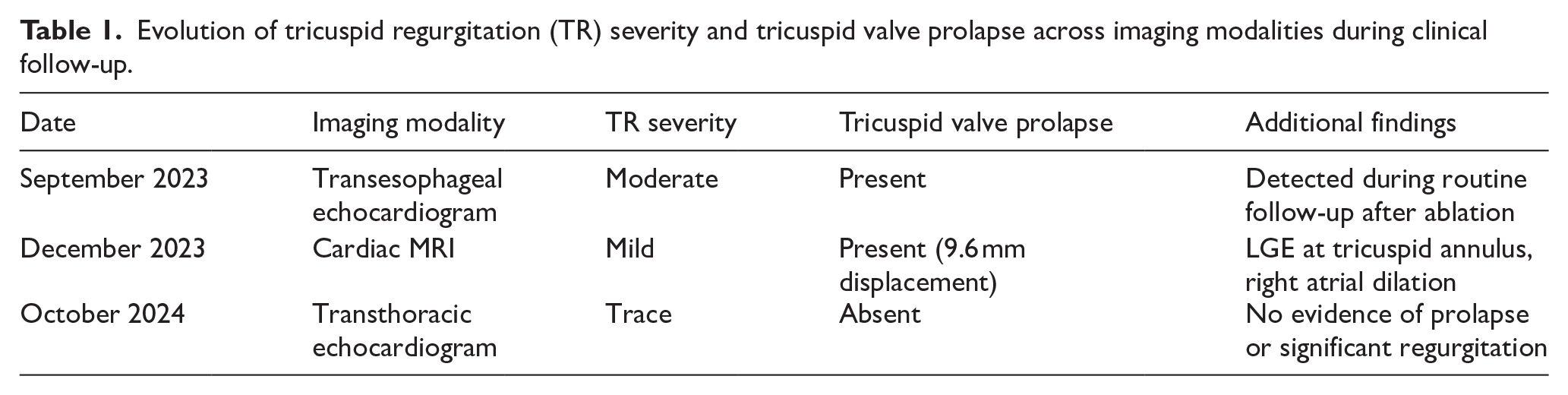
At follow-up in February 2024, the intensity of the systolic murmur had decreased to grade 2/6, with no symptoms of dyspnea or angina. The patient remained in a stable functional class and reported a good quality of life. Medical management was continued with sitagliptin/metformin, rivaroxaban, rosuvastatin, and metoprolol succinate. A follow-up transthoracic echocardiogram (TTE) performed in October 2024 showed mild tricuspid regurgitation without evidence of prolapse. A summary of the imaging findings, including the progression of TR severity and resolution of TVP, is provided in Table 1.
Evolution of tricuspid regurgitation (TR) severity and tricuspid valve prolapse across imaging modalities during clinical follow-up.
Literature Review and Discussion
A large echocardiographic study conducted at Houston Methodist by Lorinsky et al analyzed 218 943 transthoracic echocardiogram (TTE) reports observed a prevalence of 0.3% for tricuspid valve prolapse (TVP). Although formal diagnostic guidelines are lacking, TVP was defined based on leaflet displacement, specifically ⩾2 mm displacement of the anterior and posterior leaflets, and ⩾3 mm for the septal leaflet in the parasternal short axis view. Among 312 cases with subjectively identified TVP, mitral valve prolapse (MVP) was present in 75% of patients. Conversely, among individuals with MVP, only 0.04% had concurrent TVP. 2 In contrast, a study from the University of California, San Francisco by Werner et al found a 21% prevalence of TVP in patients with MVP. 3
In our case, the diagnosis of tricuspid valve prolapse (TVP) was based on standard echocardiographic criteria, as described by Lorinsky et al, which were extrapolated from the diagnostic thresholds for mitral valve prolapse (MVP). On transthoracic echocardiography (TTE), systolic displacement of the tricuspid leaflets consistent with these thresholds was observed. Cardiac magnetic resonance (CMR) imaging subsequently quantified significant prolapse, with a maximum displacement of 9.6 mm, substantially exceeding the established diagnostic criteria and confirming the diagnosis.
Unlike the studies by Lorinsky et al and Werner et al, which primarily described TVP in the context of mitral valve prolapse (MVP), our case involves isolated TVP without evidence of concomitant MVP. In the Lorinsky et al. 2 cohort, only 9 cases of isolated TVP were identified among 218 943 TTEs, and Werner et al. reported no isolated TVP cases. 3 These findings highlight the uniqueness of our case and suggest the possibility of an acquired, post-procedural mechanism. Moreover, the subsequent resolution of prolapse and reduction in TR severity suggest a transient and possibly reversible mechanism, rather than the structural myxomatous degeneration typically seen in syndromic or congenital cases. To our knowledge, this is one of the few reports to document isolated TVP potentially related to post-ablation tissue changes.
The most common morphological subtype of TVP is diffuse myxomatous degeneration, also referred to as Barlow disease. 1 A strong association has also been reported between TVP and connective tissue disorders, particularly Marfan syndrome. Cardiac involvement occurs in approximately 80% of patients with Marfan syndrome, most commonly presenting as aortic dilatation, aortic regurgitation, and mitral valve prolapse (MVP), with occasional reports of TVP. 4 In the same UCSF cohort, a subset analysis of patients with both Marfan syndrome and MVP revealed that TVP was present in 67% of these cases (4 out of 6 individuals). 3 Additionally, case reports have described TVP in patients with Ehlers-Danlos syndrome and Behçet’s disease.5,6 These associations highlight the importance of systematically evaluating both mitral and tricuspid valves in patients with connective tissue disorders and suspected valvular abnormalities.
In terms of outcomes, the study by Lorinsky et al found that TVP was associated with more severe tricuspid regurgitation, right ventricular dilation, and increased mitral regurgitation severity. However, mortality rates did not significantly differ between patients with isolated TVP and those with concomitant MVP. 2
Regarding the aforementioned presentation of TVP, catheter ablation is widely recognized as a guideline-endorsed approach for rhythm control in patients with atrial flutter. Although the procedure is generally considered safe, it is not without potential complications. A recent German registry study involving 3526 patients reported an overall complication rate of 1.4%, with the most frequent events being vascular access site complications (0.6%) and complete heart block (0.2%). 7
While neither the German registry study nor prior clinical series have reported tricuspid regurgitation or valve prolapse as procedural complications, experimental evidence suggests a potential mechanistic link. In a porcine model of cavotricuspid isthmus ablation, radiofrequency energy was associated with valvular apparatus injury: among 27 animals, 4 developed mild lesions and 3 exhibited severe endocardial disruption. Notably, these injuries occurred in animals exposed to higher catheter contact pressures and temperatures. 8 Histopathologic analyses of ablation sites have demonstrated a central zone of coagulative necrosis (with lymphocytic and plasma cell infiltrate) surrounded by a border of partially damaged tissue. 9 Additionally, phenomena such as “steam pops” (acute intramyocardial explosions caused by rapid tissue heating) have been documented and may contribute to abrupt structural disruption, including valvular involvement.10,11
Nonetheless, post-ablation fibrosis may represent a physiological response rather than a pathologic process. In a cardiac magnetic resonance study of 67 patients undergoing cavotricuspid isthmus ablation, late gadolinium enhancement (LGE) at the CTI was observed in 69% of patients 1 day after the procedure and in 84% at 1 month, despite resolution of T2 signal and wall thickening. 12 These findings suggest that LGE in this region is common and may reflect stable fibrotic remodeling. In our case, the presence of LGE at the tricuspid annulus may similarly represent localized fibrosis and/or edema secondary to ablation, potentially contributing to transient disruption of leaflet coaptation.
Tricuspid regurgitation (TR) is frequently observed in patients with atrial fibrillation and is often linked to dilation of the right atrium and tricuspid annulus. Recent studies have shown that TR may improve following atrial fibrillation ablation, likely due to reverse atrial remodeling.13,14 However, the extent to which this mechanism applies to atrial flutter remains unclear. This gap in knowledge underscores the importance of evaluating the interplay between atrial remodeling, valvular function, and potential procedural injury when considering ablation strategies. A comprehensive understanding of these factors is essential to minimizing the risk of iatrogenic valvular damage.
Implications for Clinical Care
This case highlights the critical importance of vigilant post-procedural monitoring in patients undergoing cavotricuspid isthmus ablation for atrial flutter. While catheter ablation is widely regarded as a safe and effective treatment, this report underscores the potential for uncommon but clinically significant complications, including tricuspid valve prolapse (TVP) and tricuspid regurgitation (TR). Early identification of such complications using imaging modalities—particularly TEE and CMR—is essential for timely clinical decision-making and effective management.
These findings underscore the importance of thoroughly assessing valvular structure and function prior to ablation, especially in patients with preexisting cardiac conditions or atrial enlargement. The case also illustrates the clinical value of a multidisciplinary approach—integrating electrophysiologists, cardiologists, and imaging specialists—to ensure comprehensive evaluation and optimized patient care.
Conclusions
A key strength of this case report lies in the successful implementation of medical therapy and the patient’s consistent clinical stability throughout the follow-up period. Additionally, the timely identification of the valvular abnormality enabled comprehensive characterization via CMR, supporting prompt clinical decision-making and the exclusion of other potential complications.
However, the report is limited by the scarce literature on isolated tricuspid valve prolapse (TVP), particularly in the context of post-ablation settings, as well as the absence of guideline-endorsed diagnostic criteria. While most existing studies focus on mitral valve prolapse (MVP) and its occasional association with TVP, more focused research—such as that conducted by Lorinsky et al.—is needed to better define the clinical and imaging characteristics of this entity. Another limitation concerns the 3-month delay between the initial TEE and the CMR, which may have influenced the observed severity of prolapse and regurgitation due to interval improvement.
In conclusion, this case report highlights the importance of prompt post-ablation imaging in the presence of abnormal findings on physical examination or cardiopulmonary auscultation. The transient nature of the event described suggests that delayed evaluation could result in missed diagnoses with potential therapeutic implications. The integration of modalities such as transesophageal echocardiography and cardiac magnetic resonance imaging could facilitate timely diagnosis, guide management decisions, and improve patient outcomes. Therefore, a structured post-procedural follow-up with a general practitioner, cardiologist, or electrophysiology specialist may be essential to detect early complications and prevent adverse outcomes.
Take Home Messages
Tricuspid valve prolapse (TVP) and subsequent tricuspid regurgitation (TR) may occur as complications following cavotricuspid isthmus ablation, emphasizing the importance of post-procedural imaging and monitoring for valvular changes.
Comprehensive imaging and timely medical management are key to identifying and addressing potential complications, ensuring favorable outcomes in patients undergoing atrial flutter ablation.
Footnotes
Abbreviations
Consent for Publication
Written informed consent was obtained from the patient prior to the preparation of this case report. The patient was provided with a detailed explanation of the purpose of the report, the information that would be included, and how it would be disseminated. The consent form explicitly outlined the patient’s rights, including the right to withdraw consent at any stage without any impact on their medical care. The patient voluntarily signed the consent form, indicating their agreement to participate and for their anonymized clinical data to be used for publication purposes. All ethical considerations were adhered to in accordance with the Declaration of Helsinki.
Author Contributions
Pablo Pineda-Sanabria (PPS), Catalina Arbelaez-Hoyos (CA), and Antonio Villamizar-Romero (AV) conducted the study’s conceptualization, data collection, and drafting of the manuscript. Mauricio Pineda-Gomez (MP) assessed the content in a critical manner and provided the study data and imaging interpretations. All the authors meticulously reviewed and approved the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the findings of this case report are not publicly available to ensure the privacy and confidentiality of the patient. Specific clinical details and images have been anonymized to prevent identification. Further information related to the case may be made available from the corresponding author upon reasonable request and with appropriate ethical approval, provided it does not compromise patient confidentiality.
