Abstract
Objective:
Sodium-glucose co-transporter-2 inhibitors (SGLT2-I’s) are novel oral hypoglycaemic agents, with proven decreased MACE and re-hospitalisation risk in type 2 diabetic patients with concomitant heart failure. This study aimed to assess the current practice in the use of SGLT2-I’s in general medical units at a large metropolitan health service.
Methods/Results:
A retrospective audit was conducted of patients admitted to general medicine over a 12 month period (between April 2018 and 2019). Inclusion criteria included decompensated heart failure of any aetiology and ejection fraction, and type 2 diabetes mellitus with an HbA1c ⩾ 7 within 6 months of the admission period. A total of 150 admissions fulfilled criteria. Baseline demographics and comorbidities identified an older, more comorbid population than reference trials. These included age (75% over 75 years), smoking history (46%), hypertension (83%), chronic kidney disease grade IV or V (26%), previous myocardial infarction (57%), stroke (18%), atrial fibrillation (55%) and known left ventricular ejection fraction < 50% (38%). Co-prescribed medications included ACE-I/ARB (53%), beta-blocker (67%), loop diuretic (87%), thiazide (7%), MRA (31%), insulin (57%), metformin (47%), sulphonylurea (31%), DPP-4 Inhibitor (21%), GLP-1 analogue (6%) and 15% of patients had an HbA1c > 10. There was a significant difference between patients in our study eligible for and prescribed metformin (66/111) compared to SGLT-2 inhibitors (4/25) (P = .013). A total of 26 patients had readmissions within 28 days, of which one had been discharged on an SGLT2-I.
Conclusion:
The results of this study identified significant under prescribing of SGLT2-I’s in eligible type 2 diabetic patients with heart failure admitted under general medicine.
Introduction
Sodium glucose co-transporter 2 (SGLT2) inhibitors are a novel class of oral hypoglycaemic agents, acting by promoting urinary excretion of glucose and limiting renal glucose reabsorption. Current literature supports the use of SGLT2-Inhibitors in Type 2 diabetic patients with overt cardiovascular disease, with proven decrease in cardiovascular morbidity and mortality, including reduction in Major Adverse Cardiovascular Events (myocardial infarction, stroke, cardiovascular death). In patients with concomitant heart failure, trials have indicated reduced re-hospitalisation risk for heart failure with the use of such medications.1-4
In addition, with growing evidence of clinical efficacy, most recent guideline changes in Australia have clarified new PBS indications to commence some of these agents for non-diabetic patients with symptomatic (NYHA class II-IV) heart failure with reduced ejection fraction (HFrEF) as add on therapy to standard optimal therapy. 5
There is limited epidemiological data available in regards to the current use of SGLT-2 Inhibitors in general medicine in-patients, who are typically older and have more co-morbidities than the major trial patients. As such, a review of the current practice would help identify areas for improvement in the current management of patients with heart failure and type 2 diabetes, which represent a large cohort of the patients admitted to our general medical units. This includes identifying the current practice and comparison with evidence based best practice, and appropriate and inappropriate medication prescribing according to current evidence.
This study was aimed at assessing the current practise in the use of Sodium-Glucose Co-Transporter-2 inhibitors (SGLT2-I) in the management of patients admitted with decompensated heart failure and a history of type 2 diabetes mellitus, to any general medical unit at the Royal Melbourne Hospital over a 12 months period between April 2018 and 2019.
Materials and Methods
A retrospective observational study was conducted of patients admitted to the General Medical Units at RMH with decompensated heart failure and a history of Type 2 diabetes Mellitus over a 12-month period (between April 2018 and 2019).
Inclusion criteria
- Patient admission between April 2018 and April 2019
- Admission to a General Medical Unit (AMU/MU1/MU2/MU3)
- Admission for Decompensated Heart Failure of any aetiology including Heart Failure with preserved Ejection Fraction (HFpEF)/Heart Failure with mid-range Ejection Fraction (HFmrEF/Heart Failure with reduced Ejection Fraction (HFrEF), identified via discharge ICD code
- Diagnosis of Type 2 diabetes Mellitus
- HbA1c ⩾ 7% within 6 months of the admission period
Exclusion criteria
- No documented history of Type 2 diabetes Mellitus
- No HbA1c available within 6 months of admission
Data collection
The electronic medical records of patients who fit the inclusion criteria were analysed, to collect relevant information including patient demographics, co-morbid disease, smoking history, relevant heart failure and diabetes medications (including ACE/ARB, B-Blocker, Diuretics (Loop/Thiazide/Potassium-sparing), Oral hypoglycaemic agents (Metformin/Sulphonylurea/DPP4-I/Thiazolidinedione/GLP-1 Analogue), Insulin and listing the use of an SGLT2 inhibitor on admission/discharge. Other collected information including most recent transthoracic echocardiogram details of left ventricular ejection fraction, and admission biochemistry (including haemoglobin, creatinine, eGFR and HbA1c within 6 months).
Outcome measures
The outcome measures evaluated included:
Percentage of General Medical patients with an admission for heart failure and a concurrent diagnosis of type 2 diabetes mellitus, eligible for a SGLT-2 Inhibitor based on PBS criteria at the time of admission (Including concurrent use of metformin, sulphonylurea or insulin, age ⩽75 (as a relative contraindication for patients aged >75 years) and with an eGFR ⩾45), and prescribed an SGLT2-I on admission or discharge
Percentage of General Medical patients with an admission for heart failure and a concurrent diagnosis of type 2 diabetes mellitus, eligible for metformin (eGFR ⩾30, without other contraindication) and prescribed metformin on admission or discharge
The proportion of PBS criteria based eligible patients prescribed an SGLT2-I on discharge in comparison to those eligible and prescribed metformin.
Data analysis
From previous audit work, we were aware that approximately 10% of potentially suitable patients admitted to general medicine at our institution were prescribed SGLT-2 inhibitors (P1). We hypothesised that this was due to the presence of contraindications, and that in the presence of a contraindication the proportion prescribed SGLT-2 would be 0% (P2) (HA: P1 = P2). If this were not the case, the proportion would be the same (H0: P1 ≠ P2). A sample of 148 participants was deemed to be required to provide evidence to reject the null hypothesis with 80% power at a significance level of .05.
Analysis was conducted according to pre-defined variables and was completed with Stata 15.1 (Statacorp, Texas). Statistical significance was defined as P < .05, all tests 2-sided. Categorical variables were analysed using Chi test unless the expected cell values were less than 5, in which case the Fisher’s exact test was used.
Results
A total of 150 admissions fulfilled criteria for inclusion in the study. Baseline demographics and comorbidities identified an older, more comorbid population than reference trials. These included age (75% over 75 years), smoking history (46%), hypertension (83%), chronic kidney disease grade four-fifths (26%), previous myocardial infarction (57%), stroke (18%), atrial fibrillation (55%) and known left ventricular ejection fraction<50% (38%) (Table 1, Figure 1).
Patient baseline demographics, co-morbidities and biochemistry.


Demographics and co-morbidities (%).
Co-prescribed medications included ACE-I/ARB (53%), beta-blocker (67%), loop diuretic (87%), thiazide (7%), MRA (31%), insulin (57%), metformin (47%), sulphonylurea (31%), DPP-4 Inhibitor (21%), GLP-1 analogue (6%) and 15% of patients had an HbA1c > 10 (Table 1, Figure 2)

Co-prescribed medications (%).
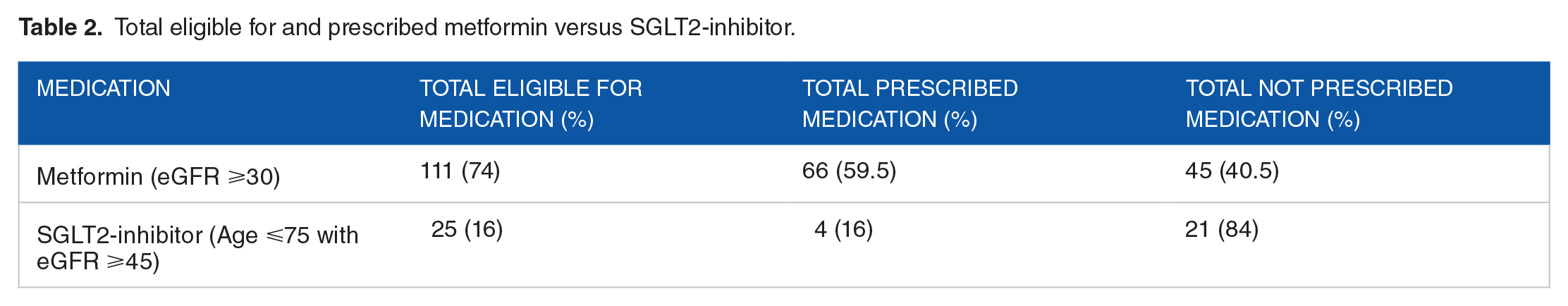
There was a significant difference between the proportion of patients in our study eligible for and prescribed metformin (66/111) compared to SGLT-2 inhibitors (4/25) (P = .013), on assessment using a Pearson’s chi analysis (Table 2 and Figure 3).
Total eligible for and prescribed metformin versus SGLT2-inhibitor.

Comparison of metformin versus SGLT-2 inhibitor prescription (%).
A total of 26 patients had readmissions within 28 days, of which one had been discharged on an SGLT2-I (Table 3 and Figure 4).
28 day readmission rates.


28 day re-admissions whilst on an SGLT2-I versus without (%).
Three patients who were admitted with an SGLT2-I had it withheld and ceased at discharge as they had acute renal impairment with CrCl < 30. None of the patients admitted with SGLT2-I were identified to have euglycemic ketoacidosis as a complication during their admission. One patient discharged on an SGLT2-I was 87 years of age, hence considered a relative contraindication for SGLT2-I use, and not accounted for in the metformin versus SGLT2-I comparison analysis.
Discussion
Sodium-glucose co-transporter 2 inhibitors have been widely studied recently with current literature supporting their use in patients with type 2 diabetes and cardiovascular disease with reductions noted in all-cause and cardiac specific mortality, reduced heart failure hospitalisation rates and reduced progression of renal disease. Meta-analyses data for empagliflozin/dapagliflozin/canagliflozin versus placebo have shown reduced major adverse cardiovascular events (MACE) risk of 86.9 versus 99.6 events/1000 patient-years, HR 0.89, 95% CI (0.83 to 0.96) and cardiovascular death or heart failure hospitalisation composite outcome rates of 48.2 versus 65.6 events/1000 patient-years, HR 0.77, 95% CI 0.71-0.84.3,4 More recently, there is even growing evidence for reduced MACE in patients with heart failure with reduced ejection fraction (HFrEF). 6 From a diabetes perspective, studies have shown a reduction in glycated haemoglobin (HbA1c) by 0.5% to 1% in comparison to placebo.7,8
Their mechanism of action is by increasing urinary excretion of glucose, by almost completely blocking glucose reabsorption by the sodium-glucose co-transporter 2 expressed in the proximal renal tubule (responsible for around 90% of filtered glucose load reabsorption). This lowers blood glucose levels and hence glycated haemoglobin A1c levels, and also the osmotic diuresis with increased renal tubular glucose excretion contribute to management of fluid overload in patients with decompensated heart failure, and also cause modest blood pressure decrease and weight loss. Adverse effects including intravascular volume depletion with polyuria (with associated hypotension, risk of syncope, dehydration and acute kidney injury) and glycosuria contributing to increased risks of genitourinary infections are also related to its novel mechanism of action. Other notable adverse effects include euglycemic ketoacidosis, increased risk of fractures and amputations. Also of note, SGLT2-Is have decreased efficacy in the setting of renal impairment. 9
The Australian Therapeutic Goods Association (TGA) first approved SGLT2-I’s in 2013, and currently empagliflozin, dapagliflozin and ertugliflozin are listed on the Pharmaceutical Benefits Scheme (PBS). Currently, there is no approval in Australia for use in Type 1 diabetic patients.
Despite the widespread literature available for these novel agents, their use in the older and multimorbid general medical patient cohort has been less well studied. Our study therefore was aimed at a cross-sectional audit of local prescription of these agents in the general medical inpatients admitted to the hospital medical units. We identified an older cohort than in the general SGLT2-I studies, with 75.3% of admissions above the age of 75 with co-existent cardiovascular co-morbidities. More than 50% of these admissions also had a concurrent diagnosis of chronic renal impairment (including 59.3% with CKD stage IIIB or higher). These factors in itself could be considered relative contraindications for initiating SGLT2-Inhibitors given limited experience in this cohort. About one-third of patients (29.4%) also had very poorly controlled type 2 diabetes with an HbA1c above 9%, though noting the general recommendations for a more individualised approach to glycaemic control in this very heterogenous elderly cohort, taking into consideration overall life expectancy, health and risk of hypoglycaemia and associated complications.10,11 It was also noted that 38% of our patients had documented mid-range to reduced LV ejection fraction on most recent transthoracic echocardiograms, and only 53% of patients were on a betablocker, 67% on an angiotensin converting enzyme-inhibitor/receptor blocker and 31% on an MRA (with known evidence for mortality benefit in HFrEF), 12 but close to 90% were on loop diuretic therapy primarily used for symptomatic benefit for fluid overload.
In terms of type 2 diabetes therapy, most patients were on insulin regimens followed by metformin or a sulphonylurea, possibly reflecting reduced tolerance or contraindications of oral hypoglycaemic agents in this older population, and perhaps reduced efficacy in comparison to insulin.
Analysis of the 8 patients (5.3% of the entire study cohort) who were admitted on SGLT2-Inhibitors revealed that 3 had the drug withheld during the admission and ceased at discharge due to acute renal impairment, however, no other side-effects including euglycaemic ketoacidosis were documented during the admission. Multifactorial acute renal impairment (primarily due to pre-renal injury) in the setting of acute decompensated heart failure is very common in the general medical inpatient cohort, and as such may explain the reduced rates of new prescription of SGLT2-I agents during an admission with decompensated heart failure. Transient decrease in eGFR is an established adverse effect during initiation of an SGLT2 inhibitor, but this is not usually expected to progress, and in fact there is evidence for longer term renoprotection, with up to a 45% relative risk reduction in nephropathy progression in comparison to placebo. 3
The primary outcome of the study identified a significant difference between the proportion of patients eligible for and prescribed metformin in comparison to SGLT-2 inhibitors based on PBS criteria and relative contraindications at the start of the admission (including age ⩽75 and eGFR ⩾45). Twenty-five patient admissions (16%) were identified as eligible and likely to have overall benefitted from an SGLT2-Inhibitor addition to their ongoing management, but only 4 (16%) were prescribed an SGLT2-I on discharge. This was in comparison to 66 (59.5%) of patients eligible for and prescribed metformin on discharge.
Potential reasons for the reduced SGLT2-I prescription could include limited physician experience in prescribing these novel agents, particularly in this specific vulnerable cohort, including concerns for increased risk of adverse effects and unclear longer-term side-effects. The widespread use of other oral hypoglycaemic agents such as metformin have more robust longer term data in comparison to the novel agents, and could further explain hesitancy in prescription. Other reasons could also be due to inclination to commence new medications when patients have improved clinical status in a more controlled outpatient setting, after recovery from decompensated disease. However, given the rapidly evolving evidence for improved clinical outcomes from a cardiovascular, renal and metabolic perspective, it would be prudent to perform individualised risk/benefit analysis in general medical patients to assist utilising these novel agents added to the heart failure and type 2 diabetes management armamentarium.
Also, 17.3% of the studied cohort had a readmission for decompensated heart failure within 28 days, and of this only 4% of these patients were on an SGLT2-I, possibly adding further evidence in regards to the efficacy of SGLT2-Is in reduced heart failure rehospitalisations, though noting overall limited prescription of these agents at discharge.
Limitations identified in this study include the low sample size affecting the power of the study, and also generalisability of the results given the clinician dependant practise variability and single centre experience.
Conclusion
Despite current literature supporting significant clinical efficacy for SGLT2-Inhibitors in both heart failure and type 2 diabetes management, the results of this study identified significant under prescribing of SGLT2-I’s in eligible type 2 diabetic patients with heart failure admitted under general medicine. Given current limited therapeutic experience in the use of these novel agents in this specific cohort of patients, further detailed studies to assist risk-benefit analysis in the predominantly older, multimorbid, general medical patient population would be essential.
Footnotes
Acknowledgements
The abstract for this study has been previously presented at the Cardiac Society of Australia and New Zealand Annual Scientific Meeting (2020), and was published in the Heart, Lung and Circulation Journal (2020) (![]() ). The current listed authors retain all rights to this manuscript and prior related publications.
). The current listed authors retain all rights to this manuscript and prior related publications.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All author contributed to the study conception and design. PP, RO and TF contributed to data acquisition, and PP, RO and PL contributed to data analysis. All authors contributed to drafting and revision of the manuscript.
