Abstract
Objective:
To evaluate the efficacy and safety of valsartan (V) or chlorthalidone (C) monotherapy in comparison with a fixed combination of valsartan and chlorthalidone (V + C).
Methods:
This 12-week multicenter randomized three-arm open-label study randomly allocated 72 patients to V or C as monotherapy or a combination of V + C. The aim was to measure changes in office systolic blood pressure (SBP) and diastolic blood pressure (DBP) and in 24-hour ambulatory blood pressure monitoring (ABPM) from baseline to week 12, in addition to medication tolerability.
Results:
The proportion of patients achieving target BP in office at week 12 was not statistically different for the three groups. However, comparisons of daytime and nighttime 24-hour ABPM values from baseline to week 12 revealed significant differences in nighttime mean SBP for the three groups, due to a significantly greater reduction in the values in patients assigned to the V + C group (−14.7 vs. −8.7 vs. −10.7, P = .042, V+C; V; C, respectively). Although patients assigned to the V + C group also had greater nighttime reduction in mean DBP values compared with those in the other groups, this difference was not statistically significant. The incidence of adverse events did not differ significantly.
Conclusion:
In patients with hypertension treated with V, C, and both medications combined, the fixed combination of V + C provided a significantly greater reduction of late night to early morning BP values when interventions were assessed with 24-hour ABPM.
Trial registration:
clinicaltrials.gov Identifier: NCT.01850160, https://clinicaltrials.gov/ct2/show/NCT01850160
Introduction
Hypertension is a major public health issue and has been considered the leading preventable cause of premature mortality and disability worldwide. 1 Hypertension is one of the most important contributors to the burden of cardiovascular disease, stroke, and kidney failure. 2 Although hypertension represents a major health problem with a high prevalence in all world regions, recent data have documented a large and widening global disparity in hypertension prevalence and control. A recent systematic analysis of population-based studies from 90 countries showed that 31.1% of the global adult population aged 20 years and older (1.39 billion people) had hypertension in 2010, of whom 74.8% (1.04 billion people) lived in a low- and middle-income country (LMIC). Additionally, from 2000 to 2010, while the age-standardized prevalence of hypertension decreased by 2.6% in high-income countries, it increased by 7.7% in LMICs. 3 In Colombia, a representative Latin American LMIC, a recent national report indicated hypertension prevalence of 31% for women and 28.8% for men. 4
Adequate and opportune treatment of hypertension is crucial for the prevention of cardiovascular events and kidney diseases. 5 Convincing evidence has shown that pharmacotherapy for hypertension positively impacts several clinically important outcomes, including nonfatal and fatal cardiovascular events.6,7 Among several therapeutic options for patients with hypertension, angiotensin receptor blockers (ARBs, e.g., valsartan) and diuretics are used widely in clinical practice because they are efficacious and well-tolerated. Therefore, both drug classes are recommended as first-line medications for the treatment of hypertension in the recent high-quality evidence-based hypertension guidelines.8,9 However, it has been estimated that more than two-thirds of the patients with hypertension do not attain and maintain the target blood pressure (BP) using one drug alone, thus requiring more than one antihypertensive agent selected from different drug classes to provide optimum control. 10 Among all various possible antihypertensive drugs/combinations, ARBs in combination with diuretics are among the most widely used, not only because of the opportunity to combine the complementary advantages of both drug classes (reduction in angiotensin II-mediated sodium retention and vasoconstriction with ARBs and volume-reducing benefits of diuretics), but also because of high efficacy. A recent systematic review of randomized clinical trials (RCTs) aimed to evaluate the treatment efficacy of antihypertensive drugs in monotherapy or in combination, and concluded that compared to monotherapy, valsartan/hydrochlorothiazide is one of the combinations resulting in the greatest mean reduction in BP. 11 However, only very few of these comparative studies have been carried out in LMICs, which have the greatest burden of the disease. 3 Chlorthalidone is another diuretic that is approximately 1.5–2.0 times more potent than hydrochlorothiazide (HCTZ), with a much longer and smoother duration of action12,13 and well-documented benefits for reduced cardiovascular morbidity and mortality 14 (therefore being potentially a more effective diuretic in combination); however, very few studies have evaluated its efficacy and safety in combination with an ARB, such as valsartan.
Accordingly, the aim of the present study was to compare the efficacy and safety of valsartan or chlorthalidone, administered as monotherapy, and a combination of a fixed dose of valsartan and chlorthalidone, evaluated by 24-hour ambulatory BP monitoring (ABPM) in a population of adult patients with mild to moderate hypertension.
Methods
Patient population
The study included eligible male and female outpatients aged between 18 and 70 years, either untreated (currently not taking antihypertensive medication) or receiving antihypertensive medication (other than valsartan or chlorthalidone), who did not have their hypertension controlled, with a documented diagnosis of hypertension defined as mean sitting systolic BP (MSSBP) ⩾ 140 and < 180 mmHg and mean sitting diastolic BP (MSDBP) ⩾ 90 and < 110 mmHg. The main exclusion criteria were severe or malignant hypertension, defined as MSSBP ⩾ 180 or MSDBP ⩾ 110 mmHg; known or suspected secondary hypertension; female patients of childbearing age not using an approved or adequate method of contraception, or those who were pregnant or planning on becoming pregnant during the time they would be participating in the study; history of myocardial infarction and/or angina pectoris in the preceding 6 months; history of cerebrovascular disease or transient ischemic attack in the preceding 6 months; severe renal disease (serum creatinine > 1.5 mg/dL); proven or suspected renal arterial stenosis; history of malignancy without complete remission in the preceding 5 years; hypokalemia or hyperkalemia; history of allergy/hypersensitivity to valsartan or chlorthalidone, or use of valsartan or chlorthalidone within the previous 14 days; history of drug or alcohol abuse in the preceding 12 months; unstable status with a history of hospitalization within 4 weeks before enrollment; participation in another clinical study 4 weeks before inclusion or during participation in the proposed study; and inability to comply with the protocol activities.
All patients provided written informed consent, and the study protocol was approved by the following independent Ethics Committees: Medplus, Bogota; Fundacion Cequin, Armenia; and Centro Diagnostico Cardiologico, Cartagena. The institutional review board at each of the three study sites was responsible for approval of the clinical study, in accordance with ethical principles and the Guidelines of the Declaration of Helsinki, the regulations and guidelines of the International Conference on Harmonisation, Harmonised Tripartite Guideline for Good Clinical Practice, and all applicable local regulations. All patients provided written informed consent prior to screening. The study was registered at clinicaltrials.gov, Identifier: NCT.01850160. https://clinicaltrials.gov/ct2/show/NCT01850160.
Study design
This was a 12-week multicenter randomized, three-arm parallel-group open-label study conducted across three centers in Colombia. Recruitment and randomization took place from April 2013 to June 2014, and the patients were followed up until October 2014.
At the initial screening visit, patients underwent a medical history review, complete physical examination, 12-lead electrocardiogram (ECG), standard laboratory analyses of complete blood chemistry and hematology, pregnancy test (for female patients of childbearing age), and 24-hour ABPM.
After the initial screening visit, each participant was assessed on three separate occasions, 6 weeks apart. At baseline and at each of the follow-up assessments, patients underwent a complete physical examination, with measurement of heart rate and BP. During baseline assessment, eligible patients were randomly allocated to 1 of 3 treatment arms: one arm using valsartan as monotherapy, one using chlorthalidone (HIDROTEN; FARMA Labs) as monotherapy, and one using a combination of valsartan and chlorthalidone (BRASARTAN CTDN; FARMA Labs) for 12 weeks. The stratified randomization sequence was generated by an investigator not involved in patient enrollment, using a table of random numbers. Investigators assigned patient numbers sequentially within each center, and each center received drug supplies with medication numbers corresponding to the center and randomization numbers.
During the second assessment, patients were evaluated to assess whether they would be eligible for titration to the next dosage level of the same medication assigned at baseline (in case of uncontrolled BP) or for maintenance at the same dose level used at baseline (in case of achievement of target BP goal) (Figure 1).
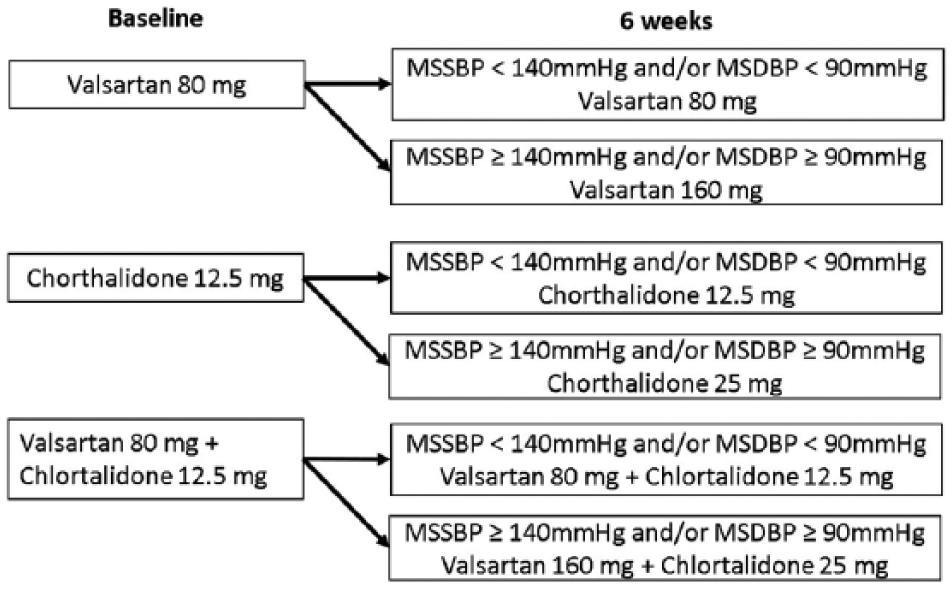
Drug allocation and time for titration.
At the final assessment, patients again underwent a 12-lead ECG, standard laboratory analyses of complete blood chemistry and hematology, pregnancy test (female patients of childbearing age), and 24-hour ABPM.
Follow-up assessment was scheduled in the morning after a 12-hour fast, and before the study medication for that day was taken, to provide BP measurements under similar conditions for all patients.
Study medications
As noted, eligible patients were randomly allocated at baseline to receive treatment with valsartan 80 mg once daily (V80), chlorthalidone 12.5 mg (HIDROTEN produced by FARMA Labs) given once daily (C12.5), or a fixed combination of valsartan 80 mg with chlorthalidone 12.5 mg (BRASARTAN CTDN produced by FARMA Labs) given once daily as a single pill (V80+C12.5) for 12 weeks. At the second assessment 6 weeks later, patients whose BP was uncontrolled (MSSBP ⩾ 140 mmHg and/or MSDBP ⩾ 90 mmHg) were titrated to the next dosage level of valsartan 160 mg (V160) once daily, chlorthalidone 25 mg (C25) once daily, or a fixed combination of valsartan 160 mg with chlorthalidone 25 mg once daily as a single pill (V160+C25), for the remaining 6 weeks of the study (Table 1). Patients achieving a target BP goal of 140/90 mmHg at the second assessment were maintained at the dose level of medication assigned at baseline.
Demographic and clinical variables of the patients included in the study, according to study treatment group.

BMI: body mass index, MSSBP: mean sitting systolic blood pressure, MSDBP: mean sitting diastolic blood pressure, ABPM SBP: 24-h ambulatory systolic blood pressure monitoring, ABPM DBP: 24-h ambulatory diastolic blood pressure monitoring, BP: blood pressure, AEs: adverse effects.
Patients were instructed to take their medication in the morning at the same time each day throughout the duration of the study. Medication compliance was assessed by medication measurement involving documenting the number of tablets dispensed and returned at follow-up assessments. The concomitant use of any other antihypertensive drug (e.g., other ARBs, angiotensin-converting enzyme inhibitors, β-adrenergic antagonists, calcium-channel blockers, and potassium-sparing diuretics such as spironolactone) was not permitted during the study.
BP and heart rate measurements
At each clinical assessment, BP and heart rate were measured by a trained research nurse. Additionally, at the initial screening visit and the final assessment, 24-hour ABPM was also measured.
BP was measured in accordance with the American Heart Association recommendations. 15 All office BP measurements were taken in the non-dominant arm after a 10-minute rest in a sitting position using World Health Organization recommendations, 16 to the nearest 2 mmHg. For diastolic BP, phase IV (disappearance of Korotkoff sounds) was used. At each center, BP was measured by the same trained research nurse in the same arm and using the same sphygmomanometer, and every attempt was made to measure BP at the same time of day each time. Heart rate was measured for 60 seconds immediately prior to BP measurements by the same trained research nurse.
Measurements of 24-hour ABPM were performed using SpaceLabs monitor 90207 (Spacelabs Medical, Inc., Richmond, WA, USA), with recordings at 20-minute intervals for 24 hours, starting between 8 and 9 AM. Patients returned to the clinic 24 hours later, when the device was removed and BP data were collected. The mean values of the hourly ambulatory SBP and DBP were calculated for each patient.
Outcomes
The primary outcome was the change in the 24-hour ABPM mean value from baseline to week 12. Secondary outcomes were a target SBP of 140 mmHg or less and/or a target DBP of 90 mmHg or less in office BP measurements 12 weeks after the start of medication administration.
Safety and tolerability assessment
Safety and tolerability were evaluated at each clinic visit (week 6 and week 12, using diary cards) and every 2 weeks by telephonic contact to monitor adverse events (AEs), including clinical, laboratory, and ECG changes.
Laboratory tests included a complete blood cell count, serum creatinine, blood urea nitrogen, sodium, potassium, fasting glucose, total cholesterol, high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), triglycerides, and uric acid.
The nature, frequency, and severity of all AEs, as well as their relationship to study medications, were evaluated by the investigators to determine whether they were mild, moderate, severe, or serious, as well as whether they were not related, possibly related, or probably related to study medications. The frequency and severity of clinical AEs, as well as the frequency of laboratory and ECG findings that were outside of predetermined ranges, were summarized and correlated with each of the study treatments.
Statistical analysis
The sample size was based on the expected difference in the proportion of patients who achieved target BP goals using combined or monotherapy, either with valsartan or chlorthalidone. A calculated sample size of 123 patients (41 patients per treatment group) would be needed to detect a 17% difference in the proportion achieving BP goals at the 0.05 level (two-sided) with 80% power, assuming a dropout rate of 15%.
Statistical analysis was performed on the full data set, based on the intent-to-treat principle. Continuous variables are presented as mean and standard deviation (SD) or median (interquartile range), as appropriate. Categorical variables are presented as numbers (percentages). Differences in continuous variables between patients who achieved target BP goals and those who did not were analyzed using the unpaired t-test or the Mann-Whitney U test, as appropriate. Associations between categorical variables and achievement or not of target BP goals were analyzed using the chi-square test or Fisher’s exact test, as appropriate. To identify factors independently associated with achievement of BP goals at week 12, we used logistic regression models, adjusting for baseline variables and other potential confounders. Similarly, to identify independent predictors of changes in sitting SBP and DBP and changes in the mean 24-hour ABPM (SBP and DBP) from baseline to week 12, we used multiple linear regression models, adjusting for baseline variables and other potential confounders. To assess the effect of each of the three treatments over time, repeated measures analyses were performed for sitting SBP and DBP and mean 24-hour ABPM (SBP and DBP). We used the Kruskal–Wallis test to analyze possible differences in daytime and nighttime changes in the 24-hour ABPM values (SBP and DBP) from baseline to week 12 between the 3 groups. Finally, associations between study treatments and the occurrence and severity of AEs were analyzed using the chi-square test or Fisher’s exact test, as appropriate.
All statistical tests were two-tailed, and the significance level used was P < .05. The data were analyzed with the statistical package Stata, version 12.0 (Stata Corporation, College Station, TX, USA).
Results
Patients and baseline characteristics
A total of 122 patients were randomized into the three treatment groups. However, 50 patients were excluded, all belonging to one of the research centers, because of technical problems in processing the information contained in the ABPM, leaving 72 patients to be analyzed. There was no statistically significant difference in the proportion of patients with technical problems in office BP measurements between the three treatment groups (P = .986). Of the total of 72 patients, 25 (34.7%) received valsartan and chlorthalidone, 23 (31.9%) received valsartan, and 24 (33.3%) received chlorthalidone. Figure 2 shows the flow chart of the participants according to treatment group. Overall, the majority of study participants were female (52.8%) and were currently receiving antihypertensive treatment (52.8%); patient age ranged from 26 to 69 years, with a mean (SD) of 50.3 (11.3) years. The demographic and baseline clinical characteristics of patients in the three treatment groups are presented in Table 1. At baseline, all three treatment groups were comparable in terms of age as well as other demographic and clinical characteristics.

Flow chart of the participants, according to treatment group.
Treatment efficacy
The proportion of patients achieving the target BP goal of 140/90 mmHg or less in office BP measurements at week 12 was not statistically different between the three groups (72.0% vs. 73.9% vs. 75.0%, for valsartan and chlorthalidone, valsartan, and chlorthalidone study treatment groups, respectively, P = .971). Given the fact that the number of patients who were available for analyses was limited, we worked backward from the fixed sample size, and estimated a 65% power to detect a 17% difference in the proportion achieving BP goals. After adjusting logistic regression models to control for baseline variables and other potential confounders, neither patients assigned to the V80 + C12.5 group (odds ratio [OR]: 0.86, 95% confidence interval [CI]: 0.15 to 5.12, P = .872), nor those assigned to the V80 group (OR: 0.61, 95% CI: 0.11 to 3.29, P = .567), when compared to patients assigned to the C12.5 group, were independently associated with the probability of achieving the target BP goal at week 12 (data not shown).
The proportion of patients achieving the target SBP of 140 mmHg or less at week 12 in the office BP measurements was not statistically different between the three groups (80.0% vs. 73.9% vs. 79.2% (V+C; V; C), respectively, P = .862). Similarly, the proportion of patients achieving the target DBP of 90 mmHg or less at week 12 for the office BP measurements was not statistically different for the three groups (72.0% vs. 78.3% vs. 79.2%, (V+C; V; C) respectively, P = .813). The changes in mean office SBP and DBP values from baseline to week 12, by study treatment group, were not statistically different (Figure 3).

Changes in mean office SBP and DBP values from baseline to week 12, by study treatment group.
With respect to the changes in the mean 24-hour ABPM values from baseline to week 12, patients assigned to all three groups had significant reductions in both SBP (difference: −9.7 ± 6.2, P = .013; −6.8 ± 3.0, P = .003; −9.5 ± 4.5, P = .004, respectively) and DBP (difference: -6.2 ± 4.8, P = .027; −4.0 ± 3.2, P = .029; −7.1 ± 3.7, P = .006, respectively). However, reductions from baseline to week 12 in 24-hour ABPM SBP and DBP were not significantly different between the three treatment groups (P = .532 and P = .400, respectively). After adjustment of multiple linear regression models, controlling for baseline variables and other potential confounders, the group to which patients were assigned was not an independent predictor of either the reduction from baseline to week 12 in 24-hour ABPM SBP (β = −1.02, 95% CI: −4.11 to 2.06, P = .511) or the 24-hour ABPM DBP (β = −0.081, 95% CI: −2.26 to 2.09, P = .941) (data not shown). However, comparison of daytime and nighttime changes in the 24-hour ABPM values (SBP and DBP) from baseline to week 12 between the three groups revealed significant differences in nighttime mean SBP values between the three groups, due to a significantly greater reduction of these values in patients assigned to the valsartan and chlorthalidone group, when compared to those assigned to the valsartan group (difference: −14.7 ± 2.5 vs. −8.7 ± 2.5 vs. −10.7 ± 1.5, P = .042, respectively) (Figure 4). Although patients assigned to the valsartan and chlorthalidone group also had a greater nighttime reduction in mean ABPM DBP values when compared with the other two groups, this difference was not statistically significantly different (difference: −9.3 ± 1.1 vs. −6.6 ± 0.6 vs. −6.3 ± 5.1, P = .466, respectively) (Figure 5).

Mean change from baseline to week 12 in mean 24-h ambulatory systolic blood pressure monitoring, by study treatment group.
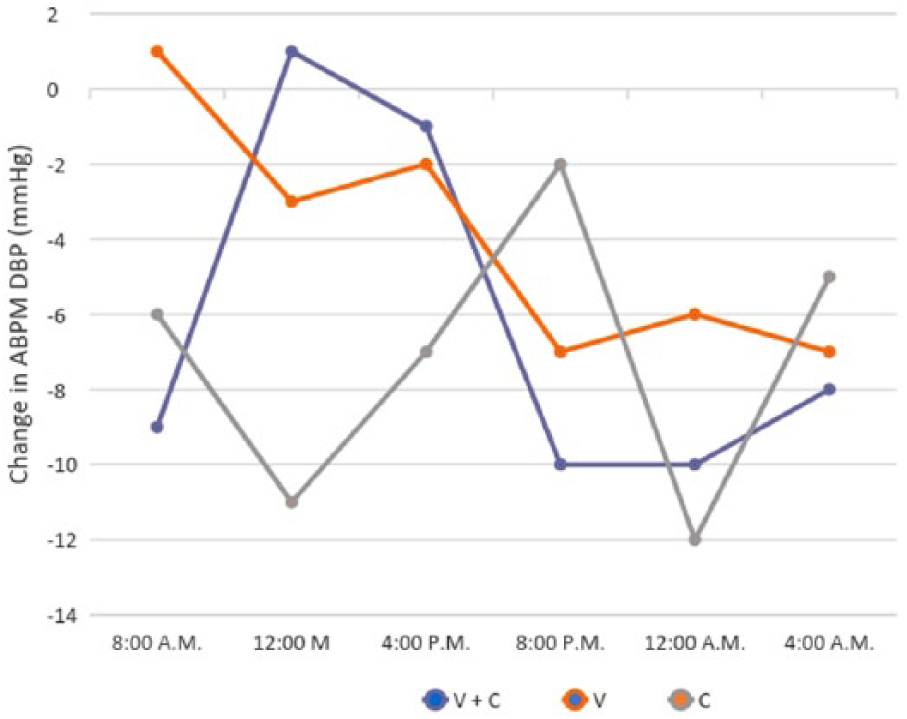
Mean change from baseline to week 12 in mean 24-h ambulatory diastolic blood pressure monitoring, by study treatment group.
Tolerability and compliance
The tolerability and rates of AEs did not differ significantly between the three groups. Of the total of 72 participants, 30 (41.7%) reported at least 1 AE (treatment-related or not) during the follow-up assessment at week 6, and 17 reported at least 1 AE (23.6%) during the follow-up assessment at week 12. In patients receiving combination therapy, 14 (35.0%) reported AEs during the V80+C12.5 treatment period, and 3 (60.0%) reported AEs during the V160+C25 treatment period. In patients receiving monotherapy with valsartan, 9 (39.1%) reported AEs during the V80 treatment period, and 3 (42.9%) reported AEs during the V160 treatment period. In patients receiving monotherapy with chlorthalidone, 13 (35.1%) reported AEs during the C12.5 treatment period, and 5 (71.4%) reported AEs during the C25 treatment period.
No clinically significant changes occurred in the laboratory values. There were no significant changes from baseline to week 6 or 12 in the values for serum creatinine, blood urea nitrogen, potassium, fasting glucose, or total cholesterol in any treatment group.
There were no significant differences in compliance rates between the three treatment groups. In the follow-up assessment at week 6, mean (SD) compliance rates were 94.8% (5.5%) for combination therapy, 94.9% (6.5%) for monotherapy with valsartan, and 95.4% (5.4%) for monotherapy with chlorthalidone. Similarly, in the follow-up assessment at week 12, mean (SD) compliance rates were 95.2% (5.5%) with combination therapy, 96.9% (4.4%) for monotherapy with valsartan, and 94.2% (8.3%) for monotherapy with chlorthalidone.
Discussion
This study provides a direct comparison of the antihypertensive effect of valsartan alone, chlorthalidone alone, and both medications combined, by using a stepped-care approach in a population of adult patients with mild to moderate hypertension. The most notable result drawn from the data is that although the proportion of patients achieving the BP control goals at week 12 was not statistically different between the three interventions when their antihypertensive efficacy was assessed with office BP measurements, a fixed combination of valsartan with chlorthalidone provided a significantly greater reduction of late night to early morning BP values when the antihypertensive efficacy of the interventions was assessed with 24-hour ABPM; this is significant because studies have shown that morning surge of SBP is a significant predictor of target organ damage and cardiovascular events such as stroke, independent of daytime office BP measurements.17–19 In addition, the findings of this study further suggest that all three interventions have similar tolerability and comparable compliance rates.
The efficacy findings of this study suggest that at least some hypertensive patients treated with valsartan have an apparently controlled BP (based on a single daytime office BP measurement), when it is actually poorly controlled during night and early morning hours, a period of time that has been identified as that within the diurnal cycle with the highest risk of stroke and other cardiovascular events. 17 Therefore, relying on daytime office BP measurements to assess the level of control in hypertensive patients treated with valsartan could overestimate the actual BP control rates, allowing physicians and patients to be lulled into a false sense of safety. An alternative explanation for these findings is that due to the exclusion of a significant proportion of patients because of technical problems in BP measurement, the study was underpowered to detect significant differences in daytime office BP measurements between the three group interventions. However, it seems highly improbable that the inclusion of all hypertensive patients in the analysis would have affected the findings, and a more logical alternative explanation is that valsartan therapy converts sustained hypertension into masked hypertension, without obtaining antihypertensive effects for a duration of 24 hours. This feature could be shared with other ARBs due to the fact that efficacy is similar. A Cochrane Database Systematic Review evaluated the dose-related trough BP reduction with 9 ARBs vs. placebo in more than 13,000 patients who had been enrolled in 46 randomized controlled trials and followed for 3–12 weeks. The average trough reduction in systolic and diastolic BP was -8 and -5 mmHg, respectively; the authors were unable to identify any single ARB that was more effective in reducing BP than others.20,21
One issue to take into account is the role of thiazide diuretics. These are among the preferred pharmacologic treatments for hypertension. HCTZ and chlorthalidone have been the two most commonly used diuretics in clinical trials and in clinical practice. However, the evidence suggests that cardiovascular outcomes are not necessarily the same with these two drugs. Chlorthalidone is around 1.5 to 2.0 times more potent than HCTZ as an antihypertensive, and chlorthalidone has a much longer elimination half-life (24-55 hours) than HCTZ (2.5 hours), a property suggested to result in more sustained BP reduction over 24 hours. 22
This longer elimination half-life could help sustain a prolonged low level of diuresis, resulting in lower mean nighttime BP, a finding which would not be readily observed in office BP measurements routinely obtained during daytime hours. Although differences in antihypertensive efficacy between HCTZ and chlorthalidone are statistically significant only with nighttime ambulatory SBP, this is a significant finding because ABPM is known to correlate more closely with the prediction of cardiovascular outcomes than office BP. In particular, nighttime ambulatory BP readings have been found to be the most predictive of cardiovascular outcomes. In the case of HCTZ, it has also been shown that office BP overestimates the antihypertensive response to HCTZ, with ABPM readings more reliably assessing antihypertensive response. 12
The findings show that the addition of chlorthalidone to valsartan positively impacts the circadian variation in BP, by producing a much better and significant decrease in late night to early morning BP values. The similar efficacy in lowering daytime BP with combined therapy and valsartan, but with greater BP lowering between midnight and early morning using combined therapy, is considered to be mainly due to the pharmacokinetic profile of chlorthalidone, a diuretic drug that has a wide volume of distribution and partitioning into red blood cells, which explains its long and smooth duration of action. 18 This action of chlorthalidone produces much better control of late night to early morning BP, which has been associated in the literature with a well-documented reduction in cardiovascular morbidity and mortality. 14 This is because ideal control of BP should guarantee a therapeutic coverage over the entire 24-hour period, especially during late night and early morning hours, because studies have shown that morning surge of SBP is a significant predictor of target organ damage and cardiovascular events such as stroke, independent of daytime office BP measurements. 19 The importance of this circadian variation in BP is such that late night and morning surge in BP has been proposed as a specific therapeutic target for preventing target organ damage and cardiovascular events in hypertensive patients. 23 Although this was a short-term study and could not establish the impact of any of the three interventions in long-term cardiovascular outcomes, we are confident that the present findings have made some progress toward enhancing our knowledge of efficacy, safety, and tolerability of two of the most commonly prescribed antihypertensive drug classes, namely ARBs and diuretics, and their combination, in a population of hypertensive adult patients. It is fundamental to note that we have gained experience in the treatment of patients with hypertension with chlorthalidone (alone and in combination with ARBs), a diuretic drug that has clear advantages over HCTZ, the most prescribed diuretic for treating patients with hypertension.
Our results are consistent with previous findings that use of office BP measurements to compare the efficacy of valsartan monotherapy and valsartan + chlorthalidone in a population of 4,555 Brazilian hypertensive patients found comparable rates of response between monotherapy and combination therapy (70.8% vs. 72.6%, respectively). 24 In the same manner, our results are in line with those reported in the VICTORY trial, in which 365 patients were treated with valsartan or a combination of valsartan and HCTZ for 16 weeks; using office BP measurements, the proportion of patients that achieved target BP at the last visit was not significantly different between the two groups (98 vs. 84%, respectively). 25 With respect to the greater reduction in late night and early morning BP, values in chlorthalidone-treated patients (alone and in combination with other agents) are consistent with those reported by Ernst et al, 12 who found a significant reduction in SBP during nighttime hours with chlorthalidone compared to that observed on using HCTZ. Similarly, Pareek et al 13 compared the efficacy of chlorthalidone and two preparations of HCTZ in patients with stage 1 hypertension, and found that compared to the baseline values, all three treatments significantly lowered office BP at weeks 4 and 12; however, at weeks 4 and 12, significant reductions in systolic and diastolic 24-hour ambulatory and nighttime BP were observed with chlorthalidone but not with HCTZ.
With respect to tolerability, the rates of AEs found in our study were comparable to those reported in other similar studies that also reported AEs, irrespective of the relationship to study medications, but greater than those reported in other studies that only reported AEs considered related to the study medications. 12 Additionally, we found that diuretics and ARBs given as monotherapy have comparable rates of AEs as those when given in combination therapy, as reported in other comparable studies. 24
The major limitation of this study is the above-mentioned low power for detection of significant differences with use of the study medications. Additionally, the open-label design could have influenced the results. However, we consider that this study provides valuable insights for clinicians and patients, providing confidence regarding the efficacy, safety, and tolerability of two of the most commonly prescribed antihypertensive drug classes, and their combination, in treatment of hypertension.
In conclusion, the current study showed that in a population of adult patients with mild to moderate hypertension treated with valsartan alone, chlorthalidone alone, and both medications combined, the fixed combinations of valsartan with chlorthalidone provided a significantly greater reduction of late night to early morning BP values when the antihypertensive efficacy of the interventions were assessed using 24-hour ABPM. The findings of this study further suggest that all three interventions have similar tolerability and comparable compliance rates.
Footnotes
Acknowledgements
The study was done at Centro Diagnostico Cardiologico, Cartagena (Colombia), Medplus, Bogota (Colombia), Fundacion Cequin, Armenia (Colombia)
Author Contributions
JDR participated in discussions of study design, data analysis, and manuscript writing. AR participated in data collection and analysis. CERM participated in statistical analysis and manuscript writing. FM designed the study and participated in data analysis and manuscript writing.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Juan Romero is a full-time employee of Farma. Dr. Carlos Rodriguez-Martinez has received partial compensation for statistical analysis. Data entry and statistical analyses were supported by Farma. Dr. Manzur and Dr. Rico report no potential conflict of interest relevant to this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by FARMA laboratories.
