Abstract
We present cardiac computed tomography (CT) findings demonstrating apical hypertrophic cardiomyopathy with dystrophic calcification of the left ventricular apex. The absence of significant epicardial coronary artery disease demonstrated by coronary CT angiography suggests that increased wall tension and decreased microvascular perfusion over time account for the dyskinetic apical myocardium, rather than myocardial infarction secondary to atherosclerotic plaque rupture. These observations support CT as the imaging modality of choice to visualize the deposition of calcium in injured myocardial tissue, a recognized occurrence in chronically infarcted myocardium.
Case Report
A 76-year-old woman with atrial fibrillation, chest pain, dyspnea, and 1 prior episode of syncope presented to our medical center for pulmonary vein and coronary artery evaluation prior to an ablation procedure. Noncontrast cardiac computed tomography (CT) showed no coronary artery calcification, however, demonstrated circumferential myocardial calcification of the left ventricular apex (Figure 1). Coronary CT angiography showed minimal (<30%) noncalcified plaque in the mid left anterior descending coronary artery. Evaluation of left ventricular morphology revealed a subtle spade-shaped cavity with mild apical hypertrophy as well as apically displaced and hypertrophied papillary muscles (Figure 2). Multiphase CT cine imaging showed partial systolic obliteration of the left ventricular apex and dyskinesis of the distal apical segments. The aforementioned findings noted on cardiac CT appeared less conspicuous on transthoracic and transesophageal and echocardiography (Figure 3).
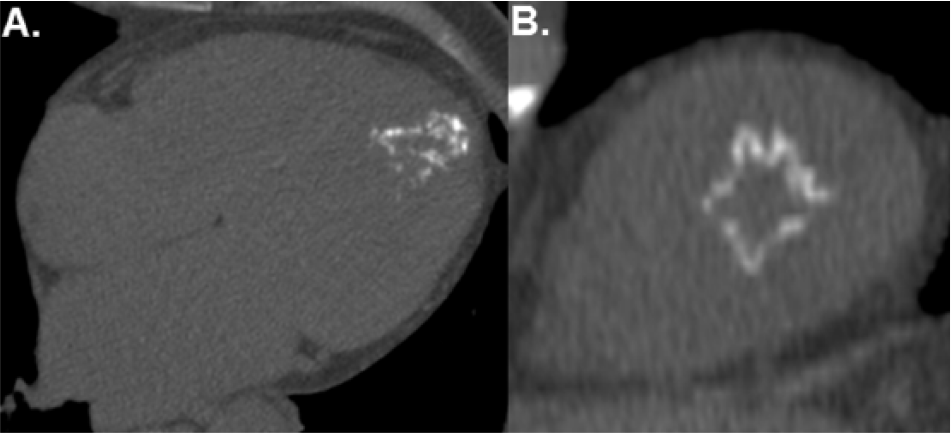
Noncontrast cardiac computed tomography. (A) The 4-chamber view shows calcification of the apical left ventricular myocardium. (B) The short-axis view of the left ventricular apex shows circumferential calcification of the apical myocardium.
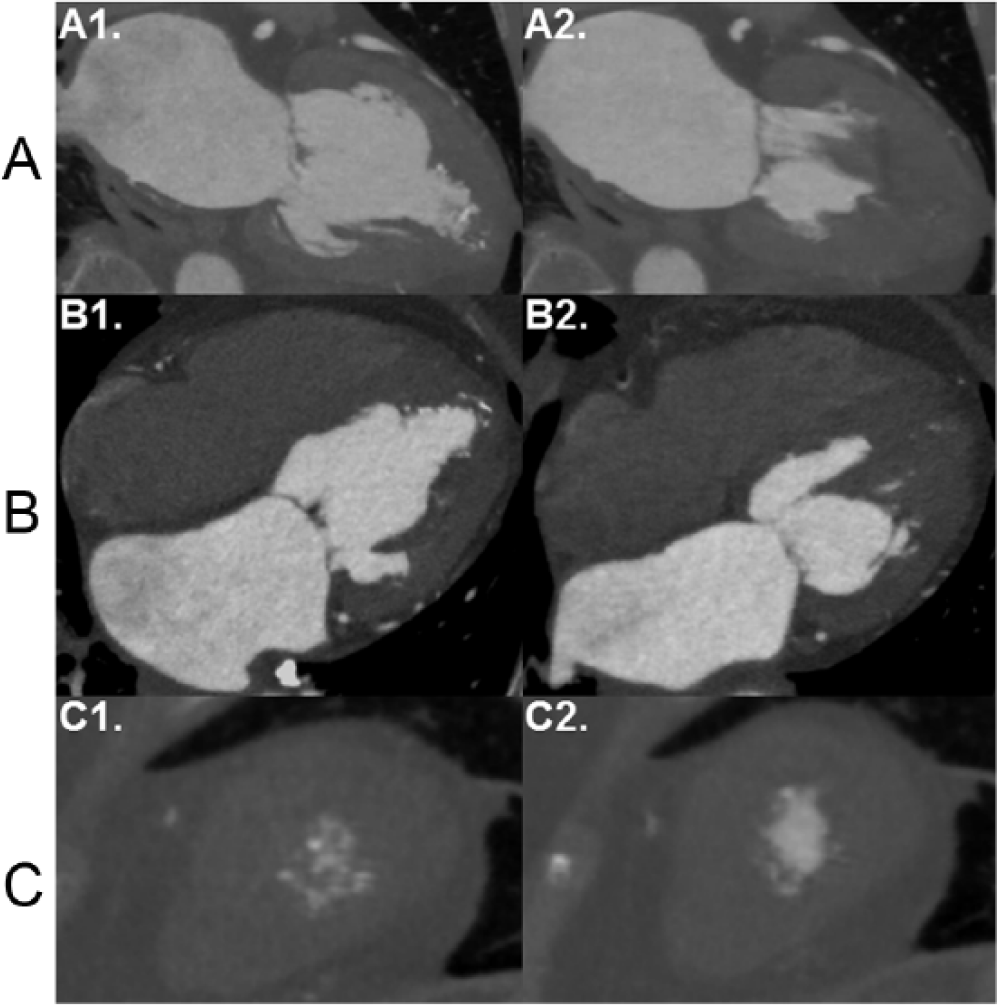
Contrast-enhanced gated cardiac computed tomography. (A) The 2-chamber view at (A1) end-diastole and (A2) end-systole shows a spade-shaped left ventricular cavity with mild apical hypertrophy and partial systolic cavity obliteration. (B) The 4-chamber view at (B1) end-diastole and (B2) end-systole demonstrates apically displaced and hypertrophied papillary muscles. (C) Contrast-enhanced short-axis views at (C1) end-diastole and (C2) end-systole show a slightly larger apical cavity at end-systole, consistent with dyskinetic myocardium.

Echocardiography. (A) Transesophageal and (B) transthoracic echocardiography demonstrate increased echogenicity at the left ventricular apex, however, less conspicuous compared with the calcification noted on noncontrast cardiac computed tomography.
Discussion
These overall cardiac CT findings demonstrate the potential hemodynamic sequelae of apical hypertrophic cardiomyopathy.1,2 Prior reports describe cases of apical hypertrophic cardiomyopathy with calcified myocardium 3 ; however, the mechanism remains unknown. We postulate that increased left ventricular filling pressure in the setting of known microvascular disease associated with hypertrophic cardiomyopathy 4 leads to decreased microvascular perfusion pressure. Episodic elevations in circulating catecholamines further increase myocardial wall tension and reduce perfusion pressure. These hemodynamic effects induce ischemia and potentially the subsequent pathophysiologic cascade of infarction, fibrosis, and dystrophic calcification. The circumferential apical calcification, the most striking finding in this case, heralded the ultimate diagnostic impression. The calcified subendocardium represents a marker of chronically infarcted myocardial tissue, 5 a finding optimally demonstrated by cardiac CT. Ascribing the apical wall motion abnormalities to the hemodynamic consequences of hypertrophic cardiomyopathy, however, necessitates evaluation of the coronary arteries. The absence of significant epicardial coronary artery disease argues against atherosclerotic plaque rupture as the underlying cause of apical dyskinesis.
Conclusions
This case of apical hypertrophic cardiomyopathy demonstrates dystrophic calcification of the apical myocardium, ultimately a pathophysiologic consequence of decreased myocardial perfusion in the absence of signficant epicardial coronary artery disease. Calcification represents a marker of chronically infarcted myocardial tissue, histologic sequelae optimally characterized by cardiac CT.
Footnotes
Peer Review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totaled 545 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SGS and NR analyzed the data. SGS wrote the first draft of the manuscript. JNM contributed to the writing of the manuscript. SGS, JNM, NR, and RJ agree with manuscript results and conclusions and made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
As a requirement of publication, author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality, and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
