Abstract
This narrative review aims specifically to explore the relationship between tobacco exposure and systemic sclerosis (SSc), idiopathic inflammatory myositis (IIM), and systemic lupus erythematosus (SLE). Relevant articles were obtained by searching key terms such as “tobacco,” “smoking,” “scleroderma,” “myositis,” “lupus,” and “Sjögren’s” in PubMed and Google Scholar databases. The selected articles ranged from the years 2010 to 2023. Inclusion criteria were based on the relevance and contribution to the field of study. Systemic sclerosis is a complex condition involving multiple immune cell lines that can be influenced by tobacco. However, the existing literature does not provide sufficient evidence to support an increased risk of SSc in smokers or the impact on treatment options. Cigarette smoking does increase the risk of skin ulcerations in SSc patients. In addition, cigarette smoking has been associated with IIM through genetic and molecular mechanisms. Smokers with dermatomyositis or polymyositis are at an elevated risk of atherosclerosis and interstitial lung disease. Similarly, smoking in patients with SLE increases the risk of organ damage, thrombosis, and disease severity compared with non-smokers. Smokers with SLE also have more difficulty in controlling disease flares compared with non-smokers. Tobacco exposure can lead to secondary complications in patients with IIM and SLE, although the course of treatment may not differ significantly. No definitive conclusions can be drawn to the clear relationship between tobacco smoking and Sjögren’s’s syndrome.
Introduction
Tobacco smoking is a prevalent phenomenon with an estimate of around 1.14 billion people worldwide smokers in 2019. 1 Tobacco smoke contains more than 4000 chemical compounds, many of which are harmful to the human health. One smoking-associated death occurs for approximately every 0.8 to 1.1 million cigarettes smoked. Tobacco smoke may cause toxic cell damage leading to intra- and extracellular reactions.2,3 Through multiple pathophysiological mechanisms, nicotine, an addictive compound of tobacco, has been linked to numerous diseases, many of which are chronic immune-mediated disorders. 4 Beside the environmental factors, genetic factors play an important role in the link between nicotine use and induction of immune-mediated diseases. 5 Systemic sclerosis (SSc) has a complex pathogenesis that usually involves innate and adaptive immunity with specific functional autoantibody production leading to irreversible damage. 6 Such a cascade can be a target to nicotine. Likewise, dermatomyositis (DM), polymyositis (PM), and systemic lupus erythematosus (SLE) has variable pathogenesis that involves numerous cells, which can be modulated by nicotine exposure. E-cigarettes have been also linked to multiple diseases, including rheumatic diseases. For example, vaping has been rarely linked to skin manifestations of SLE. 7 However, no data exist on the relationship between e-cigarettes and SSc, DM, or PM.
In this review, we shed light on the role of cigarette smoking in SSc, DM, PM, and SLE. We will discuss the pathophysiological mechanisms involved. Nonetheless, we will report significant clinical presentations that smokers with SSc, DM, PM, or SLE may have as well as management aspect differences than the routine standard of care.
Body
This is a narrative review. Relevant articles were gathered by searching key terms such as “tobacco,” “nicotine,” “smoking,” “scleroderma,” “myositis,” and “lupus” in PubMed and Google Scholar databases, covering the years 2010 to 2023. Inclusion criteria were based on relevance and contribution to the field of study, with all chosen articles in the English language. Old articles were excluded due to outdated information. The selected studies within these articles encompassed experimental (in vivo and in vitro) as well as translational studies.
Systemic sclerosis
Systemic sclerosis is characterized by widespread dysfunction of the vascular system associated with progressive fibrosis of the skin and internal organs. Although nicotine by itself does not induce SSc, diverse mechanisms can substantiate disease activity especially vascular dysfunction leading to a negative impact on the clinical manifestations.
Smoking may increase neutrophil chemotaxis by 93% compared with baseline. 8 This mechanism happens in conjunction to a significant increase in macrophage reactivity and metalloelastase manifestation. 9 Moreover, smoking induces interleukin (IL)-8 and tumor necrosis family (TNF)-α liberation. 10 Elevated IL-6 levels have also been observed in smokers. 11 Apart from inflammation, the modulation of immune system has been observed. For example, nicotine can suppress the Th1 and Th17 responses via an interaction between CD4+ T-cells and the α7 nicotinic acetylcholine receptor. 12 Nevertheless, nicotinic signaling has a definite impact on CD40 pathway influencing B-cell activation throughout different stages of cell life, 13 and has a negative outcome on the regenerative ability of mesenchymal and ligament-derived stem cells. 14
Genetic modifications, most commonly single nucleotide polymorphism, play a key role in the pathogenesis of scleroderma via the effect on various human body cells. 15 Single nucleotide polymorphisms in HLA-DQB1 and associated genes increase the risk of developing SSc.16,17 Precisely, different mutations are related to distinct autoantibodies 18 (Figure 1). Epigenetics as in DNA methylation, histone modifications, and non-coding RNA have been shown to be a critical predisposing factor for scleroderma. 19 There is not enough research showing evident effect of tobacco smoking on the genetic alterations listed above.
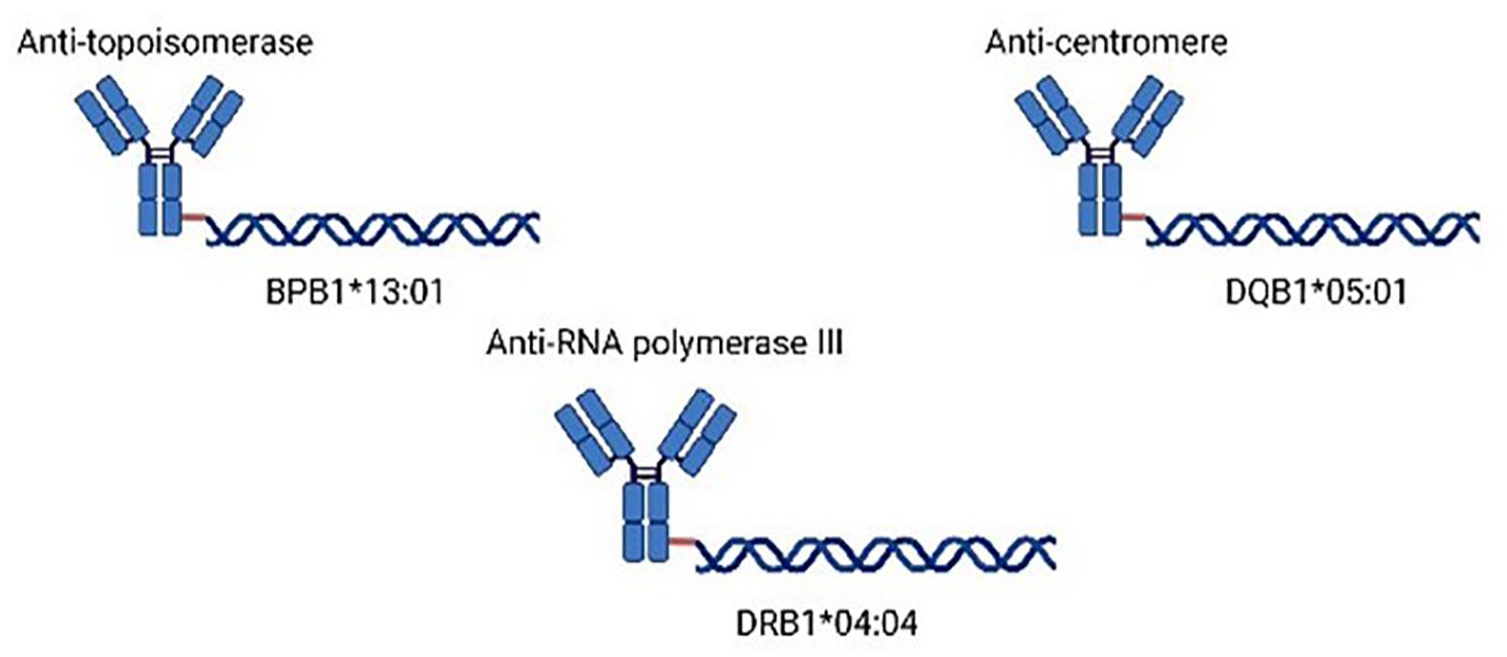
Human leukocyte antigen mutations with the respective autoantibody production in systemic sclerosis.
While some data suggest that digital ulcers are significantly more common among SSc smokers than SSc non-smokers, 20 other data indicate no relevant association or insignificant statistical association.21,22 One study reported a significant decrease in skin presentation severity, recording a 16% fall in skin factor for every unit increase in comprehensive smoking index. 23 This finding could be rationalized by the finding that nicotine leads to premature death of the skin and impaired angiogenesis,24,25 two mechanisms also affected in scleroderma. 26 Raynaud’s phenomenon pattern and severity have been also linked to cigarette use, including nicotine. 27 There is significant relationship between severity of Raynaud’s phenomenon and presence of digital ulcers in SSc smokers. 28
Besides, nicotine use significantly increases the prevalence and complications of obstructive and restrictive lung disease among SSc patients.29,30 Respiratory parameters such as total volume capacity, compliance, and static recoil are all affected in SSc smokers. 31 Interestingly, certain pieces of evidence did not find a relevant increased probability of developing pulmonary hypertension. 32 However, nicotine use increases the odds of developing lung cancer in patients with SSc, although the risk is similar to patients with no SSc. 33
Nicotine use has been linked to gastrointestinal distress in SSc. Systemic sclerosis smokers are more likely to develop poor appetite. 34 On the other hand, sleep disturbance and smoking in SSc have a significantly positive association (r = 0.27, P = .025). 35 Similarly, fatigue is more pronounced in SSc smokers. 36
Nicotine use can alter the effect of novel SSc treatments. For example, autologous hematopoietic stem cell transplantation success rate has been shown to be decreased. 37 Although biologics, in particular anti-TNF agents, were reported to be less effective in the context of smoking whenever used in patients with ulcerative colitis 38 and Crohn’s disease, 39 these agents are not used in patients with SSc.
Arthrodesis, also known as artificial ankylosing, is an effective treatment used for limb anatomical abnormalities caused by SSc. 40 No data are available to report the impact of nicotine use on arthrodesis among SSc patients.
Dermatomyositis/polymyositis
Evidence for gene-environment interactions in IIM has been found between the HLA-DRB1*03 gene locus and smoking in White populations. 41 Compared to never smokers who were HLA-DRB1*03:01 allele negative, ever-smokers with HLA-DRB1*03:01 allele positive had 16 times increased odds of anti-Jo-1 antibody positivity. 42 The exact mechanism by which anti-Jo antibodies and positive HLA associations contribute to the pathophysiology of PM and DM still needs further investigation. One of the observed mechanisms in PM and DM patients involves immune complex deposition in myocytes formed by anti-Jo-1 antibodies and RNA. 43 Immune complexes can induce endogenous production of Intereferons (IFN) in plasmacytoid dendritic cells inside myocytes which in turn promotes inflammation and the release of inflammatory mediators.
Another suggested mechanism of inflammation involves the citrullination of proteins present in endothelial and inflammatory cells in the muscles of patients with PM. 44 Myocytes of patients with PM are rich with citrullinated protein. Citrullination could be triggered by smoking which precipitates the formation of anti-citrullinated proteins/peptide antibodies in genetically susceptible individuals. 45 Yet, the exact mechanism is not clear and might be an inflammation-dependent rather than myositis-dependent phenomenon, especially as citrullinated components are reported to be found in other autoimmune diseases such as rheumatoid arthritis, Inflammatory Bowel Disease (IBD), and chronic tonsilitis. 45
In addition, maternal tobacco smoking is strongly associated with the development of juvenile DM. 46 Air pollution and tobacco smoking contain a mixture of particles and gases including carbon monoxide (CO), and a mother’s exposure to CO particularly during the third trimester may contribute to the onset of juvenile DM. The exact mechanism of how CO contributes to the pathophysiology of PM or DM is unclear. The role of CO in epigenetic modification of the human monoblastic leukemia U937 cell lines through CO of the induced change in the methylation status of histone H3 proteins and chromatin modification has been identified. 47 These results suggest a possible role of CO in the development of smoking-associated myositis that could be addressed in future studies.
There is concordant evidence that tobacco smoking increases the disease burden of patients diagnosed with PM and DM. In essence, the prevalence of cardiovascular events (such as severe coronary artery atherosclerosis, dyspnea, and chest pain) is higher in IIM patients compared with healthy controls after the adjustment of age and gender. 48 The odds of developing severe coronary artery atherosclerosis were 3.04 times higher among current smokers and 2.95 higher among former smoking in the cohort of IIM patients. The pathophysiology of smoking in promoting cardiovascular events is not clear. One hypothesis is that smoking plays a role in aggravating systemic hypertension associated with autoimmunity of IIM, which is mediated by autoantibodies against adrenergic receptors and angiotensin II. 49 These autoantibodies can promote the development of systemic hypertension while also cross-reacting with myositis autoantigens to stimulate an inflammatory response in the affected myocytes.
Tobacco smoking is also associated with the development and progression of interstitial lung diseases (ILDs) seen in patients with PM and DM. Nicotine is thought to play a role in lung fibrogenesis in a concentration-dependent manner. 50 Mechanisms of lung fibrosis include epithelial and endothelial damage, an increased number of eosinophils in the trachea with a production of pro-inflammatory cytokines, and activation of transforming growth factor-β that promotes collagen deposition in the lung parenchyma. 50 Yet, many of the above mechanisms are mainly described in murine lungs or human hepatic cell lines and need further investigation in the human lung parenchyma.
The cornerstone in the management of PM and DM starts initially with glucocorticoids. 51 The common practice is to start with prednisolone while closely following up with the patients for dose adjustment and education about the adverse effects of glucocorticoid use. Glucocorticoid-sparing therapy, such as methotrexate, azathioprine, cyclophosphamide, and calcineurin inhibitors (cyclosporine and tacrolimus), can be used in patients who are poorly responding to glucocorticoid or developing severe adverse effects related to glucocorticoid use. Similar to other autoimmune disorders, the emerging therapeutic techniques in treating different subtypes of IIM are progressing toward using biological agents as first-line treatment. Clinical trials seem to be promising and multiple immunotherapeutic agents have shown promising results. Rituximab, an anti-CD-20 (anti-B-cell), is effective in IIMs with variable efficacy among patients. 52 In addition, the same study discussed the effectiveness of monoclonal antibodies that target T-cells CD-52 in the treatment of PM.
In all cases, the choice of therapy seems to be independent of the smoking status of the patients and no evidence shows a treatment difference between smokers and non-smokers. Yet, understanding the pathophysiology of smoking-induced IIM that involves the endogenous production of IFN gamma suggests a promising target therapy. Sifalimumab, an anti-IFN monoclonal antibody, appeared to be promising in a phase 1b clinical trial in IIMs and correlated with improvement in muscle testing. 52
SLE and antiphospholipid syndrome
Systemic lupus erythematosus is a multisystem autoimmune disease characterized by the body’s immune system attacking its own healthy tissues. It has been associated with specific autoantibodies, most commonly being anti-nuclear antibodies that result in inflammation. 53 The pathogenesis of the disease depends on genetic and environmental factors. There are many ways in which environmental exposure may contribute to SLE pathogenesis.
Among the environmental factors, cigarette smoking plays a pivotal role in SLE. Early data suggested that smoking is a strong risk factor for development of SLE, 54 which has been also confirmed by recent data. 55 Tobacco smoking contributes significantly to an oxidative stress state. Such oxidative stress involves intracellular organelles, such as mitochondria and endoplasmic reticulum, with increased concentrations of inflammatory markers, impairment of T-cell and B-cell function, and epigenetic modifications that yield autoimmune-inducing effects, although there is no clear evidence for the association with SLE. 53 There are numerous toxic compounds contained in cigarette smoke that may generate free radicals. It has been estimated that cigarette smoke contains more than 1015 free radicals per puff. 56 A great deal of damage is obtained on the exposure to such chemicals that can affect endogenous proteins and DNA. 57
In addition, free radicals can elevate the count of autoreactive B-cells, and increase the FS-7-associated surface antigen (FAS) receptor (CD95) on surface of B and CD4 T lymphocytes. 53 These cells will overreact to stimuli and undergo apoptosis when not needed. This will lead to an imbalance between inflammatory and anti-inflammatory cytokines 58 (Figure 2).

Main mechanisms that cigarette smoking exposure can contribute to systemic lupus erythematosus pathophysiology.
Smoking is one of the factors that may lead to wide-ranging phenotypic differences among SLE patients. 59 Tobacco smoking is related to worse cutaneous manifestations. 60 Moreover, SLE smokers tend to have accelerated development of end-stage renal disease. 61
The Systemic Lupus International Collaborating Clinics/the American College of Rheumatology Damage Index (SLICC/ACRDI) has been used as an index to assess SLE damage. 62 By using the SLICC/ACRDI, a positive correlation between the severity of the symptoms and smoking exposure was found. Some of these adverse symptoms include nephropathy, proteinuria, and cardiovascular diseases. 63
In addition, antiphospholipid antibodies (aPL) positivity is more evident among smokers than non-smokers leading to an exacerbated thrombo-inflammation and atherothrombosis. 64 These events take place through the production of several mediators such as cytokines, adhesion molecules, and oxidized low-density lipoprotein-β2GPI complex which in turn activate different cell types that include neutrophils, platelets, macrophages, monocytes, and the complement system. Ultimately, these micro-events lead to antiphospholipid syndrome (APS) manifestations. 64 Interestingly, tobacco smoking can contribute to growth inhibition during the early stage of pregnancy leading to fetal death. 65 Nicotine exposure has been linked to criteria and non-criteria APS manifestations. It has been considered an independent risk factor for obstetric APS. 66 Nevertheless, certain non-criteria APS manifestations have been linked to smoking, one of which is livedo reticularis. 67
There is significant difference in treatment effect in SLE smokers compared with non-smokers. Tobacco smoking can decrease the efficacy of antimalarials, especially when used for cutaneous symptoms. 68 Despite no effect on the metabolism or excretion of the medication, nicotine interacts with antimalarials on the lysosomal level minimizing its intracellular effects.69-71
Belimumab is one of the effective treatments used for SLE. It is a monoclonal antibody that is targeted to neutralize the effect of a molecule that activates B lymphocytes which enhance the pathogenicity of the disease. The targeted molecule belongs to the B-cell activating factor (BAFF) family of TNF. Thus, it is suspected when belimumab neutralizes this molecule the symptoms of SLE should cutback.72-74 It was found that the effect of belimumab is limited when it is given to SLE smokers. 75 Although the effect is not very clear, tobacco smoking might decrease the effect of glucocorticoids on SLE flares. 76
Sjögren’s syndrome
Sjögren’s syndrome (SS) has been associated with multiple etiologies, such as viral infections. 77 However, tobacco smoking has never been denoted as a clear risk factor for SS. Although tobacco smoking has been rarely associated with a longer duration of the disease, parotidomegaly, polyautoimmunity, peptic ulcer disease, and coffee consumption among SS patients, smoking has been also associated with a reduced risk of developing primary SS.78,79 Nevertheless, nicotine use has been significantly associated with lower rates of focal lymphocytic sialadenitis and low rates of positive biopsy results when compared with never smokers. 80 There is no clear pathophysiological link between tobacco smoking and SS. Similarly, the guidelines of management are indifferent between SS smokers and non-smokers.
Limitations
As this is a narrative review, many limitations do exist. Authors may have selected studies that support their own viewpoints or omit studies that contradict them. In addition, there was no standardized methodology for literature search, selection, and appraisal, making it difficult to replicate the review process. There was no formal quality assessment of the included studies, which can result in the inclusion of low-quality or flawed research. Complex issues, such as pathophysiological processes, might have been oversimplified in an attempt to provide a broad overview, potentially overlooking important nuances and details.
Conclusion
Cigarette smoking is hypothesized to affect the pathophysiology of SSc, although there is no clear link regarding causality. Systemic sclerosis smokers are more likely to develop skin ulcerations and lung disease than non-smokers. Further work is needed to establish an effect of cigarette smoking on the therapeutics of SSc as well as SS. Multiple mechanisms exist to suggest a contribution of nicotine to IIM. Whether by HLA alleles, citrullination of proteins, or CO-induced epigenetic changes, there is data that suggest a strong relationship between cigarette smoking and establishment of IIM. As for clinical presentation, cigarette smoking seems to be related to ILD and coronary atherosclerosis. There is not enough evidence to suggest that cigarette smoking might affect IIM management. The visceral organ damage in SLE is mediated by free radicals which release is potentiated by cigarette smoking. Smoking exposure increases severity of SLE and increases risk of thrombosis via aPL. In addition, it might decrease the effect of glucocorticoids during flares.
