Abstract
Objective:
To scope the current published evidence on cardiovascular risk factors in rheumatoid arthritis (RA) focusing on the role of autoantibodies and the effect of antirheumatic agents.
Methods:
Two reviews were conducted in parallel: A targeted literature review (TLR) describing the risk factors associated with cardiovascular disease (CVD) in RA patients; and a systematic literature review (SLR) identifying and characterizing the association between autoantibody status and CVD risk in RA. A narrative synthesis of the evidence was carried out.
Results:
A total of 69 publications (49 in the TLR and 20 in the SLR) were included in the qualitative evidence synthesis. The most prevalent topic related to CVD risks in RA was inflammation as a shared mechanism behind both RA morbidity and atherosclerotic processes. Published evidence indicated that most of RA patients already had significant CV pathologies at the time of diagnosis, suggesting subclinical CVD may be developing before patients become symptomatic. Four types of autoantibodies (rheumatoid factor, anti-citrullinated peptide antibodies, anti-phospholipid autoantibodies, anti-lipoprotein autoantibodies) showed increased risk of specific cardiovascular events, such as higher risk of cardiovascular death in rheumatoid factor positive patients and higher risk of thrombosis in anti-phospholipid autoantibody positive patients.
Conclusion:
Autoantibodies appear to increase CVD risk; however, the magnitude of the increase and the types of CVD outcomes affected are still unclear. Prospective studies with larger populations are required to further understand and quantify the association, including the causal pathway, between specific risk factors and CVD outcomes in RA patients.
Keywords
Introduction
A large body of evidence suggests patients with rheumatoid arthritis (RA) and other autoimmune inflammatory diseases face increased cardiovascular disease (CVD) risk. 1 The precise scope of this increased risk is not clear: most current estimates indicate the range between 1.5 and 2 times higher than age- and sex-matched individuals from the general population. 2 One of the main challenges in investigating the elevated CVD risk in RA is understanding the interplay between traditional and novel CVD risk factors with RA pathology itself. 3
Although the prevalence of some traditional CVD risk factors is increased in patients with RA, extending the general population approach to CVD risk estimation in the RA population is flawed.4,5 For example, the most commonly used clinical instruments for estimating CVD risk scores, such as the Framingham Risk Score and the European SCORE system, underestimate CVD risk in RA by as much as 2-fold. 2 The main reason for this discrepancy is related to the presence of novel CVD risk factors uniquely associated with RA progression and their synergy with traditional factors in elevating CVD risk.
Treatment history can change CVD risk assessment in RA patients with some treatment regimens having moderate effect on CVD risk, while the others show more pronounced effect.6-9 For instance, conventional therapies such as methotrexate (MTX) and glucocorticoids appear to have a modest cardio-protective effect.10,11 Recently introduced biologics and small molecule disease-modifying anti-rheumatic drugs (DMARDs) may decrease CVD risk, 9 but the mechanism is poorly understood. 6
A prominent clinical feature of seropositive RA is the presence of elevated levels of autoantibodies, including, but not limited to, rheumatoid factor (RF), anti-citrullinated peptide antibodies (anti-CCP), and others. The specific role of these autoantibodies in increasing CVD risk is still under investigation. Some studies have found that elevated RF levels do not predict increased CVD risk 12 while other studies have found a direct link between increased RF levels and coronary heart disease. 13 The presence of anti-CCP titer seems to be more directly related to increased CVD risk in RA 14 ; however, the contribution of specific citrullinated proteins to atherosclerotic processes is still under investigation. 15
Our objective was to describe the current landscape of published evidence on CVD risk in RA, including traditional and novel CVD risk factors, the association between autoantibodies and CVD risk in RA and the effects of antirheumatic therapies.
Methods
Two literature reviews were conducted in parallel. A targeted literature review (TLR) was conducted to identify risk factors and describe the effects of antirheumatic therapies on the development of CVD in RA patients. Doctor Evidence LLC’s proprietary search platform (DOC SearchTM), which contains MEDLINE®, clinicaltrials.gov, WHO ICTRP, EPAR, Daily Med, and RSS feeds was used as the primary database for searches (date of search: from inception to January 2019). This was supplemented by a search of the American College of Rheumatology (ACR) and European League Against Rheumatism (EULAR) conferences for proceedings between 2017 and 2019.
A systematic literature review (SLR) was conducted to identify and characterize the association between autoantibody status and CVD risk in RA. Searches were performed on MEDLINE®, Embase, and Cochrane CENTRAL (from inception to January 15, 2019). This was supplemented by a search of conference proceedings of the ACR and EULAR, as well as the United States and European Union clinical trial registries for entries on trials with results available (2017-2019). Reference lists of previously published relevant SLRs were hand searched. Search strategies are presented in Supplemental Tables 1 to 3.
Screening for the TLR was guided by the Population, Intervention, Comparator, and Outcome (PICO) framework. 16 Population for both reviews included adult patients with RA; in addition, for the TLR the criteria required presence of CVD comorbidities and for the SLR the evidence synthesis was stratified by autoantibodies status. Interventions and comparators were any or none. Outcomes in the TLR included CVD comorbidities, presence of CVD risk factors, any additional comorbidities, and mortality. For the SLR the outcomes included incidence, prevalence, and severity of cardiovascular diseases and cardiovascular events, stratified by autoantibody status.
The titles/abstracts were screened by a senior methodologist. Eligible studies proceeded to full-text screening phase where they were assessed for inclusion by the same methodologist. Articles deemed eligible at this stage were included in the qualitative evidence synthesis.
For the SLR, title/abstract and full-text screening was conducted by 2 senior methodologists. Discrepancies were identified and resolved by a third, independent investigator. Data extraction of the included studies was performed by the same 2 investigators in duplicate. Data extracted included study and patient characteristics, autoantibodies, therapeutic interventions and comparators, and the outcomes of interest. Quality control auditing was performed to ensure the accuracy and consistency of data across the dataset. The Cochrane Collaboration’s tool 17 and Newcastle-Ottawa Quality Assessment Scale 18 were used to assess the risk of bias in randomized studies and cohort studies, respectively.
Results
A total of 69 publications (49 via the TLR and 20 via the SLR) were eligible for inclusion in the narrative evidence synthesis. The selected publications in the TLR included 19 systematic literature reviews, 28 clinical studies (21 prospective and 7 retrospective) and 2 guidelines (EULAR and ACR). For the SLR, the 20 studies selected for data extraction included 7 prospective, 11 retrospective, and 2 cross-sectional. The PRISMA diagrams for both TLR and SLR are presented in Figure 1A and B, respectively.

PRISMA diagrams for: (A) the TLR and (B) the SLR.
CVD risk factors pre-RA diagnosis
Several recent studies noted that the risk factors of CVD often predate the onset of RA.19-22 The published literature highlights that many patients diagnosed with RA already have significant CVD pathologies at the time of diagnosis. In particular, atherosclerosis of carotid vasculature, which has an established causal relationship with CVD, seems to be present early in the natural history of the CVD.23,24 Lifestyle as well as prior history of smoking and obesity were associated with an increased likelihood of developing RA. 25 In a case-control study of 547 pre-symptomatic patients in Sweden, Kokkonen et al 21 found several CVD risk factors present in individuals years before the onset of the RA symptoms and that one-third of all the pre-symptomatic individuals had at least 3 factors present. Kerola et al 20 examined subclinical cardiovascular changes in pre-RA population. Morphological evidence of arterial wall atherosclerosis was present during the first year following RA diagnosis suggesting the process started before the clinical diagnosis of RA itself. Davies et al 19 who used carotid ultrasound to determine cardiovascular risk and prevalence of subclinical atherosclerosis at the time of diagnosis of RA, found that at the time of diagnosis, over 40% of the patients had either plaque or carotid intima-media thickness >0.9 mm, both considered markers of high CVD risk. Van Boheemen et al 22 followed a cohort of 594 consecutive arthralgia patients with positivity for RF and/or anti-CCP autoantibodies over a period of 10 years to assess the time point at which CVD risk increases prior to the RA diagnosis. The authors found that although dyslipidemia present in arthralgia patients predicted the development of RA, the differences in lipid profiles were too small to have an effect on the 10-year risk of CVD morbidity and mortality.
CVD risk factors post-RA diagnosis
Traditional CVD risk factors identified post-RA diagnosis largely overlaps with the CVD risk factors in the general population as follows:
Novel CVD risk factors, those related to systemic pathological processes in RA and their relation to increased CVD risks, are as follows:
Effects of drug therapy on CVD risk in RA
Association of autoantibodies with CVD in RA
Among the 20 references included in the SLR, 18 were published full-text articles52-69 and 2 were conference abstracts.70,71 This evidence base consisted of 7 prospective observational studies,52,61-63,65,70,71 11 retrospective observational studies,53-59,66-69 and 2 cross-sectional studies.60,64
Definition of RA varied across the studies: 15 studies used the definition by ACR or EULAR criteria,53-55,57,58,60,61,63-70 2 studies identified RA through medical records,56,59 1 study reported early RA as defined by multiple symptoms and criteria, 52 1 study included patients with self-reported RA, 62 and 1 meeting abstract did not specify the RA definition. 71 In all included studies, the risk of bias was low across all categories, indicating high-quality of the included studies (Supplemental Table 4).
Cardiovascular outcomes varied across studies. Total cardiovascular events or cardiovascular diseases were the most frequently reported outcomes, stratified by autoantibody status (n = 9), followed by cardiovascular death (n = 7). Other cardiovascular outcomes included stroke (n = 4), thrombosis (n = 4), coronary artery disease (n = 2), heart failure (n = 2), ischemic heart disease (n = 2), peripheral artery disease (n = 1), diastolic dysfunction or valvular lesions (n = 1), and thromboembolism (n = 1).
Four major types of autoantibodies were evaluated for their association with CVD risk in the RA population: RF, anti-CCP, anti-phospholipid, and anti-lipoprotein (Table 1).
Studies reporting on CVD risks in RA patients.
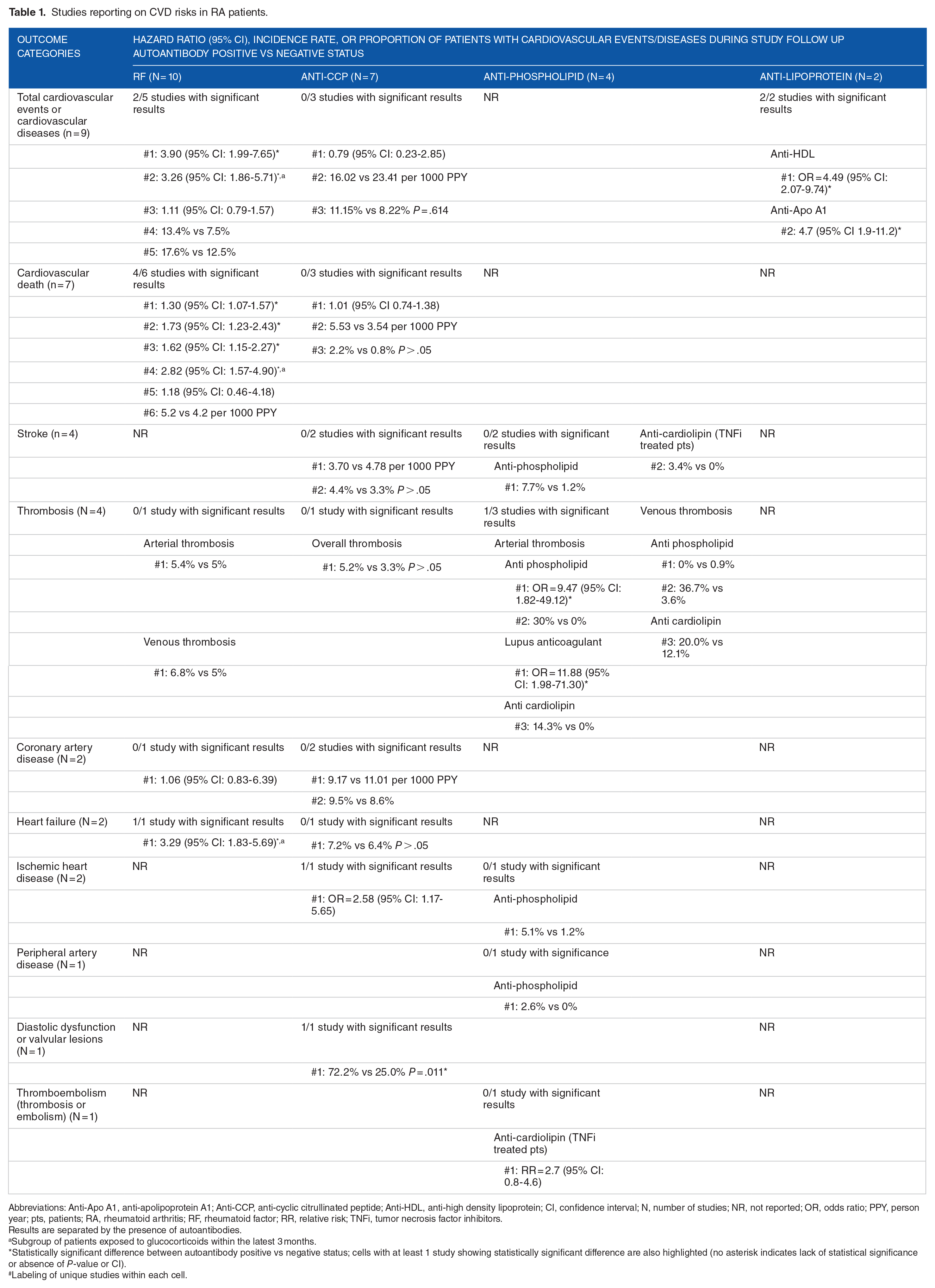
Abbreviations: Anti-Apo A1, anti-apolipoprotein A1; Anti-CCP, anti-cyclic citrullinated peptide; Anti-HDL, anti-high density lipoprotein; CI, confidence interval; N, number of studies; NR, not reported; OR, odds ratio; PPY, person year; pts, patients; RA, rheumatoid arthritis; RF, rheumatoid factor; RR, relative risk; TNFi, tumor necrosis factor inhibitors.
Results are separated by the presence of autoantibodies.
Subgroup of patients exposed to glucocorticoids within the latest 3 months.
Statistically significant difference between autoantibody positive vs negative status; cells with at least 1 study showing statistically significant difference are also highlighted (no asterisk indicates lack of statistical significance or absence of
Labeling of unique studies within each cell.
Ten studies reported cardiovascular outcomes stratified by RF autoantibody status.53-55,57,59,62,63,66-68 In RA patients, the presence of RF autoantibody was associated with a higher risk of total cardiovascular events or CVD, cardiovascular death, heart failure, and especially for patients who had recent or high dose exposure to glucocorticoids.
Seven studies reported cardiovascular outcomes stratified by anti-CCP autoantibody status.52,55,56,61,62,70,71 The presence of anti-CCP autoantibody was associated with a higher risk of ischemic heart disease, diastolic dysfunction, or valvular lesions. There was also a trend of higher risk of cardiovascular death for anti-CCP positive patients, but the difference was either not statistically significant or not tested for significance.
Four studies reported cardiovascular outcomes stratified by anti-phospholipid autoantibody status.58,60,65,66 The anti-phospholipid autoantibodies discussed include anti-cardiolipin,58,65,66 lupus anticoagulant,60,66 and general anti-phospholipid positive status defined by being positive for either of these 2 autoantibodies.60,66 The presence of anti-phospholipid autoantibodies, including anti-cardiolipin and lupus anticoagulant, were associated with a higher risk of thrombosis, especially arterial thrombosis. This finding is not surprising, since the association of these autoantibodies with thromboembolism has been found in other inflammatory and autoimmune rheumatic diseases. 72 The mean prevalence of anti-phospholipid autoantibodies in RA has been found at 28% and the median was 22%. 73 In a recently published systematic review, authors suggested that there are not enough data to support routine screening for antiphospholipid antibodies in RA, with a possible exception for pregnancy planning and thrombosis in the context a rheumatic disease. 72
Two studies reported cardiovascular outcomes stratified by anti-lipoprotein autoantibody status.64,69 The anti-lipoprotein autoantibodies discussed include anti-HDL 64 and anti-Apo A1, 69 a component of the HDL particles. The presence of anti-lipoprotein autoantibodies, including anti-HDL and anti-ApoA1, was associated with a higher risk of experiencing cardiovascular events.
Discussion
Our review contributes to the growing body of evidence of synergistic effects of traditional and novel CVD risk factors in RA patients. Our main goal was to review the cardiovascular and metabolic comorbidities in RA patients and summarize the current understanding of common mechanisms behind inflammation, atherosclerosis, and CVD diseases, in particular shared metabolic pathways involved in both pathologies. Similar emphasis on the role of inflammation can be seen in several recently published reviews. Lauper and Gabay 29 surveyed recent advancements in the understanding of the interplay between inflammation and lipid metabolism in the development of atherosclerosis in the RA population. Skeoch and Bruce 4 surveyed the role of inflammatory cytokines such as TNF alpha in modulating the traditional CVD risk factors as well as in directly impacting the vasculature. Common conclusion across these studies is that inflammation plays a crucial role in the pathogenesis of both accelerated atherosclerosis and RA and that inflammation has to be one of the targets of all therapeutic approaches.
From therapeutical point of view, 1 important finding is the fact that changes leading to the increased CVD risk start long before the clinical manifestation of RA. It is now clear that some pathological processes predate the RA diagnosis. Subclinical changes in endothelial dysfunction and atherosclerosis have been documented early in the RA disease,19,20 suggesting that the processes started before the clinical RA diagnosis. In this context, it has been suggested that elevated level of RF plays the same role in increasing cardiovascular mortality risks in early stages of arthritis as it does in later stages of the disease. In fact, RF seropositivity in the absence of arthritis has also been shown to be associated with increased rates of cardiovascular mortality.13,74
It is therefore critical to identify these early risks and initiate appropriate treatment as soon as possible. The exacerbation of atherosclerosis 75 during the course of RA progression is closely linked with chronic systemic inflammation 4 ; however, the precise mechanism leading to increased CVD risk is still under investigation. 3 One possible explanation involves the link between the level of inflammation and the disease activity, which then, in turn, is strongly predictive of both CVD events and cardiac function. 10 Clinical trials investigating arterial inflammation using FDG-PET imaging showed increased FDG aortic uptake in RA patients.76,77 The central role of the inflammation is also supported by the mitigative effects of anti-inflammatory treatment regimens on the CVD risk.7,46,49,78,79 The double-impact of inflammation on both RA progression and increased CVD risk led some authors to suggest that treatment with biologic DMARDs (such as TNF alpha inhibitors) amounts to “killing 2 birds with 1 biological stone.” 80 Currently ongoing TARGET trial (https://targetra.org) is aimed at understanding how different treatments for RA affect cardiac inflammation.
Joint evaluation of the role of autoantibodies in CVD risk assessment in our SLR is a novel contribution to the field. Considering the evidence reporting statistically significant difference between the autoantibody positive and negative patients, a few findings from this SLR warrant further evaluation. In RA patients, the presence of RF autoantibody was potentially associated with higher risk of cardiovascular death, heart failure, and total cardiovascular events or cardiovascular diseases, especially for patients who had recent or high dose exposure to glucocorticoids. The presence of anti-CCP autoantibody was potentially associated with higher risk of ischemic heart disease, diastolic dysfunction, or valvular lesions.
From a clinical perspective, this review suggests that presence of autoantibodies could potentially be associated with increased risk of specific cardiovascular outcomes, with different risk profile for different types of autoantibodies. For example, the hazard ratio for cardiovascular death between RF positive and RF negative patients was reported in 5 studies, with significant difference observed in 4 studies. No such ratio was observed in studies stratifying patients by anti-CCP status (see Table 1). Patients with anti-CCP seropositivity face increased risk of ischemic heart disease. Studies reporting on anti-phospholipid autoantibodies found significant risk increase of thrombosis (both arterial and venous) as well as stroke in seropositive patients. The increased risk of cardiovascular events associated with specific autoantibodies should be taken into consideration when deciding on a personalized treatment regimen.
A promising approach to analyzing the role of autoantibodies in RA is through genetic analysis. It is very likely that HLA-DR genes act indirectly through the regulation of autoantibody production involved in the development and outcome of RA. 43 One potential explanation for inconclusive results with autoantibodies as described in the SLR is that the RA populations consist of several serological subphenotypes defined by polymorphisms outside the HLA-DRB1 locus. 45
To ensure high sensitivity and specificity of the findings, we followed recommendations from Cochrane Handbook for Systematic Reviews of Interventions. 16 The review was focused on the most recent and relevant publications in clinical trial evidence, real-world setting, and systematic reviews therefore it did not summarize the trends over time. The goal was to capture current understanding of risk factors rather than produce an exhaustive survey of all published evidence. The review does not attempt to reconcile contradictory findings found in the literature or resolve open questions related to the relative role of CVD risk factors or the effects of therapy on CVD risk in RA. As such, it represents a snapshot of current evidence and the discussions related to the topic of CVD in RA.
Conclusion
Morbidity and mortality remain a major concern among RA patients due to increased risk of CVD. Traditional CVD risk factors, systemic inflammation, and RA disease-specific factors contribute to the accelerated risk of CVD in RA. This review suggests that the presence of autoantibodies may be associated with increased risk of specific CVD outcomes, with a different risk profile for different types of autoantibodies. The increased risk of cardiovascular events associated with autoantibodies, such as higher risk of cardiovascular death in RF positive patients, warrants further evaluation for clinical significance in RA patients. Studies in larger RA populations with a prospective design are required to further understand and quantify the association between specific risk factors and CVD outcomes in RA patients.
Supplemental Material
sj-pdf-1-amd-10.1177_11795441211028751 – Supplemental material for Cardiovascular Disease in Rheumatoid Arthritis: Risk Factors, Autoantibodies, and the Effect of Antirheumatic Therapies
Supplemental material, sj-pdf-1-amd-10.1177_11795441211028751 for Cardiovascular Disease in Rheumatoid Arthritis: Risk Factors, Autoantibodies, and the Effect of Antirheumatic Therapies by Mir Sohail Fazeli, Vadim Khaychuk, Keith Wittstock, Boris Breznen, Grace Crocket, Mir-Masoud Pourrahmat and Leticia Ferri in Clinical Medicine Insights: Arthritis and Musculoskeletal Disorders
Footnotes
Acknowledgements
We would like to thank Yuting Kuang for her scientific input and work on parts of the project.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review was funded by Bristol Myers Squibb.
Declaration of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Leticia Ferri, Vadim Khaychuk, and Keith Wittstock report employment by Bristol Myers Squibb. Grace Crocket reports employment by Joulé Inc., which is contracted by Bristol Myers Squibb. Additionally, Leticia Ferri is a Bristol Myers Squibb shareholder. Mir Sohail Fazeli, Boris Breznen, and Mir-Masoud Pourrahmat report employment by Evidinno Outcomes Research Inc., which was contracted by Bristol Myers Squibb to conduct this review.
Author Contributions
Leticia Ferri and Mir Sohail Fazeli contributed to the study design and conceptualization; Boris Breznen and Mir-Masoud Pourrahmat carried out data configuration; Boris Breznen, Mir-Masoud Pourrahmat, and Mir Sohail Fazeli wrote and prepared the original manuscript draft; and Leticia Ferri, Vadim Khaychuk, Keith Wittstock, and Grace Crocket were responsible for reviewing and editing the final draft.
Ethics Approval and Consent to Participate
An Ethical Review Committee Statement was not applicable for this study.
Availability of Data and Materials
Not applicable. The data used for analysis was retrieved from openly published studies listed in our manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
