Abstract
The consecutive reports and stored images of ultrasound examinations for 100 symptomatic ankles of 74 patients with rheumatoid arthritis (RA) were reviewed for the presence or absence of retrocalcaneal bursitis (RCB) and Achilles tendon enthesitis (ATE). The ankles were classified into 4 categories based on the presence or absence of RCB or ATE. The number of RCB(−)/ATE(−), RCB(+)/ATE(−), RCB(+)/ATE(+), and RCB(−)/ATE(+) ankles was 62, 16, 12, and 10, respectively. When classifying patients into early RA and established RA, the percentage of RCB(−)/ATE(+) ankles with early RA was significantly lower than that with established RA (P = .00595). The disease duration was significantly longer in the RCB(−)/ATE(+) ankles than in the RCB(+)/ATE(−) ankles (median [interquartile range]: 15.29 [8.69] months vs 3.6 [3.06] months, P = .0247). It was speculated that RCB precedes or accompanies ATE in the early phase of RA, which suggests that entheseal inflammation in RA arises from synovial tissues.
Introduction
Enthesitis is known as a hallmark of spondyloarthritis (SpA). However, the growing application of ultrasound (US) to various musculoskeletal disorders increases our opportunity to encounter enthesitis in arthritides other than SpA. 1 We had previously reported the frequencies of various pathologies detected by US in symptomatic ankles in patients with rheumatoid arthritis (RA) by reviewing the 74 consecutive reports of US examination for 100 symptomatic ankles. 2 Through that survey, we also recognized that Achilles tendon (AT) involvement is not rare in RA. Achilles tendon involvement including retrocalcaneal (RC) bursitis, AT enthesitis, AT tendonitis, and AT paratenonitis was detected in 39% of the symptomatic ankles examined.
Whether or not the prevalence of enthesitis is consistently higher in SpA versus RA remains controversial. 1 Regardless of the age of report or the modalities used, conflicting data have been reported until very recently.3–5 Although it has been suggested that enthesitis in RA may be secondary to articular synovial inflammation or based on the involvement of synovial membrane lining the tendon sheaths and the bursae, no direct evidence has yet been reported. 6 In this study, we aimed to find a clue to the fundamental inflammatory process underlying AT enthesis in patients with RA by analyzing the association between the presence of RC bursitis or AT enthesitis and clinical data.
Methods
The consecutive reports and the stored images of US examinations for 100 symptomatic ankles of 74 patients with RA were reviewed for the presence or absence of RC bursitis and AT enthesitis. The ethics committee of the Mitsui Memorial Hospital approved this retrospective study. All patients had fulfilled the 2010 ACR/EULAR criteria for RA and underwent US examination because of clinical need. An experienced sonographer/rheumatologist (T.S.) performed US examinations mainly using a GE LOGIQ 7 device (GE Medical Systems, Milwaukee, WI, USA) as described previously. 2 A 10- to 14-MHz linear transducer was used, mainly at 12.0 MHz for gray scale and 6.7 MHz for color mode. Power Doppler US was performed using standard methods with a pulse repetition frequency of 0.9 to 1.1 kHz.
The AT was examined with the patient lying prone and the feet hanging over the edge of the examination table at 90° of flexion according to musculoskeletal US guidelines and evaluated according to the MAdrid Sonographic Enthesis Index (MASEI).7,8
In this study, RC bursitis was defined to be present when the bursa was distended to >2 mm in the short axis with effusion and/or thickened synovium, regardless of the coexistence of power Doppler (PD) signals. Achilles tendon enthesitis was narrowly defined as active inflammation at the entheseal insertion and to be present when there was at least one of the following findings: hypoechogenicity of the enthesis, thickening of the enthesis (maximal thickness on the calcaneal insertion >5.29 mm), or the presence of PD signals up to 2 mm from the cortical bone. Bone erosions, enthesophytes, and calcifications, which are thought to represent types of damage, were excluded from the positive findings for AT enthesitis. 9 The sonographer (T.S.) determined the presence or absence of each positive finding by evaluating 100 sets of the stored images of the ankles those were anonymized and randomized to protect the identity of the individuals.
To verify intra- and interobserver reliability, randomized 33 sets of 1 to 5 images each for the ankle were rescored by 2 experienced sonographers (T.S. and Y.S.) after the acquisition of the data. Unweighted κ-statistics were calculated. The association between the presence of RC bursitis or AT enthesitis and clinical data was analyzed using the Fisher’s exact test or the Steel-Dwass test.
Results
Prevalence of US-detected RC bursitis and AT enthesitis in RA
The patients consisted of 52 women and 22 men (median age 63.3 years, range 26-83 years) with a median disease duration of 4.2 months (range 0.23 months to 19.4 years), as described previously. 2 Precisely, 74% of these patients were positive for rheumatoid factor and/or anticitrullinated protein antibody. Among the overall 100 ankles, the frequencies of RC bursitis and AT enthesitis were 28% and 22%, respectively. Both were positive in 12% of the 100 ankles. The ankles were classified by the presence or absence of RC bursitis or AT enthesitis into 4 categories as shown in Figure 1. The percentage of seropositive patients with RC bursitis-negative/AT enthesitis-positive ankles was 78%, which is comparable with that of overall patients.
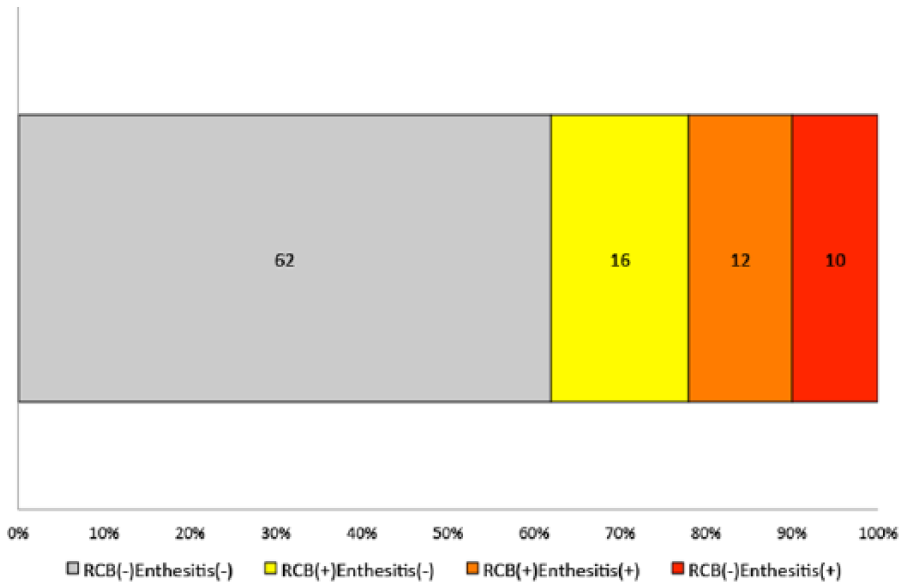
Prevalence of ultrasound-detected retrocalcaneal (RC) bursitis and Achilles tendon (AT) enthesitis among the symptomatic ankles with rheumatoid arthritis. The 100 symptomatic ankles were classified based on the presence or absence of RC bursitis or AT enthesitis into 4 categories. The number of ankles in each category is shown on the chart. RCB indicates retrocalcaneal bursitis; enthesitis, Achilles tendon enthesitis.
The intra- and interobserver κ values for the presence of retrocalcaneal bursitis (RCB) were 0.94 and 0.88, respectively. The intra- and interobserver κ values for the presence of AT enthesitis were 0.82 and 0.76, respectively. The intra- and interobserver κ values for the 4 categories were 0.88 and 0.76, respectively. Representative US images of RC bursitis and AT enthesitis are shown in Figure 2.

Representative ultrasound (US) images of the Achilles tendon (AT) enthesis of symptomatic ankles with rheumatoid arthritis. (A) Retrocalcaneal (RC) bursitis without AT enthesitis. RC bursa (*) dilatated with synovial effusion and thickened synovium is depicted. (B) RC bursitis with AT tendinitis and enthesitis. RC bursa (*) dilatated with synovial effusion and thickened synovium is surrounded by power Doppler (PD) signals. Increased thickness of AT with intratendinous PD signals adjacent to RC bursa and the enthesis insertion is also depicted (arrowhead). (C) RC bursitis with AT enthesitis. RC bursa (*) dilatated with synovial effusion and thickened synovium is surrounded by PD signals. Increased thickness and hypoechogenicity of AT adjacent to the enthesis insertion is also depicted (arrowhead). (D) AT enthesitis without RC bursitis. Calcaneal bone erosion (arrow) at the enthesis insertion with PD signal is depicted.
Difference of the prevalence of RC bursitis and AT enthesitis between early RA and established RA
When classifying patients into those with early RA (duration <6 months) and those with established RA (duration >6 months), the 100 ankles in the 74 patients were categorized as 62 ankles in 47 patients with early RA (median disease duration 2.8 months) and 38 ankles in 27 patients with established RA (median disease duration 17.4 months). The percentages of 4 categories based on the presence or absence of RC bursitis or AT enthesitis differed between the early RA and established RA as shown in Figure 3A. Interestingly, the percentage of RC bursitis-negative/AT enthesitis-positive ankles with early RA was significantly lower than that for ankles with established RA (P = .00595). Similarly, the percentages of 4 categories when classifying patients into those untreated and those already treated are shown in Figure 3B. The percentage of RC bursitis-negative/AT enthesitis-positive ankles of untreated patients tended to be lower than that of already treated patients, although the difference was not statistically significant.

Difference in the prevalence of retrocalcaneal (RC) bursitis and Achilles tendon (AT) enthesitis related to the phase or status of the disease. The 100 symptomatic ankles were classified according to (A) the disease duration (early RA or established RA) or (B) by the treatment status (untreated or already treated). The percentages of the 4 categories based on the presence or absence of RC bursitis or AT enthesitis are shown. The number of ankles in each category is shown on the chart. RCB indicates retrocalcaneal bursitis; enthesitis, Achilles tendon enthesitis.
Association between RC bursitis or AT enthesitis and clinical data
Clinical data were compared between the 4 categories classified by the presence or absence of RC bursitis or AT enthesitis. The disease duration was significantly longer in the RC bursitis-negative/AT enthesitis-positive ankles than in the RC bursitis-positive/AT enthesitis-negative ankles (median [interquartile range (IQR)]: 15.29 [8.69] months vs 3.6 [3.06] months, P = .0247; Figure 4A). Serum C-reactive protein was significantly lower in the RC bursitis-negative/AT enthesitis-positive ankles than in the RC bursitis-positive/AT enthesitis-negative ankles (median [IQR]: 0.335 [0.775] mg/dL vs 2.41 [7.30] mg/dL, P = .0479; Figure 4B).

Comparison of clinical data between the 4 categories classified by the presence or absence of retrocalcaneal (RC) bursitis and Achilles tendon (AT) enthesitis. (A) The disease duration and (B) the levels of serum C-reactive protein were compared between the 4 categories. RCB indicates retrocalcaneal bursitis; enthesitis, Achilles tendon enthesitis.
Discussion
Achilles tendon enthesis is also known as the premier enthesis, the prototype of the synovio-entheseal complex, which has been advocated in the entheseal organ concept by McGonagle et al.6,10 They have suggested that the inflammation occurs primarily at the enthesis and spreads to adjacent synovial tissues such as bursae in patients with SpA. Our cross-sectional observation of symptomatic AT revealed that active enthesitis in the narrow definition as represented by the presence of PD signals up to 2 mm from cortical bone was rarely seen without RC bursitis in the early phase of RA; instead, RC bursitis was frequently observed with or without AT enthesitis. Although this is not a longitudinal study, we can speculate from our observation that the inflammation around the AT enthesis of patients with RA occurs primarily at the synovial tissue, ie, RC bursa, and spreads to the enthesis, supporting the concept proposed by McGonagle et al.
In addition, the isolated AT enthesitis without RC bursitis in the established and/or treated patients may suggest the following possibilities: (1) enthesitis is more refractory to RA treatment than is bursitis, (2) enthesitis is partially due to the degenerative changes related to damage and deformities caused by RA synovitis, and (3) US-detected enthesitis in RA basically represents a repair process rather than ongoing inflammation. The lower levels of serum C-reactive protein are consistent with these explanations.
Conclusions
Retrocalcaneal bursitis precedes or accompanies AT enthesitis in the early phase of RA, suggesting that inflammation of the synovio-entheseal complex in RA arises from synovial tissues.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
TS conceived and designed the experiments, wrote the first draft of the manuscript, and contributed to the writing of the manuscript. TS and YS analyzed the data, jointly developed the structure and arguments for the paper, and made critical revisions and approved final version. TS, YH, and YS agree with manuscript results and conclusions. All authors reviewed and approved the final manuscript.
