Abstract
Background
New therapeutic cessation approaches are being tested in clinical trials to engage and retain people who smoke. Our team is conducting a pragmatic randomized clinical trial (RCT) to evaluate a new treatment for tobacco dependence, but enrolling participants and ensuring adherence has been more challenging than in previous trials.
Objective
To determine the predictors of enrollment and adherence in the RCT.
Design
A secondary analysis of data from a tobacco cessation RCT.
Methods
Data was collected during a two-group RCT testing the efficacy of an integrative guided imagery vs behavioral treatment, with both conditions involving six weekly, hour-long sessions over 9 weeks.
Results
Of the 1074 randomized participants, 803 (74.8%) enrolled (completed the first session), and 631 (78.6%) of those were adherent (attended all scheduled sessions). Variables associated with enrollment included age (adjusted odds ratio [AOR], 1.01; 95% CI, 1.0003-1.02; P = 0.04) and state of residence (West Virginia vs New York (NY), AOR, 0.66; 95% CI, 0.46-0.94; P = 0.02), with older participants and those from NY more likely to enroll. Variables associated with adherence included race (Black vs White) (AOR, 2.09; 95% CI, 1.05-4.16; P = 0.04), higher education (at least some college vs high school or less) (AOR, 2.27; 95% CI, 1.58-3.26; P < 0.0001), marital status (all others vs single/never married) (AOR, 1.66; 95% CI, 1.11-2.48; P = 0.01), and state of residence (Arizona vs NY) (AOR, 0.52; 95% CI, 0.34-0.78; P = 0.002). Within Arizona, older age increased enrollment, while higher education and marriage improved adherence. Within NY, higher education was associated with both increased enrollment and adherence, while higher number of household smokers, and not reporting substance use were associated with increased adherence.
Conclusion
Enrolling and retaining people who smoke in cessation trials requires novel strategies. Identifying predictors of enrollment and adherence offers valuable insights for overcoming barriers in future tobacco cessation RCTs.
Introduction
Despite the detrimental health effects of smoking, tobacco use remains the leading cause of preventable disease, disability, and death in the U.S. 1 An estimated 50.6 million or 20% of U.S. adults population uses tobacco and 4.5% of the U.S. adult population uses electronic cigarettes (e-cigarettes). 2 In 2014, the annual cost of tobacco use in the U.S. was US$332 billion and the cost continues to increase each year. 3 Although, tobacco cessation results in a nearly universal improvement in health, and evidence-based treatments for tobacco use are effective for many, there are multiple limitations, including a substantial variability in outcomes and limited reach. 4 In fact, about 66% of people who smoke attempt to quit, but only 10% achieve cessation. 5 Randomized controlled trials (RCTs) are used to test novel tobacco cessation treatments aimed at improving cessation rates. Enrolling and retaining participants in these RCTs is becoming more complex. Thus, gaining greater understanding of the predictors on enrollment and adherence is needed.
Some previous work has been done to highlight differences in participants that enroll vs not enrolled (not interested in participating), 6 and predictors of treatment adherence in smoking cessation clinical trials. 7 The limited extant literature on predictors of enrollment in smoking-cessation trials suggest that education,6,8 race, 9 intervention modality, 10 and motivation to stop smoking may influence the decision to participate in a smoking cessation clinical trial. 9 Predictors of adherence to tobacco cessation medication is associated with education, race, co-occurring mental health conditions, treatment-related side effects, and low degree of tobacco dependence. 11 Additionally, previous research indicates that adding behavioral activation components to conventional smoking cessation treatment compared to conventional treatment alone, increases adherence and overall abstinence rates. 12 However, predictors of both enrollment and adherence in smoking cessation clinical trials remain understudied.
The Be Smoke Free study is a first-of-its-kind randomized tobacco cessation clinical trial that examines the efficacy of a telephone-delivered, integrative guided imagery (GI) and behavioral treatment for smoking cessation compared with a standard, evidence-based behavioral-only treatment. 4 Details regarding the study design and methods have been previously published. 4 Research indicates that integrated approaches, when delivered remotely or online, can enhance adherence to vital components (eg, skills training, social support, and pharmacotherapy use) to improve cessation. 13 We have experienced greater challenges with enrollment into the study and lower adherence to the study protocol than in the randomized feasibility trial upon which the RCT was based. Therefore, to develop strategies to enhance future enrollment and adherence, we chose to conduct preliminary analyses from our large sample to investigate factors associated with enrollment in the study and adherence to the study protocol.
This study analyzed data from our Be Smoke Free randomized tobacco cessation clinical trial
4
to determine the predictors of enrollment into the study and adherence among enrolled participants. The following questions guided this study: 1) What are the predictive variables associated with potential participants enrolling in a smoking cessation clinical trial? 2) What are the predictive variables associated with adherence among individuals enrolled in a smoking cessation clinical trial?
Methods
Design
This was a secondary analysis of a data collected during a two-group RCT testing the efficacy of an integrative GI treatment vs an evidence-based behavioral treatment.
Data source and permissions
The data came from Gordon et al. 4 ’s Be Smoke Free randomized tobacco cessation clinical trial from October 2022 to July 2024 (Trial Registration: ClinicalTrials.gov NCT05277831, and International Registered Report Identifier (IRRID): PRR1-10.2196/48898). The Be Smoke Free study was funded in September 2021 (National Center for Complementary and Integrative Health, R01AT011500; PI: Gordon, J.) and approved by the University of Arizona institutional review board (#2103633455: Principal Investigator [PI] Gordon, J.). Participants are recruited from three states (Arizona, New York State, and West Virginia). Participants are recruited from two populations: (1) individuals who contact one of our collaborating quitlines; or (2) population-based recruitment (eg, post on social media sites, network with our contacts in each state, and use of a participant recruitment firm) among individuals who smoke in Arizona (AZ), New York State (NY), or West Virginia (WV). Participants are stratified by state and method of recruitment (eg, quitline or community) and then randomized automatically in Research Electronic Data Capture (REDCap) using a randomization table created by the study biostatistician. Statistical software was used to create a randomized-block allocation to the GI condition or the behavioral condition, stratified by recruitment method and location. The questionnaires were validated and used in a previous study.5,14 The only incentive provided was a US$10 gift card for completing the baseline survey. No incentives were provided for attending sessions.
Inclusion and exclusion criteria
The inclusion criteria for the Be Smoke Free study includes: primary tobacco use is smoking; smoke daily for the last 30 days; aged at least 18 years; speak English; have a smartphone with SMS text messaging; agree to telephone coaching and SMS text message reminders; can download an mp3 or mp4 file; live in Arizona, New York State, or West Virginia.
The exclusion criteria for the Be Smoke Free study includes: no phone, no texting capability, does not speak English, more than one person per household, psychosis, use of quitline services in the past 12 months, current use of any tobacco cessation program or medication, significant memory impairment due to dementia, traumatic brain injury, or other cognitive impairment.
Variables of interest
Enrollment
Enrollment is defined as completing the first treatment session in the study. Interested participants are screened by trained study staff over the telephone or via REDCap. Eligible participants complete the baseline survey and informed consent over the telephone or on REDCap, prior to be randomized into the study. Written informed consent was collected electronically and stored in REDCap. Study staff send condition-specific program materials to the randomized participants. Ineligible participants are transferred back to the quitline in their state of residence using a warm handoff (eg, study staff call the quitline, talk to enrollment staff, and then transfer the caller directly to the quitline enrollment staff) or a HIPAA-compliant electronic process (eg, encrypted email).
Additionally, to explore the reasons why potential participants did not enroll in the study, we also analyzed data from REDCap collected by study coaches who were responsible for scheduling and completing the first treatment session with those who were randomized in the study.
Adherence
Adherence is defined as attended all scheduled sessions, six for those who have completed the study. Additionally, to elucidate the findings associated with adherence status, we analyzed responses to the 3-month follow-up survey question: “Please tell us why you didn’t have all 6 coaching sessions”.
Participant demographic information
The demographic information that was collected from all participants included (eg, age, gender, education, primary language, number of household smokers, marital status, mood disorder, anxiety, substance use, cannabis use, state).
Data analysis
Participant’s characteristics were summarized using mean ± standard deviation for continuous variables and frequency (%) for categorical variables by enrollment/adherence status. Wilcoxon rank-sum tests were performed to compare continuous variables between groups. Chi-square tests were performed to compare categorical variables between groups. In addition, logistic regression was performed to identify the variables associated with enrollment and adherence, respectively. For both enrollment and adherence, variables with a univariate (unadjusted) analysis p of <0.05 were included in the multivariate (adjusted) analysis. The focus was on identifying the variables associated with enrollment/adherence, not better predicting enrollment/adherence or identifying the potential confounders. Also, some subgroups have a relatively small sample size. Those were the reasons a more restrictive univariate P-value of <0.05 was used to select variables to be included in the multivariate (adjusted) model. When we used a univariate P-value of <0.10 to decide the variables to include in the adjusted model, similar variables associated with enrollment/adherence were identified even if more variables were included in the adjusted model. The enrollment analysis was based on all randomized participants and by state. The adherence analysis was based on the enrolled participants and by state. R 4.4.0 was used to conduct statistical analysis. In addition, the study coaches’ notes and participants’ open-ended responses to a 3-month survey question were qualitatively analyzed to elucidate the reasons for non-enrollment and non-adherence.
Results
Sample characteristics by enrollment and adherence status
Sample Demographics.

HS = high school, AZ = Arizona, NY = New York, WV = West Virginia.
Variables associated with overall enrollment status
Multivariate Analysis of Variables Associated With Enrollment by State.

HS = high school, AZ = Arizona, NY = New York, WV = West Virginia.
Variables associated with enrollment by state
The variables associated with enrollment status for the state of Arizona included: age (older age = more likely to enroll) (adjusted OR, 1.04; 95% CI, 1.01-1.07; P = 0.002). The variables associated with enrollment status for the state of New York included: education (at least some college vs high school/less than high school) (adjusted OR, 1.57; 95% CI, 1.04-2.37; P = 0.04). West Virginia did not show any significant variables associated with enrollment status.
Additional findings associated with enrollment
To explicate the findings associated with enrollment, the REDCap data collected by study coaches was analyzed to describe why randomized participants did not enroll in the study (ie, did not complete first treatment sessions). Study coaches completed 266 participant contact notes, describing the reasons why randomized participants did not enroll in the study. We qualitatively analyzed the study coaches’ notes. Overall, the most commonly noted reasons why randomized participants did not enroll in the study (ie, did not complete first treatment sessions) were: 1) “unable to contact” (134/266, 50.4%); 2) “no longer interested (no reason given) (75/266, 28.2%); 3) “ineligible (did not smoke every day for the past 30 days/already quit or already using other cessation program/nicotine replacement therapy) (34/266, 12.8%), and 4) “did not have first session within 1 month of randomization” (23/266, 8.6%). Figure 1 displays the most frequently cited reasons for why randomized participants did not enroll in the study. For additional details, please refer to the supplementary file (Figure S1. CONSORT Diagram). Reasons for non-enrollment in the study.
Variables associated with overall adherence status
Multivariate Analysis of Variables Associated With Adherence by State.
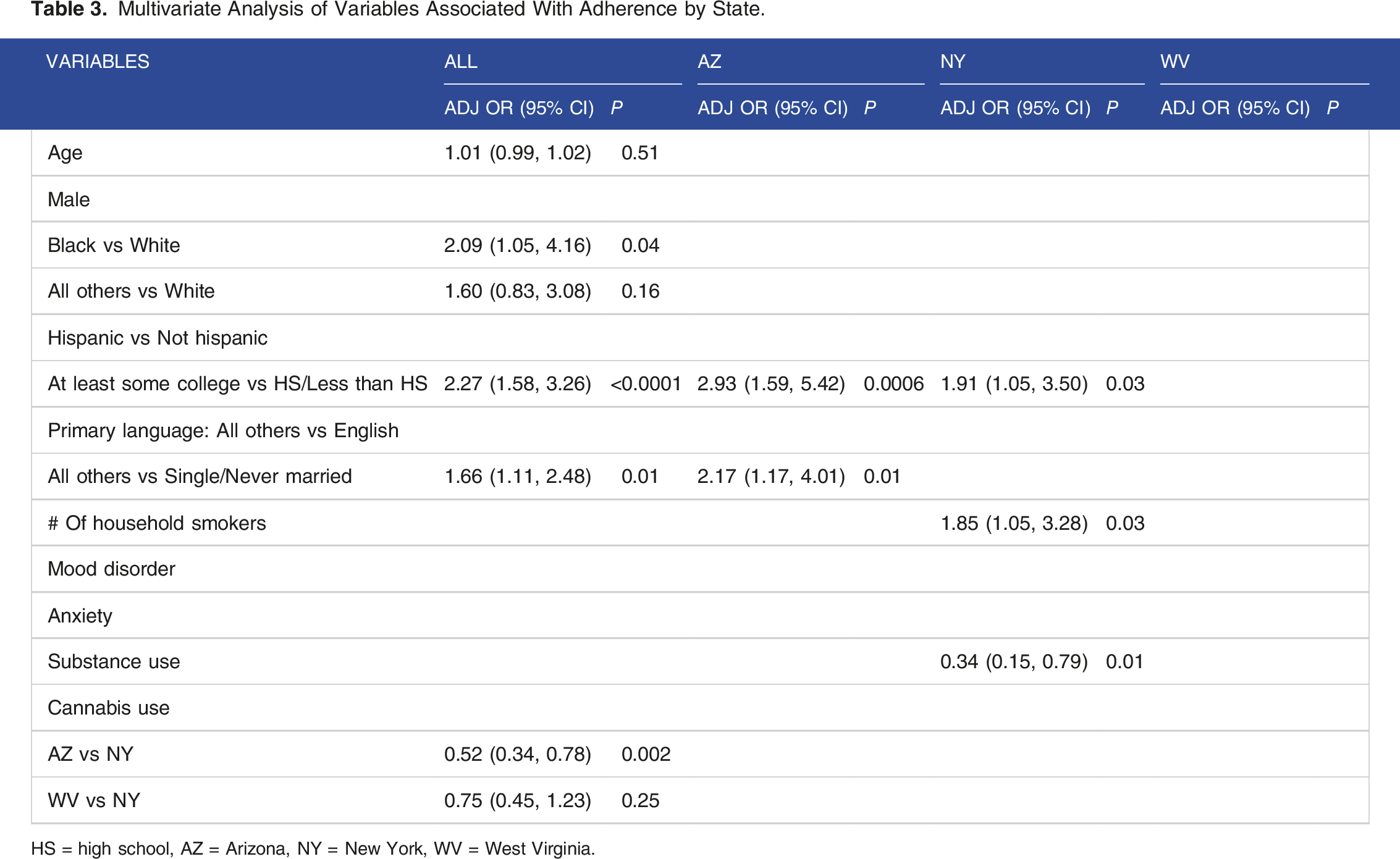
HS = high school, AZ = Arizona, NY = New York, WV = West Virginia.
Variables associated with adherence by state
The variables associated with adherence status for the state of Arizona included: education (at least some college vs high school/less than high school) (adjusted OR, 2.93; 95% CI, 1.59-5.42; P = 0.0006) and marital status (all others vs single/never married) (adjusted OR, 2.17; 95% CI, 1.17-4.01; P = 0.01). The variables associated with adherence status for the state of New York included: education ( at least some college vs high school/less than high school) (adjusted OR, 1.91; 95% CI, 1.05-3.50; P = 0.03), number of household smokers (more people who smoke in the home = more likely to adhere) (adjusted OR, 1.85; 95% CI, 1.05-3.28; P = 0.03), substance use (Yes vs No reporting substance use) (adjusted OR, 0.35; 95% CI, 0.15-0.79; P = 0.01). West Virginia did not show any significant trends or variables associated with adherence status.
Additional findings associated with adherence
To elucidate the findings associated with study adherence, the 3-month follow-up survey question, “Please tell us why you didn’t have all 6 coaching sessions” was analyzed. The question is formatted as a select all that apply, including seven possible answers: 1) Program took too much time; 2) The sessions were too long; 3) The calls were inconvenient; 4) The sessions were not helpful; 5) Personal issues; 6) The sessions were too stressful; and 7) Other (please describe). Of the 172 (21.4%) participants that were not adherent, 100 (58%) responded to the 3-month follow-up survey question. As displayed in Figure 2, participants reported that: the program took too much time (16, 16%); the sessions were too long (13, 13%); the calls were inconvenient (14, 14%); the sessions were not helpful (8, 8%); personal issues (59, 59%); the sessions were too stressful (4, 4%); and “Other (please describe)” 25, 25%. Participants’ reported reasons for non-adherence.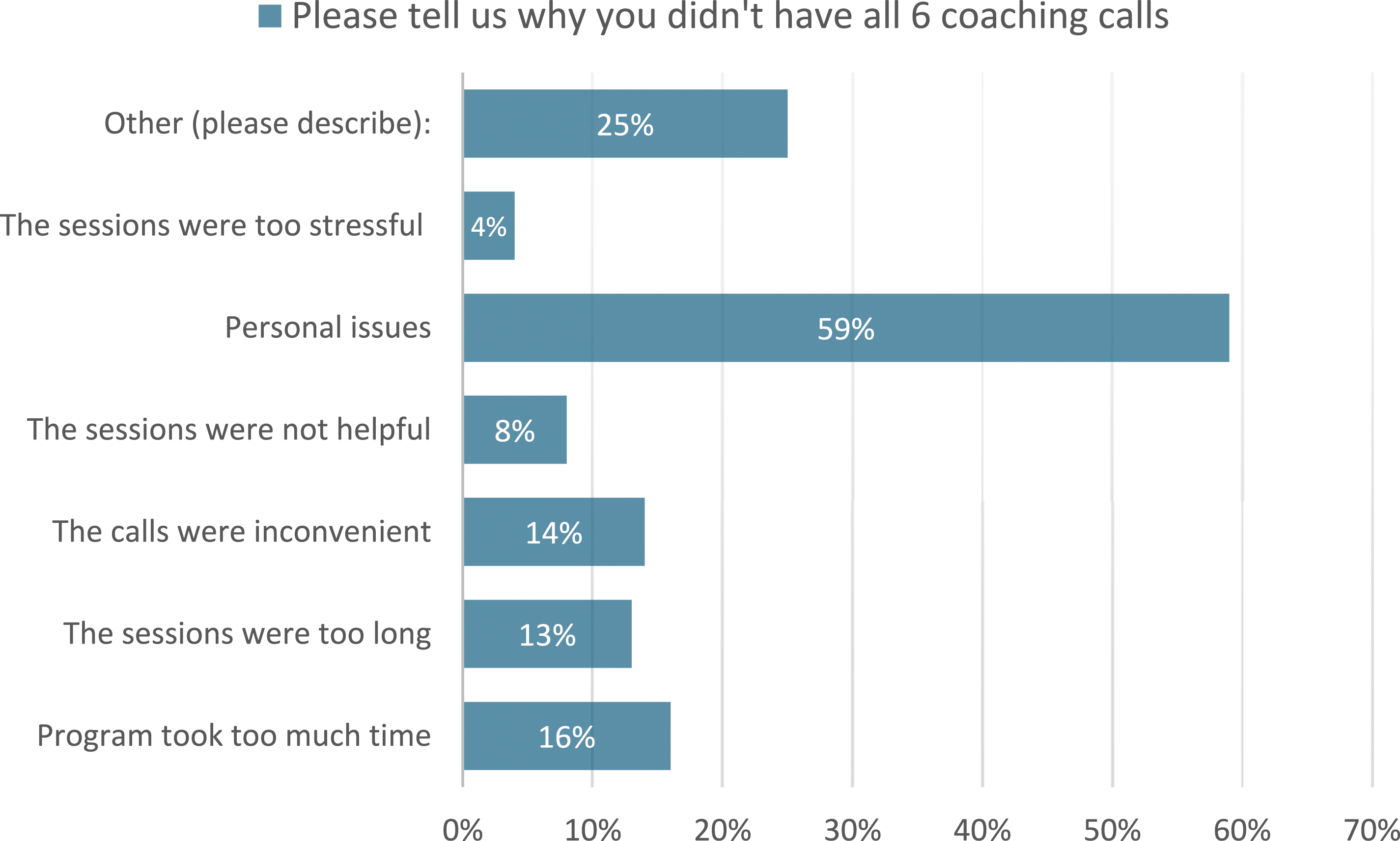
We qualitatively analyzed the seventh answer option, “Other (please describe).” We generated six common themes: 1) phone and internet issues (4/13, 30.8%); 2) coaching calls (1/13, 7.7%); 3) health related (2/13, 15.4%; 4) work and schedule conflicts (4/13, 30.8%); 5) personal (1/13, 7.7%); and 6) smoking related (1/13, 7.7%). For the theme work and schedule conflicts, participants reported: “having weeknight calls is difficult as a working mother with school age child”; “work life and not being able to drive while on the phone”; and “work schedule kept changing”. For the theme, phone and internet issues, participants reported: “I had phone issues my phone broke and shattered on the fifth week”; “lack of internet service”; and “I lost my phone.”
Discussion
Overall, we achieved an enrollment rate of 74.8% in the study, and of those enrolled, 78.6% attended all sessions. While these results compared favorably to many public health studies, they are lower than we had anticipated. In our pilot work, 5 which served as the basis for this study, we experienced enrollment rates exceeding 80%, and adherence rates over 85%. Thus, the rates observed in RCT are lower than we expected. Consequently, we conducted this secondary analysis to elucidate the current predictors associated with enrollment and adherence in order to determine ways to improve these critical study components.
We chose to recruit from Arizona, New York, and West Virginia because of the diverse sample of people who smoke across those three states. Results of this study indicate that overall, people who are older, more educated, and from New York were more likely to enroll in the study. Our within state analyses highlighted the differences between people who smoke across our participating sites. Within the three states from which we recruited, those who enrolled from Arizona were of older age and less likely to report other substance use. Those who enrolled from New York were more educated.
Overall, participants who were Black, more educated, were or had been married, and from NY were linked to better adherence in the study. Additionally, participants reported “personal issues” as another factor contributing to non-adherence. We also saw differences across states regarding adherence. From the three recruitment states, Arizona participants who were more educated and single or never married had higher rates of adherence. Among New York participants, those who were more educated, reported more household smokers, and were less likely to report other substance use had higher adherence rates.
Of note, participants from West Virginia did not have any significant predictors of enrollment or adherence, which may be due to the smaller subsample size from West Virginia than from New York or Arizona.
The findings of this study are consistent with previous smoking cessation clinical trials. Our enrollment findings are similar to those from reported by Ahluwalia et al 6 and Graham et al, 8 who found that age (eg, younger age less likely to enroll) and education (eg, less education less likely to enroll) were related to enrollment.6,8 In comparison to our findings, the study by Qin et al 11 and systematic review by Pacek et al 14 showed similar variables associated with higher adherence (eg, older age, higher education).11,15 However, our study showed that substance use was associated with non-enrollment for people from Arizona and non-adherence for participants from New York. Smoking is often not an isolated habit, but rather commonly co-occurs with substance use, mental health, and chronic pain symptoms. 16 Research indicates that individuals with co-occurring substance use should be regarded as a tobacco-related disparity group and prioritized for tobacco control interventions. 17 New research shows quitting smoking may have causal inferences and subsequent benefits to unhealthy alcohol use, cannabis use, cocaine, and illicit opioid use. 16 Therefore, more research is needed to identify effective methods for enrolling and retaining participants who have co-occurring disorders into tobacco treatment trials.
Our qualitative analyses of why randomized participants did not enroll in the study (ie, did not complete first treatment sessions) revealed that being “unable to contact”, “no longer interested”, “ineligible”, or “did not have first session within 1 month of randomization” were the most commonly reported reasons for non-enrollment. Quantitative analyses of the 3-month follow-up survey data indicated that “personal issues” was the most commonly reported reason for non-adherence, followed by work and schedule conflicts, and phone and internet issues.
We found an inverse relationship between attempts to enroll participants and adherence among those who enrolled. When project staff increased the number of contact attempts to convert potential participants into enrolled participants, many of those individuals who required multiple attempts demonstrated lower adherence. The pattern of non-response persisted throughout their participation, with difficulties in scheduling study sessions, leading to higher treatment dropout rates. When project staff limited the number of contact attempts with potential participants, we experienced lower enrollment rates, but scheduling throughout the coaching process and adherence improved. Although potential participants completed the consent and baseline survey process, motivation to quit smoking can vary, which may lead to ambivalence and make commitment to study enrollment challenging. In addition, the context and environment in which potential participants live may be barriers to study participation, including scheduling and time constraints. Thus, there is a need for feasible, holistic interventions that address these barriers and the social determinants of health. Future research aimed at enhancing motivation and designing interventions that reduce barriers to participate in tobacco cessation clinical trials is needed.
Limitations
The sample for this study was from a clinical trial that used specific inclusion and exclusion criteria, which may limit generalizability. Additionally, we were not testing some specific hypothesis; therefore, no power analysis was performed. Furthermore, we were not able to follow individuals who did not enroll after randomization to confirm their reason for not enrolling. Thus, other confounding variables could affect enrollment status. Lastly, this was a secondary analysis to determine associations between variables, and we are unable to determine the “cause” of non-enrollment and non-adherence.
Conclusion
The results of the present study show that enrolling and retaining people who smoke in cessation interventions requires pragmatic strategies for addressing common challenges. Predictors of enrollment and adherence provide insights that can be useful for developing and testing novel methods to ameliorate the barriers to enrolling in and adhering to intensive tobacco cessation interventions. The results of this study suggest the need to devise and test recruitment strategies directed toward younger people who smoke to increase enrollment, and interventions that target single and more educated people who smoke. In addition, proactively discussing possible work schedule conflicts and phone access concerns may improve rates of treatment adherence in similar remotely delivered clinical trials. Additional research is needed to develop and test interventions that address the social determinants of smoking and barriers associated with attendance of treatment sessions. Also, novel strategies to enhance engagement and retention are needed for people who smoke with co-occurring substance use. Further studies are needed to develop and test novel strategies for enhancing enrollment and adherence in intensive smoking cessation treatment clinical trials.
Supplemental Material
Supplemental Material - Predictors of Enrollment and Adherence in a Randomized Tobacco Cessation Clinical Trial
Supplemental Material for Predictors of Enrollment and Adherence in a Randomized Tobacco Cessation Clinical Trial by Zhanette Coffee, Chiu-Hsieh Hsu, Todd W. Vanderah, and Judith S. Gordon in Tobacco Use Insights
Footnotes
Acknowledgements
The authors wish to thank their colleagues at Roswell Park Cancer Center and West Virginia University. In addition, we thank First Choice and Roswell Park Cancer Center who operate the West Virginia Tobacco Quitline and New York State Smokers’ Quitline, respectively. The authors also wish to thank research coordinator Kari Marano, research assistant Yessenya Barraza, and student research assistant Kyiah Moore, and the study quit coaches Anjanette Gutierrez, Mindi Horwich, Crista Meinke, and Mary Petersdorf.
Author contribution
Conceptualization, Z.C., J.S.G.; methodology, Z.C., J.S.G.; software, C.H. validation, Z.C., J.S.G., C.H.; formal analysis, Z.C., C.H; investigation, Z.C., J.S.G.; resources, Z.C., J.S.G.; data curation, Z.C., C.H.; writing—original draft preparation, Z.C., J.S.G.; writing—review and editing, Z.C., J.S.G., C.H., T.W.V.; visualization, Z.C., J.S.G.; supervision, Z.C., J.S.G.; project administration, Z.C., J.S.G. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Be Smoke Free study was funded in September 2021 (National Center for Complementary and Integrative Health, R01AT011500; PI: Gordon, J.).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
