Abstract
The objective of the current study was to evaluate the impact of a reduced risk claim about lung cancer, presented in various formats, on smokers’ and non-smokers’ interest in trying Camel Snus and intention to purchase Camel Snus. We varied claim formats by varying advertising messages for Camel Snus in 4 ways (1) text only; (2) bar chart; (3) text/testimonial; and (4) bar chart/testimonial. 3001 participants were recruited from a web-based consumer specialty panel via an email invitation. In 2015, a second study was conducted, using similar methods, where 3001 additional participants were recruited. Overall, controlling for other factors, the presence of an MRTP claim was not significantly related to interest in trying snus [X2 (4) = 8.567, P = .073], or purchase intentions [X2 (4) = 1.148, P = .887]. Relative to a control ad where no explicit health risk claim was made, the Graphic + testimonial [OR = 1.29] or Text only [OR = 1.41] claims did significantly increase interest in trying Camel Snus. However, the adverting format did not impact interest in purchasing Camel Snus. While current smokeless tobacco users (95%) and smokers (59%) expressed interest in trying Camel Snus, non-tobacco users (7%) showed low interest in trying or purchasing Camel Snus (P < .001). Interest in trying Camel Snus was stronger in younger smokers compared to older smokers. Among current smokers, worry about lung cancer (the key focus of the reduced risk claim) was not associated with interest in trying Camel Snus or with purchase intention [OR = .91, 95% CI: .72, 1.14] or intention to purchase snus [OR = 1.07, 95% CI: .86, 1.32]. Future research should evaluate how claim and messaging formats influence perceived truthfulness and whether this effect differs among sub-groups of consumers, such as adolescents, those with tobacco-related disease, and former smokers. It will also be helpful to understand whether perceptions of ad truthfulness result in changes in product use patterns over time. In sum, giving people truthful, credible information about relative product risks, such as through authorized MRTP claims, is important, but such information is likely insufficient to get smokers to switch.
Introduction
Smoking harm reduction is a strategy that seeks to reduce smoking-caused morbidity and mortality by encouraging users who are unwilling or unable to stop smoking to switch to lower risk forms of nicotine delivery. Providing consumers with accurate information about the relative health risks of cigarettes compared with alternative (mostly non-combustible) nicotine delivery products is an important element in a smoking harm reduction paradigm.1–3 At the same time, there are legitimate concerns about tobacco product health claims coming from manufacturers given the history of cigarette companies deceiving consumers about the health risks of cigarettes overall, and the deceptive marketing of filtered and low tar cigarettes which many consumers misperceived as lower risk.4–6 In addition, even if a new product offered a health advantage for smokers, there are nontrivial concerns that claims of relative health risk advantage could potentially induce non-tobacco users, including youth and former smokers, to take up the lower risk product, and/or for such claims to forestall cessation in smokers.7–11
In 1 study, nearly half of current smokers and about 10% of former and never smokers reported on a large national survey that they would be ‘somewhat likely’ to try a tobacco product that posed lower risk of health harms or addiction compared to a conventional cigarette. 12 Other studies have reported similar findings. 13 The Food and Drug Administration (FDA) has regulatory authority over modified risk tobacco product (MRTP) advertising claims in the United States. 14 MRTP claims need to be evaluated scientifically by the FDA to determine their validity. MRTP claims can fall into 2 groups: (1) claims about reduced health risk of a product (eg, less risk of lung cancer than from smoking); and (2) claims about reduced exposure to toxicants in the product (eg, less exposure to some carcinogen or chemicals). MRTP decisions are made on a product-by-product basis, subject to applications by manufacturers. So, authorizations and denials are an evolving process. As of 2023, FDA had authorized MRTP claims for 4 products: (1) General Snus, a brand of snus smokeless tobacco manufactured by Swedish Match which was granted a reduced health risk claim; (2) iQOS, a heatedtobacco product manufactured by Philip Morris International which was granted a reduced exposure claim; (3) VLN, a reduced nicotine content cigarette which was granted a reduced nicotine exposure claim; and (4) Copenhagen, a moist snuff smokeless tobacco product which was granted a reduced risk claim for lung cancer. 15 An application proposing a reduced health risk claim for Camel Snus was also submitted, but was withdrawn by the company after being under review for several years. 15
Studies have begun examining the potential impact of hypothetical MRTP claims on perceived product risks or substance exposures and intentions to use a product, with somewhat mixed results. For example, El-Toukey and colleagues tested both reduced risk and reduced exposure claims for a hypothetical new tobacco product in national US samples and found little difference in their effects on perceived risk or exposure, but did find that the reduced risk claims had greater effects on intentions to use the product. 16 A focus group study examining a range of hypothetical MRTP claims for snus and e-cigarettes found that the claims elicited product interest in some adult smoker participants, but were also met with considerable message skepticism. 17
Indeed, the effectiveness of MRTP claim messages in terms of encouraging smokers to try a new tobacco product are likely to be related to whether the lower risk message is communicated in an effective manner and whether it is perceived as believable. The salience of the message, in this case exposure or risk reduction relative to cigarette smoking, is also an important consideration. A MRTP claim on reduced lung cancer risk should have high salience to smokers, given this is the most widely recognized health risk associated with smoking. Worry is a useful proxy for perceived risk of lung cancer and has been shown to be associated with smoking cessation and lung cancer screening behaviors.18–22 Whether a message is understood and salient may be moot, however, if such communications are perceived as coming from a source that is untrustworthy (ie, a cigarette company). The credibility and believability of a MRTP claim are important factors in determining whether a claim message will influence a behavior change.23–25 Claims may be considered more credible when the content is consistent with prior knowledge and beliefs, and may be less credible when the claim is perceived to derive from a financially motivated source.26–29
Camel Snus is 1 U.S. brand variant of snus, a class of low-nitrosamine, smokeless tobacco products with a lower cancer risk than combustible cigarettes.30,31 2 industry-supported studies have evaluated proposed MRTP claims around Camel Snus specifically. This claim noted that completely switching to Camel Snus was associated with reduced risk of lung cancer, respiratory disease, heart disease, and oral cancer, and included ‘balancing’ information about addiction and that no tobacco product is safe. Gerlach and colleagues tested a claim communicating that smokers could benefit from a reduced risk of lung cancer, respiratory disease, heart disease, and oral cancer, along with “balancing” information about addiction and a warning that no tobacco product is safe. They found that current smokers were most likely to indicate intent to purchase a product and non-tobacco users were unlikely to increase use of snus. 32 Pillitteri et al found that comprehension of the claim content was strong. 33 However, neither study included a measure to assess the credibility of the proposed claim. We previously found that while smokers exposed to a modified exposure claim were more likely to express an intention to try the product compared to non-smokers, those exposed to more elaborated versions of the reduced exposure claim (graphics, testimonial) also expressed greater skepticism about the credibility of the claim. 34 In our prior study, the claim examined was about reduced exposure to a specific carcinogen found in tobacco (TSNAs). However, it is unclear whether a reduced risk claim for a specific, well-known smoking-caused condition (lung cancer) would show a similar pattern of results.
Thus, building on our previous research, the objective of this study was to examine the impact of a reduced lung-cancer focused claim, as well as the impact of perceived claim truthfulness on interest in trying and intention to purchase Camel Snus among smokers and non-smokers. In an additional analysis limited to current smokers, we also assessed whether worry about lung cancer (the disease risk at the core of the advertising claim) influenced intentions to try or purchase Camel Snus.
Methods
Participants
Data presented are based on 2 separate survey studies. In 2014, 3001 participants were recruited from a web-based consumer specialty panel (Global Market Insite (http://www.gmi-mr.com/global-panel/index.php) via an email invitation. Participants included smokers and non-smokers and were eligible if they were between the ages of 14 and 65 years and provided informed consent. GMI’s “specialty youth panel” complies with the Children’s Online Privacy Protection Rule (16 C.F.R. Part 312) and youths’ parents were e-mailed a statement describing the risks and benefits of participation, compensation, and confidentiality prior to their child engaging in the survey. The sample targeted 2000 adults (18-65) and 1000 youths (aged 14-17) and compensated them 60 GMI “marketpoints” (20 marketpoints=1) for their time. In 2015, a second study was conducted, using similar methods, where 3001 additional participants were recruited. The study protocol was approved by the Institutional Review Board at Roswell Park Cancer Institute, Buffalo, NY.
Design and Procedure
In the 2014 study, participants were randomly assigned to view 1 of 5 different Camel Snus advertisements (see Figure 1). Four of the ads presented information related to a reduced risk for lung cancer (eg “Scientific evidence suggests that snus does not cause lung cancer) in varying formats: (1) text; (2) bar chart; (3) text/testimonial; and (4) bar chart/testimonial. The comparison condition was a current ad for Camel Snus without a claim made about reduced lung cancer risk. All participants also saw an unmodified Camel cigarette ad. Participants could see the advertisements as long or as briefly as they wanted. Following each ad presentation, participants reported on truthfulness and skepticism of ad content, perceived health risks associated with product use using measures described previously, [30] and purchase intention, assessed using a Juster scale.
35
Participants were asked to choose the product they’d be most interested in trying, choosing between 3 options: (1) Camel Snus, (2) cigarettes, and (3) neither. Among participants that expressed an interest in either Camel Snus or cigarettes, they were asked to indicate how likely they would be to purchase Camel Snus and/or cigarettes in the future. Camel Snus advertisement conditions shown to survey participants.
In 2015, a second study was conducted using similar methods. In this second study, 3001 participants were randomized to view either 1 of 3 advertising format conditions: (1) text based Camel Snus ad with a reduced lung cancer risk claim, (2) a bar chart/testimonial with reduced lung cancer risk claim, or (3) an ad without the explicit reduced risk claims (see Figure 1). The number of ads presented in the 2015 study differed in order to simplify the study design and analysis, retaining and focusing on the advertisements that had the highest rating in the 2014 study.
Survey Measures
For adults, current tobacco product use status was determined based on responses to the following questions, (1) Have you ever smoked a cigarette, even a few puffs? (2) Have you smoked at least 100 cigarettes in your lifetime? and (3) Do you now smoke cigarettes? Current smoker was defined as someone who reported currently smoking every day or some days and reported having smoked at least 100 cigarettes. Non-current smoker was defined as someone who may have had a puff or had smoked 100 cigarettes but reported not currently smoking at all. Never smoker was defined as someone who reported never having even a puff of a cigarette (non-current smokers and never smokers were combined into 1 category for analysis). For smokeless products, status was determined based on responses to the following questions: (1) Have you ever used smokeless tobacco products such as dip, moist snuff, or chewing tobacco even once? and (2) Do you now use dip, moist snuff, or chewing tobacco? These variables were then combined as a measure of current tobacco product use using the following mutually exclusive categories: (1) exclusive current cigarette smokers (those with no smokeless use); (2) current smokeless users (smokeless only and dual users of smokeless and cigarettes users); and (3) current non-tobacco users (ie, those who did not currently use either cigarettes or smokeless tobacco. Because of the small number of smokeless only users available to create a meaningful category, dual smokeless and cigarette users were treated as smokeless users. This combines all participants with any smokeless experience, who might be more predisposed to interest in smokeless products. Two measures assessed truthfulness and skepticism about the MRTP claim shown to study participants. First, participants were asked, “How likely is it that the ad you just saw contained truthful information?” with responses scored on a 1-5 scale where “1” indicated “not at all likely” and “5” indicated “extremely likely”. Second, participants were asked, “How skeptical are you about the truthfulness of the ad?” also scored on a 1-5 scale where “1” indicated “not all skeptical” and “5” indicated “extremely skeptical”. The measure of skepticism was reverse coded, so responses ran in the same direction, where higher values represent more truthfulness and less skepticism of the product claim shown to participants. Interest in purchasing and trying snus was measured on a 0-10 scale where “0” indicated “no chance or almost no chance” and “10” indicated that the participant was “certain or practically certain” to try or purchase snus. These items were recoded into categories of “no interest” (based on scores of ‘0’) vs “at least some interest” (based on scores of at least ‘1’) in trying or purchasing snus. These response options were recoded to “often or all the time” or “sometimes or rarely or never” for analysis.
Statistical Analysis
There were no significant interactions observed between age and any of the variables included in preliminary analyses. We assessed differences in truthfulness and skepticism for the 3 ad conditions replicated in both years using two-way ANOVA (year x smoking status), which indicated no significant differences on these outcomes [F (1, 4) = 1.43, P = .223]. This was the driver behind the decision to combine data across both years and focus on interest in trying a product and purchasing a product as main outcomes, with truthfulness and skepticism toward ad content as predictor variables. Among all current cigarette smokers (including cigarettes only and dual cigarette/smokeless users), a sub-analysis was performed including lung cancer worry as a predictor variable, because this question was asked only among current cigarette users. In our sample, 71.5% of participants reported that there was no chance or almost no chance that they would purchase snus following the presentation of the ads. Because of this result, we dichotomized the purchase intention variables for analysis into (1) no chance or almost no chance of purchasing vs (2) at least some interest in purchasing. Data analysis was conducted using SPSS 21.0. Generalized Linear Models were used to predict base interest in selecting a product to try or purchase (snus and cigarettes). Difference scores were calculated to measure the degree to which participants were likely to purchase Camel cigarettes and Camel Snus.
Results
Demographics and tobacco use status: overall and by survey year. a

aDeviations in the sample sizes within each variable are due to missing data.
Snus Purchase Intentions, Claim Format, and Truthfulness
Multivariate logistic regression model predicting interest in trying snus (n = 5376). a

aDeviations in the sample sizes within each variable are due to missing data.
Multivariate logistic regression model predicting interest in purchasing snus (n = 5376). a
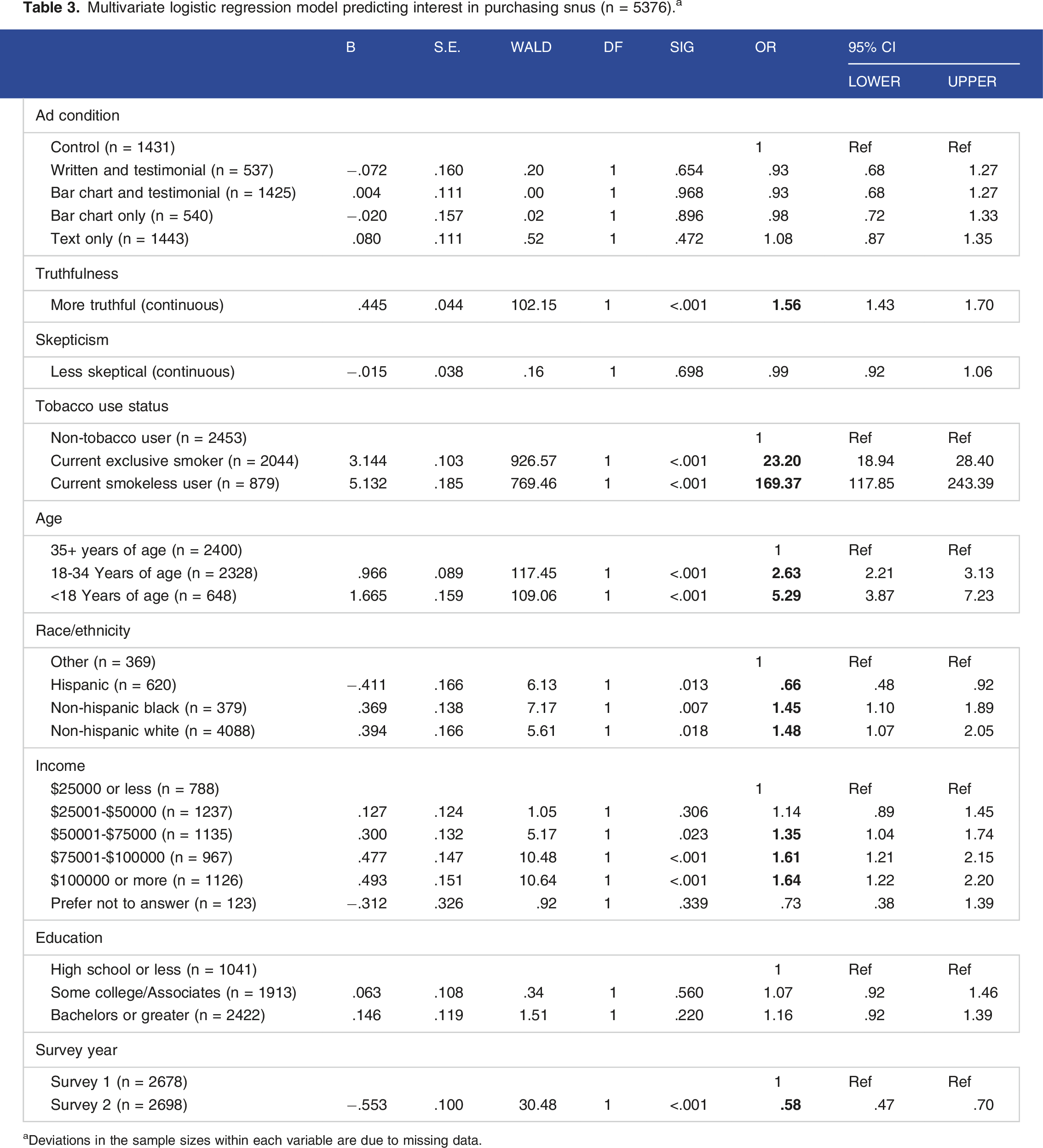
aDeviations in the sample sizes within each variable are due to missing data.
The effects of ad type on intentions to try a product did not differ by tobacco user groups. However, Interest in trying Camel Snus did vary by tobacco use status, and was higher in current users of smokeless tobacco (95.1%) followed by current smokers (58.7%) and least likely among non-tobacco users (6.9%) [P < .001]. Controlling for other factors, relative to non-tobacco users, those using smokeless tobacco currently had 8.2 times greater odds of reporting interest in trying Camel Snus, while current exclusive smokers had approximately 3.7 times greater odds of trying Camel Snus. These effects were similar with regards to intention to purchase Camel Snus. Relative to non-tobacco users, smokeless tobacco users had over 160 times greater odds of intention to purchase snus, and current exclusive smokers had 23 times greater odds. Broadly speaking, non-Hispanic White participants under age 35 with higher incomes were more likely than other participants to be interested in trying and intending to purchase snus.
Worry About Lung Cancer, Interest in Trying, and Intention to Purchase Snus
Finally, we conducted a sub-analysis among all current smokers to examine the potential effect of worry about lung cancer on interest in trying and purchase intention, given this was the focus of the claim. 1 Controlling for other factors, worry about lung cancer was not associated with interest in trying snus [OR = .91, 95% CI: .72, 1.14] or intention to purchase snus [OR = 1.07, 95% CI: .86, 1.32]. In addition, an analysis of whether greater worry about lung cancer was associated with advertisement type viewed by each participant indicated no statistically significant differences by advertisement type.
Discussion
Overall, we found that exposure to 2 out of the 4 tested ads with MRTP claims about lung cancer had positive impacts on interest in trying snus, but no significant effects on purchase intentions were observed. The format of the advertisement did seem to make a difference. Relative to a control ad where no explicit health risk claim was made, the Graphic + testimonial or Text only claims did significantly increase interest in trying Camel Snus. However, the adverting format did not impact interest in purchasing Camel Snus. While current smokeless tobacco users and smokers expressed interest in trying Camel Snus, non-tobacco users showed low interest in trying or purchasing Camel Snus. Interest in trying Camel Snus was stronger in younger smokers compared to older smokers. Among current smokers, worry about lung cancer (the key focus of the reduced risk claim) was not associated with interest in trying Camel Snus or with purchase intention. Greater perceived truthfulness and lower skepticism of the ad message were both significantly associated with increased interest in trying Camel Snus. However, only greater perceived truthfulness was associated with increase intention to purchase Camel Snus.
These findings suggest that it is important to assess how credible the health claim message is, particularly in studies attempting to stimulate consumer interest in and uptake of MRTPs among smokers. Source credibility is a key feature of health communication effectiveness, even as the functional definition of who is and is not a credible source shifts. 22 In 2019 the FDA’s Tobacco Products Scientific Advisory Committee (TPSAC) reviewed a reduced lung cancer risk claim for a moist snuff smokeless product called Copenhagen. While the single text-only statement itself was judged by TPSAC to be scientifically defensible, research by the manufacturer of Copenhagen, Altria, showed that exposure to the claim did not actually result in significant changes in risk perceptions or intentions to use Copenhagen among current cigarette smokers.36 Thus, while the study did demonstrate that participants were able to understand the claim, understanding the claim did not appear to translate into increased likelihood of intention to use the product. 37 A similar pattern of results was found for a broader set of reduced risk claims submitted by both Swedish Match for its General Snus products, and by RJ Reynolds for Camel Snus. In both instances, factually accurate reduced risk claims did not translate into substantial changes in intention to use the modified risk product by smokers.
Limitations of the current study include use of an opt-in internet panel, which may not be representative of consumers in general, and a single brief exposure to the claims. Extended exposure to claims in context might yield different results. These data were collected in 2014-2015 from online panels, and so may not represent current populations; however, the issues around modified risk claim wording and presentation continue to be relevant. While an application for Camel Snus was withdrawn 1 for Copenhagen was authorized, and it is reasonable to anticipate modified risk applications in the future for any newly deemed products (eg, e-cigarettes). Strengths of the current study include a large sample size, randomization to various claim presentation styles (as opposed to a single execution), and inclusion of both adults and youth.
While some smokers may simply not find oral tobacco products acceptable alternatives for cigarettes, it is possible that at least some smokers do not accept reduced risk claims as credible when the source is a manufacturer who has a vested financial interest in the product itself. Because noncredible claims may fail to motivate use of MRTPs, any harm reducing effects that might be possible on the population level could be diminished. Understanding and even believing in a claim of reduced risk or reduced exposure associated with use of smokeless tobacco products is probably a necessary but not sufficient condition for moving smokers to change their behavior. There are likely to be additional factors that prompt users to substitute cigarettes for something else, including price, stigma, ease of use, and availability of the alternative. Future research should evaluate how claim and messaging formats influence perceived truthfulness and whether this effect differs among sub-groups of consumers, such as adolescents, those with tobacco-related disease, and former smokers. It will also be helpful to understand whether perceptions of ad truthfulness result in changes in product use patterns over time. In sum, giving people truthful, credible information about relative product risks, such as through authorized MRTP claims, is important, but such information is likely insufficient to get smokers to switch to smokeless products.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Richard O’Connor and Brian Fix. The first draft of the manuscript was written by Brian Fix and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Cancer Institute (U19CA157345, R37CA222002).
