Abstract
Background
In a low and middle-income country (LMIC) like India, non—communicable diseases (NCDs) contribute a major proportion (61.8%) of all causes of death. Out of this, 48% of cardiovascular diseases , 23% of Chronic Respiratory Diseases , and 10% of Cancer deaths are attributable to tobacco use. Tobacco use is a major risk factor for NCDs and thus, the tobacco cessation approach is a high priority intervention to combat complications and death among NCD patients. While several interventions are available for tobacco cessation, in resource constraint countries like India, the effectiveness of low-cost, culturally specific patient-centric tobacco cessation behavioral intervention holds a potential that needs to be evaluated. A newly developed evidence-based tobacco cessation intervention package will be compared with the existing/usual care provided under the National Programme for Prevention and Control of Cancers, Diabetes, Cardiovascular Diseases, and Stroke (NPCDCS) at NCD clinics.
Methods and design
2 arm, parallel-group randomized controlled trial.
Participants
Patients aged ≥30 years suffering from NCD, currently using tobacco, and attending NCD clinics in 2 districts of Punjab, India.
Sample size
A total of 200 participants meeting the selection criteria will be recruited. They will be allocated either to the intervention arm or control (usual care) arm (100 each) using block randomization.
Intervention
For the participants, there will be 4 face-to-face disease-specific cessation counseling sessions, disease-specific pamphlets, short text messages in vernacular language, that is, Punjabi. Follow-ups will be done at the third, sixth, ninth, and 12th months.
Primary outcome
Seven-day abstinence, biochemically verified by plasma cotinine levels.
Secondary outcome
Quit attempts, number of sticks/number of times of smokeless tobacco usage in a day, and stage of behavior change in tobacco users.
Discussion
This multicomponent culturally specific-patient-centric behavioral intervention package for tobacco cessation at NCD clinic settings focusing on the individual, family, and social environment could increase the outreach of cessation services using existing resources, thereby strengthening health systems and enhancing the quality of life of NCD patients.
Trial registration
The protocol for the study has been registered with the Clinical Trials Registry in India under the registration number CTRI/2018/01/011643.
Keywords
Background
Tobacco use is a global epidemic that kills more than 8 million people annually. 1 Worldwide, the Disability Adjusted Life Years (DALY’s) attributable to tobacco smoking and smokeless tobacco (SLT) was 148.6 million 2 and 6 million, 3 respectively. Almost one-fifth of the world’s population resides in India, 4 where 28.6% (266.8 million) of the adults aged 15 years and above use tobacco in any form. 5 The number of years lived with disability (YLDs) among NCD patients increased by 61.1% between 1990 and 2017, according to the Global Burden of Disease study. 6 World Health Organization (WHO) has projected that if adequate measures are not taken to control NCDs, then the annual toll of deaths due to NCDs shall rise to 55 million by 2030. 7 In India, the contribution of NCDs to total deaths has increased from 37.9% (in 1990) to 61.8% (in 2016). 4
The consumption of tobacco in either form, smoked or smokeless, among patients suffering from NCDs has many deleterious consequences. Tobacco smoking among patients with diabetes mellitus leads to an increase in glycosylated hemoglobin (HbA1c), 8 which results in higher morbidity and premature death due to the development of macrovascular complications. 9 The prognosis among smokers with heart failure is poorer than nonsmokers with heart failure. 10 Tobacco use also poses a risk for secondary/recurrent stroke among stroke survivors 11 and predisposes patients with Chronic Obstructive Pulmonary Disease (COPD), poor health outcomes with further deterioration in primary lung disease. 12 Continued use of tobacco among patients diagnosed with tobacco-related cancers leads to reduced treatment efficacy with a higher rate of complications. 13 Further, smokeless tobacco (SLT) use has also been implicated as an established risk factor for causing various cancers, 14 and type 2 diabetes emllitus, 15 adverse cardiovascular events, reducing the likelihood of survival after a myocardial infarction (MI) or stroke. 16
The WHO identifies tobacco as a modifiable behavioral risk factor for NCDs. 17 Tobacco use is a learned behavior, and nicotine addiction is a very complex process involving biological, behavioral, psychological, and cultural factors. The interplay between these factors results in the continued use of tobacco products among the users. 18 There is plenty of evidence that has incorporated the health belief model in motivating tobacco users to quit and educating them about the impact of tobacco use and passive exposure to tobacco smoke.19,20 However, there are certain limitations of this model that suggest considerable factors that are not related to health but do impact health behavior and are not predicted by HBM. This is seen in tobacco smoking as HBM does not consider habitual behavior, and health beliefs may not have an intervening outcome among seeking health care information and intending to quit.21,22 Interventions involving the Social Cognitive Theory (SCT) process raise resistance and self-efficacy, increasing the likelihood of successful quitting. 23 However, there is also evidence that emotional factors, higher anxiety levels affect self-efficacy making the quitters more vulnerable to relapse after cessation. 24 Moreover, behavior arises from complex interactions, and the SCT ability to explain human behavior is unreal and weakens its usefulness in practical settings. 25
In this paper, we propose a cessation intervention for the tobacco users of Punjab based on the Transtheoretical Model (TTM), also called as Stages of Change model. This model has been widely used in the development of cessation interventions. 26 TTM has evolved through studies examining smokers’ experiences who quit, thus understanding their habitual behaviors in various stages of readiness and assisting physicians in administering a customized plan of action for quitting tobacco use.
The largest number of TTM-related intervention studies have focused primarily on smoking cessation27-29 and very few on smokeless tobacco among disease-specific groups, especially in India, where the burden of SLT use remains relatively high. 30 In addition, tailoring interventions incorporating modified theoretical concepts of TTM is much required to cater to different tobacco users across varying stages of behavior change.27,31 Further, there is evidence that TTM supports the cultural relevance of thematic constructs for various public health interventions such as physical activity, weight control, unhealthy alcohol use, etc.32-34 Moreover, harnessing the power of TTM of change in adapting multiculturalism has also been documented in various domains. 35 Besides, the model considers an individual level of motivation and readiness to change when setting goals in contrast to other behavior change models. The development of culture-specific cessation interventions has been reported to effectively reduce risky health behaviors and address the cultural roadblocks to change.34,36
The state of Punjab lies in the northern part of India, and the majority of residents practice Sikhism as the primary religion. The Sikh Code of Conduct strictly prohibits tobacco use for its followers and is categorized as a tabooed practice.37,38 Due to this, many users do not disclose their tobacco use status and therefore do not seek help to quit. 39 Further, the state has rich cultural diversity wherein the residents from neighboring states of the country migrate in search of better work opportunities. 40 The risk health behavior such as tobacco use of individuals from such diverse backgrounds could be addressed through a patient-centric approach. This patient-centric approach has also been demonstrated to increase the acceptability among users besides managing their craving and cutting down on tobacco use.
Moreover, the lives of most Indians are centered on the institution of the family. Therefore, the involvement of family members in developing a quit plan is centric to this approach as well. Evidence also suggests that the involvement of the family in the cessation process by harnessing their influence yields better outcomes. 41
Biochemical verification of quit status is vital in trials where social norms and personal characteristics influence reporting of tobacco use status apart from increasing the scientific rigor and validity. 42 The present study also proposes to undertake biochemical verification of tobacco use by analyzing plasma cotinine levels (long half-life, greater sensitivity, and specificity), 43 using high-pressure liquid chromatography. 44 Assessment of cotinine in blood samples produces uniform matrix measurement and does not require adjustment for hydration levels among users. 43 Moreover, literature also reports a relationship between plasma cotinine levels and nicotine dependence score. 45 Majority of the studies have reported salivary and urine cotinine levels.46,47 However, assessment of plasma cotinine among the NCD population has not been widely reported.
The existing literature from developed countries aptly demonstrates the effectiveness of tobacco cessation services among these patients at the clinics. However, the majority of these trials have been conducted among tobacco smokers and not SLT users. In an LMIC like India, where the burden of SLT remains the highest, followed by smoking, it is necessary to offer support to SLT users and smokers at every available opportunity. Nonetheless, given the growing burden of NCDs in India, the utilization of NCD clinics for delivering “opportunistic cessation support” can be enchased. These NCD clinics are established under the National Programme for Prevention and Control of Cancers, Diabetes, Cardiovascular Diseases, and Stroke (NPCDCS) launched by the Government of India in 2010–2011. 48 The feasibility and effectiveness of NCD clinics in delivering tobacco cessation package has not been evaluated. Most of the trials conducted on such disease-specific populations cater to 1 or 2 diseases, whereas an established NCD clinic of the health system would favor engaging more patients of different NCDs with infrastructure and human resources being constant.
Thus, we aim to assess the effectiveness of a patient-centric culture-specific intervention using the TTM framework among smoked and smokeless tobacco users in NCD clinics running under the NPCDCS program of the Government of India. Box-1 List of operational definitions used in study 49
Methods
Study design and period
It will be a randomized controlled trial (RCT) comparing 2 parallel groups with an allocation ratio of 1:1. The patient (participant) is the unit of randomization. The study will comprise 6 month recruitment period with intervention at the routine visits during the 1-year follow-up (at third, sixth, ninth month and a final assessment at 12th month) (Figure 1).
Study area and settings
Punjab is one of the wealthier states situated in northwest India. With a population of 27.7 million (Census, 2011), the state is divided into 22 districts. Each district is further divided into 3–4 Community Development Blocks, the rural area administratively designated for planning and development. 50 Sikhism is the most widely practiced religion, and Punjabi is the widely spoken language in the state. 51 Global Adult Tobacco Survey−2 (GATS-2, 2016–2017) reports current tobacco use prevalence in the state to be 13.4%. 5 The NCDs contribute to 66% of the total disease burden in Punjab. 4 The state-wide STEPS survey reported an overall prevalence of diabetes mellitus and hypertension as 8.3% and 40.1%, respectively, which are higher than the national average.52,53 2 NCD clinics running at the district level of Punjab will be selected for the pilot study where the intervention will be delivered.
Specific program settings
During 2010–2011, the Government of India initiated a National Programme for Prevention and Control of Cancers, Diabetes, Cardiovascular Diseases, and Stroke (NPCDCS) under which “NCD clinics” were established at Community Health Centre (CHC) level and district hospitals for comprehensive diagnosis and management of subjects suffering from Cancer, Diabetes, Hypertension, Cardiovascular Diseases, and Stroke in conjunction with dietary counseling and lifestyle management. The Punjab State has been a forerunner in implementing the NPCDCS. The State Cells for NCD Control under the Director of Health Services, Punjab, runs District NCD Control Cells in all 22 districts. These NCD clinics are staffed by a medical officer, counselor, nurse, and a data entry operator and provide screening, diagnosis, management, and counseling services for NCD’s focusing primarily upon lifestyle and dietary modifications. 48
Study participants
The participants will comprise tobacco-using patients visiting the NCD clinic to seek management of their NCD ailment.
Inclusion criteria and exclusion criteria
This shall include the patients visiting the NCD clinic who are - Age 30 years and above - Diagnosed with 1 or more NCDs (Diabetes, Hypertension and other CVDs, Stroke, Cancer, Chronic Respiratory Diseases such as COPD and asthma) - Using any tobacco product (smoked/smokeless) - Able to read simple texts in local language - Access to a mobile phone - Willingness to give informed consent to be a part of the study and involve their family members in the process.
Exclusion criteria
- Patients already on Nicotine Replacement Therapy (NRT) - Patients in the event of the end stage of cancer, on renal dialysis, psychiatric illness, multimorbidities, etc.
Intervention
Description
Roles and responsibilities of health care providers during the study.

Components
Overview of the content of tobacco cessation counseling session in the intervention arm.

Features
i. Culture-specific
The socio-cultural setting of Punjab is different from the neighboring states in north India. Punjab is a state which has a comparatively low prevalence of tobacco use. It exhibits a culture where tobacco use is ostracized. As a comparison, its neighboring states have a higher prevalence of tobacco use which is engrained in their cultural practice irrespective of gender. Thus, every culturally diverse group has distinct needs. This opens an opportunity to tailor our intervention that appropriately fits the needs and preferences of this particular cultural group. We have incorporated the strategies proposed by Kreuter and Wray, 2003
55
for culturally tailored interventions. These strategies emphasize the importance of cultural tailoring in making information more relevant and applicable to the targeted subgroup.
55
Besides, evidence identifies 5 elements/strengths of culturally tailored interventions. We have integrated these elements into the current intervention package: (1) culturally respectful and patient-centric care (Punjabi culture and PCC-based case vignettes); (2) healthy lifestyle promotion (quitting tobacco use); (3) increased family supports (encourage to bring a family member along during sessions); (4) use of technology (SMS to patients that are disease-specific and stage of change-based); and (5) increased knowledge of disease (disease-specific pamphlets that illustrate how continued tobacco use would impact their current disease).
56
The photographs from the community that will be used in the disease-specific pamphlets would be real-time and culturally relatable. Couplets and adages from local and religious contexts would be incorporated into the intervention package. The text messages and pamphlets will be translated into the vernacular language (Punjabi). The local role models would be highlighted for adopting a tobacco-free lifestyle. The tips for managing craving due to tobacco withdrawal would revolve around the available local cultural resources and real-time experiences of tobacco users from the community (Figure 2) ii. Individualized and Patient-Centric Stages of package development
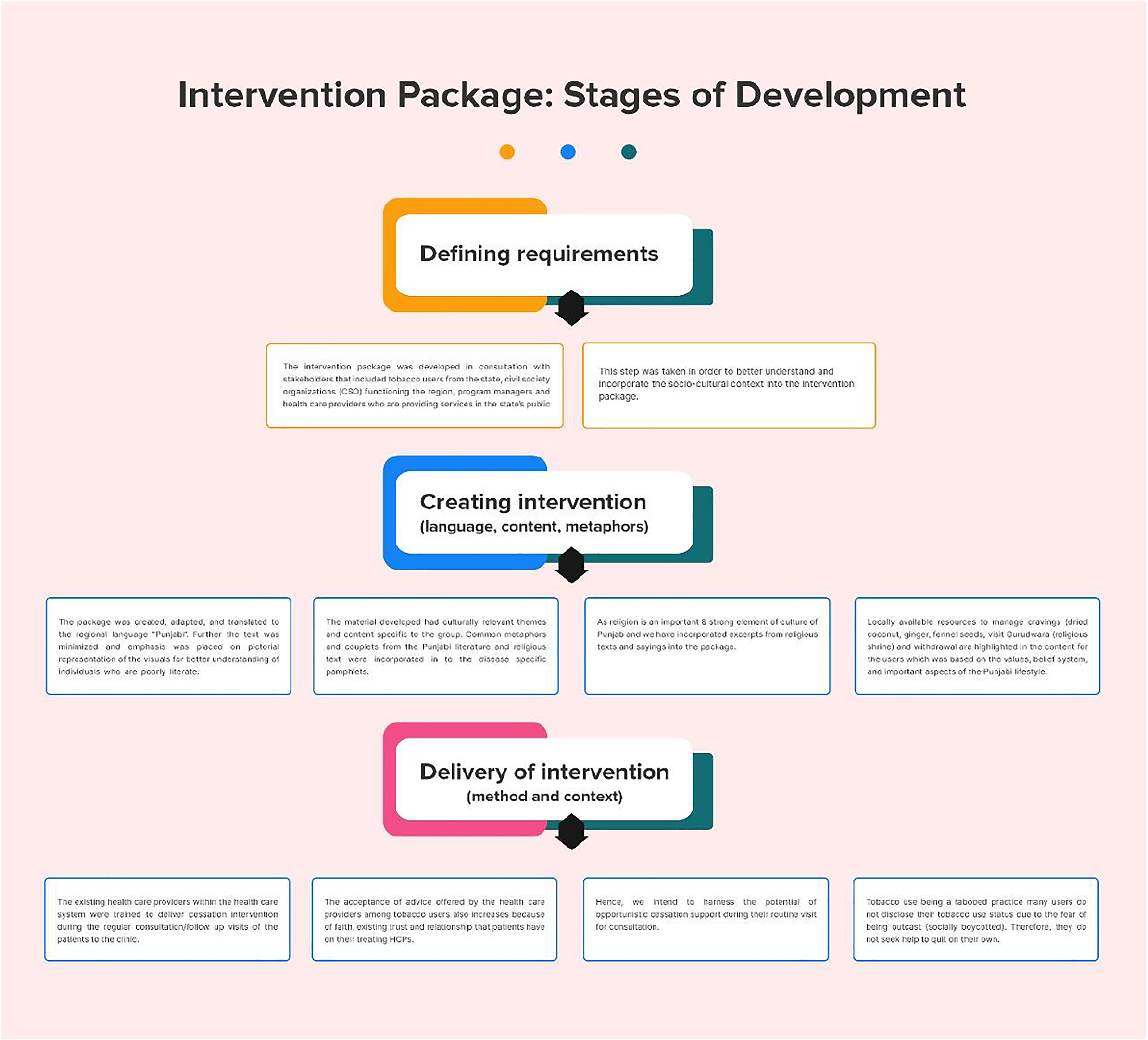
It is intended to use a client segmentation approach in the study. Since different stroke works for different people, the intervention shall be individualized based upon the disease profile of tobacco users and the stage of behavior change. The counselor will use various visual mediums depicting common complications of patient-specific NCDs exacerbated by tobacco use. For example, for a hypertensive tobacco user, how continuing tobacco use would impact the patient’s blood pressure, which could increase their risk of a cardiovascular event. The patient would be asked to draw a decision matrix for himself and list down the pros and cons of continuing and discontinuing tobacco use, and setting a quit date for them. The patient would be encouraged to bring along a family member during each visit. The accompanying family member would be motivated to provide a supportive environment and sit along with the counseling sessions. iii. Trans Theoretical Model Based Strategy
All the events in the lives of people are culturally interpreted. Whatever behavior change is contemplated by the provider, and individuals view it with the prism of culture. Moreover, harnessing the power of TTM of change in adapting multiculturalism has also been documented in various domains. Stage-based model is very relevant because all people are not a single entity and not in the same phase of behavior change. There is evidence that TTM supports the cultural relevance of thematic constructs for various public health interventions such as physical activity, weight control, unhealthy alcohol use, etc. A TTM-based strategy would be adapted to motivate tobacco users depending upon their stage of behavior change and present morbidity. This strategy would be incorporated into counseling sessions and short text messages, which will be sent on the mobile phone. For example:
Stage of Change—Precontemplation
Action intended (e.g.,: diabetic tobacco users)
Session
SMS
**Hello!!
A sensible person will quit tobacco use for himself, his family, and his kids. Start thinking about quitting tobacco at the earliest. Quitting tobacco will keep your sugar levels/Diabetes under control and eliminate/minimize its complications—heart attack, asthma, and other respiratory diseases, cancer, blindness, kidney disease, etc. will also be reduced.
“Choose life not tobacco”!! SPH-PGI, Chandigarh
** Hello would be addressed as “Sat Shri Akal” (in Punjabi) and “Namastey/Namaskar” (in Hindi) in translated text messages. The complete text of messages would be translated to vernacular language “Punjabi” and “Hindi”. Depending upon the language preference of the user, text messages in the preferred language would be sent to the participant. iv. Multimodal Behavioral Intervention Package
The current package encompasses modalities including individualized patient-centric face-to-face counseling, delivery of culture and disease-specific messages, active involvement of family during the intervention, and customized mobile SMS to the stage of behavior change of tobacco users.
Control arm
The “usual care” will be administered in the control arm. The “usual care” includes services that are being provided by the government under the National Programme for Prevention and Control of Cancers, Diabetes, Cardiovascular Diseases, and Stroke (NPCDCS). This will comprise a comprehensive examination, diagnosis, management of the disease by the doctor, and brief advice on quitting tobacco use. The nurse will assist in patient’s examination, imparting education about risk factors of NCDs to the patients and attendants with assistance in follow-up care. The counselor shall provide diet and lifestyle management counseling as well as assistance with follow-up care.
The researcher shall actively monitor both these groups for 12 months.
Outcome indicators
The primary outcome would be 7-day abstinence from tobacco use.
The secondary outcome would include a quit attempt, the number of sticks/pouches of SLT used expenditure on tobacco products, switching to any other tobacco products (if any). Flow diagram for enrollment and follow-up plan of RCT.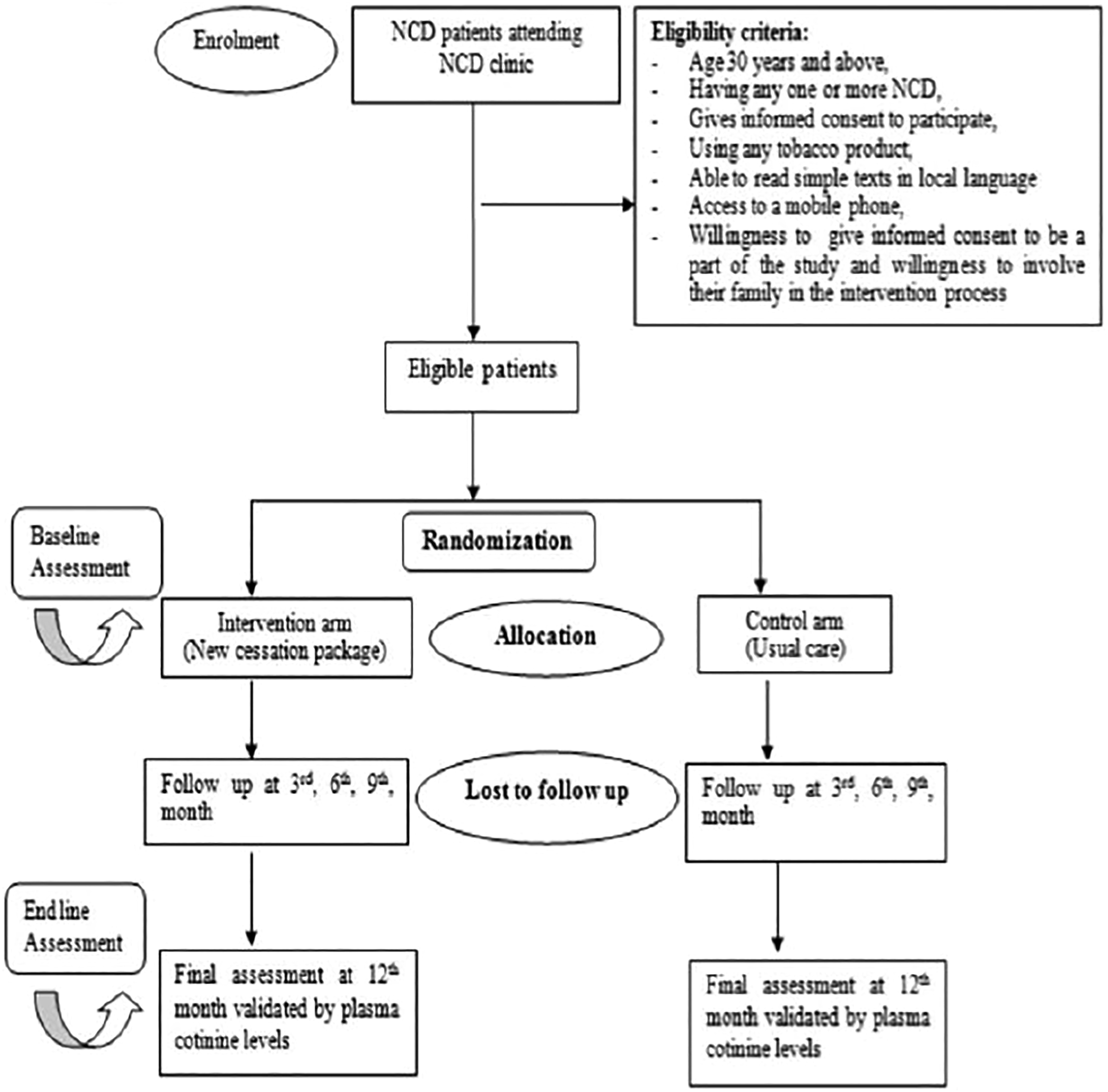
Sample size
For calculating sample size, estimated abstinence of (.9%) in the usual care group and (14.4%) 47 in the intervention group with type I error 5%, power of 90% at 95% of the confidence interval will be taken along with additional 20% of the calculated sample to compensate for the loss to follow-up. STATA version 13SE was used to calculate sample size, using power analysis for 2 sample proportions (Pearson’s chi-squared test). We arrived at a sample size of 160 for the study and multiplied with an adjustment factor for a 20% follow-up loss, resulting in 200 (.ie.,, 100 in each group).
Enrollment, randomization, and generation of random sequence
NCD patients will be screened using a screening tool based upon standardized tools (WHO-STEPS survey and GATS questionnaire),5,57 which will comprise questions on basic socio-demographics, disease history, and duration, along with the history of tobacco use. They will then be screened based upon the inclusion and exclusion criteria. Those who meet the eligibility requirements will be invited to participate during their routine visit to the NCD clinic Participant selection and enrollment.
Concealment of allocation and blinding
Block randomization method would be used to prevent anticipation of the allocation and reduce the researcher’s bias. An independent faculty not involved in the study will generate a random sequence using a computer. The researchers will enroll participants based upon the random sequence and assign them to the intervention or control arm. Participants will be unaware of their allocation; however, the researcher will be aware of the patient’s (participant’s) allocation at the time of enrollment and follow-up to allow appropriate intervention and follow-up questionnaires can be administered.
Data collection
Baseline information
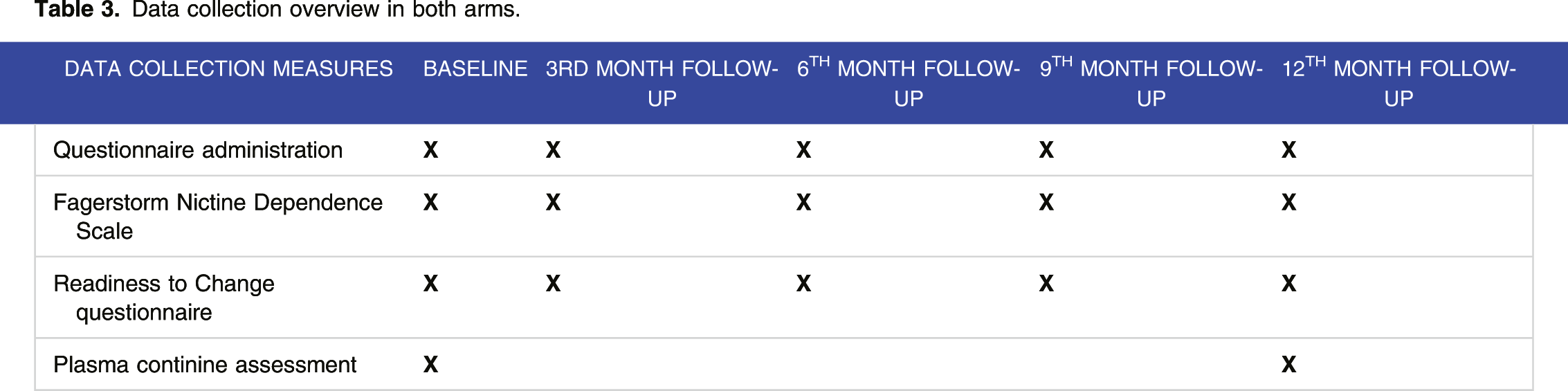
Baseline information from the enrolled patients will be collected by using a pretested questionnaire which will include their basic socio-demographic information, tobacco use history (age of initiation, years of use, type of tobacco product used, quantity and frequency, expenses on tobacco products, any previous quit attempts, etc.), history of NCD (the type of NCD, first diagnosis, number of years, and medication) in both arms. Further, each participant will be administered the Fagerstrom nicotine dependence scale58,59 to assess nicotine dependence and the Readiness to Change questionnaire to determine the stage of behavior change. 60
Follow-up
Data collection overview in both arms.
Training of health care providers
The health care providers of NCD clinics shall be trained on tobacco cessation intervention package by experts in tobacco cessation in collaboration with the Department of Health and Family Welfare, Punjab, through a 1-day workshop. The training workshop curriculum will broadly cover topics like tobacco use as a major risk factor for NCDs and its impact, benefits, and challenges of quitting 5 A’s and 5 R’s algorithm. 61 Further, the researcher will observe them while administering the package in real settings and provide valuable inputs and feedback. Also, qualitative feedback will be sought from health care providers at the NCD clinic, administrative authorities of both programs (NTCP and NPCDCS), and public health experts for improvement in the intervention package.
Data management and analysis
Each study participant will be assigned a unique identification number, and their information and blood samples collected will be stored using that unique identity number. Double data entry will be done, errors will be checked, and data will be analyzed using IBM SPSS version 22. Descriptive analysis will be undertaken for baseline and follow-up variables as appropriate. Chi-square test of the association will be used to compare socio-demographic and tobacco use characteristics between groups at baseline. Logistic regression will be used to undertake between-group comparisons on the primary and secondary outcomes at third, sixth, ninth, and 12th-month follow-up (95% confidence intervals). Multivariate models using multiple logistic regression analysis will be used to identify the correlates of quit attempts. Statistical significance will be set at two-tailed P < .05. Mann–Whitney test will be carried out to compare secondary outcomes (such as the number of quit attempts and number of sticks/SLT pouches used per day). Intention to treat analysis will be done while comparing the outcome in participants in the intervention arm with those in the control arm. To assess the difference in cessation between types of tobacco use, we would compare the overall quit rate between intervention and control groups followed by subgroup analysis among the type of tobacco users.
Discussion
A lack of intervention protocol was cited as a barrier to culturally tailored intervention research in many studies. 56 Culturally tailored intervention has immense potential to reduce health care disparities and improvise the quality of life for specific subgroups. Adopting these strategies will help us in achieving Sustainable Development Goals. This study focuses on smoked as well as smokeless tobacco users, and there is meager evidence in the country where a culture-specific patient-centric intervention package is evaluated among smokeless tobacco users. It undertakes capacity building of existing human resources, further strengthening and increasing the outreach of cessation services, with a potential to ensure suitability within the system for the long term. Further, the study proposes utilizing and encash existing NCD clinics to deliver “opportunistic cessation support.” Development and implementation of culturally tailored patient-centric cessation interventions could retain study participants and decrease dropouts. The application of mobile technology to deliver culturally, linguistically, and stage of change appropriate short text messages could prove to be an efficient method of staying connected and intervention delivery. Further assessment of implementer experiences concerning this intervention could elicit the strengths and areas of improvisation to practice and upscale interventions for culturally diverse groups. In addition, the findings could support health care policymakers to initiate large-scale projects to deliver culturally tailored patient-centric care and enhance health care among diverse socio-cultural groups.
This pilot study protocol outlines the design of a single-blind randomized controlled trial that compares a novel tobacco cessation intervention package with the existing usual care within the NCD clinics running under the NPCDCS Programme. The proposed intervention package shall increase the outreach of cessation services to the population using available resources, thereby strengthening health systems. It is a documented fact that HCPs are uniquely positioned to make a significant difference in the lives of tobacco-using patients suffering from any noncommunicable disease through minimal intervention because of their established relationship and trust with the patient. Further, there are amply documented pieces of evidence that tobacco users benefit in terms of quit rate more in case of disease-specific advice rather than generic advice. To the best of our knowledge, this RCT is the first to assess the effectiveness of utilizing NCD clinics for tobacco cessation services in India. The results of this pilot study could be pertinent to improve the quality of life of patients suffering from NCDs by supplementing efforts in their disease management through behavioral counseling and providing a base for larger-scale trials.
This intervention shall provide an opportunity to intervene when the patient’s receptivity to the advice offered is optimal and mutually benefit both the programmes (National Tobacco Control Programme and NPCDCS) resulting in reduced prevalence of NCDs and tobacco use. Thus, this pilot study shall also guide the policymakers and implementers in adopting this framework for tobacco cessation intervention in other similar settings. The intervention proposed in this study will be developed on the TTM model, which could offer cessation support to the user according to its stage of change. The intervention will be culturally specific and thus more adaptable and applicable in the given context. The involvement of a tobacco user and his family make the intervention patient-centric, where they can collectively develop an individualized quit plan and the user can also garner support from family members during the process. Box-1 List of operational definitions used in study
61

The strengths of the study are its highly rigorous RCT design, where patients are blinded to intervention to prevent contamination; use of biochemical verification of quit status; intensive behavioral counseling package involving tobacco user and his/her family; use of multimodal behavioral intervention package (face-to-face counseling sessions, pamphlets, and short text messages) which is specific to disease and stage of behavior change of tobacco users. The consideration of tobacco users’ socio-cultural and patient-centric approach in developing intervention makes it robust and adaptable in similar settings. The proposed intervention also emphasizes empowering patients to develop their quit plan, building on their needs and concerns. Through this intervention package, we also attempt to harness the power of TTM of change in adapting multiculturalism in tobacco cessation.
However, the social desirability bias attached to tobacco use in Punjab might lead to the underreporting of tobacco use status among the patients attending the NCD clinic. Further, as multispecialty and tertiary care hospitals exist within the study area, patients with cancer, stroke, and cardiovascular diseases may seek consultation and treatment in these health facilities instead of routine NCD clinics. Besides, the longer follow-up period (up to 12 months) may lead to substantial dropouts. Also, the proposed study may fall short of proposing any specific solutions for illiterates due to its limited application among them.
Conclusion
Tobacco use is a major risk factor for developing NCDs and exaggerating its complications. For LMICs and MIC’s wherein financing is a critical issue for sustaining any health programme, the accessibility and affordability of pharmacotherapy becomes a significant bottleneck in the effective delivery of tobacco cessation services. Hence, a culturally specific patient-centric intervention package focusing on behavior change appears to be a sustainable way to help NCD patients quit successfully.
Footnotes
Acknowledgments
We thank the State Tobacco Control Cell and State NCD Control Cell, Department of Health and Family Welfare, Punjab, India for granting permission to conduct the study. We also thank Prof. Amarjeet Singh, Head of Department, Community Medicine, and School of Public Health, PGIMER, Chandigarh for his valuable inputs.
Authors' contributions
SG conceptualized the idea. GB did a review of the literature. SG, RG, SG, and BM designed the study. GB drafted the paper. The draft was critically revised for important intellectual content by all authors and thereafter approved the final version. All authors have read and approved the manuscript. SG is the guarantor for all aspects of the study ensuring those questions related to the accuracy or integrity of any part of the work that is investigated.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported GB is funded by the Indian Council of Medical Research (No. 3/1/3/JRF-2016/HRD), New Delhi and Junior Research Fellowship (JRF) for undergoing PhD program.
Ethics approval
Ethical approval for the study was sought from the Institute Ethics Committee, Post Graduate Institute of Medical Education and Research, Chandigarh, India. (IEC no. INT/IEC/2017/1361). Permissions are also obtained from State Tobacco Control Cell and State NCD control Cell, Department of Health and Family Welfare, Punjab India for carrying out the study. The study protocol is registered with Clinical Trials Registry, India. The registration number is CTRI/2018/01/011 643. Data obtained will be kept confidential and this study will contribute to the PhD thesis of the first author (GB). The eligible participants will also be given a Participant Information Sheet (PIS) containing details about the procedures that would be adopted during the study.
Informed consent
Informed written consent will be requested from the eligible participants in the language which they understand. The participants will be free to decide their participation in the study (Autonomy). The participants in each arm of the study will receive routine health services and those in the intervention arm will receive some additional trial-specific benefits like counseling services, mobile messages, and disease-specific pamphlets. There will be no risk to the participants. There may be a reduction of complications related to tobacco use leading to improved quality of life through tobacco cessation intervention (Beneficence). The trial has no ethnical basis for inclusion criteria in the study (Equity). In case of any amendments, the relevant authorities would be informed. Any unintended effects of trial interventions or trial conduct or reporting any adverse events, from the trial shall be reported to the Institute Ethics Committee (IEC) of PGIMER, Chandigarh, India. All contact details of IEC shall be mentioned on the Participant Information Sheet (PIS) given to the patient before recruiting them to the trial. To protect confidentiality before, during, and after the trial, all information collected about the enrolled participants would be saved and stored using a unique identity number.
