Abstract
Background:
Biomarkers for psychological stress have been examined and the “gut–microbiota–brain axis” is currently attracting attention. An intervention study reported improvements in both the intestinal environment and psychological stress. However, the relationship between psychological stress scores and urinary 5-hydroxytryptamine (u-5-HT), produced by enterochromaffin cells in the intestinal tract, has not yet been investigated over time in healthy subjects under psychological stress. Therefore, the present study examined the relationship between subjective psychological stress (depression and anxiety) scores and u-5-HT levels over time in healthy women.
Methods:
The effects of the objective structured clinical examination (OSCE), considered to be a uniform source of psychological stress, on u-5-HT levels were assessed in 16 third-year female medical university students (21.3 ± 2.1 years old) in Japan with a normal menstrual cycle. A self-administered questionnaire consisting of Zung’s Self-rating Depression Scale (SDS) and State-Trait Anxiety Inventory (STAI) was used to evaluate subjective stress 1 month, 1 week, and 1 day before and 1 week after the OSCE. Pearson’s product-momentum correlation coefficient was used to calculate the correlation coefficient between u-5-HT levels, STAI, and SDS for each examined period.
Result:
On the day before the OSCE, u-5-HT levels correlated with SDS and STAI (SDS: r = .524, P = .037, State-Anxiety: r = −.718, P = .002).
Conclusion:
A correlation was observed between subjective psychological stress scores and u-5-HT levels in healthy women under psychological stress.
Keywords
Background
Biomarkers for stress have recently been examined and the relationship between intestinal bacteria and brain/central nervous system functions, called the “gut–microbiota–brain axis,” has recently attracted attention. 1 Serotonin, biosynthesized from tryptophan, exists in both the brain and gut as a neurotransmitter involved in the gut-brain connection. It also exerts the physiological functions of the brain and gut through various receptors. Approximately 90% of serotonin is stored in intestinal enterochromaffin cells, that is, neuroendocrine cells, and it is also widely distributed throughout the body, including the brain, platelets, pineal gland, kidneys, and spleen. Urinary 5-hydroxytryptamine (u-5-HT), which is mainly produced by enterochromaffin cells in the intestinal mucosa, released into the blood, and excreted in the urine, does not correlate with cerebrospinal fluid serotonin, 2 which may be associated with viral infection and intestinal bacteria.3 -5
Audhya et al and Kema et al demonstrated that u-5-HT levels did not increase as a result of psychological stress in patients with fibromyalgia or carcinoid tumors.6,7 Benton et al conducted a randomized controlled trial on the effects of dairy products containing Lactobacillus casei Shirota to investigate the relationship between psychological stress and intestinal bacteria in healthy subjects. 8 In their analysis of subjects with a high depression score, improvements in depressive mood were significantly greater in the intervention group than in the control group. Sawada et al conducted a randomized controlled trial with a probiotic preparation containing Lactobacillus gasseri CP2305 in long-distance runners, and reported a significant improvement in the State-Trait Anxiety Inventory (STAI) score, an anxiety scale, in the intervention group. 9 Therefore, interventions with intestinal bacteria improve psychological stress through the “gut–microbiota–brain axis” in healthy subjects.10,11 In an observational study of healthy subjects, psychological stress loading was suggested to affect the “gut–microbiota–brain axis” through the regulation of intestinal enterochromaffin cells.3,4 However, the relationship between psychological stress scores and the level of u-5-HT, produced by enterochromaffin cells in the intestinal tract, has not been investigated over time in healthy subjects under psychological stress.
The male-to-female ratio for psychiatric diseases caused by psychological stress is reportedly 1:2. 12 Good mental health in young females is important for preventing psychiatric diseases. An assessment of psychological stress is urinary cortisol by the hypothalamic-pituitary-adrenal (HPA) axis. Urinary cortisol is high upon waking and is influenced by waking time.13,14 It is also known to fluctuate widely during the day. In women, estradiol administration decreases cortisol levels in the blood by decreasing adrenocorticotropic hormone secretion. 15 In fact, women cannot deny that urinary cortisol may be affected because the amount of estradiol regulates (suppresses) the function of the HPA axis. On the other hand, serotonin is secreted more during the day and is assumed to fluctuate less during the day. 16 And it is reported to decrease 2 to 3 hours after lights out. Audhya et al reported that urinary 5-HT concentration has an association with the 5-HT level in cerebrospinal fluid. 6 We have employed the objective structured clinical examination (OSCE; a test that objectively evaluates skills and attitudes prior to clinical practicums) as a uniform psychological stress,17,18 and our findings suggest that human herpesvirus-6 in saliva (s-HHV-6) is applicable to an objective assessment of fatigue caused by psychological and physical stress in young females, including that produced not only by knowledge of, but also skill and attitude aspects of the OSCE. In addition, an increase in u-5-HT levels was detected 1 week after the examination. 19 We examined the relationship between subjective psychological stress (depression and anxiety) scores and u-5-HT levels in healthy women before and after OSCE psychological stress loading over time. This comparison before and after psychological stress can capture changes in the association between subjective psychological stress and u-5-HT levels under psychological stress.
Methods
Description of the data reanalyzed in this study
The data used in this study were those published by Iida et al. 19
Participants
Participants were third-year female students at a medical university in Japan. Volunteers were recruited under the condition that their menstrual cycle was between 26 and 38 days based on the cumulative pattern of menstrual cycles presented by Taylor et al. 20 Based on this menstrual cycle, 2 students were excluded before the survey and 2 students were excluded at the time of the survey. One student who was in the PMS period (within 3 days of menstruation) at the time of the survey was excluded. Sixteen students who participated in a meeting to explain the study were ultimately included (mean age ± standard deviation [SD]: 21.3 ± 2.1 years, height: 160.3 ± 4.3 cm, weight: 53.1 ± 5.3 kg; Figure 1). Based on their self-report, none were smokers. Heavy physical exercise and alcohol consumption the day before sample collection were restricted. The survey was conducted between December 2015 and January 2016 and time was divided into 4 periods: 1 month, 1 week, and 1 day before and 1 week after the OSCE. In addition, psychological stress (Zung’s Self-rating Depression Scale [SDS] and State-Trait Anxiety Inventory [STAI]) scores were previously shown to be significantly higher 1 day before than 1 month before the OSCE. 19 The present study was conducted in accordance with the Declaration of Helsinki and was approved by the Ethical Review Board of the Faculty of Health and Welfare, Prefectural University of Hiroshima (approval No. 14MH045).

Subject enrollment flow diagram. (Previous Study 19 )
The OSCE
The OSCE is a practical examination to objectively evaluate whether clinical skills and attitudes have reached a certain standard. In this study, the OSCE was administered to students studying physical therapy before they began their clinical training. In the OSCE, students were divided into groups of 5 and each group visited 3 different testing stations. While at a station, each student took the practical test for a total of 10 minutes. Specifically, the examinee was initially given 1 minute to read the case description and then 9 minutes to demonstrate the role of a therapist. Two faculty members who were licensed physiotherapists observed each student’s performance and evaluated it according to a pre-prepared checklist. The evaluation was published 2 weeks after the OSCE, which was after the survey period of this study.
The OSCE was used as a final examination given after the completion of a series of courses. Students were required to attend 15 lectures of 90 minutes each and then pass the OSCE to receive credit for the course. Passing the OSCE requires communication skills as well as professional evaluation and treatment skills, which the students had studied for approximately 15 weeks. They were given a large number of possible questions in advance, and based on these questions, practiced intensively during and outside of the lecture times to prepare for the OSCE. On the day of the OSCE examination, each student was expected to be in a state of chronic physical fatigue due to preparation for the examination as well as mental tension from anticipation. Before and after the OSCE, variables of autonomic modulation based on an analysis of heart rate variability in the temporal, frequency, and non-linear domains, the subjective perception of distress train, and academic performance were assessed. Additionally, decreases in sympathetic activity were measured. 21
Survey measures
A self-administered questionnaire survey consisting of SDS 22 and STAI23 -26 was conducted as a measure of subjective stress, and stress-related biomarkers (urine samples) were collected 1 month, 1 week, and 1 day before and 1 week after the OSCE. In consideration of the effects of premenstrual syndrome, the 3 days before the next menstruation (self-reported) were avoided. 27 U-5HT levels were measured as a stress-related biomarker. All urine samples and questionnaires were collected between 12:00 and 13:00 before the participant had lunch. Prior to sample collection, it was confirmed orally that the participant had not engaged in any strenuous physical activity the day before. Urine specimens used to measure u-5HT levels were centrifuged at 1500 rpm for 5 minutes after collection, with the supernatant being frozen and stored at −20°C. Regarding u-5-HT measurements, urine samples were examined with an All Species Serotonin ELISA kit (LifeSpan BioSciences, Inc., Seattle, WA, USA). The calculated u-5-HT value for each sample was obtained by dividing by the amount of creatinine (u-5-HT: ng/mgCr). All concentration measurements were triple-checked to maintain accuracy (R 2 = .92-.96, CV = 0.022-0.025).
Statistical analysis
The sample size was selected according to the change in STAI (state anxiety) scores by the OSCE using G power. Based on the results of a previous study conducted by Iida et al 19 on the relationship between 1 day and 1 week before the OSCE and after considering a correlation coefficient = .62, two-sided α = .05, and power = 0.8, the minimum number of participants was 14, which was increased to 16 to take into account potential attrition.28,29 The normality of SDS and STAI scores as well as u-5-HT levels was confirmed with a histogram together with a Shapiro-Wilk test for normality (P > .20). Pearson’s product moment correlation coefficient was used to calculate correlation coefficients between u-5-HT levels, STAI, and SDS. SPSS 25.0 J (IBM Japan, Tokyo) was used for analyses, with the level of significance set at P < .05.
Results
Relationship between SDS scores and u-5-HT levels
SDS scores received from participants ranged between 23 and 49 (1 month before: 24-48, 1 week before: 29-49, 1 day before: 23-47, 1 week after: 24-46). Two participants had an SDS score of 40 points or higher (cut-off value with SDS used for screening 30 ) 1 month before, seven 1 week before, nine the day before, and three 1 week after the OSCE. A correlation was observed between SDS scores involving all participants (n = 16) and u-5-HT levels on the day before the OSCE (correlation coefficient = −.524, P = .037; Figure 2C). However, SDS scores did not correlate with u-5-HT levels 1 month before (correlation coefficient = −.037, P = .890), 1 week before (correlation coefficient = −.321, P = .226), or 1 week after the OSCE (correlation coefficient = −.189, P = .484).
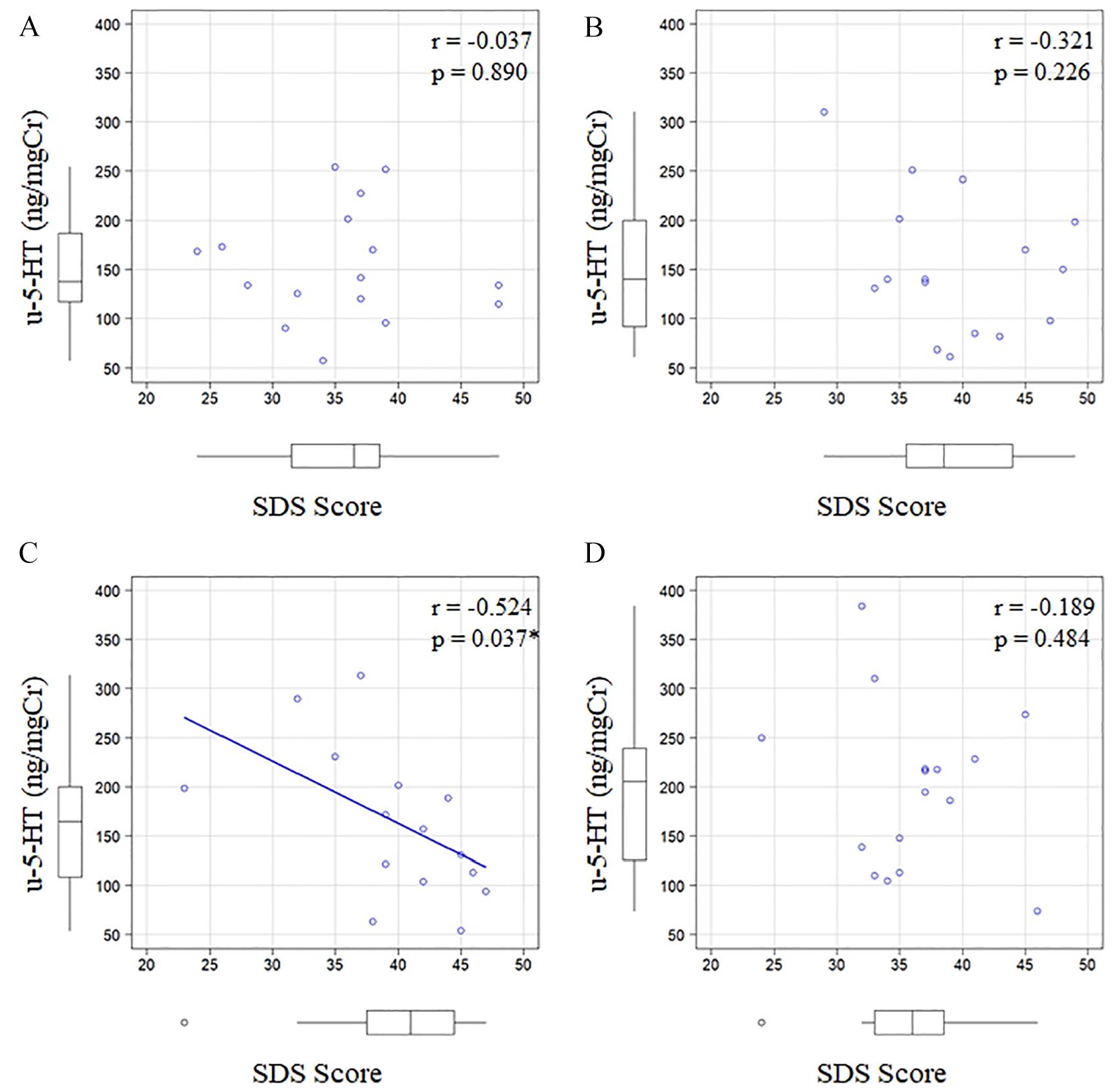
Relationship between SDS scores and u-5-HT levels by the objective structured clinical examination (OSCE) over time. (A) 1 month before the OSCE, (B) 1 week before the OSCE, (C) 1 day before the OSCE, and (D) 1 week after the OSCE.
Relationship between State Anxiety scores and u-5-HT levels
State Anxiety scores received from participants ranged between 23 and 68 (1 month before: 23-47, 1 week before: 28-66, 1 day before: 33-68, 1 week after: 23-46). Four participants had a State Anxiety score of 42 points or higher 31 1 month before, nine 1 week before, nine the day before, and six 1 week after the OSCE. A correlation was noted between State Anxiety scores involving all participants (n = 16) and u-5-HT levels on the day before the OSCE (correlation coefficient = −.718, P = .002; Figure 3C). However, State Anxiety scores did not correlate with u-5-HT levels 1 month before (correlation coefficient = −.012, P = .963), 1 week before (correlation coefficient = −.108, P = .691), or 1 week after the OSCE (correlation coefficient = −.259, P = .333).

Relationship between State Anxiety scores and u-5-HT levels by the objective structured clinical examination (OSCE) over time. (A) 1 month before the OSCE, (B) 1 week before the OSCE, (C) 1 day before the OSCE, and (D) 1 week after the OSCE.
Relationship between Trait Anxiety scores and u-5-HT levels
Trait Anxiety scores received from participants ranged between 31 and 68 (1 month before: 31-50, 1 week before: 35-64, 1 day before: 37-68, 1 week after: 31-53). Four participants had a Trait Anxiety score of 45 points or higher 31 1 month before, eight 1 week before, nine the day before, and six 1 week after the OSCE. A correlation was observed between Trait Anxiety scores involving all participants (n = 16) and u-5-HT levels on the day before the OSCE (correlation coefficient = −.496, P = .050; Figure 4C). However, SDS scores did not correlate with u-5-HT levels 1 month before (correlation coefficient = .140, P = .605), 1 week before (correlation coefficient = .002, P = .994), or 1 week after the OSCE (correlation coefficient = −.178, P = .509).

Relationship between Trait Anxiety scores and u-5-HT levels by the objective structured clinical examination (OSCE) over time. (A) 1 month before the OSCE, (B) 1 week before the OSCE, (C) 1 day before the OSCE, and (D) 1 week after the OSCE.
Discussion
The present study investigated the relationship between subjective psychological stress (depression and anxiety) scores and u-5-HT levels by the study period of psychological stress loading in healthy women using the OSCE as a psychological stress load. To consider the effect of BMI on u-5-HT levels, a partial correlation analysis adjusted for BMI was performed using data from the day before the OSCE (Table 1). SDS, State Anxiety, and Trait Anxiety scores, all scores of psychological stresses, showed a significant partial correlation when adjusted for BMI. Using data from the day before the OSCE, the discriminatory ability between u-5-HT levels and psychological stress scores (SDS, State Anxiety, and Trait Anxiety scores) was analyzed by ROC analysis (Figure 5). This ROC analysis also determined the area under the curve (AUC) adjusted for BMI. The AUC without BMI adjustment analysis was significant for State Anxiety (AUC = 0.897, P < .001) and Trait Anxiety (AUC = 0.778, P = .033) scores, but not for SDS (AUC = 0.730, P = .109) score. In the BMI-adjusted analysis, SDS (AUC = .794, P = .015), State Anxiety (AUC = 0.968, P < .001), and Trait anxiety (AUC = 0.857, P < .001) scores, all scores showed significant AUC. The results obtained demonstrated a high correlation coefficient between State Anxiety and SDS scores on the day before the OSCE and u-5-HT levels, but not during the other times examined. This may be partially explained by stress-induced inflammatory changes in the intestinal flora. 32 Oxidative stress caused by stress-induced inflammation and viral inflammation increase the expression level of indoleamine 2,3-dioxygenase (IDO), which opens and split the indole ring of tryptophan. 33 IDO is activated by inflammatory cytokines, such as interferon-γ (IFN-γ), 34 thereby inducing immune tolerance and enhancing viral infection. 35 As a result of the activation of IDO, the tryptophan kynurenine pathway may have become predominant, resulting in a decrease in the serotonin pathway. 36 We previously demonstrated an increased HHV-6 level the day before the OSCE and an increased u-5-HT level 1 week after the OSCE. These findings are inconsistent with the present results. However, this may be explained by IFN-α produced by HHV-6 reactivation (induces antiviral proteins in hepatocytes and enhances the expression of human leukocyte antigen [HLA] class-I antigen in virus-infected hepatocytes) peaking at 2 to 5 days. 37 IFN-α, generated by HHV-6 reactivation, activated T cells and produced 5-HT through interactions with enterochromaffin (EC) cells in the intestinal mucosa, resulting in a time lag.2,3,11 Stress-induced inflammation associated with the intestinal flora increased kynurenine from the intestinal flora into the blood and decreased serotonin. 38 As inflammation remitted, changes in the intestinal flora may have been restored by a greater abundance of Lactobacillus spp., leading to an increase in serotonin.32,39 Due to the close proximity of the microflora, the EC cells, and the immune cells in the intestinal mucosa, the interactions between these components may have an influence on the pathophysiology of intestinal inflammation. 40 5-HT functions in the inflammatory direction in pathological conditions and expresses IL-1β, IL-6, and IL-8 via 5-HT3 and 5-HT4.41,42 In relation to the kynurenine pathway, 5-HT is converted to 5-hydroxykynuramine by IDO1, which enters part of the kynurenine pathway and inhibits 5-HT-induced platelet aggregation. 43 However, metabolites of the kynurenine pathway function primarily immunosuppressive and anti-inflammatory, 44 and 5-HT in the intestinal mucosa also functions anti-inflammatory due to EC cell-mediated melatonin synthesis in the basal state.43,45 This mechanism is also substantiated by reactive oxygen species derived from lactic acid bacteria potentially suppressing the kynurenine metabolism of hosts by inhibiting the expression of the metabolic enzyme IDO1 in the gut. 38 Teradaira et al studied acute stress loading by experimental VDT work, and they reported that plasma serotonin increased significantly, but kynurenine did not show significant changes. 46 The study found that serotonin in peripheral blood is released into plasma by platelet activation, but kynurenine produced by IDO was not increased by VDT stress. Kynurenine is produced by IDO in many cells, including leukocytes, vascular endothelial cells, and the gut. According to Teradaira et al study, 46 it is thought that IDO (Kynurenine) changes in the periphery are minimal unless there is a significant disease such as inflammation or depression. Therefore, the association between psychological stress score and urinary serotonin concentration in this study is presumed to indicate the severity of psychological stress. However, this study does not aim to elucidate the kynurenine-mediated mechanism, but this mechanism is only speculative. Therefore, we would like to conduct additional studies to clarify. More than 90% of peripheral 5-HT is synthesized from the essential amino acid tryptophan in the EC cells of the intestinal wall, stored within the EC cells, and then released into the mucosal and mesenteric layers of the gut.47,48 5-HT has an important function in the stimulation of peristalsis and other intestinal processes. 5-HT stored in EC cell vesicles is released into the mucosal lamina propria or lumen when EC cells are exposed to intraluminal pressure or chemical and mechanical stimuli. 49 Released 5-HT is metabolized locally by uptake by intestinal epithelial cells and then by vascular endothelial cells, lung, and liver after entering the systemic circulation. 50 5-HT is primarily metabolized by monoamine oxidase A (MAO-A) to 5-hydroxyindoleacetic acid (5-HIAA). However, not all 5-HT is excreted as 5-HIAA in the urine. Increased 5-HT production in the gut, as seen in carcinoid syndrome, leads to elevated platelet 5-HT levels and increased urinary excretion of 5-HIAA. 51 Separately, Mulder et al reported that u-5-HIAA and u-5-HT were highly correlated in normoserotonemic and hyperserotonemic autistic patients. 52 These findings do not prove whether u-5-HT is proportional to intestinal serotonin synthesis. However, it is speculated that much of the u-5-HT is derived from the intestinal chromaffin cells. Plasma serotonin is derived from platelets as well as the gut. Teradaira et al reported significant increases in plasma serotonin levels and plasma platelets derived growth factor (PDGF) in a pre- and post-comparison of acute stress loading from experimental VDT work. 53 PDGF is released from alpha granules in platelets. Their study shows that the increase in plasma serotonin levels due to acute stress load includes platelet-derived increases. This results might be due to the possibility that oxidative stress from cortisol induced by the HPA axis activates platelets 54 and the presence of glucocorticoid receptors (GR) in the platelet cytoplasm. 55 On the other hand, kynurenine was not increased by VDT work stress. 46 From these findings, it might be inferred that the increased u-5-HT levels in the present study are a response to medium-term psychological stress rather than an acute stress response, although the intestinal or platelet origin cannot be clearly established. In this study, we did not measure intestinal microflora by next-generation sequencing, short-chain fatty acids (colon mucosal environment index), or urinary indoxyl sulfate (indole concentration produced from tryptophan due to deterioration of the intestinal environment), which are indicators of the intestinal environment.56,57 Future studies are needed.
Relationship between urinary 5-HT levels and SDS, state anxiety, and trait anxiety on the 1 day before the OSCE.
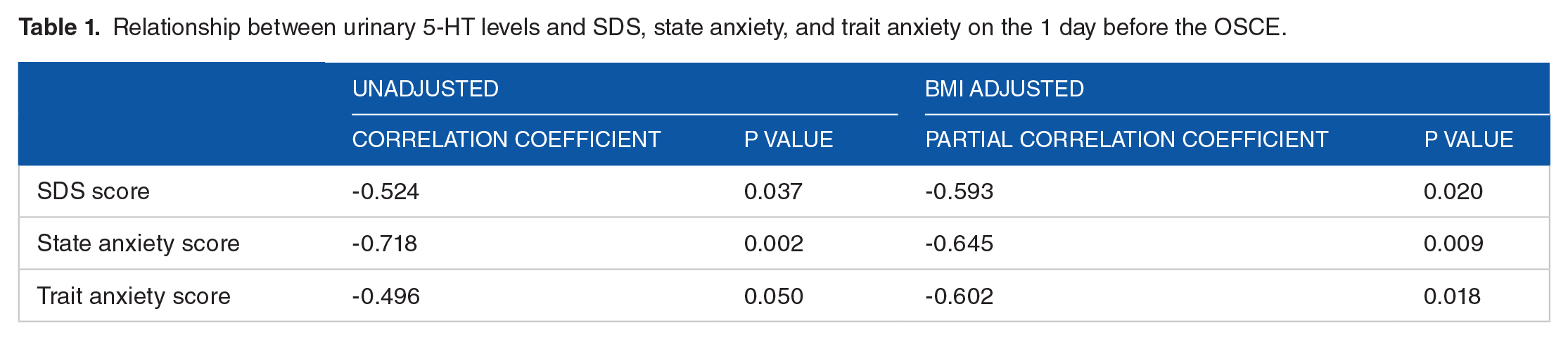

ROC curves at 1 day before the OSCE for each SDS, state anxiety, and trait anxiety score. The solid line dhows the results for the urine 5-hydroxytryptamine, the dotted line those for the urine 5-hydroxytryptamine adjusted for BMI. The AUCs (p-values) in urine 5-hydroxytryptamine and urine 5-hydroxytryptamine adjusted for BMI for each SDS, state anxiety, and trait anxiety score were 0.730 (0.109), 0.794 (0.015) (SDS); 0.897 (<0.001), 0.968 (<0.001) (state anxiety); 0.778 (0.033), 0.857 (<0.001), respectively.
An increase in cortisol through the responsiveness of the HPA axis, caused by psychological stress, enhances the expression of tryptophan 2,3-dioxygenase in the liver, which, similar to IDO, increases blood kynurenine and suppresses serotonin, resulting in a decrease in the urinary excretion of 5-HIAA. 58 Koenig et al compared urinary 5-HIAA levels between schizophrenia patients and healthy individuals as a control group, and found lower levels in the former than in the latter. In addition, lower urinary 5-HIAA levels in schizophrenia patients were more strongly associated with symptom severity and a poor prognosis. 59 Furthermore, urinary 5-serotonin levels correlated with 5-HIAA, while serum 5-HT levels correlated with 24-hour urinary 5-HIAA.60,61 These findings substantiate the correlation observed between psychological stress and u-5-HT levels in the present study.
Ohmatsu et al suggested that an exercise intervention reduced the amount of activity in the anterior cingulate gyrus and increased urinary serotonin levels. 62 Bench et al reported decreased cerebral blood flow in the anterior cingulate gyrus in depressed patients. 63 Therefore, psychological stress may reduce the amount of activity in the anterior cingulate gyrus, resulting in the decrease in urinary serotonin levels found in the present study. Yoshihara et al also demonstrated that the functional relationship between the amygdala and anterior cingulate cortex was critical for the fear-induced activation of sympathetic nerves, that is, the greater the degree of fear, the stronger the cooperation between the left amygdala and anterior cingulate cortex. 64 Specifically, increasingly severe psychological stress may have activated sympathetic nerve activity, aggravated unpleasant emotions, and increased the amounts of activity in the amygdala and anterior cingulate gyrus, resulting in a decrease in urinary serotonin levels. The present study longitudinally examined psychological stress in healthy subjects, revealing a correlation between psychological stress scores and u-5-HT levels only under psychological stress.
This study has several limitations that need to be addressed. The subject cohort comprised female students from a single university; therefore, the results obtained cannot be generalized even though their height and weight were similar to those in a national survey conducted in Japan. 65 In addition, the lifestyles of the students were likely to be similar, unlike the general population, which may also be related to the higher internal validity of the results obtained. Moreover, smoking history relied on self-reporting and nicotine levels in urine may need to be measured in future studies. In consideration of the effects of premenstrual syndrome, no survey was conducted within 3 days before menstruation. However, since premenstrual syndrome may begin up to 10 days before menstruation, its effects cannot be excluded. Although full consideration was given to the confidentiality of the survey content, psychological stress, smoking, and premenstrual syndrome are known to be related.12,66 Another limitation is that psychological stressors were only assessed with SDS and STAI, and participants were not examined by a psychiatrist; therefore, some participants may have been affected by depression. Self-efficacy may also have attenuated SDS and STAI. 67 In a future study, it may be necessary to determine psychological stress in conjunction with the findings of psychiatric examination, in addition to the investigation of urinary kynurenine and short-chain fatty acids.
Conclusion
The present study investigated the relationship between subjective psychological stress (depression and anxiety) scores and u-5-HT levels using the OSCE as a psychological stress load in healthy women. Psychological stress was examined over time in healthy women, and the results obtained revealed a correlation between the severity of subjective psychological stress and u-5-HT levels under psychological stress. The results of this longitudinal study were obtained only through a comparison of before and after psychological stress loading.
Footnotes
Author Contributions
TI conceived the original idea for the study, performed analyses of the data, and drafted the manuscript. YI measured the biomarkers and provided comments regarding the manuscript. SM and YY contributed to the interpretation of data and critical comments, and revised the manuscript. AT commented on results from a psychological perspective, and revised the manuscript. All authors have approved the final version.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI (grant numbers JP18K10583 and JP26350876).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent for Publication
All respondents provided consent to publish the results prior to completing the questionnaires.
