Abstract
For the fluorometric determination of picolinic acid in human serum, HPLC-postcolumn UV irradiation using zinc acetate has been developed. Picolinic acid in serum sample was separated on a Capcell Pak C18. The mobile phase consisted of 0.1 mol/L sodium phosphate solution (adjusted to pH 3.0) containing 3.0 mmol/L zinc acetate and 3.5 mmol/L trimethylamine, and delivered at a flow rate of 0.8 mL/minutes. In order to stabilize the retention time (6.5 minutes), a back pressure tube (0.4 m × 0.13 mm i.d.) was attached after the photoreaction tube. Column effluent was irradiated with ultraviolet light to produce fluorescence, excitation wavelength of 336 nm and emission wavelength of 448 nm. The calibration graph for picolinic acid showed linearity when the amount was in the range of 0.89 to 455 pmol, and the detection limit (S/N = 3) was determined to be 0.30 pmol. The pretreatment of serum sample consisted of deproteinized by perchloric acid, potassium hydroxide, and mobile phase. The mean recovery of picolinic acid from serum was 99.0%. Using this procedure, the concentration of picolinic acid in serum of a healthy subject was determined.
Keywords
Introduction
Picolinic acid (PIA: 2-pyridinecarboxylic acid) is a metabolite in the kynurenine pathway of tryptophan. PIA has been reported to enhance: Immune competence by inducing apoptosis,1-4 brain, function,5,6 bone metabolism, 7 and therapeutic agent for diabetes mellitus by utilizing the chelating effect.8-10 Therefore, PIA may be of significant diagnostic value in disease detection. However, the utility of PIA as a diagnostic marker relies on development of sensitive and reliable quantification methods capable of quantifying varying PIA levels in complex samples. PIA levels have previously been obtained using various quantitative methods: HPLC,11,12 GC/MS, 13 and LC/MS. 14 Although LC/MS has high sensitivity and peak reliability, it has several disadvantages such as complicated pretreatment due to the matrix effect and periodic maintenance of the equipment. In the present study, it was found that PIA in a zinc acetate reagent produces a fluorophore upon irradiation with UV light. Apply this method, developed a fluorometric system for determination of dipicolinic acid in fermented soybeans. 15 That method, phosphate-citric acid buffer solution, in which zinc acetate hardly precipitates, was used as the mobile phase, but it was difficult to separate the peaks of serum contaminants and picolinic acid in this buffer solution. Therefore, a measurement method of picolinic acid in serum was established by using sodium phosphate solution which is easy to precipitate zinc acetate but is easy to separate from contaminants, and by combining a pretreatment method with good washability and recovery. In addition, the HPLC system was applied back pressure to use a mobile phase free of organic solvents, and the relative standard deviation (RSD) of retention time for at least 5 hours was 0.9%.
Experimental
Chemicals and serum
PIA (2-pyridinecarboxylic acid), DPIA (2,6-pyridinediarboxylic acid), quinolinic acid (QIA: 2,3-pyridinedicarboxylic acid), quinaldinic acid (QA), kynurenic acid (KA), xanthurenic acid (XA), and kynurenine (KY) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Zinc acetate and other chemicals were purchased from Fujifilm-Wako Pure Chemical (Osaka, Japan). All the chemicals were of analytical reagent grade, and used without further purification. The handling of serum was approved by Teikyo University Ethical Review Board for Medical and Health Research Involving Human Subjects (Ethical committee approval No. 21-104 and approval list of 25 February 2011). Blood was collected from 1 healthy subject and serum was prepared at 4°C. Human serum was stored at −30°C until analyzed. PIA in frozen serum is stable for at least a week.
Measurement of PIA fluorescence spectra using the manual method
Table 1 consists 1 mL of 0.3 mol/L zinc acetate aqueous solution were added to 2.0 mL of PIA (2.2 nmol) solution or its analog mentioned in Chemicals section. In a 6 cm diameter Petri dish, the mixture was irradiated for 5 minutes with germicidal light of 15 W, from a distance of 15 cm. Table 2 compares the various metal salts, it is the same procedure except that metal salts in sodium phosphate solution (pH 3.0) and irradiation for 20 minutes. The excitation (ex.) and emission (em.) spectra was measured by a Fluorescence Spectrophotometer RF-5000 (Shimadzu, Kyoto, Japan).
Relative fluorescence intensity (ex. 336 nm, em. 448 nm) of similar compounds under the various conditions.
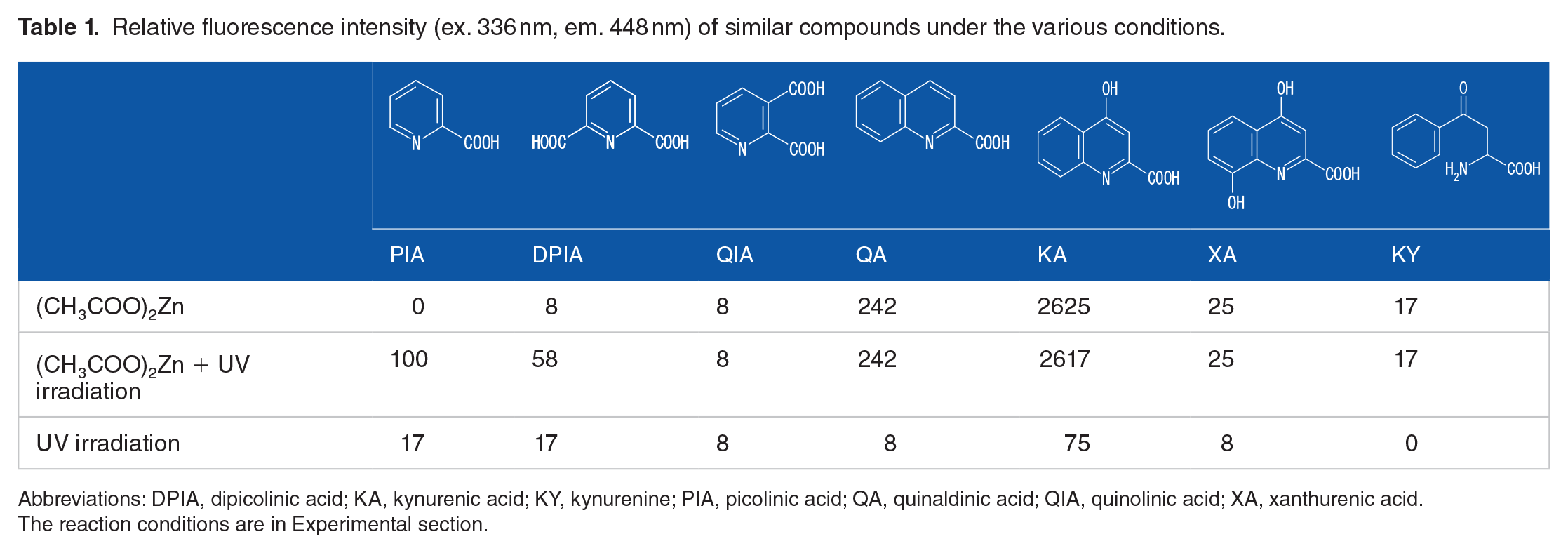
Abbreviations: DPIA, dipicolinic acid; KA, kynurenic acid; KY, kynurenine; PIA, picolinic acid; QA, quinaldinic acid; QIA, quinolinic acid; XA, xanthurenic acid.
The reaction conditions are in Experimental section.
Effect of metal salts on the fluorescence intensity of picolinic acid by manual method.

The reaction conditions are in “Experimental” section.
0.1 mmol/L.
Chromatographic system
The chromatographic system used in this study consisted of a high pressure pump (Model LC-20AT, Shimadzu, Kyoto, Japan), a sample injector fitted with a 100 μL loop (Model 7125, Rheodyne, Berkeley, CA, USA) an analytical column (250 mm × 4.6 mm i.d.) with Capcell Pak C18 (particle size 5 μm, Type II, Osaka Soda, Osaka, Japan), photoreactor by UV irradiation, fluorescence spectrophotometer RF-10AxL (Shimadzu) and a Chromatopac C-R6A (Shimadzu), recorder-integrator.
As a post-column reaction system with a single pump for the addition of zinc acetate to the mobile phase, the reduction of sensitivity due to diffusion was prevented. The mobile phase, 500 mL of 0.1 mol/L sodium dihydrogen phosphate solution and 500 mL of 0.1 mol/L disodium hydrogen phosphate solution (adjusted to pH 3.0 with phosphoric acid) containing 3.5 mmol/L zinc acetate and 3.5 mmol/L triethylamine, was delivered at a flow rate of 0.8 mL/minutes. The retention time of PIA was 6.5 minutes. UV irradiation was carried out in a co-polymer of ethylene and tetrafluoroethylene (ETFE) tube (10 m × 0.25 mm i.d. × 1.5 mm o.d.; GL Sciences Inc., Tokyo, Japan) which was wound around a germicidal light (Model GL-15; 15 W, NEC, Tokyo, Japan). After the eluate passed through the reaction coil, a PEEK tube (0.4 m × 0.13 mm i.d.) and a PTFE tube (1.0 m × 0.25 mm i.d.) were installed to cool and apply back pressure. In this condition, the back pressure was 30 kg/cm2. The fluorescence was measured with ex. and em. wavelengths of 336 and 448 nm, respectively.
Pretreatment of human serum
Fifty microliters of 1.0 mol/L perchloric acid was added to 100 µL of the serum in a polypropylene tube (1.5 mL). The mixture was then vortex mixed and centrifugation (9600×g) for 1 minutes. Subsequently, the entire mixture was added to 20 µL of 1.5 mol/L potassium hydroxide and 50 μL of 0.1 mol/L NaH2PO4 to 0.1 mol/L Na2HPO4 (1:1) buffer. After vortex-mixing and centrifugation, a 100 µL of the pretreatment fluid supernatant was injected into the chromatograph.
Results and Discussion
Optimization of fluorometric derivatization
The fluorescence reaction due to the addition of metal salts were reported by Iinuma et al. who observed increased fluorescene of KA 16 and XA 17 after addition of zinc acetate or calcium nitrate without UV irradiation, respectively. In Table 1, it compares the fluorescence intensity due to photoirradiation at the same conditions as KA without UV irradiation. The detection wavelength was fixed at ex 336 nm and em 448 nm, and the fluorescence intensity of PIA was set at 100 by the manual method. It also determined zinc acetate relative fluorescence intensity with and without UV-irradiation in compounds with PIA and the analog: DPIA, QIA, KY, QA, KA, and XA. PIA emitted fluorescence by UV-irradiation in the presence of zinc acetate. Likewise, DPIA also emitted fluorescence. The fluorescence intensity of QIA did not change in the absence and presence of zinc acetate and UV irradiation. For QA, KA, XA, and KY, fluorescence intensity increased the addition of zinc acetate solution even in the absence of UV irradiation. Although the fluorescence intensities of QA, KA, and XA were high sensitivity, the retention time was very late and the serum concentration was very low, so there was no effect on the determination of PIA. Table 2 shows the effects of various metal salts on fluorescence intensity. The relative fluorescence intensity with zinc acetate was much higher than that with magnesium oxide, cobalt chloride, chrome (III) acetate, iron (III) chloride, and calcium nitrate using the manual method. PIA has been shown to form chelates with iron (III) ion and then decarboxylate to form pyridine radicals. 18 However, irradiation of PIA with the iron ion did not produced fluorescence. Figure 1 shows the excitation (ex.) and emission (em.) maxima were observed at 336 and 448 nm. Thus, zinc acetate was employed as the fluorogenic reagent for the HPLC-fluorescence method used to analyze PIA in serum. The fluorescence derivatization reaction occurred with the 2-pyridinecarbonyl compound, but was not observed with 3-pyridinecarboxylic acid.

Excitation and emission spectra of the fluorescence produced by PIA upon UV irradiation in the presence of zinc (II) acetate. Bottom line represents sample solution: blank control.
Separation conditions
Figure 2 shows the effect of pH on the fluorescence intensity by the HPLC. Precipitation occurred above pH 3.8 in the phosphate solution. The sensitivity was high at pH 3.5, but the peak of PIA shows a broad peak at pH > 3.6 (Figure 3), it optimized HPLC system by adjusting the mobile phase to pH 3.0. Standard PIA and DPIA peaks, eluted at 6.5 and 7.8 minutes, respectively, are shown in Figure 4.
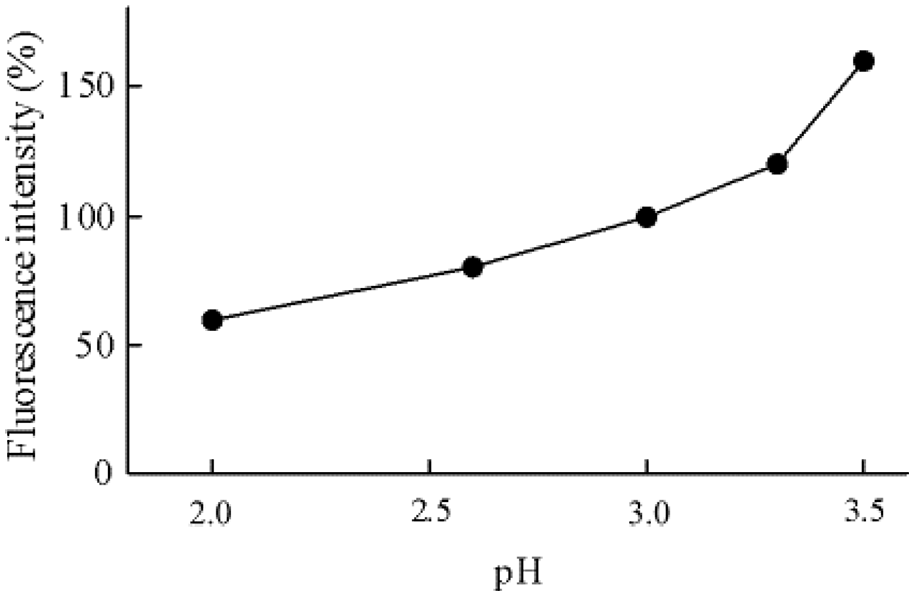
Effect of pH on the fluorescence intensity of picolinic acid.

Chromatograms obtained with picolinic acid.

Chromatogram of PIA and DPIA standard samples.
The highly polar QIA, 3-hydroxyanthranic acid and 3-hydroxykynurenine eluted at approximately 5.0 minutes, and QIA had no effect on the PIA peak due to its low sensitivity. Even further, anthranilic acid, tryptophan, KA and XA had a long retention time and had no effect on the isolation of PIA. The mobile phase containing no organic solvent, it has been reported that the eluent does not sufficiently enter the pores of the filler, and the original separation ability is not obtained.19,20 In this method, the back pressure was applied at 30 kg/cm2, and it became 130 kg/cm2 by the column mounting. When the standard sample of PIA was injected 5 times at 70 minutes intervals without stopping the mobile phase flow, the RSD of retention time was 0.9% (mean = 6.54 minutes, range = 6.45-6.63 minutes) and this retention time was not uniformly decreased. After the end of the measurement, the column was washed with water and then replaced with methanol. The similar results were obtained on the next use.
Reaction conditions
Figure 5 shows the zinc acetate concentration of 8.0 mmol/L in phosphate solution was the most sensitive, but was chosen to be 3.0 mmol/L, which did not precipitate during storage of the mobile phase. The optimum conditions were found to be 3.0 mmol/L of zinc acetate in the ETFE tubing of 10 m length for further photoderivatization. The addition of triethylamine to the mobile phase reduced the precipitation of zinc acetate and contributed to baseline stabilization. The addition of 3.5 mmol/L triethylamine increased assay sensitivity by 20%.

Effect of zinc acetate concentration on the fluorescence intensity of picolinic acid.
Analytical validation
The PIA calibration was linear in a range of 19.6 to 10 010 pmol/mL (amount of 0.89-455 pmol, y = 0.986 x + 2.45, with a correlation of 0.999; y and x are the peak area and the amount of PIA, respectively), and within-day variation, relative standard deviation (RSD) was 3.4% (n = 6) at 19.6 pmol/mL and 0.64% (n = 6) at 640 pmol/mL. Between-day variation was 5.4% (n = 5) at 19.6 pmol/mL and 6.0% (n = 5) at 1144 pmol/mL. The detection limit (S/N = 3) was determined to be 6.6 pmol/mL (amount of 0.30 pmol).
Pretreatment of serum and conditioning
After deproteinization with perchloric acid, potassium hydroxide and buffer were added and the pH of supernatant was about 2. When the supernatant was made neutral or basic, the recovery decreased and column washing took a long time. Therefore, the deproteinized solution was made acidic. At least 50 injections of serum samples did not affect the separation performance. After the injection of the serum sample, a single injection of methanol did not adequately wash the column. The solutions were injected in the following order for repeated analysis. Inject 100 µL of methanol at a flow rate of 1.2 mL/minutes, and after 3 minutes, 20 µL of 0.1 mol/L NaH2PO4 to 0.1 mmol/L Na2HPO4 (1:1) buffer. Fifteen minutes later, 100 µL of methanol. After 20 minutes, set the flow rate to 0.8 mL/minutes. The conditioning recycling time was about 40 minutes.
Recovery from serum
Figure 6 shows the chromatograms of irradiated and non-irradiated serum samples. The chromatograms of standard (a), serum (b), and spiked serum (c) were obtained using photoirradiation, and the chromatogram (d) was obtained by cutting off the UV-irradiation. The chromatogram (d) has no peak at the location of the PIA peak. To determine recovery, PIA was added to the serum sample as 116 pmol/mL in 0.1 mol/L NaH2PO4 to 0.1 mol/L Na2HPO4 (1:1) buffer. The recovery (%) ± SD of PIA added to serum in 2 subjects were 99.4 ± 3.3% (n = 6) and 99.0 ± 3.1% (n = 6).
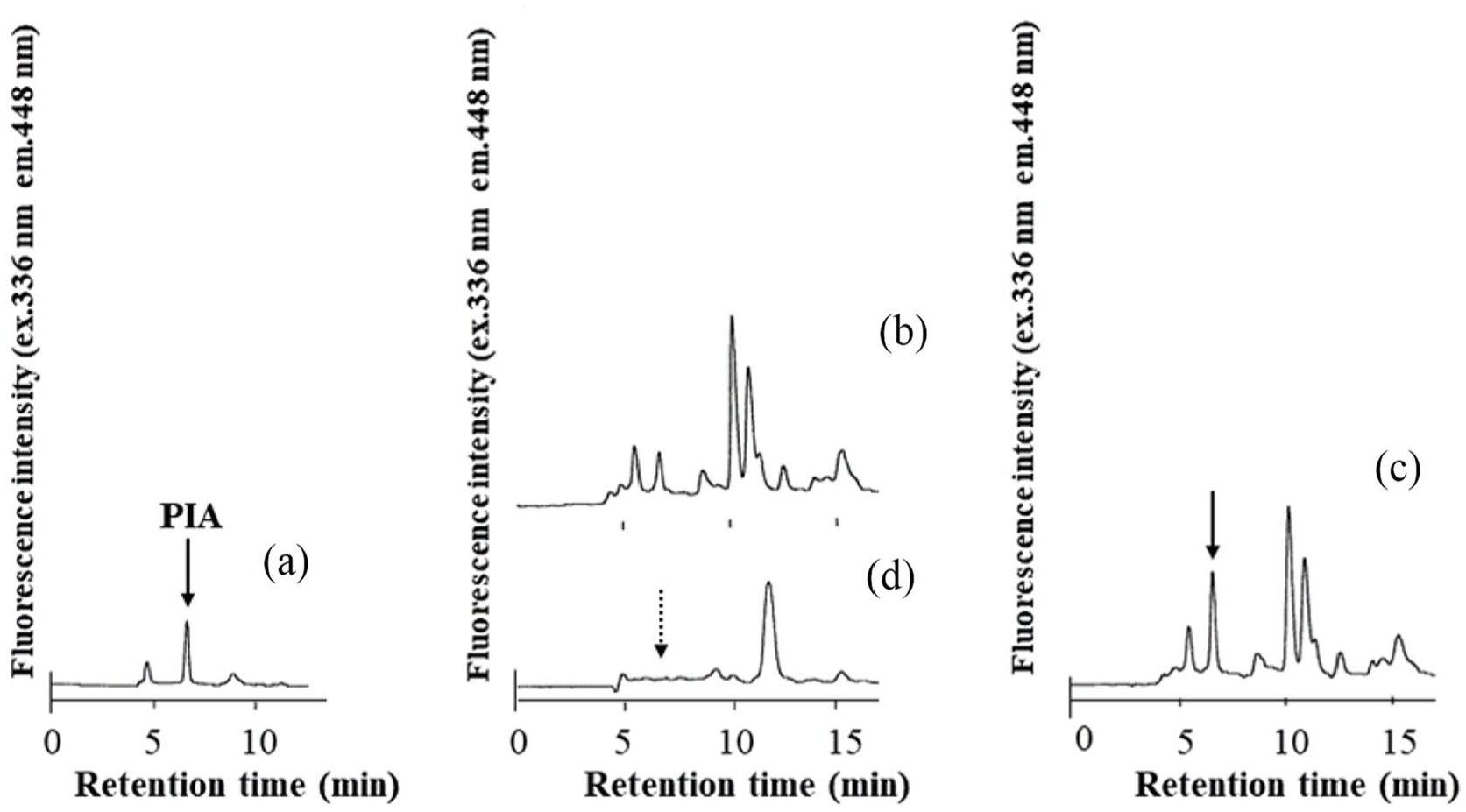
Chromatograms obtained with standard and serum samples. Irradiation with UV light: (a) deproteinized PIA standard (concentration of 57.2 pmol/mL); (b) serum (38.7 pmol/mL in serum); (c) serum spiked PIA (94.8 pmol/mL in serum); and (d) the same sample as in (b), but without irradiation.
Determination of PIA in serum samples from a healthy donor
Table 3 shows PIA contents in serum from 6 healthy subject. The mean ± SD-PIA serum concentration was 41.5 ± 10.5 pmol/mL and a similar value was obtained for human plasma mean concentration determined using LC-MS (43.9 pmol/mL). 14 Variation in the data was thought to be due to differences in diet and beverages.
Picolinic acid contents in the serum.

Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
KM designed the study, and wrote the drafting of the manuscript. YT contributed to analysis and interpretation of data, and assisted in the preparation of the manuscript. All other authors have contributed to data collection and interpretation. All authors approved the final manuscript.