Abstract
A system was developed for determining dipicolinic acid in “natto” using liquid chromatography with fluorometric detection. The compound was separated by reversed-phase chromatography using a mobile phase of 0.1 mol/L disodium hydrogen phosphate, 0.05 mol/L citric acid buffer (adjusted to pH 3.0) containing 3.0 mmol/L zinc acetate and 35 mmol/L perchloric acid. The compound in the column effluent was irradiated with ultraviolet light to produce fluorescence. This fluorescence was monitored at an excitation at 336 nm and an emission at 448 nm. The calibration curve for dipicolinic acid was observed to be linear in a range of 0.2 to 112 ng. The dipicolinic acid content of natto was 7.24 ± 0.54 mg/100 g (wet weight, mean ± standard deviation [SD], n = 6).
Keywords
Introduction
Dipicolinic acid (DPA; 2,6-pyridinedicarboxylic acid) is a tryptophan metabolite of microorganisms and is found in natto, a foodstuff consisting of fermented soybeans. Dipicolinic acid has various biological activities,1,2 such as affecting the heat resistance of bacterial spores3,4 and inhibiting platelet aggregation. 5 Consequently, a method for determining DPA levels would help elucidate DPA’s biochemical mechanism and also to assess the quality of commercially available natto.
The DPA quantification has been carried out using a colorimetric assay, 6 gas–liquid chromatography, 7 fluorometry,8,9 and liquid chromatography. 10 However, these methods require a time-consuming pretreatment. In this work, a method for the determination of DPA by high-performance liquid chromatography (HPLC) coupled with postcolumn photoirradiation with zinc acetate has been developed. To provide a DPA assay by separating admixture components, using a Capcell Pak C18 (type of MG III) column and in the derivatization reaction, zinc acetate is added to a 2-picolinic acid compounds, and ultraviolet (UV) light is emitted to generate fluorescence. Natto is made into a paste, subsequently weighed, dissolved, and deproteinized with perchloric acid to prepare a sample for injection. Thus, the system could be adapted for determining DPA in natto by a simple pretreatment.
Experiment
Chemicals
DPA (Figure 1) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Zinc acetate and other chemicals were purchased from FUJIFILM Wako Pure Chemical (Osaka, Japan). All chemicals were analytical reagent grade.

Structure of dipicolinic acid (DPA).
Natto sample
Commercially available natto was used as an analytical sample until the flavor expiration date and was stored at 4°C until analyzed.
Fluorescence spectra of UV-irradiated DPA
Fluorescence spectra were recorded using a fluorescence spectrophotometer (F-7000; Hitachi, Tokyo, Japan). Reaction conditions: to 0.5 mL of DPA solutions (10 μg/mL) was added 2.5 mL of the mobile phase in a petri dish. The mixture was irradiated with germicidal light (Model GL-15BL) for 20 minutes.
Chromatographic system
The chromatographic system (Figure 2) comprised a high-pressure pump (Model LC-20AD, Shimadzu, Kyoto, Japan), a sample injector (Model 7125, Rheodyne), an analytical column (250 × 4.6 mm i.d.) of Capcell Pak C18 (type of MG III; OSAKA SODA, Osaka, Japan), an RF-20Axs fluorescence spectrophotometer, and a Chromatopac C-R6A data processor (Shimadzu, Kyoto, Japan). Ultraviolet irradiation was carried out in an ethylene tetrafluroethylene tube (8.0 m × 0.25 mm i.d.), which was wound around a “germicidal light” source (Model GL-15). The mobile phase of 0.1 mol/L disodium hydrogen phosphate, 0.05 mol/L citric acid buffer containing 35 mmol/L perchloric acid and 3.0 mmol/L zinc acetate (pH 3.0), was delivered at a flow rate of 0.8 mL/min. Fluorescence was measured at excitation and emission wavelengths of 336 and 448 nm, respectively.
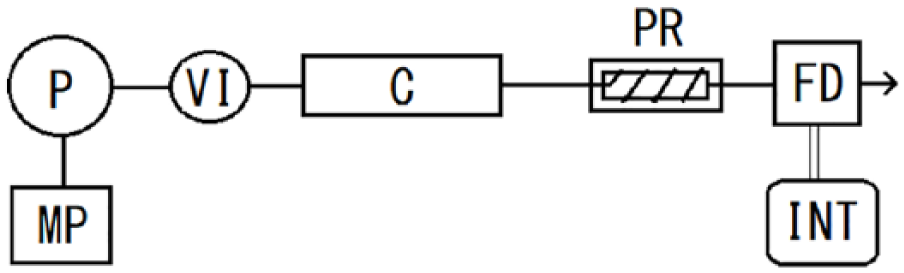
Flow diagram of postcolumn photoderivatization system. Conditions are given in the “Experiment” section.
To avoid any cross-contamination and regenerate the column, the flow rate was increased to 1.2 mL/min and 100 μL of tetrahydrofuran (THF) were injected into the column. After 10 minutes, flow rate was decreased to 0.8 mL/min and 100 μL of mobile phase were injected.
Analytical validation
Calibration graphs were based on the analysis of standard solution of DPA with injection amounts of 13.3 to 7467 ng/mL by injecting a 15 μL volume. The detection limit was determined as three times the baseline noise. Intraday and interday precisions for the developed method were measured in terms of relative standard deviation (%) with 240 and 333 ng/mL DPA, respectively. To determine the recovery, diluted sample of natto was prepared by adding 160 ng/mL DPA standard solution.
Pretreatment of natto
Five grams of natto were grinded into paste using a mortar and pestle. And, 0.5 g of paste was suspended in 100 mL of deionized water and sonicated for 1 minute at room temperature in an ultrasonic cleaner. The suspension (200 μL) was then deproteinized by adding 100 μL of 0.5 mol/L perchloric acid. Excess perchlorate was removed by adding 100 μL of 0.5 mol/L potassium chloride. Prior to injection, the deproteinized suspension was 0.2-μm-filtered and 15 μL was injected into the HPLC.
Results and Discussion
Fluorescence spectra of UV-photoirradiated DPA
Figure 3 shows the excitation and emission spectra of the fluorescence produced by picolinic acid and DPA on UV irradiation in the presence of zinc acetate. The excitation (ex.) and emission (em.) maxima were observed at 336 and 448 nm, respectively.
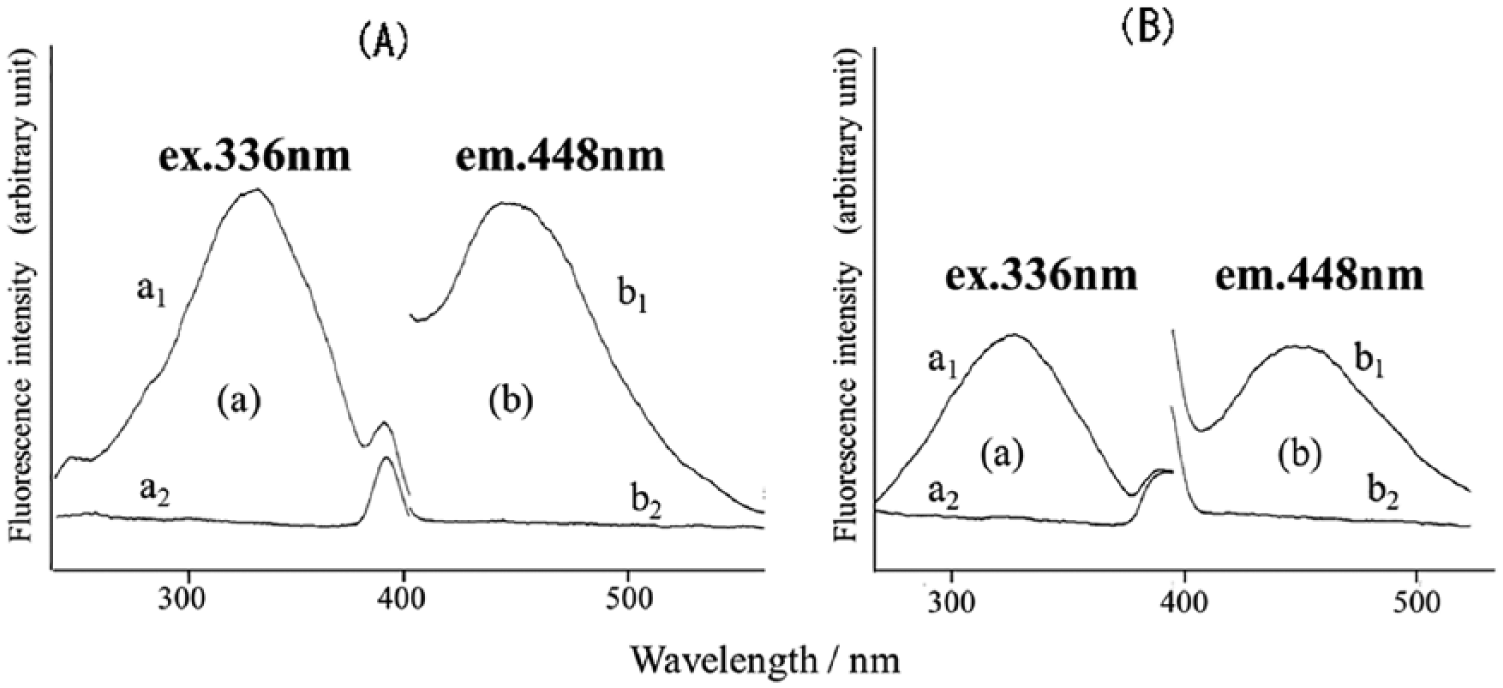
Fluorescence excitation and emission spectra of picolinic (A) and dipicolinic acid (B) obtained by the method of “Experiment” section. a1 and b1, DPA; a2 and b2, sample blank for a1 and b1, respectively.
When the metal salts made the fluorescence intensity of zinc acetate 100% by UV-irradiation, magnesium (II) sulfate was 18%, and calcium (II) acetate, iron (III), and iron (II) chloride did not show the fluorescence. Although the fluorometry using zinc acetate is an example of kynurenic acid developed at our laboratory, 11 the presented method does not fluoresce without photoirradiation. In the pyridine-2-carboxylic acid derivatives, eg, pyrazine-2-carboxamide (PZ) and glipizide (GPZ), they produced fluorophore with photoreaction using zinc acetate. Pyrazine-2-carboxamide and glipizide were observed at ex.367 nm, em.419 nm and at ex.364 nm, em.416 nm, respectively. The photoreaction of 2-pyridinecarboxylic acid was reported including chelate componds. 12 Sugimori et al 13 reported the formation of 2,2′-bipyridine in the photoreaction of 2-pyridinecarboxylatoiron (III) complex. In this method, the fluorophore of DPA was still unknown.
Chromatographic conditions and photoderivatization
The mobile phase did not contain an organic solvent, and the C8 column had insufficient separation of DPA and impurities. Furthermore, in Capcell Pak ADME filled with an adamantyl group, DPA peak observed a broader peak than C18 column. Capcell Pak C18 was able to obtain a sharp peak by adding perchloric acid to the mobile phase.
Figure 4 depicts a typical chromatogram of a standard DPA sample. The retention time of DPA was 7.7 minutes. In addition, retention time of picolinic acid was 8.7 minutes.
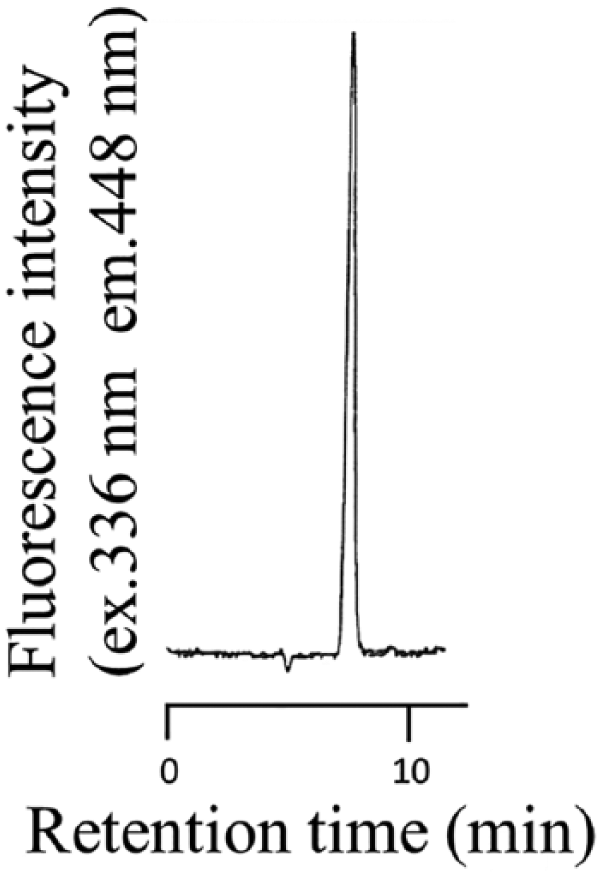
Chromatogram of DPA standard sample. The photoirradiation reaction was carried out as described in the “Experiment” section using amount of 5.0 ng DPA.
In the mobile phase, the concentration of zinc acetate for the maximum fluorescence intensity was observed at 3.0 mmol/L. Figure 5 shows the maximum fluorescence of pH was observed at pH 4.0, but the pH 3.0 was adopted, because of became clouded in the mobile phase at pH 4.5 or higher. The optimum condition of coil length was 7.0 to 9.0 m; here, 8.0 m length was adopted.

Effect of (A) pH and (B) coil length for DPA on the fluorescence intensity.
Validation of the chromatographic system
Table 1 shows the validation results for this method. The intraday and interday precisions for DPA was <7% (n = 6). The DPA calibration was linear in a range of 0.20 to 112 ng (y = 10.7x − 0.44, r = 0.999) and relative standard deviation was 3.6% at 3.6 ng (n = 8). The recovery (%) of DPA was >92% (n = 6).
Validation results for DPA.

Abbreviation: DPA, dipicolinic acid.
S/N = 3.
Mean ± SD.
Figure 6 depicts chromatograms of DPA in natto. The DPA peak was observed at the corresponding retention time after irradiation (Figure 6A), but not when the sample was not irradiated (Figure 6B), demonstrating the specificity of the method.

Chromatograms obtained with natto samples. (A) Natto sample irradiated with photoreactor. (B) The same sample as in (A), but without irradiation. Conditions are given in the “Experiment” section.
More peaks were detected at higher retention times. They were not sufficiently removed by methanol injection and took several hours to analyze the next sample. It was thought that octadecyl silylated silica (ODS) group of packing material in the column became a ligand collapse in mobile phase containing methanol14,15 and stood up in THF. Therefore, to avoid any cross-contamination and regenerate the column, THF were injected into the column. After 100 μL of mobile phase were injected, column was conditioned for 40 minutes before new sample injection (Figure 7).

Chromatogram illustrating column regeneration method. Injection volume of tetorahydrohuran (THF) and mobile phase is 100 μL, respectively.
The mean ± SD of DPA in a commercially available natto sample was 7.24 ± 0.54 mg/100 g (wet weight, n = 6) by the presented assay. Sumi and Ohosugi 16 reported that the amount of DPA in natto without and with an autoclaving; 6.64 and 20.55 ± 13.67 mg/100 g, respectively. The value without autoclave treatment is similar to the value by presented assay. The autoclave was used to break spores; however, in our experiments, the amount of autoclaved standard DPA decreased to 14.2% and 17.7% at 121°C, in 20 and 30 minutes, respectively. Therefore, pretreatment with the autoclave may not be appropriate.
This method facilitates easy pretreatment and is highly sensitivity and efficient for determining DPA levels in natto, indicating that it could be useful in nutritional and biochemical studies.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
KM designed the study, and wrote the drafting of the manuscript. MA and FN contributed to analysis and interpretation of data, and assisted in the preparation of the manuscript. All other authors have contributed to data collection and interpretation. All authors approved the final manuscript
