Abstract
The crosstalk between central nervous system (CNS) and gut microbiota plays key roles in neuroinflammation and chronic immune activation that are common features of all neurodegenerative diseases. Imbalance in the microbiota can lead to an increase in the intestinal permeability allowing toxins to diffuse and reach the CNS, as well as impairing the production of neuroprotective metabolites such as sodium butyrate (SB) and indole-3-propionic acid (IPA). The aim of the present study was to evaluate the effect of SB and IPA on LPS-induced production of cytokines and tryptophan metabolites in human astrocytes. Primary cultures of human astrocytes were pre-incubated with SB or IPA for 1 hour before treatment with LPS. Cell viability was not affected at 24, 48 or 72 hours after pre-treatment with SB, IPA or LPS treatment. SB was able to significantly prevent the increase of GM-CSF, MCP-1, IL-6 IL-12, and IL-13 triggered by LPS. SB and IPA also prevented inflammation indicated by the increase in kynurenine and kynurenine/tryptophan ratio induced by LPS treatment. IPA pre-treatment prevented the LPS-induced increase in MCP-1, IL-12, IL-13, and TNF-α levels 24 hours after pre-treatment, but had no effect on tryptophan metabolites. The present study showed for the first time that bacterial metabolites SB and IPA have potential anti-inflammatory effect on primary human astrocytes with potential therapeutic benefit in neurodegenerative disease characterized by the presence of chronic low-grade inflammation.
Introduction
Neurodegenerative disease is becoming an increasing problem due to growing mortality and morbidity worldwide and major socioeconomic impacts. 1 Individual neurodegenerative disorders, for example, Alzheimer’s disease (AD), amyotrophic lateral sclerosis (ALS), Huntington’s disease (HD), and Parkinson’s disease (PD) are heterogeneous. Although they often have overlapping features and etiology, including, genetic mutations, protein aggregations, infections, and neuronal damage, which are frequently associated with chronic activation of the innate immune response and neuroinflammation. 2
Although the immune response is essential in protecting organisms against infections, a chronic, systemic inflammation, even low-grade, has been associated with the development and progression of neurodegenerative diseases.3,4 Inflammatory mediators can cross the blood brain barrier (BBB) and activate innate immune cells, including microglia and astrocytes. These cells recognize endogenously produced heat-shock proteins (HSPs), viral and bacterial antigens, among others, by binding to receptors such as the toll-like receptors (TLRs) which then leads to the production of inflammatory cytokines and chemokines in the CNS. 5
In addition, there is accumulating evidence that alterations of the gut microbiome are associated with the development and progression of neurodegenerative diseases.6,7 Major bacterial species in the gut microbiota, such as gram-negative bacilli Bacteroides fragilis and Escherichia coli, secrete endotoxins, exotoxins, and lipopolysaccharides (LPS),8,9 that in dysbiosis (the perturbation or imbalance of the normal microbiota) can lead to an increase in intestinal permeability (“leaky gut”) allowing these toxins to diffuse and reach the CNS.10,11 Bacterial LPS have been found in lysates of hippocampus and neocortex from AD patients 12 colocalized with Aβ1-42 in amyloid plaques of AD patients. 13
LPS is known to trigger and perpetuate the neurodegenerative processes associated with inflammation in neurological diseases, and is a useful tool to mimic brain inflammation in vitro. 14 LPS can induce the activation of the key rate limiting enzyme of the kynurenine pathway (KP), indoleamine 2,3-dioxygenase (IDO1) in the brain. 15 This enzyme is responsible for catabolizing tryptophan (TRP) to N-formyl-L-kynurenine, which is subsequently hydrolyzed to kynurenine (KYN) producing downstream neurotoxic and/or neuroprotective metabolites, such as kynurenic acid (KYNA).16,17 The activation of the KP is well characterized in primary human brain cells18,19 and leads to an increase in KYN/TRP ratio that indicates increased IDO1 activity which is evident in major neurodegenerative diseases such as, PD,20,21 AD22,23 and ALS. 24
Furthermore, microbiota have been shown to modulate circulating concentrations of TRP by upregulating the synthesis of serotonin in host enterochromaffin cells. 25 In addition, gut microbiota can directly metabolize TRP limiting its availability to the host with direct implications for its metabolism and CNS. 26 Differences in commensal microbiota might impact human health and disease through the production of many metabolites, such as indole-3-propionic acid (IPA) which is catabolized from TRP. IPA production is completely dependent on the presence of gut microflora and colonization with the bacteria Clostridium sporogenes. 27 IPA has been investigated as a possible treatment for AD, 28 as IPA has a potent antioxidant effect against Aβ 1-42 in vitro, 29 and preventing aggregation and deposition of Aβ monomers. 30 In interferon-beta (IFN-β) activated murine astrocytes, IPA showed anti-inflammatory activity by reducing the expression of chemokine (CC Motif) ligand 2 (CCL2) and nitric oxide synthase 2 (NOS2). 31
Sodium butyrate (SB) is another bacterial-derived metabolite with potential as a treatment for neurodegenerative diseases. Butyrate is a short-chain fatty acid produced by the fermentation of no digestible polysaccharides by bacterial flora inside the colon. SB has been studied for its neuroprotective effect in an AD mouse model 32 and in vitro33–35 where it has shown an ability to protect against DNA damage in aged mice.36,37 Low levels of key butyrate-producing bacteria, in addition to higher relative abundances of taxa known to cause pro-inflammatory states, were found in stool samples from AD patients in comparison with samples from non-dementia subjects. 38
Although the evidence suggests that IPA and SB have protective effects with potential therapeutic implications for neurodegenerative diseases, there are no studies evaluating the anti-inflammatory effects of these compounds in vitro on human primary astrocytes. The aim of our study was to evaluate the effects of IPA and SB on inflammation related cytokines levels and TRP metabolites in human fetal astrocytes challenged with LPS.
Materials and Methods
Cell culture
Human fetal brains were obtained from 16 to 20-week-old fetuses collected following therapeutic termination with informed consent. This protocol has been approved by the human research ethics committee of Macquarie University (ethic approval: 5201600719). To prepare the astrocytes, a small piece of brain was washed twice in Dulbecco’s phosphate buffered saline (DPBS) (Invitrogen, Australia) to remove contaminating blood and then the tissue was placed in a 50 mL falcon tube containing 20 mL RPMI medium (Invitrogen, Australia) supplemented with 10% FBS, 1% L-glutamine, 1% Glucose, and 1% antibiotic/antimycotic. Repeated trituration of the mixture with a mixing cannula attached to a syringe resulted in a homogenous cell suspension. The cell suspension was then transferred to a culture flask and incubated at 37°C in a humidified atmosphere containing 95% air and 5% carbon dioxide. Medium changes were done twice per week until cells grown to confluence. Astrocyte cultures became confluent after 10 to 15 days. After cells reached confluence, they were subjected to successive trypsinisations (3-4 passages) with 0.25% Trpysin-EDTA (Invitrogen, Australia) for the removal of contaminating cells. Cultures were maintained for up to 8 weeks in total.39,40 Prior to receive any treatment the astrocyte cultures were allowed to equilibrate for 24 hours at 37°C in a humidified atmosphere containing 95% air/5% CO2.
Assessment of cellular toxicity
Astrocytes were plated in black 96-well culture plates at a density of 3 × 104 cells/mL. SB (Sigma-Aldrich) and IPA (Sigma-Aldrich) were used for the cell treatment. SB was diluted in DPBS (stock solution) and then subsequently diluted in RPMI medium to produce 0.05 mM, 0.5 mM, 1 mM, and 5 mM working solutions. IPA was diluted in DMSO (stock solution) and subsequently diluted in RPMI medium to produce 1 µM, 10 µM, 20 µM, and 50 µM working solutions. The highest concentration of IPA represents 0.05% DMSO in the final concentration, which did not cause toxicity to the cells (data not shown). Astrocytes were pre-treated with SB and IPA, and incubated for 1 hour at 37°C and before treatment with LPS (10 ng/mL). Alamar blue (ThermoFisher) was used to evaluate cell toxicity and was added to the culture 4 hours prior to assay end in accordance with manufacturer’s guidelines. The fluorescence signals were measured in a PHERAstar Microplate Reader at an excitation wavelength at 540 to 570 nm and an emission wavelength at 620 nm at 24, 48, and 72 hours after the pre-treatment.
Astrocytes treatment and supernatant collection
The cells were plated in 24-well culture plates at a density of 1 × 105 cells/mL in a total volume of 500 µL (50 000 cells/well). The treatment was performed 24 hours after seeding. The cells were pre-treated with RPMI medium (control), SB or IPA working solutions (section 2.2) and incubated for 1 hour at 37°C. After 1 hour, LPS (10 ng/mL) or RPMI medium (control) were added to the culture for 24 hours, after which supernatants were collected by pipetting and centrifuged at 3000g prior to aliquot to remove debris and immediately frozen at −80°C for analysis.
Analysis of KP metabolites
All reagents used for ultra-high-performance liquid chromatography (uHPLC) and high-performance liquid chromatography (HPLC) were of analytical grade and purchased from Sigma-Aldrich unless otherwise specified. The methodology was conducted as previously described.18,39 Cell supernatants were deproteinized with equal volume of 10% trichloroacetic acid (1:1) and filtered through a 0.22 μm PTFE syringe filter (Merck-Millipore, CA, USA). TRP and KYN quantification was performed using an Agilent 1290 infinity uHPLC coupled with temperature-controlled auto-sampler and column compartment. A diode array detector and fluorescence detector were used for the analysis of these metabolites with a 20 μL sample injection volume. Separation of metabolites was performed under a stable temperature of 38°C for 12 minutes, using 0.2 mM sodium acetate (pH 4.65) as mobile phase, with an isocratic flow rate of 0.75 mL/min in an Agilent Eclipse Plus C18 reverse-phase column (2.1 mm × 150 mm, 1.8 μm particle size). TRP was detected using fluorescence intensity set at Ex/Em wavelength of 280/438 and KYN was detected using UV wavelength at 365 nm; standards of TRP and KYN were used to generate a 6-point calibration curve in order to interpolate metabolite concentration. Agilent OpenLAB CDS Chemstation (Edition C.01.04) was used to analyze the chromatogram. KYNA was measured using an Agilent 1200 series HPLC system following injection (10 µL) onto a Poroshell RRHT C-18, 1.8 μm 2.1 × 100 mm column (Agilent Technologies, Inc, Santa Clara, CA) and quantified via fluorescence detection (excitation and emission wavelengths of 344 nm and 388 nm, respectively. The mobile phase consisting of 50 mM sodium acetate with 0.25 M of zinc acetate and 2.25% (v/v) acetonitrile. The inter- and intra-assay coefficient of variation is within the acceptable range of 3% to 8% calculated from the repeated measures of the metabolite standards incorporated during the sequence run.
Cytokine assay
The cytokine array assay was performed on the astrocyte supernatant (section 2.3) by Eve technologies (evetechnologies.com; Calgary, Canada). Levels of GM-CSF, IL-1beta, IL-2, IL-4, IL-5, IL-6, IL-8, IL-10, IL-12(p70), IL-13, MCP-1, TNF-α were evaluated in order to assess the inflammatory profile of the astrocyte supernatants.
Statistical analyses
Data from astrocyte toxicity assays are expressed as percentage of the control ± standard error of the mean, results from the analysis of KP metabolites are expressed as percentage of the control ± standard deviation and results from cytokine assay results are expressed as mean ± standard deviation. All analyses were performed using 2-way ANOVA followed by Bonferroni post hoc test, P < .05 was considered significant. All statistical analysis was performed using GraphPad Prism version 5.01.
Results
The effect of SB and IPA on astrocyte toxicity was assessed using alamar blue. SB or IPA showed no significant toxic effect on astrocytes at concentrations tested, with or without LPS challenge, at incubation periods up to 72 hours (Figure 1A and B).

Effect of SB (A) and IPA (B) on viability in LPS-stimulated primary human astrocytes assessed using alamar blue. The data are expressed as percentage of the untreated control group ± standard error of the mean from 3 independent experiments using cultures derived from 3 different fetal brains.
Next, we assessed the effects of SB alone or as pre-treatment prior to LPS challenge on cytokine levels 24 hours after treatment (Figure 2). LPS challenge did not cause a significant change in IL-2, IL-4, IL5, or IL-10. LPS challenge induced an increase in the proinflammatory cytokines, GM-CSF, MCP-1, IL1-β, IL-6 (P < .0001), IL-8 (P < .001), IL12 (P < .0001), IL-13 (P < .001), and TNF-α (P < .0001), as assessed by 2-way ANOVA. SB pre-treatment attenuated the LPS-induced increases in GM-CSF and MCP-1 at the doses 1 mM (P < .05 for both drugs) and 5 mM (P < .001 and P < .01, respectively); IL-6 at the dose 5 mM (P < .05); IL-12 at all tested doses (P < .001); and IL-13 at the doses 0.05 mM, 0.5 mM (P < .001) and 1 mM (P < .01). Interestingly, SB pre-treatment 5 mM (P < .001) increased the levels of IL-8 compared to LPS treated control. However, none of the tested SB doses attenuates the levels of IL1-β, TNF-α.

Effect of SB on cytokine levels in LPS-stimulated primary human astrocytes 24 hours after treatment. nd = not detected. The data are expressed as mean ± standard deviation from 3 independent experiments using cultures derived from 3 different fetal brains.
Similarly, we examined the effect of IPA pre-treatment on cytokine levels 24 hours after LPS challenge (Figure 3). LPS challenge did not cause a significant change in IL-2, IL-4, IL-5, or IL-10. LPS challenge induced an increase in the cytokines GM-CSF and MCP-1 (P < .0001), IL1-β (P < .01), IL-6 and IL8 (P < .0001), IL-12 (P < .001), IL-13 (P < .01), and TNF-α (P < .001). IPA pre-treatment attenuated the LPS-induced increases in MCP-1 at the dose 50 µM (P < .01); IL-12 at all tested doses (P < .001); IL-13 at the doses 1 µM, 10 µM (P < .01), and 50 µM (P < .05); and TNF-α at the doses 20 µM (P < .05) and 50 µM (P < .01). However, none of the tested IPA levels attenuates the levels of IL1-β, IL-6, or IL8. Is important to mention that most of the data from IL1-β, IL-2, IL-4, IL-5, IL-10, IL-12, IL-13, and TNF-α are extrapolated by semi-log regression, even though some FI is below the first standard point this is not below the assays lowest point of detection for the analyte. However, it can affect the variation and accuracy of the data.

Effect of IPA on cytokine levels in LPS-stimulated primary human astrocytes 24 hours after treatment. nd = not detected. The data are expressed as mean ± standard deviation from 3 independent experiments using cultures derived from 3 different fetal brains.
Finally, we assessed the ability of SB and IPA pre-treatment to modulate changes in the KP induced by an LPS challenge in astrocytes. Treatment of astrocytes with LPS treatment increased KYN/TRP ratio, potentially indicating induction of IDO1 (Figure 4A). SB pre-treatment at 0.5 mM, 1 mM, and 5 mM (P < .01) significantly attenuated KYN/TRP ratio induced by LPS, which returned to non-LPS control levels (Figure 4A; 2-way ANOVA, interaction [F (4, 10) = 5.170; P = .01], followed by Bonferroni post hoc correction). KYN levels (untreated: 0.50 µM ± 0.11; LPS: 0.70 µM ± 0.22) were decreased by SB pre-treatment at doses of 0.05 mM (0.46 µM ± 0.04; P < .05) and 0.5 mM (0.34 µM ± 0.03), 1 mM (0.35 µM ± 0.02) 5 mM (0.34 µM ± 0.01) (P < .001), compared to the LPS control group; interaction [F (4, 10) = 3.62; P = .04] (Figure 4B). There were no statistical differences in TRP, ANOVA interaction [F (4, 10) = 0.29; P = .87]; (untreated: 20.3 µM ± 0.15; LPS: 20.14 µM ± 0.46) (Figure 4C) or KYNA levels [F (4, 10) = 0.29; P = .87], (untreated: 102 nM ± 6.07; LPS: 100.01 nM ± 7.17) (Figure 4D). Although the trend of IPA pre-treatment on KP metabolites in LPS challenged astrocytes was similar to SB pre-treatment, reductions in KYN/TRP ratio (Figure 5A) and KYN (Figure 5B) were not significant; ANOVA interaction [F (4, 10) = 2.34; P = .12] and [F (4, 10) = 1.29; P = .33], respectively. It possibly reflecting the low number of fetal brains used in KP experiment (n = 2) due to unavailability of tissue. There were no statistical differences in the TRP levels (Figure 5C), ANOVA interaction [F (4, 10) = 0.06; P = .99], or KYNA levels (Figure 5D), interaction [F (4, 10) = 0.07; P = .99].
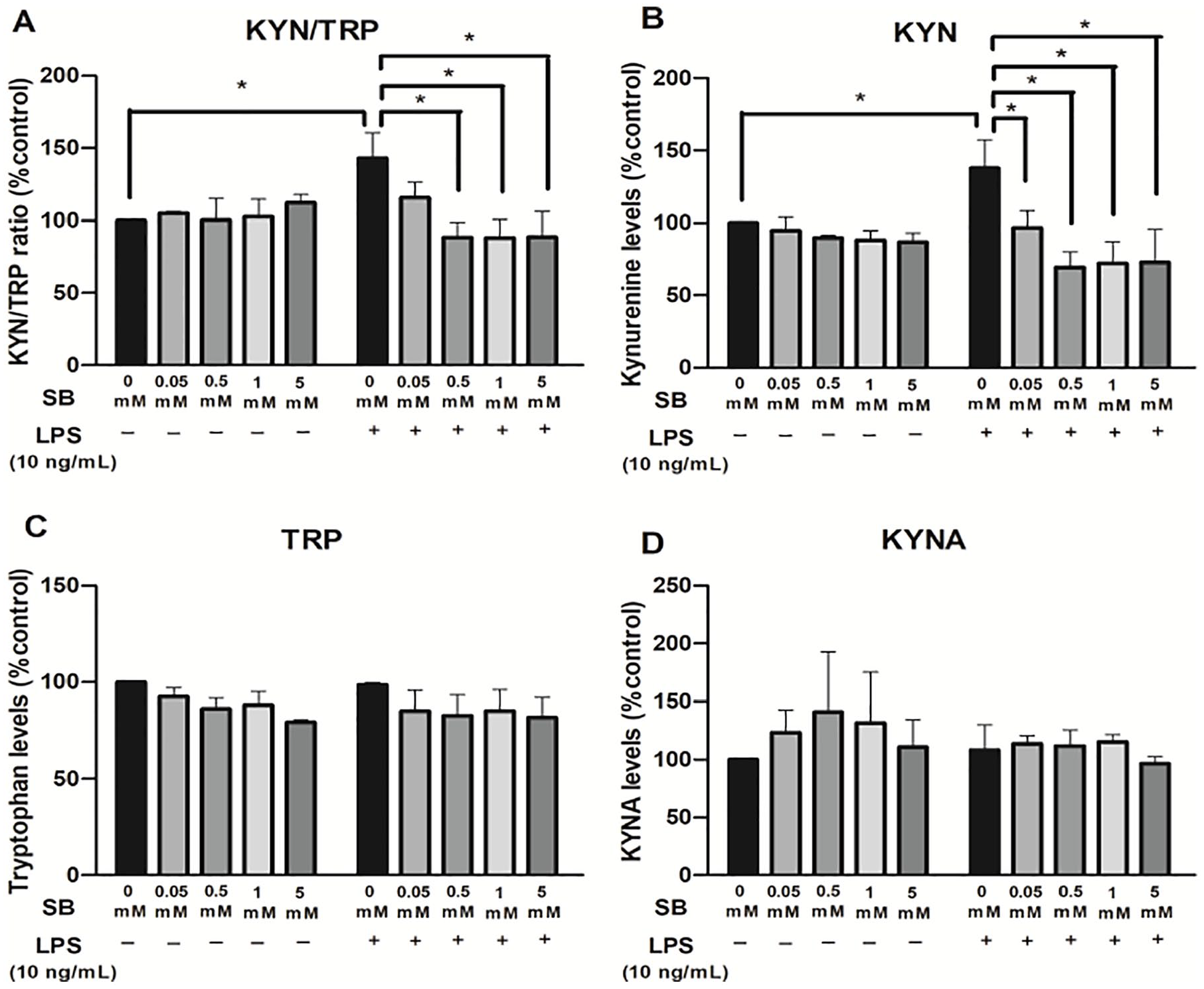
Effect of SB on (A) KYN/TRP ratio and (B) KYN, (C) TRP and (D) KYNA levels in LPS-stimulated primary human astrocytes 24 hours after treatment. The data were analyzed and expressed as percentage of the untreated control group ± standard deviation from 2 independent experiments using cultures derived from 2 different fetal brains.

Effect of IPA on (A) KYN/TRP ratio and (B) KYN, (C) TRP, and (D) KYNA levels in LPS-stimulated primary human astrocytes 24 hours after treatment. The data were analyzed and expressed as percentage of the untreated control group ± standard deviation from 2 independent experiments using cultures derived from 2 different fetal brains.
Discussion
Astrocytes represent approximately 30% of glial cells in the mammalian CNS. 41 Reactive astrocytes respond to pathological stimuli in the CNS and undergo dramatic morphological changes and alterations in their gene expression. This change can be harmful or beneficial depending the insult and the level of astroglial activation. For example, reactive astrocytes in ischemia exhibited a molecular phenotype suggesting a neuroprotective role (A2), whereas reactive astrocytes induced by LPS exhibited a phenotype (A1) that suggests that they may be detrimental. 42 LPS have been associated to trigger inflammation in neurodegenerative diseases and have been used as a tool to mimic brain inflammation in vitro. Even though it was proposed that human astrocytes do not respond to LPS, 43 others have found increased IL-1β gene expression 44 or reported an extensively inflammatory proteomic phenotype after LPS stimulation, which included cytokine release from human astrocytes. 45
As observed in the present study, LPS did not affect astrocyte viability, corroborating with other studies showing that LPS does not affect the viability of primary rat astrocytes 46 or humans astrocytes. 47 Unsurprisingly, we found that LPS caused an increase in proinflammatory cytokine production that has the potential to disturb the CNS homeostasis maintained by astrocytes. Since healthy astrocytes play a key role in maintaining and modulating normal neuronal communication, synaptic physiology and energy metabolism, 48 these modulations may exert deleterious effects on neuronal integrity and network functions.
In the present study, we demonstrated that SB and IPA were able to reduce the levels of proinflammatory cytokines produced by LPS-activated astrocytes. These results show that both SB and IPA have an anti-inflammatory effect on astrocytes experiencing inflammation. Since chronic neuroinflammation is common and highly deleterious phenomenon, 49 this work has relevance in the investigation for new therapeutic approaches for neurodegenerative diseases.
Using primary cultures of human astrocytes, we showed that SB prevents the LPS-triggered increase of GM-CSF, MCP-1, IL-6 IL-12, and IL-13. In particular, GM-CSF, MCP-1, and IL-6 play a role in mediating BBB permeability, where LPS activation of the innate immune system enhances HIV transport across the BBB.50,51 In neurodegenerative diseases, BBB leakage is increasingly being shown to play a significant role during its progression.
The anti-inflammatory effects of SB have been previously evaluated in a few studies. SB (1.2 mM) reduced TNF-α levels in mesencephalic neuron-glia cultures from rats pre-treated 24 hours prior to stimulation with LPS (10 ng/mL). 33 SB (5 mM) also prevented IL-6, TNF-α and nitric oxide increase induced by LPS in co-cultures of rat cerebellar granule neurons, astrocytes and microglial cells. 34 In the present study, we also observed decreased IL-6 levels using the same SB dose (5 mM), but we did not observe the same effect on TNF-α level. This may be because our study did not use a neuron-astrocyte co-culture, but pure primary human astrocytes.
IL-12 and IL-13 are major cytokines involved in the type 1 and 2 immune response to infection respectively. These cytokines promotes differentiation of various T-cell populations to contain infection and assist with the resolution of local inflammation. 52 However, a prolonged chronic or unresolved infection would results in constant recruitment of immune cells that would be detrimental to the local area. Our study shows that, when SB and IPA is used as a pre-treatment, IL-12 and IL-13 levels return to control levels, perhaps indicating a shift to an overall non-inflammatory environment, 53 and can be a new avenue for anti-inflammatory treatment.
In addition, SB appeared to be able to prevent IDO1 activation and the resulting increase of the KYN/TRP ratio resulting from LPS stimulation. In addition to IDO1, Tryptophan 2,3-dioxygenases (TDO2) also metabolizes TRP, TDO2 is strongly expressed in the liver and, at lower levels in astrocytes.54,55 TDO2 is mostly regulated by glucocorticoids; however, a study found IL-1β exposure upregulated its content in astrocytes. 56 Therefore, TRP is metabolized through the KP resulting in the production of neurotoxic metabolites, such as 3-hydroxykynurenine (3-HK) and quinolinic acid (QUIN) by monocytic cells and neuroprotective metabolites; KYNA and picolinic acid (PIC) by astrocytes and neurons.17,57 However, as human astrocytes lack the KP enzyme kynurenine monooxygenase (KMO) they can only synthetize the early metabolites KYN and KYNA. During inflammatory conditions, large amounts of KYN are produced by activated astrocytes, which can be used by local activated macrophages and/or microglia to increase QUIN biosynthesis. QUIN has been demonstrated to kill/damage neurons by at least 6 different mechanisms, the main one being excitotoxicity. 58 In this way, astrocytes could indirectly and inadvertently promote a key neurotoxic process. 18 KP induction with IDO1 activation has been observed in most of the major neurodegenerative diseases including AD,59,60 PD,20,21 and ALS. 61 Thus, during neuroinflammatory conditions, the ability of SB to limit IDO1 activation and KYN production, could be protective.
To our knowledge, there are no previous studies evaluating the effect of the IPA on cytokine levels in human brain cells. Our findings showed, for the first time, that IPA mediates anti-inflammatory effects in primary cultures of human astrocytes through the attenuation of increased MCP-1, IL-12, IL-13, and TNF-α levels induced by LPS. There was only one other article that evaluated anti-inflammatory effect of IPA in the brain, showing that IPA was able to reduce the gene expressions of CCL2 (controlling MCP-1) and NOS2 (activated in inflammatory conditions) in astrocytes isolated from an autoimmune encephalomyelitis mouse model. 31 Another study by Zhao et al evaluated the cytokine levels in hepatic macrophages from mice pre-treated with IPA (250 μM and 500 μM) followed by LPS treatment. IPA was found to inhibit NF-kB and reduced the levels of IL-6, IL-1β, and TNF-α. 62
Since IPA production is dependent on gut bacteria, it can be suggested that changes in the bacterial composition and hence production of bacterial metabolites, such as IPA, could affect cytokine levels in the organism including in the brain.27,63 Although studies on physiological concentrations of butyrate in the brain are scarce, the concentration used in the present study for both treatments are higher than physiological condition. However, the concentration of SB and IPA can vary depending on the diet and gut microbiota composition. 37
In conclusion, we demonstrated for the first time the significant anti-inflammatory effects of SB and IPA through their respective ability to reduce cytokine production in activated human primary astrocytes. In addition, SB can limit IDO1 activation, reversing the increased KYN/TRP ratio in activated astrocytes. As these 2 compounds are currently being assessed as a therapeutic for neurodegenerative diseases,7,64,65 these results provide valuable insights into their neuroprotective mechanisms of action and add to argument that these compounds may have a potential therapeutic effect in neurodegenerative diseases and neurological disorders, in which an inflammatory profile is present.
Footnotes
Acknowledgements
The authors are thankful to the National Council for Scientific and Technological Development (CNPq- Brazil) for the first author’s research fellowship at Macquarie University, Sydney, Australia. Prof Guillemin’s research is funded by the Australian Research Council (ARC), the National Health and Medical Research Council (NHMRC) and Macquarie University. We thank Dr David Lovejoy for his comprehensive editing of the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Garcez is thankful to the National Council for Scientific and Technological Development (CNPq- Brazil) for the first author’s research fellowship at Macquarie University, Sydney, Australia. Prof Guillemin’s research is funded by the Australian Research Council (ARC), the National Health and Medical Research Council (NHMRC), the Handbury Foundation and Macquarie University.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MLG performed all the experiments and wrote the manuscript. BH and VXT have helped MLG with experiments and with the manuscript. GJG supervise the project and help to write the manuscript.
