Abstract
Melatonin (MLT) is a well-known pineal hormone possessed with remarkable biological activities. However, its low oral bioavailability and high first-pass metabolism rate are important pharmacokinetics problems. Therefore, 5 MLT derivatives (
Introduction
Melatonin (
Many studies have examined the pharmacokinetics of MLT and its bioavailability. The pharmacokinetic studies found that exogenously administrated MLT displays poor bioavailability (3%-56%) as a consequence of its extensive hepatic first-pass metabolism. The half-life of exogenous MLT is between 12 and 48 minutes. Cytochrome P450 enzyme CYP1A2 plays a major role in the metabolism of MLT. 7 This hepatic enzyme converts MLT to 6-hydroxymelatonin, which is subsequently bound to sulphate and glucuronide and excreted in the urine.8,9 Our group has designed the derivatives of MLT with the aim to overcome the pharmacokinetic problems of MLT (Figure 1). Lipophilic derivatives of MLT demonstrated a remarkable antioxidant activity and have been proposed to be neuroprotective agents.10,11 In this study, we investigated the MLT release profile of MLT derivatives to identify whether these compounds behaved like drugs or prodrugs. In addition, we employed computational methods to investigate the nature of the interaction between the derivatives and the major metabolising enzyme CYP1A2 and to determine their physicochemical parameters and the drug-likeness of the compounds.

Structure of all compounds in this study.
Materials and Methods
In silico pharmacokinetics prediction
The pharmacokinetic properties of all compounds were predicted by an online accessible web tool: SwissADME program of the Molecular Modelling Group of the Swiss Institute of Bioinformatics, 12 which is publicly available at http://www.swissadme.ch. The software computed various pharmacokinetic properties and descriptors: the octanol/water partitioning coefficient, aqueous solubility, brain/blood permeability, and human gastrointestinal absorption (HIA) capability. Percentage of oral absorption (%ABS) was calculated according to the method of Zhao et al 13 using the following equation:

where TPSA was topological polar surface area
Metabolism prediction
To further investigate the binding mode of MLT derivatives, molecular docking calculations were performed using Autodock VINA.
14
The crystal structure of human microsomal P450 1A2 (CYP1A2) in complex with alpha-naphthoflavone (PDB code: 2HI4, retrieved from the RCSB Protein Data Bank) was used as the receptor.
15
The bound inhibitor and water molecules were removed and only the coordinates of the protein complexed with heme molecules were kept. The 3D structures of ligands were built and their geometry optimisations (HF/3-21G) were conducted utilising the Gaussian03 program.
16
The AutoDockTool (ADT) version 1.5.6 was used to prepare all the docking input files.
17
All hydrogen atoms were added to the enzyme. The Kollman united atom and Gasteiger-Marsili charges were assigned for protein and ligand, respectively.
18
All of the compounds were docked into the active site of CYP1A2 with the centre of 1.50, 22.90 and 17.50 along x, y and z axis, respectively. A box size of 30 × 30 × 30 Å with a grid spacing of 1 Å was set up. During the docking calculation protocol, the protein was treated as a rigid body while rotation and translation of ligands were allowed. For each complex, the predicted binding free and mode of binding for the selection of the docking pose were based on information from previous work.
19
To avoid the methylation process, the distance between the site of metabolism (SOM) of the ligand and the Fe(II) of heme should not be less than
Determination of the hydrolysis percentages of compounds
All the synthesised compounds were preliminarily evaluated for their ability to release MLT using an 80% human plasma assay by a method slightly modified from previous reports.21,22 Briefly, a solution of 5 mM of each MLT derivative was prepared in acetonitrile (1 mL) and added to 9 mL of 80% human plasma in phosphate buffered solution (PBS) pH 7.4. This solution was incubated and gently shaken at 37 ± 0.5°C in a water bath. An aliquot of 400 µL was withdrawn from each tube after 0.5, 1, 2, 3 and 4 hours. The solution was added to cold acetonitrile (400 µL) and placed in an ice bath in order to stop any further hydrolysis. The mixture was centrifuged at 15 000 ×g and 4°C for 10 minutes. The clear supernatant (500 µL) obtained from each tube was filtered and analysed by HPLC. Chromatographic conditions were a stationary phase Venusil® C18 Plus (4.6 × 250 mm) column, 5 μm particle size (Bonna-Agela Technologies, China) at room temperature. The mobile phase consisted of acetonitrile and water (50:50) with a flow rate 1.0 mL/min. The excitation and emission wavelengths of fluorescence detection were set at 286 nm and 346 nm, respectively. Compound concentrations and percentages in the mixture were quantified by peak area calculation and analysis. Human plasma in this study was obtained from the blood bank of Srinagarind Hospital, Khon Kaen University. The protocol for the hydrolysis study of MLT derivatives was approved by the Khon Kaen University Ethics Committee for Human Research (HE621275).
Results and Discussion
ADME properties and drug-likeness
The physicochemical properties and drug-likenesses of the derivatives were calculated by the SwissADME program (Table 1). The derivatives presented
In-silico ADME properties of MLT, 5-MT and compounds

Abbreviations: %ABS: percentage of oral absorption; cLogP: calculated octanol/water partition coefficient; cLogS: solubility parameter; HBA: number of hydrogen bond acceptors; HBD: number of hydrogen bond donors; Lipinski’s violation: 0 is good and 4 is bad; MW: molecular weight; NORTB: number of rotatable bonds; TPSA: topological polar surface area.
The cLogP and TPSA values of the compounds were plotted to predict human intestinal absorption (HIA) and blood brain barrier (BBB) access (Figure 2). The egg-shaped plot is divided into 3 parts including a grey region (no HIA or BBB access), a white area (HIA) and a yolk (BBB access). MLT, 5-MT and all derivatives are present in the yolk part of the plot, indicating that the molecules probably permeated the BBB. This boiled-egg model also predicted whether MLT, 5-MT and derivatives were substrates of P-glycoprotein (PGP). Red dots (PGP–) represent compounds that are not substrates of the PGP CNS efflux transporter, while, blue dots (PGP+) represent compounds that are substrates of PGP and predicted to pass through the CNS. As shown in Figure 2, MLT, 5-MT and all derivative compounds are represented by red dots and not PGP substrates. Therefore, all derivatives passed the pharmacokinetic requirements for drug-like compound behaviour, suggesting that these compounds had good oral bioavailability and could be considered as CNS drugs.
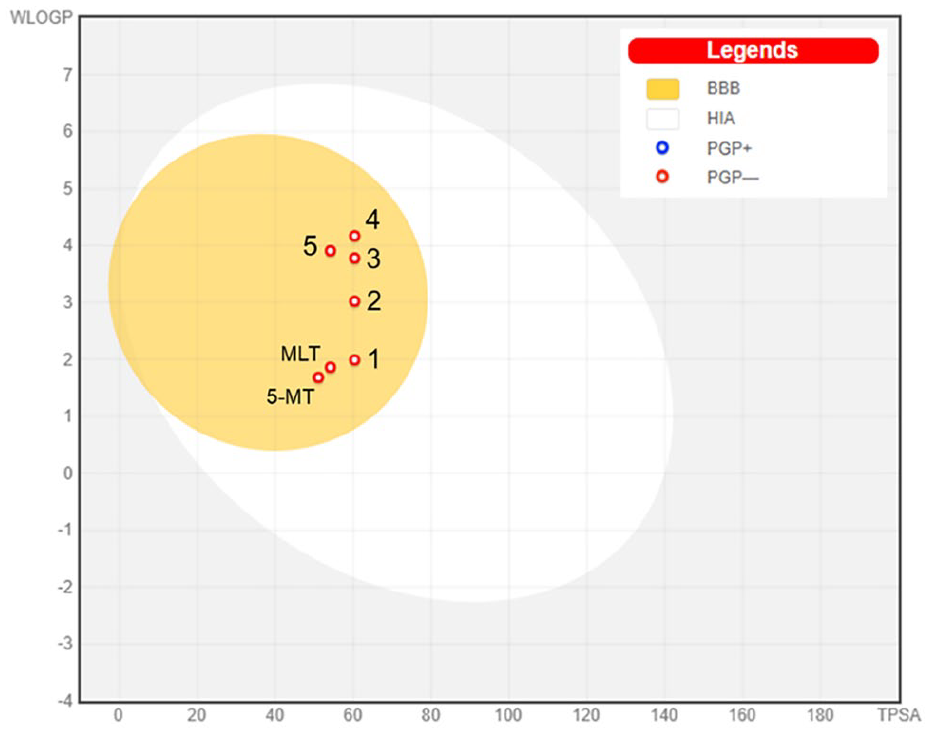
The boiled-egg plot of MLT, 5-MT and compounds
Molecular docking
It has been reported that CYP1A2 principally hydroxylates MLT at the 6-position of the indole ring in in vitro and in vivo studies and in clinical trials.7,25-27 Therefore, this hepatic enzyme was selected for metabolism prediction by computer modelling. Moreover, 5-MT which is a metabolite of tryptophan is also known as a substrate of this enzyme. 7 All derivatives were investigated for their coordination with the metabolising enzyme CYP1A2 to determine the effect of the lipophilic substituents on metabolism. The coordination protein-ligand binding energies and the distances between the SOM and the Fe(II) ion of heme are given in Table 2, while the representative binding modes of each coordinated complex are illustrated in Figure 3. The molecular docking results showed that MLT could undergo metabolism at the 6-position of the indole ring to form a 6-hydroxylated metabolite with binding energy of –9.2 kcal/mol. The NH of MLT formed hydrogen bonding interaction with a backbone oxygen of Asn312. The indole part made a face to face π-π interaction with Phe226 while acetamide moiety established hydrophobic interactions with sidechains of Phe125 and Ile117. Moreover, hydrophobic interactions between methoxyl group linking to indole ring and the non-polar residues Ile386 Leu497 were also observed. The coordinated distance between the SOM and the Fe(II) ion of heme was 8.60 Å. This accords with the study of Kesharwani et al. 19 which showed that the plot of the experimental distance against the frames from the molecular dynamic simulations showed that this distance could fluctuate between 6 and 8 Å. The orientation of MLT showed that 5-methoxyl group faced to the heme in the binding pocket and the indole ring interacted with Phe residues. In addition, the distance between the SOM and the Fe(II) ion of heme could be lower to 4.15 Å when different crystal structure (PDB code: 1FAG) was used.26,28
Binding energy and distance between SOM of MLT derivatives and Fe atom of heme of the representative pose.

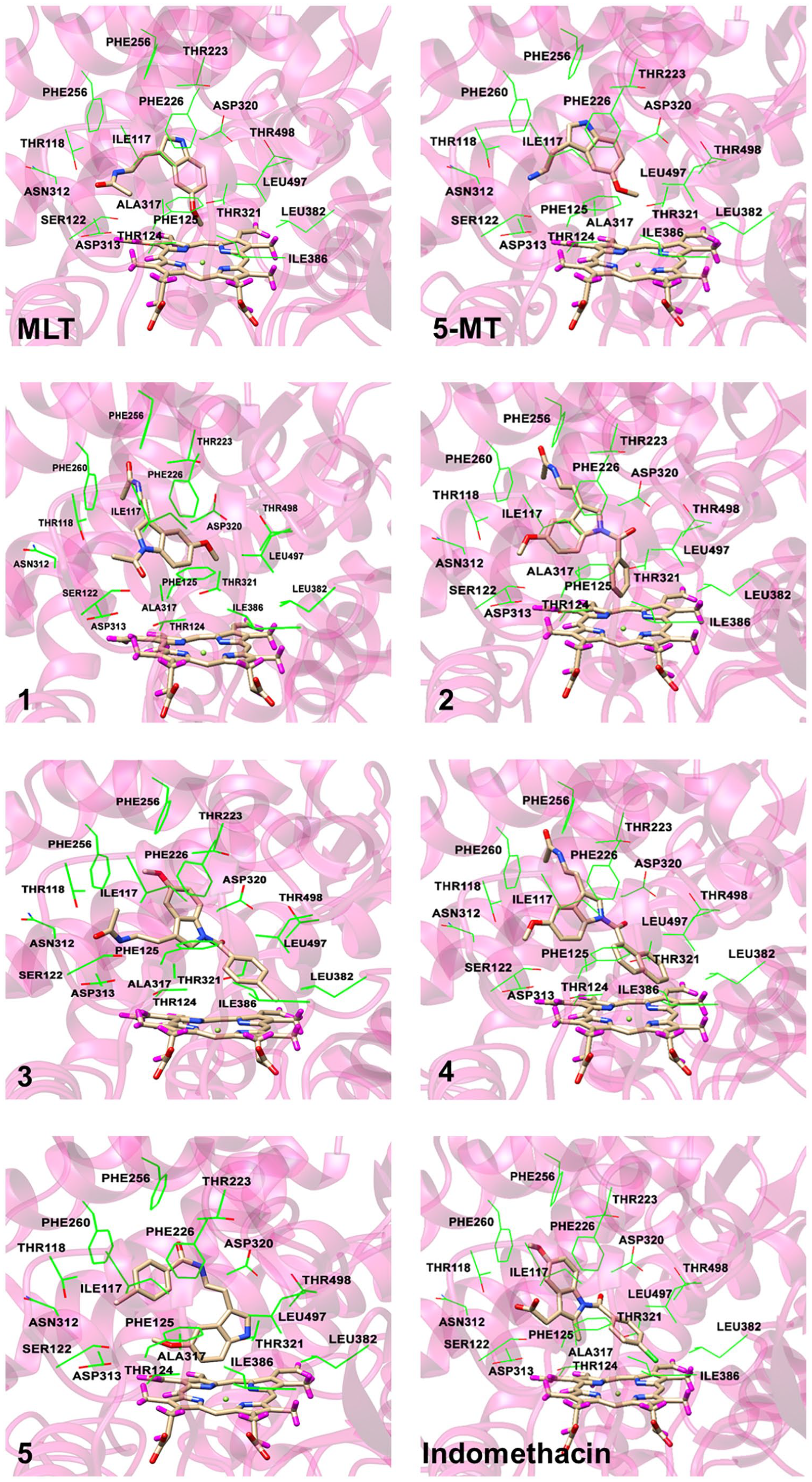
Selected pose to represent the CYP1A2-ligand binding. Some surrounding amino acids near to the ligand are shown in wireframe while the ligand and the heme are represented by stick model.
The modelling showed that the acetylated derivative (
In contrast, the modelling indicated that the lipophilic substituted derivatives (
Hydrolysis of MLT derivatives
All compounds were investigated for their ability to release MLT using 80% human plasma. Each compound was incubated with 80% human plasma in PBS (pH 7.4). An aliquot of 400 µL from each compound mixture was withdrawn after 0.5, 1, 2, 3 and 4 hours and the MLT releasing rate was determined by HPLC. Under the chromatographic conditions used, a melatonin peak was clearly detected at the retention time of 3.8 minutes. For the blank plasma, no endogenous compounds were seen at the MLT retention time. After 4 hours of incubation, the percentage of MLT released from the derivatives was less than 2.5% for all compounds (Figure 4). Compounds
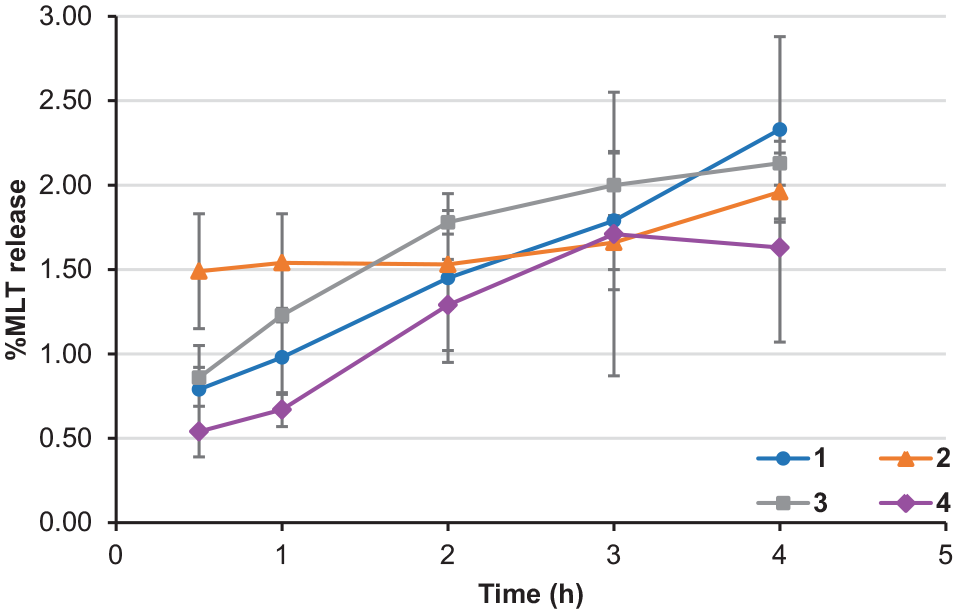
Hydrolysis profile of MLT derivatives after each interval of time.

Selected chromatograms of blank plasma, MLT and compound
Conclusion
Lipophilic substituted MLT derivatives were designed to overcome the pharmacokinetic limitations of MLT. The
Footnotes
Acknowledgements
The authors thank Dr. Glenn Borlace, Faculty of Pharmaceutical Sciences, Khon Kaen University for English language assistance.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Research and Academic Services from Khon Kaen University (RP63002), Graduate Research Fund Academic Year 2019 from National Research Council of Thailand (NRCT), The Office of the Higher Education Commission (Total Synthesis of Melatonin Project) and The Thailand Research Fund (DBG6080006), Thailand.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
PP (Ploenthip Puthongking) study conceptualization, methodology, formal analysis, investigation, resources, writing—original draft preparation, writing—review and editing, visualization, supervision, project administration and funding acquisition; PP (Panyada Panyatip) formal analysis, investigation, resources, writing—original draft preparation and writing—review and editing; NN methodology, formal analysis, investigation, resources, writing—original draft preparation and visualization.
