Abstract
Background:
Type 1 diabetes mellitus is associated with adverse maternal and neonatal outcomes. We aimed to evaluate the impact of CGM use on glycemic control and neonatal and maternal outcomes.
Methods:
This was a single-center study with prospective longitudinal data collection of pregnant women with T1DM allocated to 1 of 2 monitoring methods: Capillary blood monitoring and interstitial fluid glucose monitoring.
Results:
A total of 30 patients were enrolled. The average age was 31.26 ± 3.39 years, with an average gestational age of 9.4 ± 3.63 weeks at the first consultation. The average diabetes duration was 15.6 ± 7.36 years, with a mean preconception HbA1c of 8.67 ± 0.95%. The average BMI was 25 ± 2.88 kg/m2, and the average weight gain throughout pregnancy was 8.26 ± 5.84 kg. There was a substantial decrease in TBR compared to the control group. The control group had a slightly greater rate of pregnancy-induced hypertension, toxemia, eclampsia, and premature labor (33%, 13%, 7%, and 40%, respectively) than the CGM group (26%, 7%, 0%, and 26%). The differences were not statistically significant. Furthermore, the control group had a greater rate of preterm birth, neonatal hypoglycemia, NICU admission, and congenital abnormalities (27%, 40%, 46%, and 6.7%, respectively) than the CGM group (20%, 33%, 33%, and 0%, respectively), with no significant differences. The rates of macrosomia (20%), LGA (13%), neonatal respiratory distress (33%), and stillbirth (7%) were comparable between the groups. However, hydramnios occurred slightly more frequently in the CGM group (46% vs 40% in the control group).
Conclusion:
Early implementation and sustained use of CGM in pregnant women with T1DM may optimize glucose control and mitigate maternal-fetal risks.
Plain Language Summary
Pregnancy in women with type 1 diabetes can be challenging because high blood sugar levels increase the risk of health problems for both the mother and baby. A device called “continuous glucose monitor” (CGM) can track blood sugar levels throughout the day and night, which may help women keep their diabetes under better control during pregnancy. In this study, 30 pregnant women with type 1 diabetes were followed. Half of them used CGM, while the others used traditional methods to check their blood sugar. Women using CGM had better overall blood sugar control. However, when comparing pregnancy complications and newborn outcomes—such as high blood pressure in pregnancy, early birth, low blood sugar in babies, or admission to intensive care—there were no big differences between the two groups. This means that while CGM helps women manage their blood sugar more effectively, it did not lead to less complications in this small study. More research with larger groups of women is needed to see whether CGM can improve both blood sugar control and pregnancy outcomes.
Keywords
Introduction
Type 1 diabetes mellitus (T1DM) is a common endocrine disorder globally and poses a significant public health challenge. According to the International Diabetes Federation (IDF), approximately 537 million adults aged 20 to 79 had diabetes in 2021. Projections indicate that this number will rise to 643 million by 2030 and 783 million by 2045, which represents 12.2% of the population. 1 Among these cases, pregnancies in women with T1DM present a complex clinical scenario, with a high risk of serious complications for both mothers and their babies. 2 The IDF estimated that in 2021, 21.1 million live births (16.7%) resulted from pregnancies associated with diabetes, of which 10.6% were from women with pre-existing diabetes. 1
Pregnant women with T1DM face a higher risk of maternal complications, including gestational hypertension, preeclampsia, and the need for cesarean delivery. 3 For the fetus, the associated risks include macrosomia, neonatal hypoglycemia, prematurity, and congenital malformations.4,5 These complications are closely connected to glycemic control during pregnancy, underscoring the necessity for rigorous monitoring of blood sugar levels. 6 However, assessing glycemic control during pregnancy can be challenging due to the unreliability of HbA1c measurements and the lack of standardization in fructosamine testing. Moreover, research indicates that pregnancies complicated by T1DM demonstrate greater glycemic variability compared to those in women with T2DM, GDM, or in non-diabetic women.7,8 Standard self-monitoring of blood glucose (SMBG) through capillary blood testing does not effectively capture these glycemic fluctuations in real time. 9 Consequently, several professional organizations highlight the importance of continuous glucose monitoring (CGM) as a valuable alternative for managing pregnant women with diabetes.10-12
The emergence of CGM devices has transformed the landscape of diabetes management, enabling real-time and highly accurate tracking of interstitial blood glucose levels. 9 Landmark studies, including the CONCEPTT trial, compellingly demonstrate that the use of CGM during pregnancy not only enhances glycemic control but also significantly reduces the risk of serious neonatal complications, such as macrosomia and neonatal hypoglycemia. This technology represents a crucial advancement in ensuring healthier outcomes for both mothers and their infants. 13
Despite recent advances, the use of CGM in clinical practice remains limited, particularly in developing countries such as Tunisia. This study is the first in Tunisia aimed to address this gap and to evaluate the effects of glucose monitoring methods, specifically CGM versus capillary blood glucose testing on maternal and fetal outcomes in pregnant women with T1DM. The findings from this research could provide valuable evidence to inform clinical practices and enhance the management of pregnant women with T1D, particularly in resource-limited settings.
Methods
Study Type, Location, and Duration
This was a non-randomized controlled clinical trial involving pregnant women with T1DM. The study was conducted at the UR17SP02 research unit at Department C of the National Institute of Nutrition and Food Technology in Tunis between July 2021 and January 2023. This unit provide diabetes care with appointments scheduled every 1 to 2 weeks based on medical status. We collaborate with other specialties, mainly obstetric gynecology to oversee the monitoring of pregnant women with DM.
Population
For interstitial blood glucose measurement, only patients with a smartphone compatible with the continuous glucose monitoring (CGM) system application were included.
Patients’ Allocation
Patients were divided into 2 groups.
Collected Data
Clinical and Biological Assessment
We collected data, including age, education, family and personal medical history, surgical history, obstetric and gynecological history, and diabetes characteristics. We measured the baseline weight in kilograms (Kg) and height in centimeters (cm) and calculated the pre-gestational body mass index (BMI) using the formula: BMI = weight/height2 (expressed in kg/m2). Pre-gestational BMI was interpreted according to the World Health Organization (WHO) classification. 14 Weight gain was regularly monitored throughout the pregnancy. Excessive weight gain was defined as excessive if it exceeded the Institute of Medicine (IOM) recommendations. 15
Blood pressure (BP) was measured at each visit. The diagnosis of arterial hypertension was based on the definition outlined in the American Diabetes Association (ADA) on 2022. 16 Pregnancy-induced hypertension and toxemia were diagnosed based on the American College of Obstetrics and Gynecology classification. 17 A fundoscopic examination was conducted early in pregnancy, followed by examinations every 3 months if there was no diabetic retinopathy (DR), and monthly if DR was present, regardless of its stage. 18 HbA1c and FBG assays were performed quarterly at the same institution laboratory. Additionally, 24-hour microalbuminuria tests and urine cytobacteriological examinations (UCBE) were conducted monthly.
Glycemic Control Assessment
Blood glucose measurements were obtained using 2 distinct methodologies. Participants in the CGM group employed the Guardian Connect system in conjunction with a smartphone compatible with the Guardian-CGM application. Monitoring was performed at the onset of each trimester, with each monitoring period averaging 5 days in duration. Following this initial monitoring period, blood glucose levels were checked weekly.
The CGM System: The Guardian™ Sensor constitutes a critical component of the CGM system continuously converting small amounts of glucose from the interstitial fluid beneath the skin into electronic signals. These signals facilitate the provision of real-time glucose values. In the present study, the Medtronic Guardian™ Connect system was employed, with sensor insertion standardized through the administration by a single investigator. To optimize the CGM accuracy, calibration was performed approximately 2, 6, and 12 hours following transmitter activation, and subsequently upon receipt of a “Calibrate” alert. Notably, each sensor is single-use with a maximum operational duration of 14 days, whereas the Guardian Connect transmitter is capable of recording data for up to 7 days prior to automatic shutdown. The Guardian Connect application synchronizes recorded data with CareLink Personal, thereby enabling both patients and healthcare professionals to access the data via the CareLink Personal website. Upon sensor removal, users may retrieve comprehensive recordings from the website. Participants in this group received instruction on essential calibration procedures, including capillary blood glucose measurement and accurate data entry within the Guardian Connect application. Metrics assessed included time in range (TIR), time above range (TAR), and time below range (TBR), which were interpreted in accordance with the recommendations of the American Diabetes Association.13,16
The control group used a blood glucose meter, either ACCU-CHEK or Bionime. Participants were monitored with a weekly glycemic cycle (7 times per day, before and 2 hours after each meal and at bedtime). We assessed the point in range (PIR), point above range (PAR), and point below range (PBR), interpreting them according to the ADA recommendations. 16
All patients should document the composition and the timing of their meals, the insulin doses administered, and their capillary blood glucose levels. Training on the schedules for measuring capillary blood glucose throughout the glycemic cycle and on managing hypoglycemia was also provided. All patients were seen by the dietitian to provide an individualized dietary plan based on their pre- gestation weight.
Gynecological Assessment
Ultrasound monitoring was performed at specific intervals: first trimester (T1) between 12 and 14 weeks, second trimester (T2) between 22 and 24 weeks to detect fetal malformations, and third trimester (T3) between 32 and 34 weeks to assess fetal biometry, amniotic fluid volume, fetal presentation, and placental location. From 32 weeks onward, fetal vitality was monitored through regular recording of the fetal heart rate (FHR). Hydramnios is defined as an excessive volume of amniotic fluid, identified on ultrasound when the amniotic fluid cistern measures greater than 8 cm according to the Chaberlain method.
Patients were either visited in the maternity ward or contacted by telephone to assess the mode of delivery, due date, and to screen for potential neonatal complications. The following outcomes were assessed: Macrosomia (Defined as a birth weight greater than 4 kg), 19 Large for Gestational Age (LGA; Defined as a birth weight greater than the 90th percentile for gestational age and sex), 19 preterm birth (Defined as delivery before 37 weeks of gestation), Neonatal Respiratory Distress (NRD; Defined as the requirement for oxygen therapy to maintain saturation above 85% after the first 24 hours of life), Neonatal Hypoglycemia (Defined as a blood glucose level less than 0.4 g/l in term newborns 19 and less than 0.35 g/l in preterm newborns), 15 stillbirth (Defined as the death of a newborn before the 28th day of life).
Statistical Analysis
SPSS version 27 was used for statistical analysis. The Shapiro-Wilk test was used to analyze the normality of the data. Frequencies and percentages were calculated for qualitative variables to summarize data distribution. Means, standard deviations, and extreme values were determined for quantitative variables. The Student’s t-test for independent samples was applied to compare means between groups. When sample sizes were small, the nonparametric Mann-Whitney test was used. The chi-square test was used to analyze percentages across independent groups, and Fisher’s exact test was applied when chi-square test assumptions were not met. A significance level of .05 was set for all statistical tests.
Ethical Considerations
Approval from the National Institute of Nutrition of Tunis Ethics Committee was obtained for this study. The purpose of the research was explained to each patient, and written informed consent was obtained from all participants. Access to continuous glucose monitoring (CGM) data was restricted to the patient and the healthcare team. Authors declare not to have conflicts of interest.
Results
A total of 42 patients were initially enrolled for the study. After applying of the inclusion, non-inclusion and exclusion criteria, a total of 30 patients were selected for participation: 15 patients were allocated to the Continuous Glucose Monitoring (CGM) group, and 15 to the control group (Figure 1).

Flowchart illustrating the patients’ allocation process.
The sociodemographic and anamnestic characteristics of patients in both the CGM and control groups are presented in Table 1.
Sociodemographic and Anamnestic Characteristics of Patients in Continuous Glucose Monitoring (CGM) and the Control Group.

Characteristics and degenerative complications of T1DM are detailed in Table 2.
Characteristics and Degenerative Complications of T1DM.
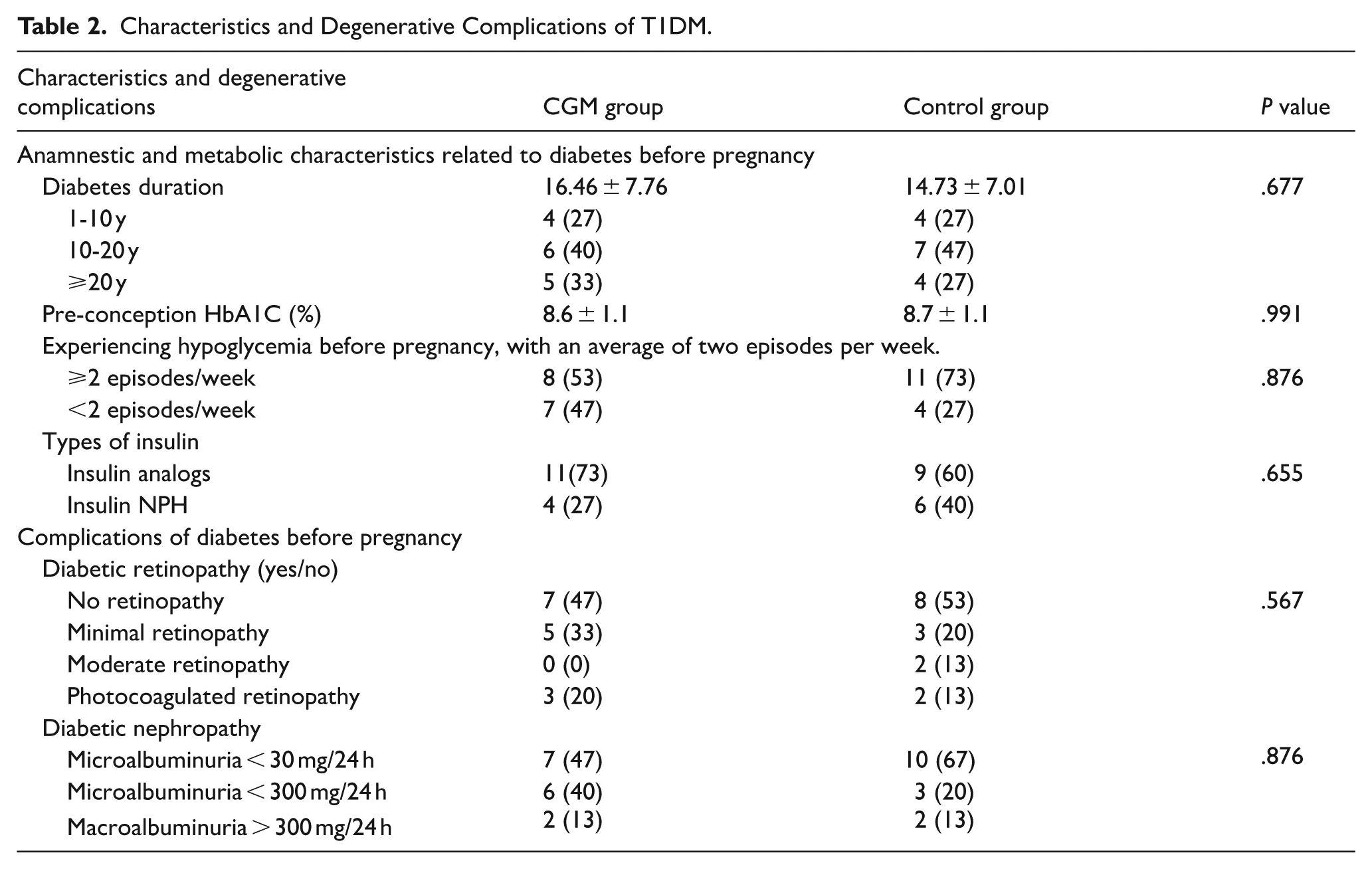
Table 3 outlines the characteristics associated with pregnancy and its progression.
Characteristics Related to the Course of Pregnancy and Its Progression.

Table 4 details the evolution of glycemic control during pregnancy in the CGM group.
Evolution of Glycemic Control During Pregnancy in the CGM Group.
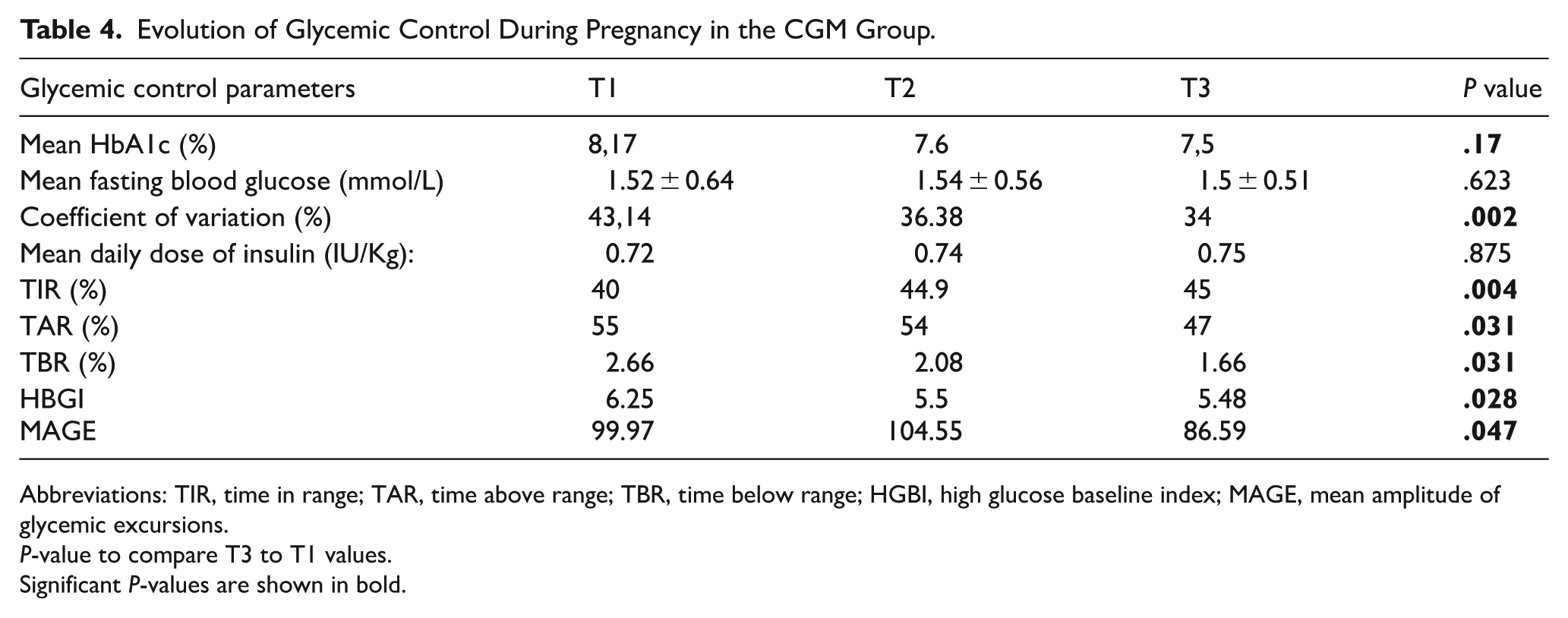
Abbreviations: TIR, time in range; TAR, time above range; TBR, time below range; HGBI, high glucose baseline index; MAGE, mean amplitude of glycemic excursions.
P-value to compare T3 to T1 values.
Significant P-values are shown in bold.
Table 5 details the evolution of glycemic control during pregnancy in the control group.
Evolution of Glycemic Control During Pregnancy in The Control Group.

P-value to compare third trimester to first trimester values.
Significant P-values are shown in bold.
Table 6 outlines a comparative analysis of glycemic parameters between the 2 groups throughout pregnancy.
A Comparative Analysis of Glycemic Parameters Between the 2 Groups Throughout Pregnancy.

Abbreviation: T1, first trimester; T2, second trimester; T3, third trimester, FBG, fasting blood glucose.
The CGM group had significantly lower TBR than the control group at T1 and T2 (P < .001; P = .003). At T3, the TBR of the CGM group was significantly lower than the PBR of the control group (P = .001).
The main indications for cesarean section in our population were acute fetal distress, macrosomia, and scarred uterus in respectively 33%, 26% and 16% of cases. Table 7 illustrates maternal and fetal adverse outcomes in the population.
Maternal and Fetal Adverse Outcomes in the Population.

Discussion
Assessment of Glycemic Control During Pregnancy
Research on pregnancies in patients with DM indicates that poor glycemic control before and during pregnancy significantly increases the risk of maternal and fetal complications. Additionally, pregnancies in women with T1DM are more likely to result in fetal health problems even when glycemic control is optimal and no other risk factors exist. This concerning trend largely stems from significant glycemic variability, which has been consistently documented in research. 20 Indeed, pregnancies in women with T1DM experience greater glycemic fluctuations than those with other type of DM, highlighting the need for close monitoring and proactive management to mitigate risks for mothers and their babies.7,8
Our study demonstrated a compelling, statistically significant improvement in mean HbA1c levels across each trimester of pregnancy. Our findings are consistent with an investigation involving 50 pregnant patients—20 of whom had type 1 diabetes—that utilized continuous glucose monitoring (CGM) for 48 hours at the onset of each trimester. This research revealed a significant improvement in HbA1c levels at T3 compared to T1. 8
In the control group, changes in HbA1c levels were inconsistent. The values remained nearly the same throughout the entire pregnancy. A comparison of HbA1c levels between the 2 groups revealed a non-significant decrease in HbA1c from T1 to T2 (P = .348) and from T1 to T3 (P = .378) in the continuous glucose monitoring (CGM) group compared to the control group. Additionally, we did not find a significant difference in mean HbA1c levels at T3 between the 2 groups. Similar findings have been reported in the literature, including a study conducted by Secher et al. 21 A study conducted by Voormolen et al. found no statistically significant difference in HbA1c levels at T3 between the CGM group and the control group. 19 However, the CONCEPTT strongly indicated a significant disparity in HbA1c levels between T1 and T3, with a P-value of .0372, highlighting the effectiveness of the intervention. 13 Furthermore, research conducted by Murphy et al. on 46 pregnant patients with T1DM provided additional compelling evidence, demonstrating a significant difference in HbA1c levels between T3 and T1, reflected in a P-value of .007. 10 These findings underline the critical impact of monitoring methods on diabetes management outcomes in pregnant patients.
The CGM group’s notable improvement in HbA1c levels can be attributed to the patient’s increased motivation and the strict therapeutic adjustments based on CGM results. Indeed, insignificant results can be attributed to the small sample size. The observed improvement in HbA1c levels should be interpreted with caution. The physiological changes during pregnancy, such as accelerated red blood cell turnover and decreased affinity for glucose, can impair the sensitivity of HbA1c as an indicator of glycemic control. 22 HbA1c levels typically fall in the first trimester, as red blood cell count rises. Conversely, during the second and third trimesters, HbA1c levels rise, which is related to increased iron deficiency and insulin resistance at that stage of pregnancy. 23 Furthermore, HbA1c does not accurately reflect fetal glycemic exposure since it does not assess postprandial glycemia nor account for the time spent beyond the target glycemic range. 24 As a result, the same HbA1c level can be associated with very different ambulatory glycemic profiles.25,26 In contrast, self-monitoring of blood glucose (SMBG) using a glucometer provides instantaneous spot glucose values, which are required for insulin dosage adjustment. This method is commonly used for diabetes self-management. 27 However, multi-daily SMBG remains challenging. 28 A national cross-sectional study conducted in France in 2014 found that only 29.4% of patients with T1DM or T2DM on basal-bolus insulin therapy did a sufficient number of SMBG tests as recommended by the High Authority for Health. 29 These limitations of HbA1c and SMBG have prompted the development of other technologies to enhance glycemic monitoring for diabetic patients, such as Continuous Glucose Monitoring (CGM). The effectiveness of CGM in T1DM has been demonstrated in various randomized controlled studies, showing improvements in both glycemic control and glycemic stability.30,31 This technique reliably reflects glycemic variability 32 and enhances the confidence of individuals with T1DM in managing hypoglycemia. 33 During pregnancy, CGM serves as an accurate tool for monitoring blood glucose levels in women with T1DM,34,35 and its use is supported by international recommendations.17,36,37 Despite the advantages of CGM for blood glucose monitoring, its adoption remains limited owing mostly to the technology’s high cost. 27 The reduced daily insulin doses in the CGM group compared to the control group are consistent with the better glycemic control in this group. This evidence underscores the effectiveness of continuous glucose monitoring in managing diabetes more efficiently in our population.
Mode of Delivery
In our study, cesarean deliveries occurred in 93.3% of cases, a rate significantly higher than those reported in other studies from Sweden (41% and 46%),38,39 and the CONCEPTT study (67%). 13 Our findings align closely with the research conducted by Guarnotta et al., which reported a cesarean rate of 93%. 40 In Tunisia, a study involving 200 diabetic patients reported a lower cesarean delivery rate of 50.2%. 41 The primary reasons for cesarean deliveries in this study were macrosomia and a history of a scarred uterus, which are consistent with our findings regarding the main indications for cesarean delivery. The high cesarean rate in our study may be attributed to its single-center design.
Additionally, we did not observe any significant differences in delivery term, neonatal weight, or cesarean section rates between the CGM group and the control group. These results align with those reported in the CONCEPTT study, 13 as well as findings from Secher et al. 42 and Murphy et al. 10 The small sample size may have contributed to the lack of significant results.
Maternal and Fetal Complications
The incidence of maternal-fetal complications varies across different studies. Diabetes is an established risk factor for preeclampsia, which is influenced by maternal endothelial dysfunction linked to diabetes and by abnormal placental development.43,44
The interest in CGM for assessing maternal-fetal complications varies across different studies. In our research, we observed that the rates of preeclampsia, preterm birth, and macrosomia in the CGM group were lower than in the control group, although these differences did not reach statistical significance. Our results are consistent with the results of the CONCEPPT study. 13
Our findings indicate that there was no significant difference in the rates of LGA and newborn hypoglycemia between the CGM group and the standard group. In contrast, the CONCEPTT trial reported a statistically significant difference in both the LGA and newborn hypoglycemia rates in the CGM group compared to the control group (P = .02; P = .025). This discrepancy could be attributable to improved glycemic control observed in the CONCEPTT study. Specifically, improvements in TIR and reductions in TAR during the second and third trimesters are associated with a lower risk of newborn hypoglycemia. 45 Therefore, our data support the implementation of CGM for monitoring pregnant women with T1DM. By leveraging CGM technology, we can significantly reduce the risk of maternal and fetal complications, ultimately enhancing outcomes for both mothers and their babies.
The current study has several strengths. Remarkably, it is the first study in Tunisia to assess the benefits of CGM for pregnant patients with T1DM, featuring comprehensive longitudinal follow-up up to delivery. We employed the Guardian Connect 3 system for glycemic monitoring, which boasts an impressive Mean Absolute Relative Difference (MARD) of just 9.1% at the CGM insertion site in our patients—an indication of its strong analytical performance given that a MARD under 10% is widely recognized as excellent.46-48 Furthermore, the Guardian Connect 3 system meets European standards for use during pregnancy, ensuring its reliability and safety. Through meticulous analysis of CGM glycemic traces and cycles, we developed an insulin therapy regimen specifically tailored to meet the unique needs of each patient, enhancing personalized care and management of their condition.
Limitations
Our study presents several limitations that warrant attention. To begin, the single-center design raises the possibility of selection bias, as all patients were recruited from the same department. Consequently, the findings may not accurately reflect the population of pregnant women with T1DM. Additionally, the small sample size—restricted to just 30 patients—exacerbates this issue. This limitation partly arises from the high cost of blood glucose sensors and the stringent inclusion criteria we adhered to. Furthermore, the absence of randomization in patient recruitment could have also influenced the selection bias. However, it is crucial to emphasize that although participants used different blood glucometers, each woman consistently used the same glucometer throughout her pregnancy, thereby lowering the likelihood of measurement bias related to device variability. These considerations taken together highlight the need for cautious interpretation of our findings and underscore the need of future studies with more extensive and diverse samples. It is crucial that upcoming research is designed with sufficient statistical power to thoroughly investigate the benefits of CGM in this specific population.
Conclusion
Our data support the use of CGM to monitor pregnant women with T1DM. While the improvements in maternal-fetal complications were not statistically significant, there was a substantial decrease in time below range throughout the second and third trimesters compared to the control group. However, larger trials are needed to evaluate whether the early use of real-time CGM prior to pregnancy can reduce the risk of severe hypoglycemia and improve pregnancy outcomes. Additionally, research should explore whether continuous CGM use throughout pregnancy can enhance glycemic management and significantly reduce maternal-fetal complications.
Footnotes
Author Note
This study has not been previously presented.
Ethical Considerations
This study has been approved by the ethics committee of the national institute of nutrition of Tunis.
Consent to Participate
At time of inclusion participants signed a written consent to participate in this study.
Consent for Publication
At time of inclusion participants signed a written consent including consent to publish the collected data in peer reviewed manuscript.
Author Contributions
Chaima Jemai: Conceptualization, Data curation, Validation, Writing - original draft Writing - review & editing; Yosra Htira: Formal analysis, Validation, Writing - review & editing; Amal Salem: Data curation, original draft Writing; Zohra Hadj Ali , Olfa Lajili, and Imen Hedfi: Software, Writing - original draft writing; Y; Sarra Ben Amara: Data curation; Faika Ben Mami: Conceptualization, Methodology, Project administration, Supervision, Validation, Writing - review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The collected data are available upon request from the corresponding author of the study (
