Abstract
Objectives:
This study examines the hormonal mediators of the effect of iodized salt in pregnancy on child cognition.
Methods:
Sixty districts across 6 zones in the Amhara region of Ethiopia were randomly allocated to a control or intervention arm of early market access to iodized salt. Twenty-two villages per arm were randomly selected for this sub-study. A total of 1220 pregnant women who conceived after the intervention began were enrolled and assessed for their iodine and iron status. Data were collected once on the household socio-demographic status and iodized salt use, and maternal urinary iodine during pregnancy. Then, infants’ diet, urinary iodine level, cognitive development (Bayley III), serum hormonal levels, iron status, and inflammation markers were measured between 2 and 13 months of age.
Results:
The median maternal urinary iodine concentration was adequate and significantly higher in the intervention mothers than that of the controls (163 vs 121 µg/L, P < .0001). Intervention children compared to the control children had lower thyroid-stimulating hormone (TSH) (mean: 2.4 ± 1.0 µIU/mL vs 2.7 ± 1.0 µIU/mL, effect size = 0.18, P < .01) and thyroglobulin (Tg) (41.6 ± 1.0 ng/mL vs 45.1 ± 1.0 ng/mL, effect size = 0.14, P < .05). There was an interaction between the intervention and iron stores such that cognition was higher with iron (effect size = 0.28, 100 vs 94 IQ points). TSH was a partial mediator (12%) of the effect of the intervention on child cognition (Sobel z-score = 2.1 ± 0.06, P < .05).
Conclusion:
TSH partially mediated the effect of the iodized salt intervention on child cognition.
Introduction
Iodine is an essential part of the thyroid hormones produced by the thyroid gland. Iodine requirement increases during pregnancy due to a 50% increase in maternal thyroxine (T4) production needed for the mother and the fetus. Due to elevated estrogen, there is an increase in iodine transfer to the fetus from the second trimester, an increased renal iodine clearance, and iodine binding to thyroid binding globulin. 1 After an initial reduction in serum thyroid-stimulating hormone (TSH) due to increased levels of human chorionic gonadotropin in the first trimester, serum TSH levels increase progressively in more than 80% of pregnant women. This change is accompanied by an increase in serum thyroglobulin (Tg), resulting in a situation of chronic thyroid hyperstimulation and an increase in thyroid volume by 20% to 30% during gestation. 2 Even mild-to-moderate iodine deficiency during pregnancy results in maternal subclinical hypothyroidism (an increased concentration of TSH in the second trimester) and hypothyroxinaemia (a free T4 concentration less than the tenth percentile at 12-weeks gestation) that may impair fetal growth and brain development.
Studies have shown that children’s mental development scores are lower when iodine is deficient in pregnancy. A review of studies that examined the effects of iodine supplementation during pregnancy on infant and young child’s mental development in iodine-deficient areas, found overall similar effect sizes regardless of design (d = 0.49). 3 However, only 2 of the studies were randomized controlled trials (RCT). The review of RCTs showed that a lack of quality evidence regarding the effects of pregnancy supplementation and none on preconception iodine supplementation and child development.
A meta-analysis of iodine supplementation or fortification RCTs found 8 studies that supplemented either the pregnant mother or the infant. 4 Only a few of the studies assessed the intervention effect on T4, triiodothyronine (T3), TSH, and Tg; none found an effect on T4 and T3. If iodine deficiency occurs with iron deficiency, there is a reduction of heme-dependent thyroperoxidase activity in the thyroid, which might blunt the function of iodine in the body. 5 Consequently, where findings are mixed concerning the biomarkers TSH and Tg after iodine supplementation in pregnant mothers and infants, iron deficiency may be a factor to consider. Iodized salt has not been used in most iodine RCTs. 6 The only randomized trial in pregnancy that used iodized salt and reported thyroid levels did not find any difference in TSH, free T4, and free T3 between iodized salt and potassium iodate intake groups, and there was no significant difference in children’s cognitive development. 7 However, in that study there was no control group and both the intervention and potassium iodate groups were iodine sufficient.
Women should ideally be provided with ample iodine intake (at least 250 µg/d) for a long period before conception to ensure adequate stores to support the increased needs in pregnancy. 8 This study examined the effect of an intervention to provide early access to iodized salt, before conception, on young children’s biomarkers of iodine and how they mediated mental development. The research was part of a broader study evaluating the effect of iodized salt on child development in the Amhara Region of Ethiopia.
Methodology
Research design and participants
The broader study was a randomized cluster trial, with districts defining clusters and units of randomization. 9 Sixty districts within 6 zones in the Amhara Region of Ethiopia were selected by a random selection process. Thirty of the districts were randomly assigned to the intervention arm and 30 to the control arm, and 1 village was randomly selected from each district to be part of the evaluation. For this specific data collection, 22 of the 30 districts per study arm were randomly chosen. The initial report of this paper has been published. 10
Participants were recruited by trained students and Health Extension Workers (HEW) within each selected village where they conducted a census of all pregnant women. Eligible women were those who: (i) became pregnant at least 1 month after the salt iodization intervention had started, (ii) had not received iodine capsules in the past year, (iii) spoke Amharic, (iv) were 18 years or older, (v) had lived in the selected village for at least a year, and (vi) planned to continue living in the selected village for at least another year to allow follow-up. Eligible mothers were enrolled after signing the informed consent form.
Sample size
This paper is part of a larger study evaluating the effect of iodized salt on children’s cognitive development. Two previous intervention trials with pregnant women in moderate iodine-deficient areas gave an effect size of 0.02 and 1.38 (95% CI: 0.45, 2.32) for mental development in children.11,12 This study’s group sample size was calculated with a small effect size (d) of 0.2 and power of 0.8 using the formula: n = [2 (Zα/2 + Zβ) 2]/d2; where n = number per group, α = 0.05, β = 0.2, Zα/2 = 1.96 and Zβ = 0.84. 13 The sample size was 393 per group. The intra-cluster correlation (ICC) was assumed to be 0.04 and the variance inflation factor (VIF) = 1 + (cluster size−1) × ICC. 14 From a preliminary study, the average number of pregnant women per village was 30. A conservative cluster size of 18 was used, then accounting for the cluster: VIF = 1 + ((18–1) × 0.04) = 1.68. Hence the adjusted sample size per group was 393 × 1.68 = 660. An adjusted cluster size of 30 would give a minimum of 22 (660/30) clusters per group. A total of 660 pregnant women per intervention arm were required. Subsequently, only 1220 pregnant women were available and recruited for data collection from which 1035 of their children were involved in the final data collection.
Intervention
The intervention involved the facilitation of iodized salt supply to the intervention villages (see previous paper for more details10,15). As salt was gradually being iodized in the country, the main distributors were contacted and assisted by the project to supply iodized salt first to the intervention markets. Staff at the salt monitoring offices within each district were trained and provided with rapid test kits to monitor and stop the channel of non-iodized salt into intervention markets and villages. A salt monitoring officer was hired and trained by the project to monitor samples of salt present in the market areas of the intervention communities. He also monitored the quality of samples of the salt supplied by salt distributors, those sold within the market, and those used in the village. Salt in control markets and villages was left to market forces but periodically sampled for information purposes. Iodized salt reached intervention communities 4 to 6 months before control communities. 15 The research assistants involved in data collection were blinded to which of the communities were intervention or control.
Outcome measures and data collection
Data was collected at recruitment (during pregnancy), a field visit post-recruitment was done to collect blood from mothers who were still pregnant, and the final data collection was done 2 to 13 months after the mothers delivered their babies. Refer to the consort flow diagram in our previous publication. 10
Measures during the pregnancy
A household salt was sampled in salt bags and urine specimen of the pregnant women were collected using urine cups and transferred into urine tubes to assess iodine status. 16 The urine samples in tubes were preserved with 29.2 g/L boric acid 17 and were subsequently sent to the project office and frozen at −20°C, and then transferred to the Ethiopian Public Health Institute (EPHI) for urinary iodine analysis using the Sandell-Kolthoff reaction. 16 The hemoglobin (Hb) of a sample of women who were available and still pregnant during a post-recruitment field visit was measured using the HemoCueTM 201+ portable photometer. Those below the cutoff of 110 mg/dL were considered anemic. 18
Post-partum measures
Final data collection was done 2 to 13 months after women gave birth. Home visits were made by research assistants to collect data on the socio-demographic background of households, hygiene practices, and water and sanitation practices. Household salt samples were taken and checked for iodine using the rapid test kits (MBI Kits International, Chennai, Tamil Nadu, India). Randomly selected 20% of salt samples that tested positive for iodine were taken to EPHI to analyze iodine concentrations by titration method using the modified Sandell-Kolthoff reaction. 16
Children’s mental development was assessed using the Bayley Scales of Infant and Toddler Development (version III) by trained psychology graduate students, overseen by clinical psychologists. 19 Four subscales of the Bayley tests were used, namely cognitive, expressive language, receptive language, and fine motor skills. A drop of capillary blood was used to test the Hb of the children by the azide methemoglobin method with a HemoCueTM 201+ portable photometer. 20 Venous blood was also taken to evaluated thyroid biomarkers. Children’s urine samples were collected with urine bags and stored in urine tubes at −20°C. All urine samples were analyzed in duplicate. Levels less than 100 μg/L for children and 150 μg/L for pregnant women were considered inadequate. The laboratory participated in the Centers for Disease Control and Prevention’s Ensuring the Quality of Urinary Iodine Procedures (EQUIP) standardization program for quality assurance related to iodine testing. 21
The procedure used to collect data on children’s thyroid biomarkers was as follows. Venous blood was taken in vacutainer tubes and then centrifuged. The serum was then transferred into mineral-free cryogenic vials, labeled, and transported back to the project office in ice packs to keep them frozen. The vials were stored at −20°C before being transported to EPHI and stored at −80°C before serum analysis. The serum was analyzed for markers of iron status including serum ferritin and soluble transferrin (sTfR), thyroid hormones including T3, T4, TSH, and Tg, and inflammation markers, namely serum C-reactive protein (CRP) and alpha 1-acid glycoprotein (AGP). The thyroid hormones, ferritin, and CRP were analyzed using the electrochemiluminescence immunoassay (ECLIA); sTfR and AGP were analyzed using the immunoturbidimetric method. Quality control was ensured by strictly following the analytic procedures, and running the controls, and duplicates, with every run to control procedural and instrumental performance. The details of the procedures and quality control used in analyzing the serum samples were published elsewhere. 22
Data analysis
A total of 1220 respondents were enrolled (651 intervention and 569 control) and 1035 (84.8%) were included in the final analysis. The consort flow diagram was published in an earlier publication. 10 Main outcomes and related variables (hemoglobin, urinary iodine levels, mental development scales) were first checked for normality and skewed variables were transformed (squared to transform negatively skewed variables and log to the base 10 to transform positively skewed variables). The results of the analysis using transformed and original variables did not vary. World Health Organization cut-off points for deficiency were used to classify the serum indicators for Hb (110 mg/dL 17 ) and ferritin (12 ng/mL 23 ). The cutoff for CRP (5 mg/L) and sTfR (8.3 mg/L) were obtained from other published works.24,25 A cutoff of below 2.5th percentile score was used for low levels of T3 and T4, and above 97.5th percentile score was used for high TSH and AGP. The cutoff for Tg (18 ng/mL) was obtained from the manufacturer and the calculated TfR-F index (sTfR ÷ log Ferritin, the lower the better) used a cutoff of 7.7 (using sTfR of >8.3 mg/L and ferritin of <12 ng/mL) to define the deficiency of iron stores. 25
Group differences of the socio-demographic variables were analyzed with cluster-adjusted PROC MIXED analyses from SAS 9.4 to identify covariates of the main outcomes (mental development and serum hormonal variables). Cluster-adjusted logistic regression analyses were used to analyze dichotomous serum variables. Cluster-adjusted PROC MIXED was used to analyze the treatment effect, adjusting for identified covariates and conventional covariates from the literature (household assets, water and sanitation, maternal education, child age, and hygiene for mental development indicators; household assets, child sex, age, hygiene, CRP, and AGP for serum outcome indicators). Mixed models were used to analyze the interaction between serum indicators and intervention on Bayley scores. Modifiers tested include Hb (low: <110 mg/dL), serum T3 (low: <1.55 ng/ml), T4 (low: <7.57 µg/dL), TSH (high: >6.68 µIU/mL), TG (low: <18 ng/mL), ferritin (low: <12 ng/mL), sTfR (high: >8.30 mg/L), TfR-F index (high: >7.70), AGP (high: >1.20 g/L), and CRP (high: >5.00 mg/L). Only significant interactions are presented in results tables before running stratified analyses to obtain separate effect sizes.
Mediation analyses were conducted to examine serum hormonal levels as potential mediators of the effect of the intervention on mental development; that is, to be an intermediate in the relationship between the 2 variables. First, conditions to support mediation were examined to establish that there was an association between the intervention and mental development and between hormones and mental development. Secondly, the Sobel test was used to check the significance of the mediation effect (Figure 1). 26 Bootstrapping mediation analysis in R was then used to calculate the percentage mediated. Finally, means were reported with standard deviations, and mixed models were reported with least-squares means and standard error.

TSH mediation of the intervention effect on cognition.
Results
The participants were mostly subsistence farmers from low socioeconomic households; however, the intervention communities had fewer household assets than those from the control communities (2.3 vs 2.5 out of 10 items, respectively; P = .04). Although water and sanitation practices were better in the intervention communities (P = .02), good hygiene practices did not differ in the 2 communities. The educational level of the mothers was very low (77% with no schooling) and this did not vary between the 2 study arms.
At enrollment, there were slightly fewer intervention households using iodized salt than the control households, though both were above 90% (Table 1). The proportion of households using adequately iodized salt (iodine concentration >15 ppm) was similar in the 2 groups. Maternal urinary iodine (UI), however, was higher in the intervention communities than the controls, and the median UI was adequate. Additionally, there were fewer pregnant women with severe iodine deficiency (UIE <50 µg/L) in the intervention communities (11%) compared to those in the control communities (15%, P < .05).
Characteristics of participants in intervention and control communities in the Amhara region of Ethiopia.
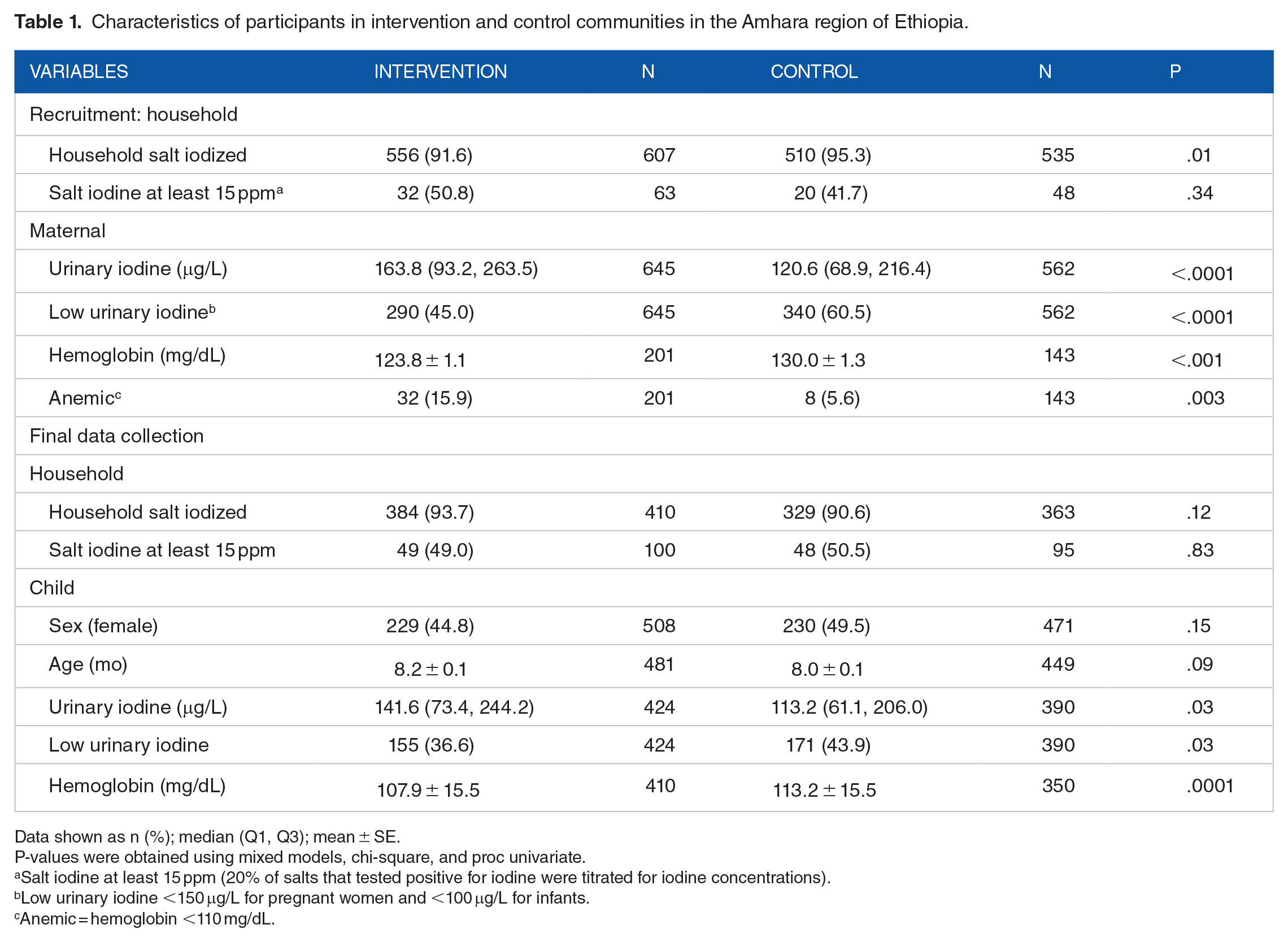
Data shown as n (%); median (Q1, Q3); mean ± SE.
P-values were obtained using mixed models, chi-square, and proc univariate.
Salt iodine at least 15 ppm (20% of salts that tested positive for iodine were titrated for iodine concentrations).
Low urinary iodine <150 μg/L for pregnant women and <100 μg/L for infants.
Anemic = hemoglobin <110 mg/dL.
Some of the children were lost to follow-up and the final serum samples of others (30%) were not obtained because either their caregivers refused blood draw or our attempt to draw venous blood was unsuccessful. The latter was observed more frequently in younger infants and led to a higher age of those with serum samples used in these analyses compared to those without serum samples taken (P < .001). Children with serum analysis data had significantly higher length-for-age z-scores (P < .01) and lower wasting odds ratio (OR (95%CI): 0.38 (0.21, 0.69)) compared to those without data used for this serum analysis.
At the final postpartum data collection, the number of households using iodized salt and having adequately iodized salt was not significantly different between the intervention and control sites (Table 1). Also, there were no group differences in the sex nor age of the children. However, the urinary iodine concentrations and adequacy of iodine status (>100 μg/L) were better in the intervention children compared to the controls (P < .05). More intervention children were anemic as defined by their low Hb levels (53% vs 39%, OR (95% CI): 1.9 (1.4, 2.5)) and low serum ferritin levels (55% vs 44%, OR (95% CI): 1.6 (1.2, 2.2)) than the controls, with the intervention children almost twice as likely to be anemic. In addition, more intervention children recorded high TfR-F index values (an indicator for depleted iron stores) than the controls (36% vs 29%, OR (95% CI): 1.5 (1.1, 2.1)). The serum level of T3 correlated positively with infants’ fine motor skills and their combined Bayley score, while TSH negatively correlated with cognitive development. Serum soluble transferrin negatively correlated with fine motor and combined Bayley scores, and depleted iron stores also negatively correlated with fine motor scores. Acute inflammation marker c-reactive protein levels also correlated negatively with receptive language (Table 2).
Correlation between serum indicators and mental development scores.
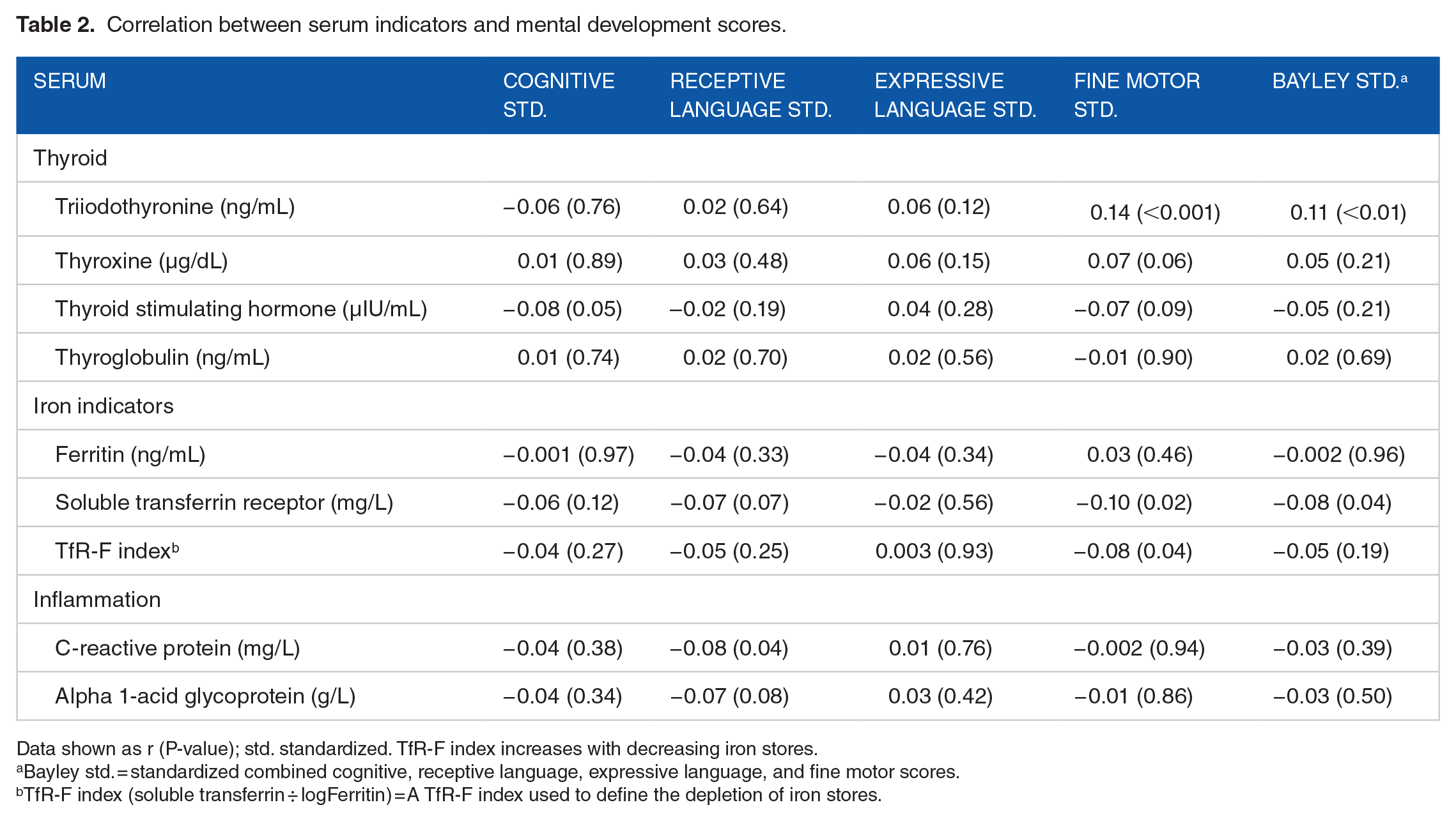
Data shown as r (P-value); std. standardized. TfR-F index increases with decreasing iron stores.
Bayley std. = standardized combined cognitive, receptive language, expressive language, and fine motor scores.
TfR-F index (soluble transferrin ÷ logFerritin) = A TfR-F index used to define the depletion of iron stores.
A mixed model comparing the continuous variables in the 2 groups revealed that serum concentrations of T3 and T4 were not significantly different between the 2 groups, but serum TSH and Tg were lower in the intervention compared to the control arm after adjusting for covariates and clusters (P < .05) (Table 3). The serum ferritin concentration and TfR-F index of the intervention children were also lower than those in the control communities, but the difference lost significance when adjusted for covariates and clusters. There were no significant differences in the concentrations of sTfR, TfR-F index, or the serum inflammation markers (AGP and CRP) of the children in the 2 intervention arms.
Results of the PROC MIXED model evaluating the effect of an iodized salt intervention on serum indicators in 2 to 13-mo-old children in the Amhara region of Ethiopia. a

Abbreviations: ICC: intraclass correlation coefficient; LSM: least-square mean; SE: standard error.
Covariates for serum indicators were child sex, age, hygiene, C-reactive protein, and Alpha 1-acid glycoprotein as well as household assets and water and sanitation); covariates for Bayley scales were child age and hygiene, maternal education, and household assets and water and sanitation.
TfR-F index (soluble transferrin ÷ logFerritin) = A TfR-F index used to define the depletion of iron stores.
Bayley scores = Bayley scales of infant and toddler development III (2006) reflect the competencies expected of infants and young children in the first 3.5 years: 4 scales were used: cognition, receptive language (ability to understand language), expressive language (ability to express themselves), and fine motor (hand-eye coordination). The results have been earlier published. 10
The mean cognitive score of intervention children was significantly higher than that of control children after controlling for covariates and clusters (effect size = 0.17). Other subscale scores from the Bayley were not significantly different between the groups. Modifier effects were examined using each serum indicator interacting with the intervention. There was only a significant modifier effect of iron stores (TfR-F) on the intervention effect on cognition, such that the intervention effect was higher (effect size = 0.28) among children with lower TfR-F values, meaning higher iron stores (Table 4).
ANCOVA results evaluating the interaction between the effect of the intervention on child cognitive development and an indicator of iron stores (TfR-F index).

Abbreviations: ANCOVA: analysis of covariance; LSM: least square mean; SE: standard error; TfR-F: sTfR ÷ logFerritin.
Low TfR-F index = high iron stores. High TfR-F index = low iron stores.
Bayley scores = Bayley scales of infant and toddler development III (2006), reflects the competencies expected of infants and young children in the first 3.5 years: 4 scales used; cognition (reasoning ability), receptive language (ability to understand language), expressive language (ability to express themselves) and fine motor (hand-eye coordination). Sum of the standardized scores gives the Bayley standardized score which is out of 80.
TfR-F index (soluble transferrin ÷ logFerritin) = A TfR-F index value of >7.7 (using sTfR of >8.3 mg/L and ferritin of <12 ng/mL) was used to define the depletion of iron stores.
In the mediation analysis, the intervention had a significant effect on maternal and child UI, TSH, and Tg as well as on cognition (P < .05). TSH was the only indicator that showed the potential of being a mediator of the effect of the intervention on child cognition. A Sobel test confirmed TSH was a partial mediator (Figure 1). TSH mediated approximately 12% of the effect of the intervention on cognitive; that is, the mediation coefficient effect was 12.4% of the total effect (both direct and indirect).
Discussion
This study demonstrated that increased access to iodized salt led to better iodine status of both pregnant women and their subsequent children as well as improved child cognition. The serum concentrations of TSH and Tg were significantly lower in the intervention children compared to their controls; there was no difference in the other thyroid hormones or serum iron and inflammation markers. The cognitive difference between groups was partially mediated by their serum concentration of TSH but not Tg. Anemia and Hb values were not associated with Bayley scores. However, the intervention interacted with child iron stores such that cognition was higher in intervention children with higher iron stores compared to their controls but not in those with low iron stores. Low iron stores in particular, rather than anemia, are seen as an important measure that could mask the effect of universal salt iodization on child cognition. 5
The adequate median UI concentrations of women and children in the intervention arm showed that the intervention was successful in meeting the iodine requirement of pregnant women. The control communities recorded inadequate median maternal and child UI concentrations and more of the mothers and their children from the control communities had low UI excretion compared to those in the intervention arm. Considering there was only a 4 to 6 months difference in the iodized salt exposure between the intervention and control pregnant women, the difference in the iodine status of the women reflects on the importance of getting iodine to women substantially before they conceive.
Serum TSH and Tg are better surveillance indicators of an iodine intervention than T4 and T3.27,28 The serum TSH is mainly used as a marker for neonates and pregnant women. The concentration of TSH is higher in iodine deficiency due to the reduction of serum T4 concentration. Serum Tg concentration reflects iodine nutrition over months or years. 27 Both TSH and Tg concentrations were lower in our intervention children indicating their better current and prenatal iodine nutritional status compared to the control children. The overlap of concentrations of T4 and T3 between iodine deficient and sufficient populations is large enough to make them insensitive indicators of iodine deficiency27,29 Also, there is a circadian rhythm of T3 and T4 hormones, with their concentration varying between 9% and 40% in the course of a day. 30 Hence, depending on the time the serum samples were taken there can be large enough variation in their levels to make them insensitive to our intervention.
Anemia can affect iodine metabolism and hence affect response to an iodine intervention. 5 We have seen earlier on that iron stores in the body affect the efficacy of iodine intervention. In this study, we observed iron indicators were negatively associated with some of the mental development indicators. Even though the levels of serum indicators of iron status (ferritin, sTfR, and TfR-F index) did not vary between treatment groups, the proportion of those at the lower levels did vary. The intervention children recorded the highest prevalence of anemia and low iron stores. This indicates that the effect of the intervention observed could have been reduced by the children’s anemic state and low iron stores. The interaction between the intervention and iron stores observed in this study shows that the true effect of the exposure to iodized salt on child development may be much higher than reported in this study.
Our RCT yielded a moderate cognitive benefit with an intervention effect size of 0.17 (103 vs 99 IQ points in the intervention vs control children, respectively). The cognitive advantage of the intervention children was only seen among children with higher body iron stores (low TfR-F index) with an effect size of 0.28 (or 6 IQ points) 25 . We went further to see what the mediators to this cognitive benefit of iodized salt were and found that TSH, rather than Tg, partially mediated the benefit of iodized salt on cognition. This implies that iodized salt reduced the concentrations of TSH, which partly accounted for the higher cognitive score of children in the intervention communities compared to the control communities. At this point, we can only speculate as to why TSH rather than Tg emerged as the better mediator. T4 is very important in the metabolism of iodine and the cognitive-developmental process. Unlike serum Tg, T4 is largely present within its main target cells and in the thyroid gland rather than in the serum. Unlike serum levels of these hormones, the serum TSH level is a good indicator of T4 adequacy. The negative correlation between TSH and cognitive observation could be because lower levels of TSH imply adequate levels of T4 and hence it’s the effect on cognition. It may also be possible that there are other mechanisms involved that are unknown at this stage. This is because there are several cases where TSH concentrations may be normal in hypothyroidism (very low T4 concentrations) or hyperthyroidism (very high T4 concentrations). There is a need for further investigation into the exact mechanisms involved in the TSH partial mediation of cognition in children after exposure to iodized salt.
The use of iodized salt in pregnant women is highly recommended, to protect the development of the next generation. We also recommend researchers investigating iron nutrition and cognition in children should concentrate on measuring iron stores and not only hemoglobin levels. The study however is not without limitations. The primary limitation was the contamination of the control villages by the suddenly active national program that rapidly pushed iodized salt into all markets, preventing us from having a true control with no iodized salt during preconception. Also, mothers were not recruited and measured at the same stage of pregnancy and children at the same age; assessment at the same stage/age would have reduced the effect of physiological stage and age variation among the participants. Blood was collected from only a fraction of the pregnant women and the study did not obtain serum samples for all children. Considering the fact that children whose serum samples were collected were older and better nourished (>length-for-age z-score), there could be a difference between the samples obtained compared to that of the overall study participants leading to an under-or over-estimation of the intervention effect on the serum indicators.
In conclusion, the intervention that facilitated earlier market access to iodized salt during the preconception period improved the iodine status of pregnant women and their children and children’s serum concentration of TSH and Tg. The intervention effect on children’s cognitive development was modified by iron stores and improved partially through the reduction of their serum TSH concentrations. It is recommended that iron stores in children should be prioritized in rolling out universal salt iodization programs to ensure improvement in child cognition.
Footnotes
Acknowledgements
The staff of the nutrition unit of the Ethiopian Public Health Institute are duly acknowledged for their role in the urine and salt analyses. To Kimberly B Harding and Micronutrient Initiative Ethiopia staff, we say thank you for all the support and guidance throughout the project. We would also like to acknowledge the field supervisor Biruk Alemu, and the data collection team for their tireless effort to see the successful completion of the project. Finally, we thank the staff of the national, regional, district, and village offices of the Amhara Region of Ethiopia and the participants of this project.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was funded by the Government of Canada through Global Affairs Canada, with funds delivered through the Nutrition International (previously, Micronutrient Initiative), Canada.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authorship
HM was responsible for the design of the study, field data collection, data analysis and interpretation, and manuscript preparation. GM assisted with the conception and design of the study as well as the data analysis and interpretation. FA assisted with the conception, design, and implementation of the study, the data analysis and interpretation of the results, and the preparation of the final manuscript. KB was responsible for the management of the field data collection, data entry, and analysis. AS was responsible for the laboratory analysis at EPHI and local coordination of project activities with Ethiopian institutions. All authors reviewed and approved the final manuscript.
Ethical Approval
Ethical approval was sorted from the McGill University (IRB Study Number A01-M09-11B), Ethiopian Public Health Institute (EPHI), and the Ethiopian National Research Ethics Review Committee (NRERC) (C.H.N.R.16.13/938) review boards. All procedures performed were done according to the ethical standards of the above review boards and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was registered at ClinicalTrials.gov, with the registration No: NCT01349634. Details of the ethical concerns were addressed in the earlier paper. 10
Informed Consent
Permission from the Regional Health Bureau of Amhara Region, the zonal, districts, and village offices, and all the Trade and Transport Bureau offices within the selected districts were also obtained. Informed consent forms were signed by mothers before participating in the study. All identifiers were removed from the publication of the results.
