Abstract
There are few studies of cognitive behavioral therapy for women with antenatal depression including qualitative and quantitative data, and yet, individual cases can provide valuable information on personal experiences of treatment effectiveness and acceptability. The purpose of this case report is to explore the long-term qualitative outcomes following cognitive behavioral therapy for antenatal depression. A pregnant woman with a Diagnostic and Statistical Manual of Mental Disorders diagnosis of depression was allocated to receive seven sessions of cognitive behavioral therapy in a randomized controlled trial. We describe her experiences and mood during treatment, at 12 weeks, 9 months, 2 years, and 7 years postpartum, as well as markers of her child’s development. The woman’s mood symptoms were dramatically improved after treatment and remained in the mild to moderate range until 7 years postpartum. Her child showed overall age-appropriate development, with strengths highlighted in his nonverbal and problem-solving ability. Relative weaknesses were in the communication domain and his processing speed. This case report suggests that psychological treatment for depression during pregnancy can be both acceptable to women and potentially protective in the long term.
Introduction
Studies have reported rates of up to 20% for increased depressive symptomatology in pregnant women. 1 Antenatal depression is positively associated with postnatal depression 2 and high-risk health behavior. 3 Moreover, antenatal depression is associated with later behavior problems, cognitive deficits, and psychopathology in children.4,5 Treatment of antenatal depression might thus benefit the health of both mother and child. Qualitative research has received recognition as an important source of information. 6 A systematic review of intervention programs for antenatal depression concluded that quantitative and qualitative results are not always in line and that longer-term assessment is required. 7 To illustrate the potential long-term benefits of treatment, we use a mix of qualitative and quantitative data to report a case study of a pregnant woman presenting with a major depressive disorder and generalized anxiety disorder. We track her progress after treatment and describe some markers of her child’s behavioral, temperamental, and cognitive development, up to 7 years postpartum.
Case
Methods
This case study is drawn from a randomized controlled trial (RCT) that recruited pregnant women less than 30-weeks pregnant, screened with the Edinburgh Postnatal Depression Scale (EPDS),
8
and assessed potential recruits via the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders (4th ed.;
Case presentation
We describe a 33-year-old woman, who was 28-weeks pregnant with her second child. Over the preceding months, she had been feeling increasingly depressed and anxious and expressed recurrent thoughts about death. She resided with her husband and her 2-year-old daughter, after whose birth, she had experienced severe depression. At the start of this pregnancy, she was prescribed sertraline 50 mg but stopped as it made her feel “numb.” She had experienced sexual abuse as a child and described her parents as not very affectionate. She appeared restricted in her affect and met criteria for a major depression and a generalized anxiety disorder. Risk assessment was consistent with a current low risk of suicide although monitoring was indicated. She was allocated to the CBT group, consisting of seven individual sessions and one partner-inclusive session.
Summary of weekly cognitive behavioral sessions: content and goals set
In the first session, she described her goals as being able to exercise, eat well, and make lifestyle changes. In the second session, she recognized that she “took on more than she could chew” and an additional goal was to take on less. In the third session, she had gained insight in how to develop a positive thinking style and be more assertive. By the fourth session, she felt she had put changes in place and experienced fewer mood swings. Her goals were to wind back on helping others and to put less pressure on herself. In the fifth session, she had lowered her expectations. In the partner session, she said that she experienced her partner as supportive, and they expected there would be a lot of juggling to manage once the baby was born. In the seventh session, she stated that she had found a balance between positive and negative thoughts. In the final session, the strategies she described as helpful were being aware of negative feelings, trying to do more pleasurable things, expecting to be tense on busy days, doing less per day, looking at the positives, adjusting expectations, and letting things go. She felt ready to end the sessions.
Follow-up maternal outcomes
Maternal symptom scores of depression and anxiety from baseline to 7 years postpartum are illustrated in Figure 1. The symptom scores had dropped dramatically during the treatment. At 12 weeks postpartum, the woman’s score on the EPDS showed no indication for depression. She had not experienced the “blues” with her new baby. However, she had decided to again use sertraline 50 mg as she felt irritable, and a lack of sleep precluded her implementing CBT strategies at this early stage. She felt much better than with her first child and was balancing pleasurable activities. At 9 months’ follow-up, she reported that the program had made a difference in how she had been coping and that she was no longer using antidepressants, with similar reports at 2 and 7 years’ follow-up.
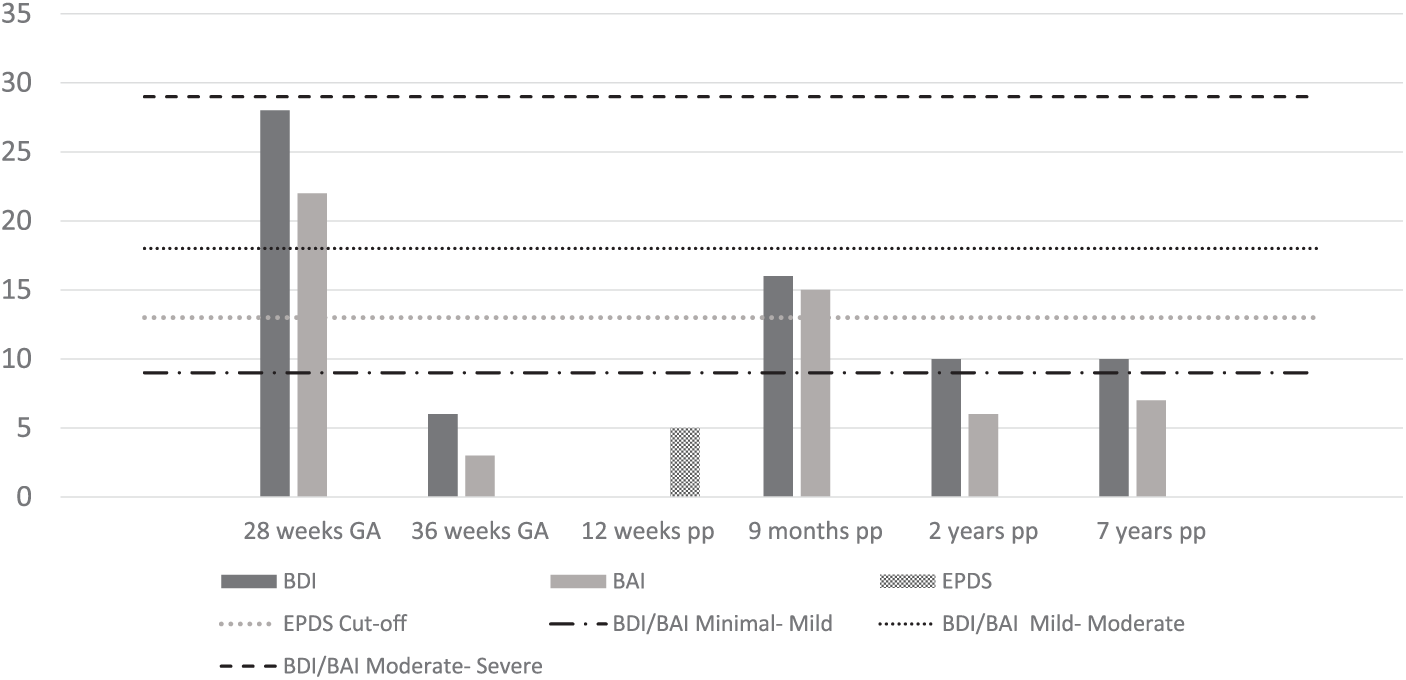
Mood symptoms scores from baseline up to 7 years postpartum of a case of a woman with antenatal depression, before and after treatment with cognitive behavioral therapy in a randomized controlled trial.
Follow-up child outcomes
At 9 and 12 months, developmental assessment indicated scores in the average range of development with good problem-solving and interpersonal skills, with communication skills and attention as areas of relative weakness. At 2 years, a cognitive assessment again indicated that the child was performing in the average to high average range with a significant strength in the cognitive domain. At 7 years, his nonverbal (performance) abilities were in the average range. A significant weakness however was his processing speed, which relies on the ability to attend to information without distraction. His scores on the CBCL at 7 years reflected age-appropriate behavior. Scaled scores of the Bayley-III at 2 years and the WPPSI-III at 7 years are presented in Table 1.
Developmental outcomes at 2 and 7 years of a case of a child after prenatal maternal depression treatment with cognitive behavioral therapy in a randomized controlled trial.

WPPSI-III: Wechsler Preschool and Primary Scale of Intelligence – III; IQ: intelligence quotient.
Discussion
With this case report, we have illustrated the positive outcomes that followed pregnancy-specific CBT in a woman with a major depressive disorder and generalized anxiety disorder during pregnancy. Some lasting benefits were clearly evident. In this case, the woman indicated she found several techniques from the sessions helpful, and she indicated that the program had made a difference in how she had been coping. Treating maternal mood symptoms during pregnancy may reduce the reported impact of depression and anxiety on fetal development. 20 We cannot know how her child would have developed if his mother had not been treated, or if she had been treated earlier, but his development showed that overall, he was in the normative range, with some skills better developed. It would have been insightful to compare her son’s development to that of his older sister. However, as there was no information available on the woman’s mood trajectory during her pregnancy with her daughter, and we did not have any data on her daughter’s development early postpartum, we were unable to establish valid comparisons. In future studies, adding sibling comparisons would be highly informative to distinguish the effects of CBT during pregnancy on child development from environmental and genetic confounding factors. We also know from the literature that depression treatment antenatally may reduce unhealthy life choices 3 and the risk for postnatal depression. 21 In this case, the woman did not experience postnatal depression of the severity reported with her previous child, and this may have been the direct result of antenatal treatment. Admittedly, she had been using sertraline, both early in pregnancy as well as in the postpartum period, at least for a couple of weeks as a short-term solution, and the positive effects on her mood are also likely (partially) attributable to the antidepressant use. Nevertheless, in this case, the woman had a clear preference for psychological therapy above medication use, as she experienced unpleasant side effects of sertraline. We argue that the use of antidepressants in pregnancy should not be discouraged per se but that careful consideration of the advantages and disadvantages of different treatments for depression, especially during pregnancy, is important. The efficacy of CBT with gradual, guided discontinuation of selective serotonin reuptake inhibitors (SSRIs) under medical management for depression during pregnancy is currently under study. 22 In conclusion, this case report suggests that psychological treatment for depression can be both acceptable to women and potentially protective in the long term.
Footnotes
Acknowledgements
The authors thank the participating women and staff at the hospitals involved. They also gratefully acknowledge Carmel Ferretti for her contribution to the interpretation of the infant developmental assessment outcomes.
Author notes
L.S.B. was the examiner of the 7-year postpartum follow-up of this case. The study was approved by the Human Research Ethics Committees of Austin Health and Northern Health, Melbourne, Australia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from Human Research Ethics Committees of Northern Health, Austin Health, and Mercy Health, Melbourne, Australia (trial registration: ACTRN12607000397415).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The randomized controlled trial and follow-up were funded by the MBF/BUPA Foundations, Australian Rotary Health, Urquhart Charitable Fund and Austin Medical Research Foundation and the Brain and Behavior Research Foundation (NASRAD Young Investigator Grant, project 22975). L.S.B. was supported by DynaHealth funding (grant agreement no. 633595, Horizon 2020).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
