Abstract
This pilot study examined the effects of freeze-dried mango (Mangifera indica L.) supplementation on anthropometric measurements, lipid parameters, and inflammatory mediators in obese individuals. A total of 20 obese (body mass index [BMI]: 30-35 kg/m2) adults (11 men and 9 women), aged 20 to 50 years, received 10 g/d of ground freeze-dried mango pulp for 12 weeks. Anthropometrics, lipids, and inflammatory mediators were assessed at baseline and after 12 weeks of mango supplementation. There were no differences between baseline and final visits in inflammatory mediators, lipids, diet, physical activity, and anthropometrics. Relationships were present at baseline and final visits between adiponectin and high-density lipoprotein cholesterol and between leptin and fat mass. Correlations were found after 12 weeks of mango supplementation between leptin and the following variables: waist-to-height ratio, BMI, percent fat, and fat mass. Our findings demonstrate that 12-week consumption of freeze-dried mango by obese individuals has no impact on obesity-related inflammation.
Introduction
Overweight and obesity, characterized by excessive adipose tissue, are major public health problems that present a serious challenge in both developed and developing countries.1,2 The World Health Organization 3 reported in 2014 that due to the high prevalence rates of overweight and obesity in both children and adults, global obesity has doubled since 1980. Of the public health challenges with overweight and obesity, there is substantial evidence of increased health risks from chronic inflammation and the development of various chronic diseases, including diabetes and cardiovascular disease (CVD).4 –6 The impact on the health care expenditures required to treat obesity and its comorbidities will continue to be high without targeted intervention. 7 Effective strategies need to be identified to aid in the prevention of overweight, obesity, and the chronic diseases associated with excess adipose tissue and inflammation. Interestingly, it has been demonstrated that when incorporated into the diet, certain fruits with bioactive components, such as mangoes (Mangifera indica L.), may reduce obesity-related disorders, including glucose intolerance and/or excess body fat, conditions that are associated with inflammation.8–10
Excessive adipose tissue induces complex interactions physiologically and metabolically, which promote a pro-inflammatory state and the development of obesity-related comorbidities.4,11 Under normal conditions, anti-inflammatory cytokines, such as adiponectin, are secreted in the body to reduce inflammation and maintain homeostasis. 12 However, with obesity, there is an enhanced production of pro-inflammatory adipocytokines and a decrease in anti-inflammatory cytokines, which gives rise to a low-grade inflammatory state. 13 Some of the mediators involved in the relationship between obesity, inflammation, and chronic disease are leptin, adiponectin, interleukin (IL)-1b, IL-6, IL-17A, tumor necrosis factor α (TNF-α), monocyte chemoattractant protein-1 (MCP-1), superoxide dismutase (SOD), and C-reactive protein (CRP). 4
Even though the scientific literature on the interplay between obesity and inflammatory continues to evolve, effective strategies for intervention must consider reduction in inflammation and excessive adiposity for the prevention of obesity-related chronic diseases. 14 Intervention strategies have included increased physical activity and a healthy diet because lack of physical activity and diets high in fat are known contributors to the development of overweight and obesity. 15 In addition, consumption of refined carbohydrates has also been evaluated for their influence on obesity, blood sugar, and inflammation.16,17 Foods that have a greater content of carbohydrates and added sugars are known to negatively affect blood glucose and insulin, and, hence, may cause an increase in inflammatory mediators. 18 Studies have shown that fruits containing natural carbohydrates and not added sugars are shown to positively affect inflammation and obesity.19,20 As an example, a proprietary blend of mangosteen juice decreased serum CRP and had a modest reduction in body mass index (BMI) in obese individuals. 10 In addition, our preclinical study has demonstrated that mango supplementation reduced adiposity and lowered blood glucose concentrations in a diet-induced obesity model. 9
The mango fruit is a good source of dietary fiber and contains several bioactive compounds and antioxidant nutrients, such as polyphenols (eg, mangiferin), carotenoids, and ascorbic acid, that have exhibited anti-inflammatory and/or antidiabetic properties in human and/or animal models.9,21,22 Our recent human pilot study reported that mango powder supplementation lowered blood glucose in obese individuals. 23 Because higher levels of blood glucose are thought to trigger an inflammatory response, reduction in blood glucose, as seen with mango supplementation in both the animal and human studies, may also lead to reduction in inflammation. 24
In view of our findings regarding the effects of mango supplementation on blood glucose, this study investigated the effects of freeze-dried mango consumed daily for 12 weeks on anthropometric measurements, serum lipids, and inflammatory markers in obese individuals. Because mango supplementation has been demonstrated to reduce blood glucose, we hypothesize that the incorporation of mango into the diet will also have positive effects on body composition, lipid profile, and inflammatory response in obese individuals.
Methods
Participants and recruitment
In this pilot study, 20 obese (BMI = 30-35 kg/m2) adults, between 20 and 50 years of age, were recruited from 2011 to 2013 in Stillwater, Oklahoma, and the surrounding area. Study characteristics for inclusion were adults who did not use any form of tobacco, drank less than 1 oz of alcohol per day, and were obese with normal liver (normal range for aspartate aminotransferase [7-40 U/L] and alanine aminotransferase [10-45 U/L]) and kidney function tests (blood urea nitrogen: 5-20 mg/dL and normal serum creatinine: adult male: 0.6-1.2 mg/dL, adult female: 0.5-1.1 mg/dL). Participants were excluded if they had the following preexisting medical conditions: CVD, cancer, diabetes, liver, or renal disorders; were pregnant or lactating females; were consuming mega doses (>1 g/d) of antioxidants and/or fish oil supplements; and/or abnormal hemoglobin concentration (normal range: 12.0-18.0 g/dL), white blood cell count (normal range: 4.0-11.0 k/mm3), or platelets (normal range: 140-440 k/mm3).
Study design
The study was approved by the Institutional Review Board at Oklahoma State University (Stillwater, OK, USA). The design included a prescreening interview by telephone, email, or in person and 4 visits to Oklahoma State University’s Department of Nutritional Sciences for evaluation. After the prescreening interview and obtaining consent from the participant, visit 1 consisted of: (1) baseline anthropometric measurements, (2) completion of health/medical and physical activity questionnaires, and (3) instructions from trained personnel on how to document dietary intake in a 3-day food record. At visit 2, which was approximately 1 week after visit 1, participants turned in their first completed 3-day food record, had a fasting baseline blood sample drawn, and underwent a baseline body composition scan by dual-energy x-ray absorptiometry (DXA; Discovery A S/N 84671, Hologic Inc., Bedford, MA, USA). At this visit, participants were provided 6 weeks of prepackaged (10 g/packet) freeze-dried ripe “Tommy Atkins” mangoes. They also received instructions on how to best consume the mango supplement and how to record daily mango supplement intake in the provided study calendar. Participants were also instructed to store the mango supplement in the freezer until consumption. At visit 3, participants returned all mango packets (both empty and not consumed) and the calendar used for recording mango supplement consumption. Participants were then given the second 6-week supply of mango supplement and a new calendar. Visit 4 was the last visit, which concluded with the final fasting blood draw, DXA scan, and physical activity questionnaire administration. At this time, the final calendar of supplement intake, all supplement packets received at visit 3, and the last 3-day food record were turned in. Throughout the study, participants were reminded to maintain their normal dietary intake and performance of physical activities while also continuing to take any regularly prescribed medications. Compliance was managed for each participant through the following measures: one 6-week supply of mango supplement was received at visits 2 and 3, records of mango supplement consumption were kept in study calendars, both empty and unused packets of mango supplement were returned at visits 2 and 3, and random telephone contact was made to promote adherence to daily consumption of the mango supplement.
Dietary intervention
In this study, we used the same mango preparation, variety, and equivalent dose that were employed in our animal model of diet-induced obesity. 9 The 10 g/d freeze-dried mango used in this study is equivalent to the amount used in our earlier animal study that showed mango supplementation improved glucose response and body composition in mice fed a high-fat diet. 9 The dose of 10 g/d freeze-dried mango is approximately 100 g fresh mango or half of a fresh mango per day.9,25 The 10 g/d freeze-dried mango powder provided approximately 39 kcal and contained 89.6% carbohydrate, 4.01% protein, 1.62% fat, and 13.4% fiber (analyzed by NP Analytical Laboratories, St. Louis, MO, USA).
“Tommy Atkins” was the mango variety chosen because it can easily be found in US markets, with peak times during the months from March to July and October to January. To prepare the supplement, ripe “Tommy Atkins” mangoes were acquired from a local supermarket, peeled, and the pulp was freeze-dried and ground into a powder. The mango powder was preweighed, placed in opaque plastic bags (10 g/bag), and stored in the freezer until ready for distribution.
Anthropometric and clinical parameters
Height, weight, circumferences of waist and hip, blood pressure, and DXA scan of each participant were taken at baseline and at the end of 12-week supplementation by trained personnel. When undergoing evaluations, participants wore no shoes and light indoor clothing (scrubs) provided by the study to maintain consistency of weight. Each participant was weighed to the nearest ±0.1 kg while standing on a flat surface using the Health O meter Physician Balance Beam weight scale (Continental Scale Corp., Chicago, IL, USA). A scale-mounted Shorr Board Stadiometer (Shorr Productions, Olney, MD, USA) measured the standing height of each participant to the nearest 0.1 cm. Body mass index (weight in kg/height in m2) or Quetelet index was used to delineate appropriate weight or mass per height. 21 Body mass index was categorized as healthy weight (BMI: 18.5-24.9 kg/m2), overweight (BMI: 25-29.9 kg/m2), and obese (BMI: 30 kg/m2 and greater). 26
Using standard methods and a tape measure, waist and hip circumferences were measured to the nearest 0.1 cm. 27 The waist circumference, an indicator of excess fat around the abdomen, was measured at the narrowest area between the inferior portion of the costal margin and the top of the iliac crest. Increased risk of obesity-related CVD was defined as a waist circumference of 102 cm (40 in) or more in men or 88 cm (35 in) or more in women.28 –30 Hip circumference was measured at the greatest distance around the hips. Waist-to-hip circumference ratio (WHR; increased cardiovascular risk if >1.0) and the waist-to-height ratio (WHtR; increased cardiovascular risk if >0.53 for men and >0.49 for women) were calculated.31,32
After participants rested in a quiet environment for approximately 5 to 10 minutes, blood pressure was measured at 2 different times on the same arm, using a digital blood pressure monitor (ReliOn; Oncue HealthCare Inc, Bennockburn, IL, USA). Blood pressure levels were categorized as follows: normal (systolic: ≤120 mm Hg, diastolic: ≤80 mm Hg), prehypertension (systolic: 120-139 mm Hg, diastolic: 80-89 mm Hg), and hypertension (systolic: ≥140 mm Hg, diastolic: ≥90 mm Hg).33,34
A DXA scan assessed each participant’s body composition at baseline and after 12 weeks of mango supplementation. During the DXA scan, the participant wore light indoor clothing and laid in the standardized supine position (arms at sides).
Laboratory measurements
After an 8- to 10-hour fast, a venous blood sample was obtained from each participant by a certified phlebotomist or registered nurse at baseline and at the end of the 12 weeks of mango supplementation. An independent laboratory (Stillwater Medical Center, Stillwater, OK, USA) performed the analyses for plasma triglycerides, total cholesterol, high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, very-low-density lipoprotein (VLDL) cholesterol, glucose, and insulin. The blood chemistries were evaluated based on the following normal reference values: total cholesterol: <200 mg/dL, triglycerides: <150 mg/dL, HDL cholesterol: ≥40 mg/dL for men and ≥50 mg/dL for women, LDL cholesterol: <130 mg/dL, VLDL cholesterol: ≤30 mg/dL, HDL/LDL: <3.5 to 1.0, blood glucose: <100 mg/dL, and insulin levels between 10 and 20 μU/mL.35 –37
To assess plasma levels of adiponectin, leptin, and CRP, enzyme-linked immunosorbent assay kits from EMD Millipore Corporation (St. Charles, MO, USA) were used. Bio-Plex Pro cytokine assay kits from Bio-Rad Laboratories, Inc. (Hercules, CA, USA) were used to measure IL-1β, IL-6, IL-10, IL-17A, MCP-1, and TNF-α. Superoxide dismutase was assessed using a kit from Cayman Chemical Company (Ann Arbor, MI, USA).
Health, medical, dietary, and physical activity assessment
A health and medical history questionnaire was completed at baseline by each participant. This questionnaire focused on social aspects (eg, smoking, alcohol), education, allergies, medications, supplements, hospitalizations, and medical conditions. Dietary intake was assessed through a self-reported 3-day food record at baseline and after 12 weeks of mango supplementation. The food record consisted of all food and beverage consumed by the participant more than 3 consecutive days, including at least 1 weekend day. Analysis of dietary intake was accomplished by the Diet Analysis Plus 10.0 software (Cengage Learning, Inc., Independence, KY, USA).
The physical activity questionnaire used in the study was the Physical Activity Scale 2 by Andersen et al. 38 Trained personnel administered the questionnaire to participants at baseline and at the final visit. The participant’s physical activity was evaluated as to the time spent each day in various activities, such as sleeping, sitting, watching TV, reading, standing, walking, heavy physical work, and transportation to and from work. The amount of time spent weekly in performing light, moderately strenuous, and strenuous physical activity was also determined.
Statistical analysis
Data were analyzed using PC SAS version 9.4 (SAS Institute, Cary, NC, USA). Descriptive statistics were determined to describe the socio-demographics, laboratory measurements, and the clinical characteristics of the participants. Pearson linear correlation coefficients were calculated regarding the relationships between obesity parameters and inflammatory markers. Paired t-tests were conducted, overall and by sex, between clinical parameters and inflammatory mediators to assess the effects of the 12-week mango supplementation on obese adults. The level of significance was set at P < .05 for all analyses.
Results
Participants
Initially, there were 22 participants enrolled in the study, but 2 male subjects were not able to complete the study protocol due to either being absent for a visit or by not providing the required documentation of mango supplement consumption. Aside from the 2 study participants who did not finish the study due to compliance issue, the study participants reported to consume the mango supplement daily as shown by their calendar records and return of both empty and unused packets of mango supplement. In addition, participants did not report any adverse effects from consuming the freeze-dried mango supplement daily for the duration of the study.
There were 20 adults (11 men [55%], 9 women [45%]), who finished the study and consumed freeze-dried mango for 12 weeks. The mean age was 36.5 ± 9.1 years (men: 34.8 ± 9.6 years; women: 38.6 ± 8.4 years). Of the 20 participants in the study, 4 were receiving treatment for hypertension and 1 participant each for gastroesophageal reflux disorder, menopause, or seasonal allergies. No significant changes were found in the dietary consumption and physical activity of the participants when comparing baseline with the end of the 12-week mango supplementation as we have previously reported. 23 The average daily caloric intake (kcal) at baseline and after the 12-week mango powder supplementation for all the participants, as well as separated by sex, was as follows: overall: 2200 ± 640 and 1995 ± 472 kcal (baseline and final, respectively); men: 2341 ± 858 and 2102 ± 372 kcal (baseline and final, respectively); and women: 2096 ± 545 kcal and 1823 ± 545 kcal (baseline and final, respectively).
Anthropometrics, body composition, and clinical parameters
Descriptive statistics and paired t-tests, overall and by sex, regarding the effects of 12-week mango supplementation on participant weight, BMI, waist circumference, WHR, WHtR, percent fat, and fat mass are shown in Table 1. There were no significant differences, overall and by sex, in anthropometrics and body composition of the participants. A more complete description has been previously reported. 23 The WHRs of both male and female participants were within normal limits; however, based on the mean values of waist circumference and WHtR, study participants were at increased risk of obesity-related CVDs, as defined by the National Heart, Lung, and Blood Institute (NHLBI). 30
Effects of 12-week freeze-dried mango supplementation on anthropometric measurements and body composition of obese adults a .

Abbreviation: BMI, body mass index.
Data reported as mean ± SD. Values in brackets are the minimum and maximum values.
The average blood pressures for participants were 133 ± 15 mm Hg for systolic blood pressure and 85.0 ± 8.0 mm Hg for diastolic blood pressure, both of which fall in the range of the prehypertensive category, as per NHLBI guidelines. 37 There were no significant differences in paired t-tests between baseline and final visits observed in either systolic (P = .197) or diastolic blood pressures (P = .198).
Lipid profile
The mean descriptive values of the complete lipid profile, overall and by sex, were within normal limits (reference range noted in the methods section and reported in Table 2). There were also no significant differences between baseline and final levels of the lipid profile, overall and by sex, after 12 weeks of mango supplementation.
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; VLDL, very-low-density lipoprotein.
Data reported as mean ± SD.
These values were previously reported. Adapted from Evans et al. 23
Inflammatory mediators
Overall baseline and final descriptive statistics are shown in Table 3 and Figure 1. Descriptive statistics and paired t-tests of the inflammatory mediators by sex are shown in Table 3. Paired t-tests, overall and by sex, between baseline and post 12-week mango supplementation levels of the inflammatory mediators were not significantly different. Overall and by sex, the baseline and final levels of one of the most commonly used indicators of inflammation, CRP, were elevated above the reference range, which is indicative of increased risk of atherosclerosis (reference range: 0-3 μg/mL). 39
Effects of 12-week freeze-dried mango supplementation on anti-inflammatory and pro-inflammatory mediators in obese individuals, overall and by sex a .
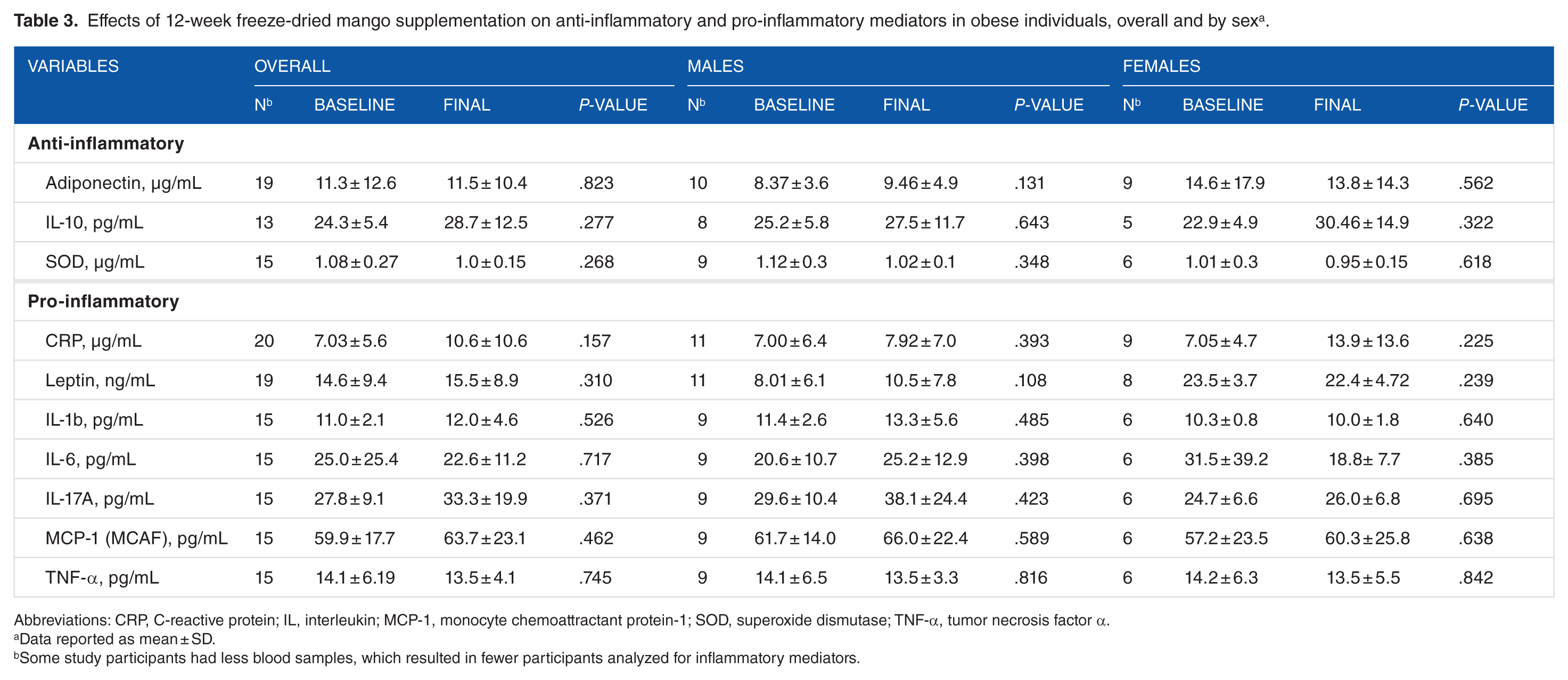
Abbreviations: CRP, C-reactive protein; IL, interleukin; MCP-1, monocyte chemoattractant protein-1; SOD, superoxide dismutase; TNF-α, tumor necrosis factor α.
Data reported as mean ± SD.
Some study participants had less blood samples, which resulted in fewer participants analyzed for inflammatory mediators.

Overall baseline and final inflammatory mediators in obese individuals before and after 12 weeks of mango supplementationa. Abbreviations: CRP, C-reactive protein; IL, interleukin; MCP-1, monocyte chemoattractant protein-1; SOD, superoxide dismutase; TNF-α, tumor necrosis factor α.
Pearson correlations between serum inflammatory mediators (i.e., leptin, adiponectin, IL-6, and TNF-α) and anthropometrics and body composition after 12 weeks of mango supplementation are shown in Table 4. Positive correlations significant at both baseline and final visits were between leptin and fat mass (baseline [r = .622, P = .004] and final [r = .690, P = .001]) and adiponectin and HDL cholesterol (baseline [r = .697, P = .001] and final [r = .704, P = .001]). After 12 weeks of mango supplementation, there were no significant correlations between adiponectin, IL-6, and TNF-α and anthropometrics and body composition measures; however, positive significant Pearson correlations were found between leptin and the following anthropometrics and body composition measures: WHtR, BMI, and percent fat. In addition, the remaining inflammatory mediators evaluated were not significantly correlated (data not given).
Pearson product-moment correlation coefficients (r) between serum inflammatory mediators, anthropometrics, and body composition in obese individuals before and after 12 weeks of mango supplementation.

Abbreviations: BMI, body mass index; IL, interleukin; TNF-α, tumor necrosis factor α.
Some study participants had less blood samples, which resulted in fewer participants analyzed for the correlations.
Correlation is significant at the .05 level (2-tailed); **correlation is significant at the .01 level (2-tailed).
Discussion
The relationship between the effects of bioactive substances on obesity and levels of anti-inflammatory and pro-inflammatory mediators is being investigated to develop appropriate interventions to lower the risk of obesity-related chronic conditions induced by inflammation, such as diabetes and CVD.4,8,9,17,40,41 Strategies targeting reduction in body fat, lipids, and inflammation would be useful not only in the treatment of obesity but also in prevention of obesity-related chronic conditions triggered by the inflammatory response.14,42 It has been shown that certain foods and their bioactive components, such as the mango fruit and its many polyphenolic compounds and vitamins (eg, mangiferin, ascorbic acid, lycopene, β-carotene), can help reduce body fat and/or modulate inflammation.9,10 To the best of our knowledge, this study is the first to investigate the effects of mango supplementation on anthropometrics, lipids, and levels of inflammatory mediators in obese individuals.
Research has established that excessive abdominal or visceral adiposity and chronic low-grade inflammation are causally involved in the development of obesity-related chronic conditions, such as diabetes and CVD. 11 Measurements, such as waist circumference and, in particular, the 2 ratios of WHR and WHtR, can be used to define abdominal or visceral obesity and to assess risk for disease.43,44 In this study, obese individuals were found to have abdominal or visceral adiposity based on waist circumference and WHtR. These indicators, when categorized, fell within the range for an increased risk of obesity-related CVD.
When evaluating obesity-related chronic conditions, the inflammatory response related to overweight and obesity appears to stem from the excessive number and increased size of adipocytes, which, consequently, cause oxygen depletion and cell death in adipose tissue. 40 If cellular hypoxia and death persist, there is overstimulation of the immune system with a continual release of free fatty acids into the blood that subsequently results in a production of both pro-inflammatory and anti-inflammatory cytokines. If left unresolved, an elevated level of pro-inflammatory cytokines or mediators leads to chronic inflammation and ultimately to undesirable conditions, such as CVD and metabolic dysfunction. 45 Unfortunately, concentration of free fatty acids in the blood was not assessed in this study and so this relationship cannot be confirmed in our study.
Certain bioactive substances, particularly in fruits, have been found to influence inflammation and/or obesity.8 –10,17 Of these substances, several are found in the pulp, leaves, and bark of mango, which are thought to be responsible for this fruit’s reported positive health effects. 46 In comparison with other tropical fruits, mango consumption has demonstrated beneficial effects on postprandial glucose and insulin response in type 2 diabetes. 47 Analysis of mango reveals that it is a good source of dietary fiber, ascorbic acid, carotenoids, and phenolic compounds, which are thought to aid also in the reduction or prevention of obesity and its associated chronic inflammatory conditions. 46 Of special interest, studies on mango supplementation have shown mango contains bioactive substances that exhibit both anti-inflammatory and antidiabetic characteristics.9,21 Substances, such as ascorbic acid, carotenoids, and phenolic compounds, in mango may play an active role in reducing obesity-related inflammation by acting as agents that lower oxidative stress and interfere with pro-inflammatory mediator signaling pathways.48 –50 The interaction between bioactive substances and the various inflammatory mediators is complex and requires extensive scrutiny.4,17,21,40,51
Of the inflammatory mediators assessed in this study, the adipocytokines, leptin and adiponectin, are especially important in the interplay between bioactive substances, inflammation, and metabolism in obesity.52,53 It has been determined that leptin and adiponectin are exclusively secreted by adipocytes and that the level of expression is dependent on the degree of adiposity present. 54 In obesity, the level of leptin is usually higher, whereas adiponectin is lower.
Adipocytes secrete the protein leptin, a hormone proposed to be a suppressor of appetite and regulator of body weight. 12 Serum leptin concentrations differ by sex and, as in other studies, female participants’ mean level of leptin was greater than that of the male participants. In addition, as expected, leptin was positively correlated with total fat mass at both baseline and final visits. The concern with leptin is that, in the presence of metabolic dysregulation, this adipocytokine can exert pro-inflammatory properties. As an intervention to lower the negative effects of leptin, consumption of foods rich in anti-inflammatory bioactive substances, such as those in mango, could prove to be protective. For example, lycopene, a carotenoid in mango, has been reported by de Azevedo Melo Luvizotto et al. 55 to lower leptin levels when using a tomato oleoresin-maize oil mixture in an animal model. However, after 12 weeks of mango supplementation, there were no significant changes in leptin levels but there were positive associations of leptin with percent fat, WHtR, and BMI.56,57
Adiponectin is another adipocytokine that participates in various metabolic interactions, which include both anti-inflammatory and anti-atherogenic functions.53,58 Although prior research has reported an association of adiponectin and abdominal or visceral adiposity, no associations were determined between adiponectin and the anthropometric measures of weight, BMI, waist circumference, WHR, and WHtR in the study participants. 59 With respect to effects on lipid profile, a positive correlation was seen between adiponectin and HDL cholesterol (r value reported in results) at both baseline and post mango supplementation in obese individuals, which has similarly been demonstrated in other studies.60,61 Although the interaction between adiponectin and HDL cholesterol has not been fully investigated, it would appear to reduce the risk of CVD.58,60,62 Because adiponectin has beneficial effects but may be reduced in obesity, intervention strategies to increase adiponectin may include consumption of foods high in polyphenolic compounds, such as mangiferin in mango. In fact, Saleh et al. 63 reported that intraperitoneal administration of mangiferin in diabetic insulin-resistant rats increased serum adiponectin levels via activation of the peroxisome proliferator–activated receptor γ (PPAR-γ).
Another important inflammatory mediator assessed in our study is TNF-α. The concern regarding TNF-α is that when elevated, it can damage cellular contents and upregulate production of other pro-inflammatory cytokines. 64 Saleh et al. 63 also found that mangiferin reduced levels of TNF-α via activation of PPAR-γ in diabetic insulin-resistant rats. In this study, no significant changes where observed in TNF-α before and after mango supplementation in both female and male participants. However, we observed a positive association between TNF-α with BMI, as noted in other studies, but there were no associations with any other anthropometric parameters. 65
Finally, CRP is a commonly measured nonspecific inflammatory marker that, when increased, is indicative of inflammation stemming from acute and chronic conditions (eg, obesity, high blood pressure, diabetes, cigarette smoking, medication therapy). 66 Untreated hypertension, diabetes, and smoking were exclusions in this study; however, 1 female participant was taking estrogen replacement therapy. Fiber, found in significant amounts in mango, has been demonstrated to lower CRP and oxidative stress through inhibition of the inflammatory process, as reported in a study by Xie et al. 67 on the effects of water-soluble fiber supplementation (added to rice) in hemodialysis patients. When comparing baseline and final assessment, there was no significant rise or decrease in CRP, either overall or by sex. However, it should be noted that the level of CRP remained above the reference range (0-3 μg/mL), which represents an increased risk in CVD.39,68 –70
Before and after 12 weeks of mango supplementation, associations were demonstrated between inflammatory mediators, anthropometrics, and lipids in obese individuals. There were no significant differences between baseline and final inflammatory mediators, but this may be related to the findings that the obese participants had normal lipid profiles and no history of preexisting chronic medical conditions. Of interest, study parameters indicated that the participants were at increased risk of developing obesity-related chronic diseases, such as CVD. This may suggest a need for a longer time and/or different dose of mango supplementation in obese individuals to bring about positive effects in inflammatory mediators. Our findings demonstrate that freeze-dried mango fruit supplementation had no effects on the inflammatory response related to obesity.
Strengths and Limitations
In this study, there are limitations that should be under consideration for their potential impact. Some of these limitations include factors such as, the small sample size, the lack of a control group, and the dose and duration of mango supplementation. For example, outliers in a small study may overtly affect the study outcomes, due to this being an exploratory research, all data were included. One must also bear in mind that both dietary intake and physical activity level were self-reported but; however, participant responses were obtained using validated questionnaires and professionally administered by trained personnel. Because consumption of mango supplementation took place for a duration of 3 months, this may not have allowed enough time to initiate an effect and suggests that the time of supplementation could be extended in future research. In addition, we only used one dose of mango supplement and higher dose might be needed to see an effect on inflammatory mediators. Future studies should consider the limitations of our study.
Conclusions
Mango supplementation in the diet may play a vital role in the prevention of obesity and obesity-related chronic inflammatory conditions, as shown in prior studies.9,21 In this study, when assessing baseline and final inflammatory mediators, anthropometric measurements, and lipid parameters, the 12-week mango supplementation in obese individuals had no effects. With consumption of mango, relationships were demonstrated between leptin, a pro-inflammatory mediator, and measurements of WHtR, BMI, percent fat, and total fat mass, along with adiponectin, an anti-inflammatory mediator, and HDL cholesterol. Future research may consider a longer consumption duration and/or different dose of mango supplementation to investigate its effects on inflammatory mediators in obese individuals previously diagnosed with chronic diseases.
Footnotes
Acknowledgements
The authors would like to thank all the study participants and the staff at the Stillwater Medical Center for their support during this study. They also acknowledge the Oklahoma State University Department of Nutritional Sciences for their assistance in this research.
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1185 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A grant from the National Mango Board supported this study, but the Board was not involved in the study’s design, data collection, data analysis, manuscript preparation, or decision in manuscript submission. Additional support was also received through the Oklahoma State University College of Human Sciences.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
EAL, BJS, and PP-V contributed to the experimental design; PP-V to the preparation of the freeze-dried mango powder; SFE, EAL, MB, MM, SJ, and HE to the study procedures; MB, MM, SJ, HE, and SP to the laboratory analyses; and MP to the statistical analyses. SFE prepared the first draft of the manuscript. EAL, SFE, MP, BJS, and PP-V contributed to joint revisions and final approved version of paper.
Disclosures and Ethics
This paper is not under consideration by any other publication nor had any prior publication. The authors have also provided to the publisher documents confirming compliance with legal and ethical obligations, as a requirement of publication.

