Abstract
Background:
Sickle cell disease (SCD) patients are an important risk group for Staphylococcus aureus (S. aureus) carriage and infections. Little is, however, known about the nasopharyngeal carriage epidemiology of the pathogen in this vulnerable population.
Aim:
The aim of this study was to evaluate S. aureus and methicillin-resistant S. aureus (MRSA) nasopharyngeal carriage prevalence, carriage determinants, and antimicrobial resistance among SCD adults in Ghana.
Methodology:
Nasopharyngeal swabs, obtained from 200 SCD adults recruited at the Korle Bu Teaching Hospital, were cultured for S. aureus, and these isolates were subjected to antimicrobial susceptibility testing via the Kirby-Bauer method.
Results:
The prevalence of S. aureus carriage was 41.5% (n = 83), and that of MRSA carriage was 1.0% (n = 2). Moreover, carriage of coagulase-negative Staphylococcus (CoNS) was the only determinant of S. aureus carriage identified (OR = 0.012, P < .0001). However, neither this variable nor the other features of the participants emerged as a determinant of MRSA carriage. The antimicrobial resistance rates decreased across penicillin (98.8%, n = 82), tetracycline (54.2%, n = 45), gentamicin (32.5%, n = 27), ciprofloxacin (21.7%, n = 18), erythromycin (18.1%, n = 15), clindamycin (10.8%, n = 9), amoxicillin-clavulanic acid (10.8%, n = 9), teicoplanin (1.2%, n = 1), and linezolid (0.0%, n = 0), and the multidrug resistance rate was 45.8% (n = 38).
Conclusion:
The nasopharyngeal carriage prevalence of S. aureus in the current study was high, while that of MRSA was low. The isolates were highly resistant to several of the antibiotics tested, but not teicoplanin and linezolid, making these antibiotics suitable for treatment of S. aureus infections among the SCD population.
Introduction
Staphylococcus aureus (S. aureus) is a ubiquitous commensal and an important cause of invasive and other diseases, such as meningitis,1 -3 endocarditis,2 -5 bacteremia,1,4,6,7 septicemia,1,2,5,6,8,9 skin and deep tissue infections,10,11 bacterial conjunctivitis, 5 osteomyelitis, 2 surgical wound infections,1,5 lower respiratory tract infections,2,9 and medical implant infections.12 -14 Its infections are preceded by its carriage, which could be intermittent or persistent. 5 Although S. aureus carriage principally occurs in the anterior nares, the nasopharynx is another major carriage site of the pathogen.5,15,16 The high clinical significance of S. aureus has been potentiated by its propensity for antimicrobial resistance acquisition, cardinal of which is methicillin resistance. Methicillin resistance is primarily mediated by the acquired cassette-borne mecA gene, and S. aureus subpopulations that exhibit this trait are called methicillin-resistant S. aureus (MRSA). 17 MRSA is resistant to several routinely-prescribed antibiotics, particularly the beta-lactams (including penicillin, amoxicillin, and oxacillin), and like most multidrug-resistant organisms, it significantly increases the economic burden of healthcare, morbidity, and mortality.18 -20 In the past 2 decades, the fraction of S. aureus infections attributed to MRSA has been on the rise, with high fatalities.21 -25
Sickle cell disease (SCD) patients are an important risk group for S. aureus carriage and infections, partly owing to their immunocompromised state and their frequent visits to healthcare facilities.26 -30 Moreover, they often take in antibiotics, either for treatment of infections they acquire, or as prophylaxes, and this could select for multidrug resistance among S. aureus and other commensal bacteria they harbor. Nonetheless, less attention has been paid to them in terms of antimicrobial resistance (AMR) surveillance. Particularly, little is known about them in relation to S. aureus nasopharyngeal carriage epidemiology. To help fill these knowledge gaps, the current study evaluated S. aureus and MRSA nasopharyngeal carriage prevalence, carriage determinants, and antimicrobial resistance among sickle cell disease (SCD) adults in Ghana.
Methods
Study site, design, and sampling
This cross-sectional study was conducted at the Korle Bu Teaching Hospital (KBTH) in Accra. KBTH is a major referral health facility, serving a population of over 1.6 million individuals. The hospital houses the Ghana Institute of Clinical Genetics (GICG), which operates a sickle cell disease clinic from Mondays to Fridays, and annually, receives about 10 000 visits from patients. Archived nylon-tipped nasopharyngeal swab (Copan Flock Technologies Srl, Italy, Lot L.70VE00) specimens previously collected between September 2016 and June 2017 from 200 SCD adults in the pneumococcal epidemiology study of Dayie et al 31 were the samples used in this study.
S. aureus (and MRSA) isolation, identification, and antimicrobial susceptibility testing
As summarized in Figure 1, the swab specimens were bacteriologically cultured, and the resultant isolates were identified and subjected to antimicrobial susceptibility testing, as described by Dayie et al. 15 First, each archived specimen, contained in 1 mL skim-milk tryptone glucose glycerol (STGG) medium, was retrieved from the −80°C storage and thawed at room temperature, followed by vortexing and subsequent pre-enrichment in 5 mL of brain heart infusion broth. Twenty-four hours post-incubation, each suspension was plated on Mannitol salt agar (Oxoid, Basingstoke, Hants, UK) and incubated aerobically at 37°C for 18 to 24 hours. Isolate identification was based on a combination of colonial morphology, Gram reaction, and catalase and tube coagulase tests. Among the catalase-positive isolates, those that tested positive to coagulase were identified as S. aureus, and those that tested negative, as coagulase-negative Staphylococci (CoNS). S. aureus ATCC 25923 was used as a positive control.
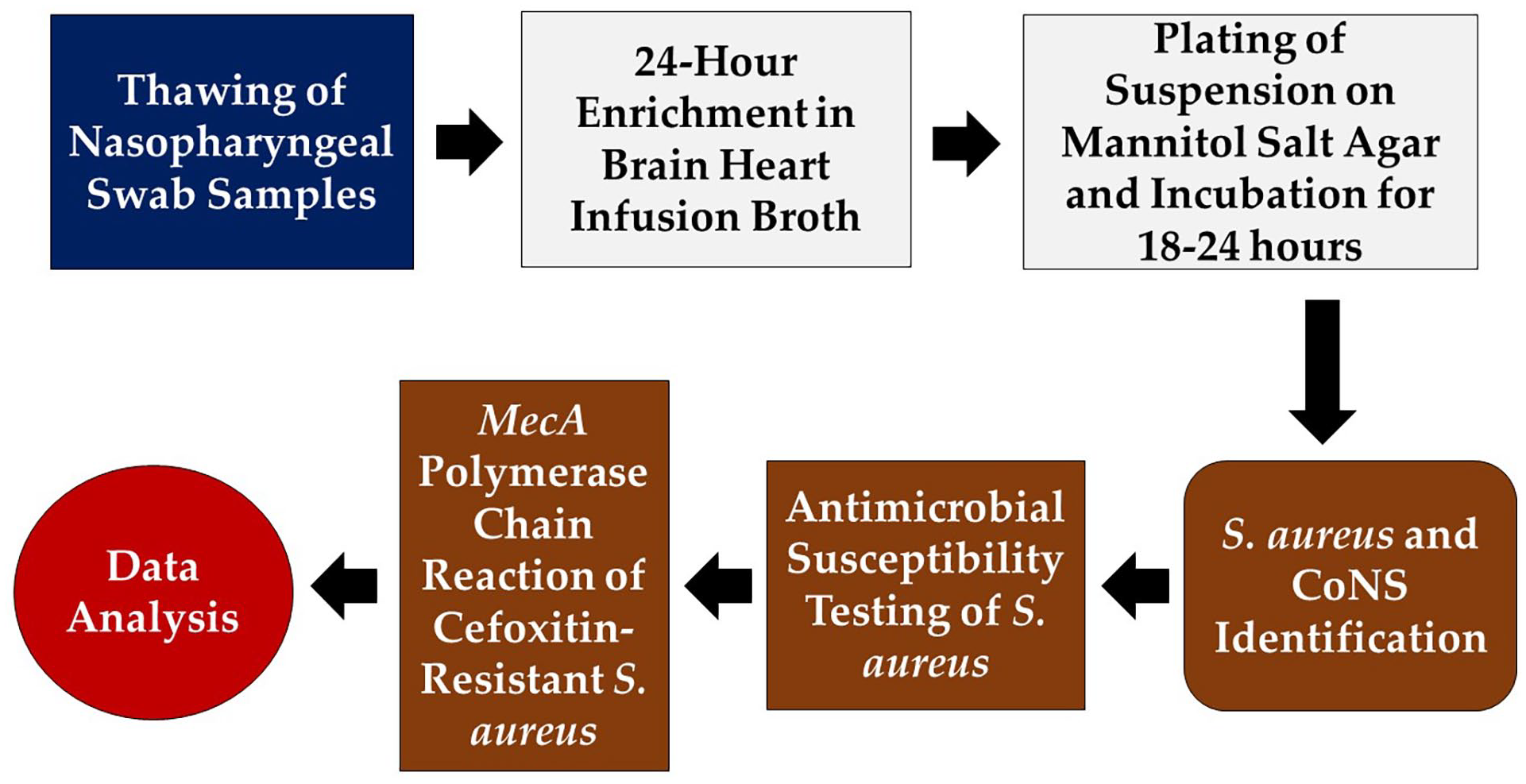
A scheme of the methodology.
Antimicrobial susceptibility testing (AST) was done on Mueller-Hinton agar (Oxoid, Basingstoke, Hants, UK) plates using penicillin (1 unit), tetracycline (30 µg), gentamicin (10 µg), ciprofloxacin (5 µg), erythromycin (15 µg), clindamycin (2 µg), amoxicillin-clavulanic acid (20/10 µg), teicoplanin (30 µg), linezolid (30 µg), and cefoxitin (30 µg) (Oxoid, Basingstoke, Hants, UK), via the Kirby-Bauer method, in accordance with the Clinical and Laboratory Standards Institute (CLSI) 32 recommendations. S. aureus ATCC 25923 was used as a positive control. The inoculums used were of 0.5 McFarland standard equivalent turbidity. The AST plates were read after aerobic incubation at 37°C for 18 to 24 hours, and interpreted according to the breakpoints of (CLSI). 32 Isolates that were cefoxitin-resistant were presumptively identified as MRSA, and this identity was confirmed via polymerase chain reaction targeting the mecA gene, using an in-house mecA-positive MRSA isolate as a positive control, as described by Dayie et al. 15
Data analysis
The data were analyzed using Statistical Products and Services Solutions (SPSS), version 25. The participants’ data were summarized with descriptive statistics, and univariate and multivariate analyses were used to identify determinants of S. aureus and MRSA colonization, at an alpha level of .05.
Ethical approval
The study was given approval by the Ethical and Protocol Review Committee of the College of Health Sciences, University of Ghana, with protocol identification number “CHS-Et/M.9–P 4.3/2015–2016.”
Results
Sociodemographic and clinical features of the study participants
The participants’ ages ranged from 16 to 82 years. Additionally, more than half of them were in the age group of 21 to 40 years (56.0%) and were females (62.0%) (Table 1).
Sociodemographic characteristics of the study participants.
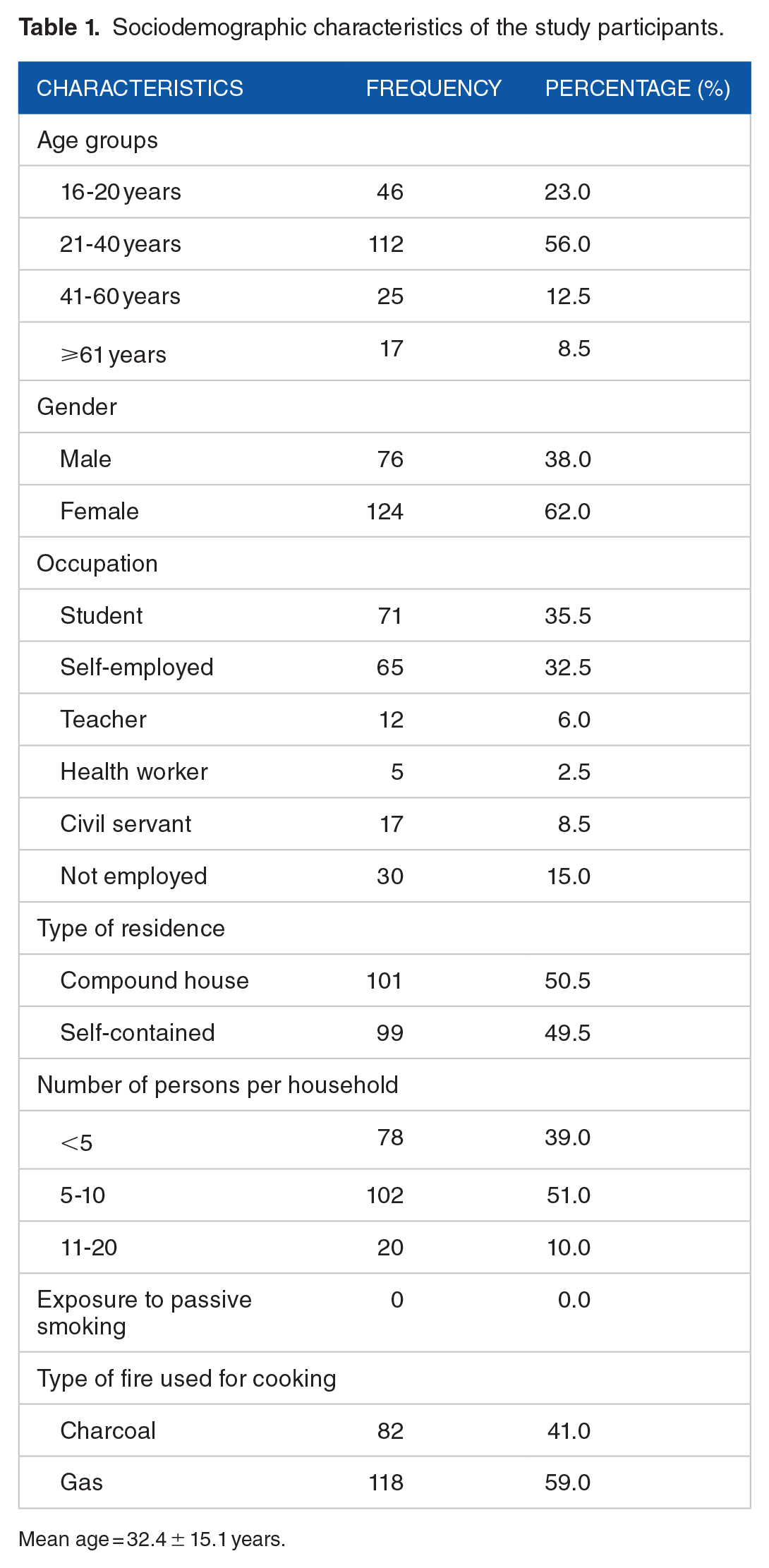
Mean age = 32.4 ± 15.1 years.
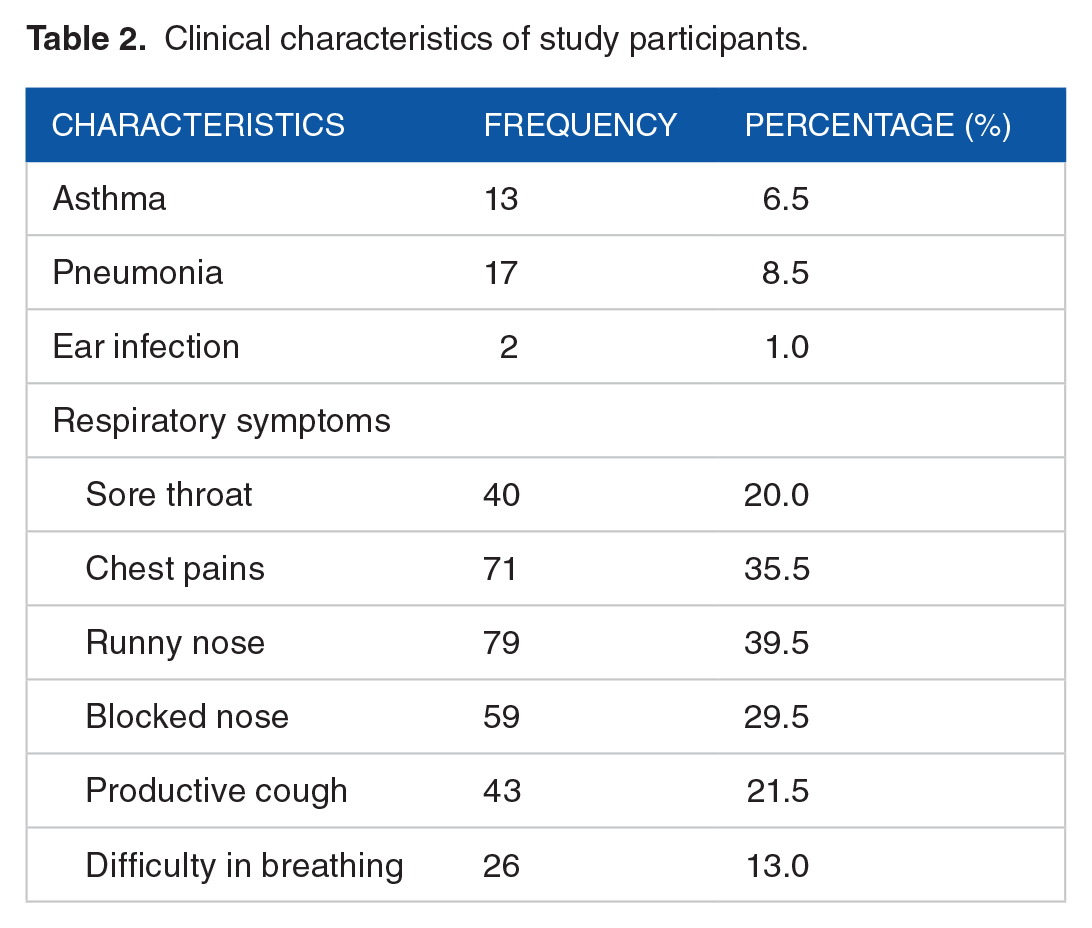
A few of the study participants had pneumonia (8.5%), asthma (6.5%), and ear infection (1.0%), and the highest proportion of respiratory symptoms was accounted for by runny nose (39.5%) (Table 2).
Clinical characteristics of study participants.
Prevalence and determinants of S. aureus nasopharyngeal carriage
The prevalence of S. aureus carriage was 41.5% (n = 83), and that of MRSA carriage was 1.0% (n = 2). The carriage prevalence of CoNS was 43% (n = 86). Moreover, carriage of CoNS was the only determinant of S. aureus carriage identified—it protected against S. aureus carriage (OR = 0.012, P < .0001). However, neither this variable nor the other features of the participants emerged as a determinant of MRSA carriage.
Antimicrobial resistance among the S. aureus isolates
The highest antimicrobial resistance rate was observed in penicillin (98.8%, n = 82), followed by tetracycline (54.2%, n = 45), gentamicin (32.5%, n = 27), ciprofloxacin (21.7%, n = 18), erythromycin (18.1%, n = 15), clindamycin (10.8%, n = 9), amoxicillin-clavulanic acid (10.8%, n = 9), teicoplanin (1.2%, n = 1), and linezolid (0.0%, n = 0), and the multidrug resistance rate, resistance to 3 or more antimicrobial classes, was 45.8% (n = 38).
Discussion
The study evaluated nasopharyngeal S. aureus carriage among adults with SCD in Ghana, and appears to be the first of its kind, somewhat mirroring its parallel investigation among SCD children. 15 It was observed that the prevalence of S. aureus carriage was high (41.5%) while that of MRSA carriage was low (1.0%). By comparison, both prevalence are lower than the 57.9% and 3.5% reported for S. aureus and MRSA, respectively, by Dayie et al. 15 It is noted that the children in that study had received PCV-13 vaccination, and hence had had significant Streptococcus pneumoniae clearance from their nasopharynx. 15 Therefore, it is not surprising that they had higher prevalence of S. aureus and MRSA in the nasopharynx than did the SCD participants of the current study. That notwithstanding, the S. aureus nasopharyngeal carriage prevalence observed in the current study could be considered high, at least, for 2 reasons. First, in Dayie et al’s 16 study that evaluated S. aureus nasopharyngeal carriage among children under the age of 5 who had also received PCV-13 vaccination, a 23.2% S. aureus prevalence was observed, and this is markedly lower than the prevalence observed in the current study. Second, the prevalence is higher than the prevalence reported in the anterior nares—the ecological niche of S. aureus–in the general population (21%) 33 and risk groups for S. aureus carriage, such as SCD children (33.3%) 26 and HIV-infected individuals (8%). 34 The prevalence is also comparable to that reported in the anterior nares by Donkor et al 35 (44.9%) involving another risk group for S. aureus carriage—HIV-infected children. Thus, although lacking a control group of non-SCD patients that could assist in estimating the risk of carriage attributable to SCD, the study seems to confirm SCD individuals as a risk group for S. aureus carriage, as noted by Appiah et al. 26 It is, however, difficult to attribute the high S. aureus nasopharyngeal carriage prevalence observed in this study to indirect effects of PCV vaccination, unlike has been done in previous studies,36,37 although the specimens investigated in this study were collected several years post-PCV introduction in Ghana. This is because besides the absence of concurrent anterior nasal sampling, data was not collected on whether or not the participants had close associations with vaccinated individuals. Future studies could include this angle of hypothesis in their research designs.
It was observed that colonization with CoNS protected against S. aureus colonization among the SCD participants. A similar finding was made by Dayie et al, 15 cited earlier, as did Kotey et al 38 and Donkor et al 35 among infants and HIV-infected children, respectively. Indeed, Olson et al 39 and Iwase et al 40 have reported previously that the coagulase-negative organism Staphylococcus epidermidis is antagonistic to S. aureus. This phenomenon has been attributed to the autoinducing peptide produced by CoNS, which obstructs agr quorum sensing in S. aureus. 41
In this study, a high resistance rate was recorded against penicillin, the highest in comparison with all the other antibiotics tested. This was predictable, given that in several studies conducted in the country previously, such high rates have been reported against the antibiotic.15,16,26,34,35,38 Besides, in Ghana, the antibiotic has a wide coverage, and it is routinely prescribed for SCD individuals as a prophylactic agent. In contrast, all the S. aureus isolates were susceptible to linezolid, an antibiotic whose use is primarily restricted to treatment of MRSA, and is rarely used in the country. Consequently, several preceding studies in the country have recorded low resistance rates against linezolid.15,35,38,42-44 This mutual observation of those studies and the current one suggests that the antibiotic can retain its usefulness in anti-MRSA therapy. Similar to the case of linezolid resistance, teicoplanin resistance was low, and hence could be retained as part of the spectrum of anti-S. aureus arsenals. Moreover, the high rates of resistance recorded against tetracycline, gentamicin, ciprofloxacin, erythromycin, clindamycin, and amoxicillin-clavulanic acid are similar to what have been reported in prior studies in the country.15,35,38,42,43 The multidrug resistance rate was high in this study as well, as has been the case for several studies conducted in the country.15,35,38,42,43 The antimicrobial resistance trend observed in the current study, besides being useful in the review of local treatment guidelines, adds to the several lines of evidence that support the need to fast-track the development of newer antimicrobials.
Conclusion
It is concluded that the nasopharyngeal carriage prevalence of S. aureus in the current study was high (41.5%), which may suggest a high risk of developing S. aureus infections among the participants; MRSA carriage was, however, low (1.0%). Moreover, the isolates were highly resistant to several of the antibiotics tested, while demonstrating minimal resistance against teicoplanin and linezolid. Thus, linezolid is still useful as an anti-MRSA agent, and teicoplanin could be retained in the spectrum of antibiotics for treatment of S. aureus infections. Furthermore, it is necessary to sustain S. aureus antimicrobial resistance surveillance efforts, particularly among risk populations, to continuously obtain contemporary data with which to update treatment guidelines.
This study is limited by the fact that the nasopharynx is not the principal carriage site of S. aureus, and hence, the S. aureus carriage prevalence reported may be an underestimation. Also, a control group of non-SCD patients was not recruited, and hence it is difficult to estimate the risk of carriage attributable to SCD.
Footnotes
Author Contributions
Conceptualization: NTKDD. Methodology: NTKDD, FCNK, DNKS, ADD, and ESD. Validation: NTKDD, FCNK, M-MO, PBT-Q, and ESD. Data curation: NTKDD, FCNK, and DNKS. Formal analysis: NTKDD, ADD, and FCNK. Investigation: NTKDD, DNKS, M-MO, PBT-Q, and ESD. Resources: NTKDD, M-MO, ADD, and ESD. Visualization: NTKDD, FCNK, and ADD. Software: FCNK, ADD and PBTQ. Supervision: NTKDD, ADD, PBT-Q, and ESD. Project administration: NTKDD, PBT-Q, and ESD. Writing—original draft preparation: NTKDD and FCNK. Writing—review and editing: NTKDD, FCNK, ADD, PBT-Q, DNKS, M-MO, and ESD. All authors have read and approved the final version of the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported with funds from the Office of Research, Innovation and Development (ORID), University of Ghana.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
