Abstract
Purpose:
The morbidity and mortality due to typhoid fever can be significantly reduced with the use of effective antibiotics. At present, fluoroquinolones, third generation cephalosporins, and azithromycin are widely used to treat typhoid fever. However, changing antibiotic susceptibility among Salmonella Typhi and Salmonella Paratyphi poses a particular challenge to the therapeutic management of enteric fever. The objective of this study was to assess the antibiotic susceptibility pattern of Salmonella Typhi isolates.
Patients and Methods:
A total of 706 blood specimens were collected from febrile patients attending the outpatient department of Kathmandu Model Hospital during June to September, 2018. The antibiotic susceptibility testing for 11 different antibiotics (nalidixic acid, ciprofloxacin, ofloxacin, levofloxacin, cefixime, ceftriaxone, cefotaxime, azithromycin, cotrimoxazole, chloramphenicol, and amoxicillin) was performed by disk diffusion method. Furthermore, minimum inhibitory concentration (MIC) values of ciprofloxacin, ofloxacin, and azithromycin were determined by agar dilution method. Mutation at gyrA ser83 associated with reduced susceptibility to fluoroquinolones was determined by PCR-RFLP.
Results:
Out of 706 blood samples, 6.94% (n = 49) were culture positive for Salmonella enterica (S. Typhi, n = 46). It was revealed that 97.8% S. Typhi isolates were susceptible to conventional first-line antibiotics (ampicillin, chloramphenicol, and cotrimoxazole), 97.3% to cephalosporins and 95.7% to azithromycin. S. Typhi were either resistant or intermediately susceptible to fluoroquinolones: 97.8% to ciprofloxacin, 91.3% to ofloxacin, and 89.1% to levofloxacin. The MIC of ciprofloxacin, ofloxacin, and azithromycin for S. Typhi ranged from 0.008 to 32, 0.03 to 16, and 2 to 8 μg/mL, respectively. Out of 46 S. Typhi isolates, 44 (95.65%) had gyrA ser83 mutation.
Conclusion:
Fluoroquinolones have poor activity against Salmonella Typhi. The trends of increasing azithromycin MIC value among S. Typhi might limit its use for the treatment of typhoid fever. Effectiveness of conventional first-line antibiotics in vitro suggests considering their clinical use after large-scale studies.
Keywords
Introduction
Salmonella Typhi, the causative agent of typhoid fever, is a human-restricted pathogen transmitted via fecal-oral route. Typhoid fever is a significant public health problem in low- and middle-income countries (LMICs). The burden of typhoid fever is higher among children and young adults in Southeast Asia.1-3 In 2017, approximately 14.3 million cases of enteric fever were observed globally, of which typhoid fever accounted for 76.3% of the cases with 0.95% case fatality rate (CFR). 4 Early diagnosis and timely treatment with an effective antibiotic can serve as important measures in reducing the morbidity and mortality associated with enteric fever. The CFR in untreated cases or treated with inappropriate antibiotics ranges from 10% to 20%, while among cases treated with effective antibiotics can be only 1% to 4%. 5
Nepal remains endemic to enteric fever. 6 In fact, S. Typhi and S. Paratyphi are the most common etiological agents in blood cultures from the patients with febrile illness in Nepal.7-9 Studies have shown Kathmandu valley to have sustained prevalence of typhoid fever over the years, mainly accounted for high population density and lack of wide access to clean water and sanitation. 6 Chloramphenicol, co-trimoxazole and ampicillin were conventional first-line antibiotics to treat typhoid fever in developing countries including Nepal. 10 However, following the emergence of classical-multi drug resistant (MDR) Salmonella (isolates resistant to conventional first-line antibiotics) in the late 1980s, fluoroquinolone (FQ) was recommended as alternative antibiotics.11,12 The widespread and indiscriminate use of FQs during the 1990s and 2000s led to the emergence of Salmonella isolates with reduced susceptibility to FQs. 13 Studies have shown that patients infected with S. Typhi having reduced susceptibility to FQs have longer fever clearance time and more frequent therapeutic failure.14-17
With the emergence of multi-drug resistant (MDR) Salmonella and an increase in resistance to FQs, broad-spectrum cephalosporins and azithromycin have become the mainstay antibiotics to treat typhoid fever in Nepal and other typhoid endemic regions.18,19 However, there have been sporadic reports of Salmonella Typhi resistant to azithromycin from Nepal 20 and different parts of the world including Bangladesh, 21 India,22,23 and Pakistan. 24 Similarly, cephalosporin resistance among Salmonella isolates has also been reported from different countries including India, 25 Pakistan, 24 and China. 26
The minimum inhibitory concentration (MIC) determination tests in most of the resource-limited diagnostic laboratories are not routinely performed as these tests are labor-intensive, time-consuming, and not cost-effective. We conducted a laboratory-based cross-sectional study of Salmonella Typhi isolates from the febrile patients attending a tertiary care hospital which is located at the center of Kathmandu valley and receives patients from all over the Kathmandu and other regions of Nepal. Antibiotic susceptibility patterns of Salmonella Typhi isolates to different antibiotics were determined by disk diffusion along with MIC determination of ciprofloxacin, ofloxacin, and azithromycin. Here, we report the antibiotic resistance profile of Salmonella Typhi in Kathmandu valley during 2018 which might assist future studies to analyze emerging resistance patterns among Salmonella in Nepal.
Materials and Methods
Study design and selection of participants
A laboratory-based cross-sectional study was conducted in blood specimens from clinically suspected enteric fever patients attending the outpatient department of Kathmandu Model Hospital during June to September, 2018. Patients with fever for >14 days, pregnant women, and patients already under antibiotic therapy were excluded from the study. Clinical signs and symptoms were examined by the physician. About 5 mL of blood specimen for patients of age >5 years and 3 mL for age ⩽5 years was collected for culture.
Blood culture, bacteria isolation, and identification
Blood specimens were collected in Brain Heart Infusion broth (BHI; HiMedia, India) in the blood to BHI ratio 1:10. Blood cultures were incubated at 37°C for 72 hours and subculture was performed in every 24 hours on Blood Agar, Xylose Lysine Deoxycholate Agar, and MacConkey Agar (HiMedia, India). Salmonella Typhi was identified based on colony morphology on culture media and biochemical tests. S. Typhi serotypes were confirmed by serotyping using polyvalent and monovalent anti-sera (Denka Seiken, Japan).
Antibiotic susceptibility testing
Antibiotic susceptibility tests were performed on Mueller-Hinton Agar following the modified Kirby-Bauer disk diffusion method. The antibiotics nalidixic acid (30 µg), ciprofloxacin (5 µg), ofloxacin (5 µg), levofloxacin (5 µg), ceftriaxone (30 µg), cefotaxime (30 µg), cefixime (5 µg), azithromycin (15 µg), co-trimoxazole (25 µg), chloramphenicol (30 µg), and amoxicillin (10 µg) (HiMedia, India) were tested. Clinical and Laboratory Standards Institutes (CLSI) guidelines 27 and Sjölund-Karlsson et al 16 (for levofloxacin and ofloxacin) were used as reference standards for the interpretation of the diameter of the zone of inhibition. ATCC 25922 Escherichia coli, ATCC 29213 Staphylococcus aureus, and ATCC 27853 Pseudomonas aeruginosa were used for quality control test of antibiotics.
Determination of minimum inhibitory concentration (MIC)
MIC of azithromycin, ciprofloxacin, and ofloxacin for Salmonella Typhi were determined by agar dilution method. The protocol was adapted from Andrews, 28 Wiegand et al, 29 and Clinical and Laboratory Standards Institute guideline. 30 Dilution range 0.25 to 128 µg/mL was used for azithromycin, 0.016 to 128 µg/mL for ciprofloxacin, and 0.004 to 128 µg/mL for ofloxacin. CLSI guideline 27 and Sjölund-Karlsson et al 16 were used as the reference standards for the interpretation of MIC values. ATCC 25922 E. coli and ATCC 29213 S. aureus were used for quality control tests.
PCR of gyrA gene and detection of ser83 mutation
DNA was extracted from S. Typhi using alkali lysis method as described by Hartas et al. 31 In brief, a loopful of the overnight culture of S. Typhi was suspended in 100 µL of 50 mM NaOH. The suspension was then kept in water bath at 97°C for 3 minutes followed by refrigeration at 4°C for 5 minutes. Then, 16 µL of 1 M Tris-HCl was added to the suspension and centrifuged at 8000 rpm for 2 minutes. Finally, the supernatant was collected and stored at −20°C until further use.
PCR amplification of the gyrA gene was performed using the primers set (Eurofins Genomics, India) described previously. 32 Reaction mixture of total volume 25 µL was prepared using 3 µL of template DNA, 0.5 µL each of forward and reverse primers, and 21 µL of PCR master-mix (QIAGEN, Germany). Amplification was done in Techne Prime thermal cycler with the following thermal conditions: initial heat activation at 95°C for 15 minutes, followed by 30 cycles at 94°C for 30 seconds, 60°C for 90 seconds, 72°C for 60 seconds, and final extension at 60°C for 10 minutes.
The PCR-product 630 bp gyrA was subjected to restriction digestion by HinfI enzyme (Promega Corp., USA). Fifteen µl of the amplified product was digested using 2 µL bovine serum albumin (BSA), 2 µL of reaction buffer, and 10 units of HinfI for 1 hour at 37°C. The digested product was resolved by 2% (w/v) agarose gel at 120 volts for 45 minutes.
Data analysis
Data management and statistical analyses were performed using IBM SPSS for Windows Version 25.0. Demographic characteristics were presented as mean, median, or frequency distribution as applicable. The P-value of <.05 was considered as statistically significant in bivariate analysis. Rate of categorical agreement was used to analyze the discrepancy between agar dilution and disk diffusion tests for ciprofloxacin, ofloxacin, and azithromycin. Agar dilution was used as the reference antibiotic susceptibility test method over disk diffusion and the following errors were evaluated: minor error (mE; intermediate result in agar dilution but susceptible or resistant in disk diffusion), major error (ME; false-resistant by disk diffusion), and very major error (VME; false-susceptible by disk diffusion). Scatterplots of MIC versus zone diameter were prepared in OriginLab 2018.
Ethics statement
The study protocol was reviewed and approved by Institutional Review Committee of phect-Nepal (Kathmandu Model Hospital). Written informed consent was obtained from the adult (⩾18 years) participants. For participants under the age of 18 years, written informed consent was obtained from their parents/guardians.
Results
Demographic and clinical data
Out of 706 blood specimens, 6.94% (n = 49) were culture positive for Salmonella enterica. Among 49 enteric fever cases, Salmonella Typhi accounted for 93.87% (n = 46) cases and Salmonella Paratyphi accounted for remaining 3 cases. The median age of culture-positive typhoid fever patients was 22 years (range 1-30 years) (Table 1). Majority of S. Typhi isolates were from the patients 11 to 30 years of age (91.30%, P-value <.05). Even though about 45% of the total blood specimens collected was from the patients of age ⩾31 years, none of the patients in this age group were positive for typhoid fever.
Distribution of typhoid fever cases by patient’s age.
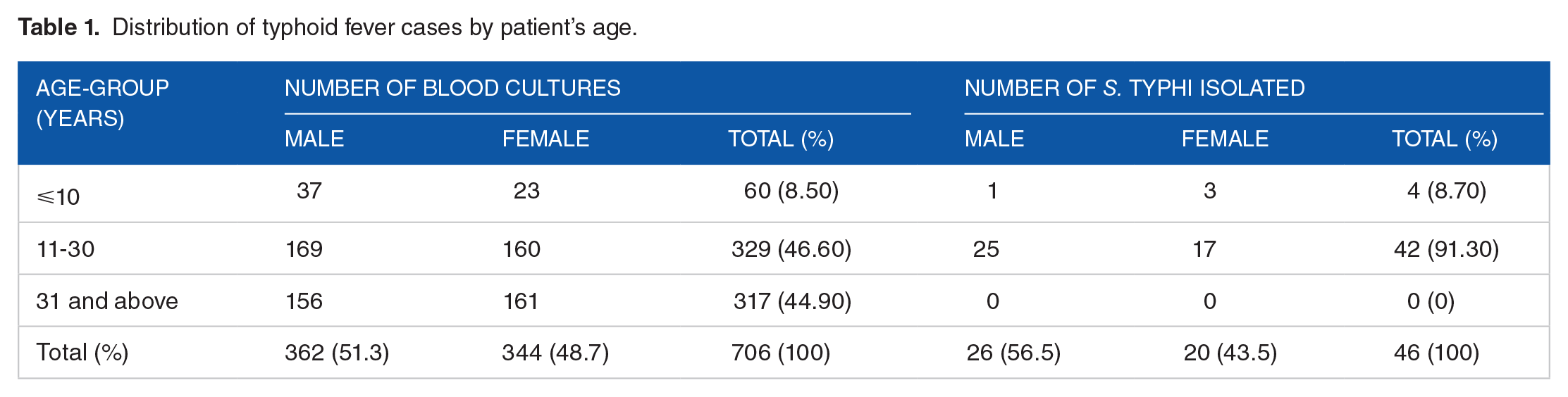
The median duration of fever before the hospital visit was 3 days (inter-quartile range: 2-5 days). Headache, chills, rigor, and evening rise of fever were among the most common clinical presentations of typhoid fever patients (Supplemental Material 1).
Antibiotic susceptibility pattern of Salmonella Typhi
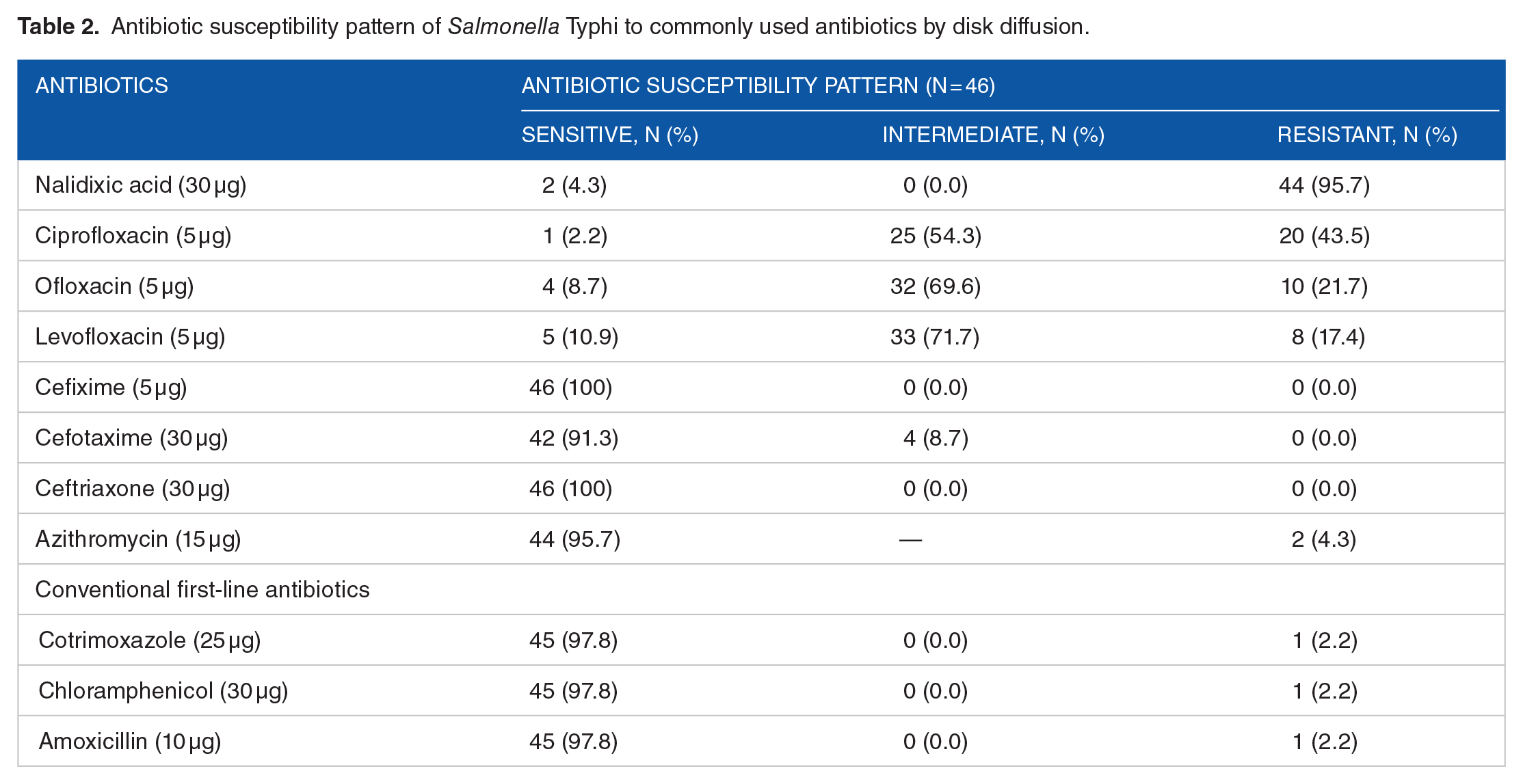
Among the 46 Salmonella Typhi isolates tested, 97.8% and 95.7% were susceptible to conventional first-line antibiotics (cotrimoxazole, chloramphenicol, and amoxicillin) and azithromycin, respectively. All the isolates were susceptible to cefixime, ceftriaxone, and cefotaxime. Among them, 4 S. Typhi had intermediate susceptibility to cefotaxime. About 95.7% isolates were nalidixic acid resistant (NAR). Only one of the isolates was susceptible to all 3 FQs (ciprofloxacin, ofloxacin, and levofloxacin). Most of the isolates had intermediate susceptibility to FQs: 54.3% to ciprofloxacin, 69.6% to ofloxacin, and 71.7% to levofloxacin (Table 2).
Antibiotic susceptibility pattern of Salmonella Typhi to commonly used antibiotics by disk diffusion.
Minimum inhibitory concentration (MIC)
MIC of ciprofloxacin, ofloxacin, and azithromycin against S. Typhi isolates were within the range of 0.008 to 32, 0.03 to 16, and 2 to 8 µg/mL, respectively (Table 3). Out of 46 isolates, 78.3% (n = 36) and 76% (n = 35) had intermediately susceptible MIC of ciprofloxacin and ofloxacin, respectively. Furthermore, 19.6% (n = 9) S. Typhi had resistant MIC of ciprofloxacin and ofloxacin. Out of 2 NAS isolates, 1 had susceptible ciprofloxacin MIC and both had susceptible ofloxacin MIC. Although 2 of the isolates were resistant to azithromycin by disk diffusion, none of the isolates exhibited resistant azithromycin MIC.
MIC value of ciprofloxacin (CIP), ofloxacin (OF), and azithromycin (AZM) against Salmonella Typhi.
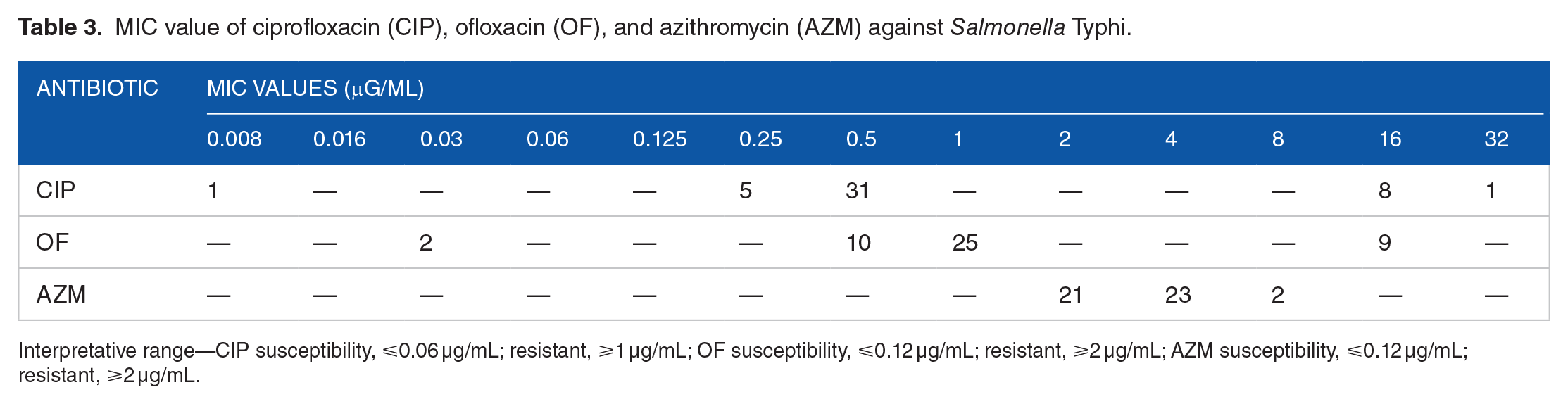
Interpretative range—CIP susceptibility, ⩽0.06 µg/mL; resistant, ⩾1 µg/mL; OF susceptibility, ⩽0.12 µg/mL; resistant, ⩾2 µg/mL; AZM susceptibility, ⩽0.12 µg/mL; resistant, ⩾2 µg/mL.
Comparison of agar dilution and disk diffusion
The categorical agreement rates of 76.09%, 93.48%, and 95.65% were observed between agar dilution and disk diffusion susceptibility interpretation of ciprofloxacin, ofloxacin, and azithromycin, respectively (Figure 1). Considering agar dilution as a reference test method, a minor error (mE) rate of 23.91% and 6.52% was observed for ciprofloxacin and ofloxacin disk diffusion, respectively. Meanwhile, azithromycin disk diffusion had a very major error (VME) rate of 4.35%. No major errors (ME) were found in the disk diffusion test of any of the 3 antibiotics.
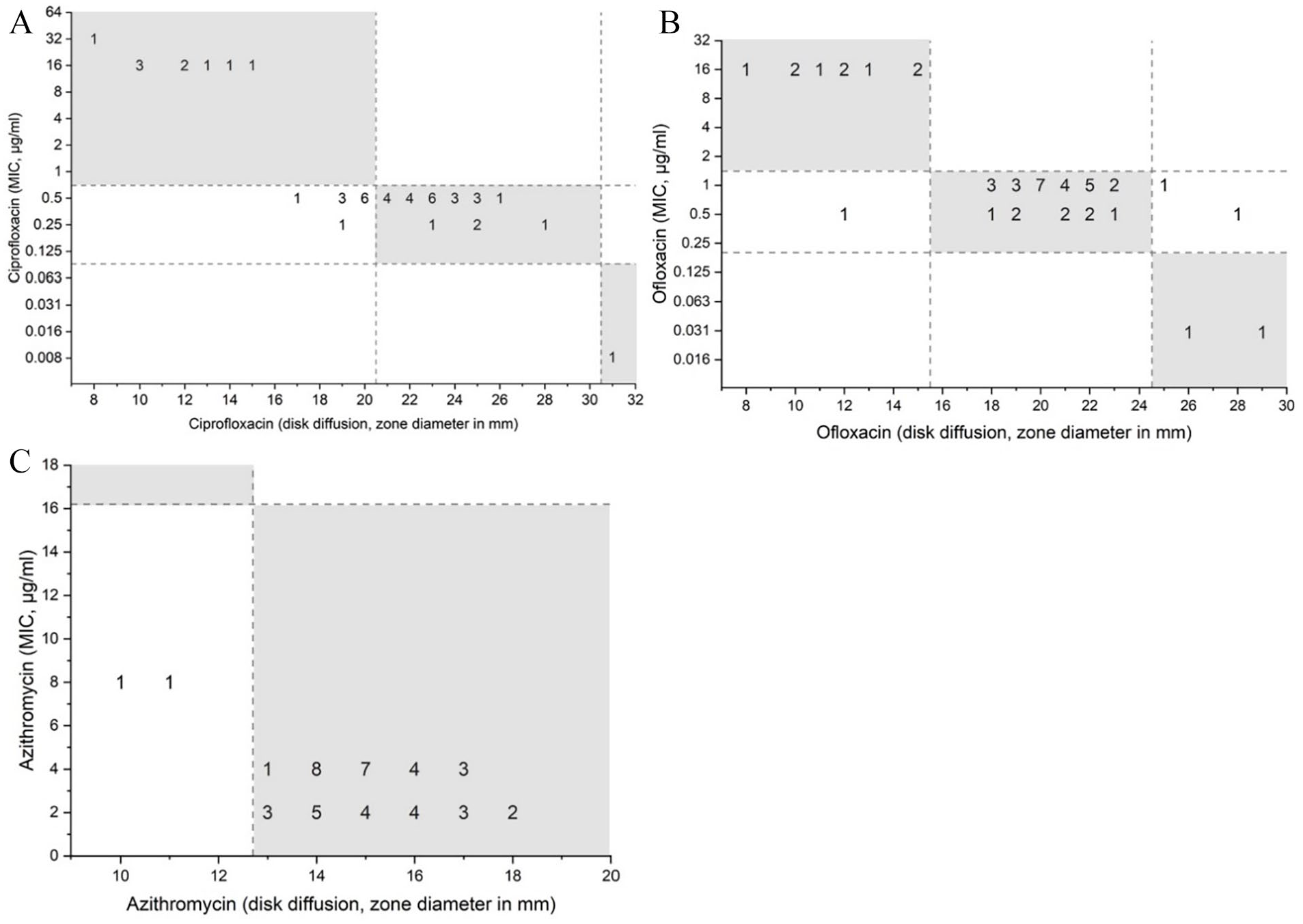
Scatterplot of MIC versus zone inhibition diameter of (A) ciprofloxacin, (B) ofloxacin, (C) azithromycin. Ciprofloxacin susceptible MIC: ⩽0.06 µg/mL, diameter: ⩾31 mm; ofloxacin susceptible MIC: ⩽0.12 µg/mL, diameter: ⩾25 mm; azithromycin susceptible MIC: ⩽13 µg/mL, diameter: ⩾25 mm). Plotted numbers represent the number of Salmonella Typhi isolates.
Ser83 mutation in gyrA gene
Two different restriction patterns were revealed on HinfI restriction digestion of gyrA PCR products: pattern A (wildtype) and B (gyrA ser83 mutated). Three intact bands of fragmented gyrA were visible for wildtype strains and 2 intact bands were visible for mutated strains (Figure 2). Two out of 46 S. Typhi isolates (4.34%) which were also nalidixic acid-susceptible (NAS) showed “pattern A” (Table 4, Figure 2). Remaining 44 (95.66%) isolates showed “pattern B” regardless of wide range of different fluoroquinolones MIC (Table 4; Figure 2).
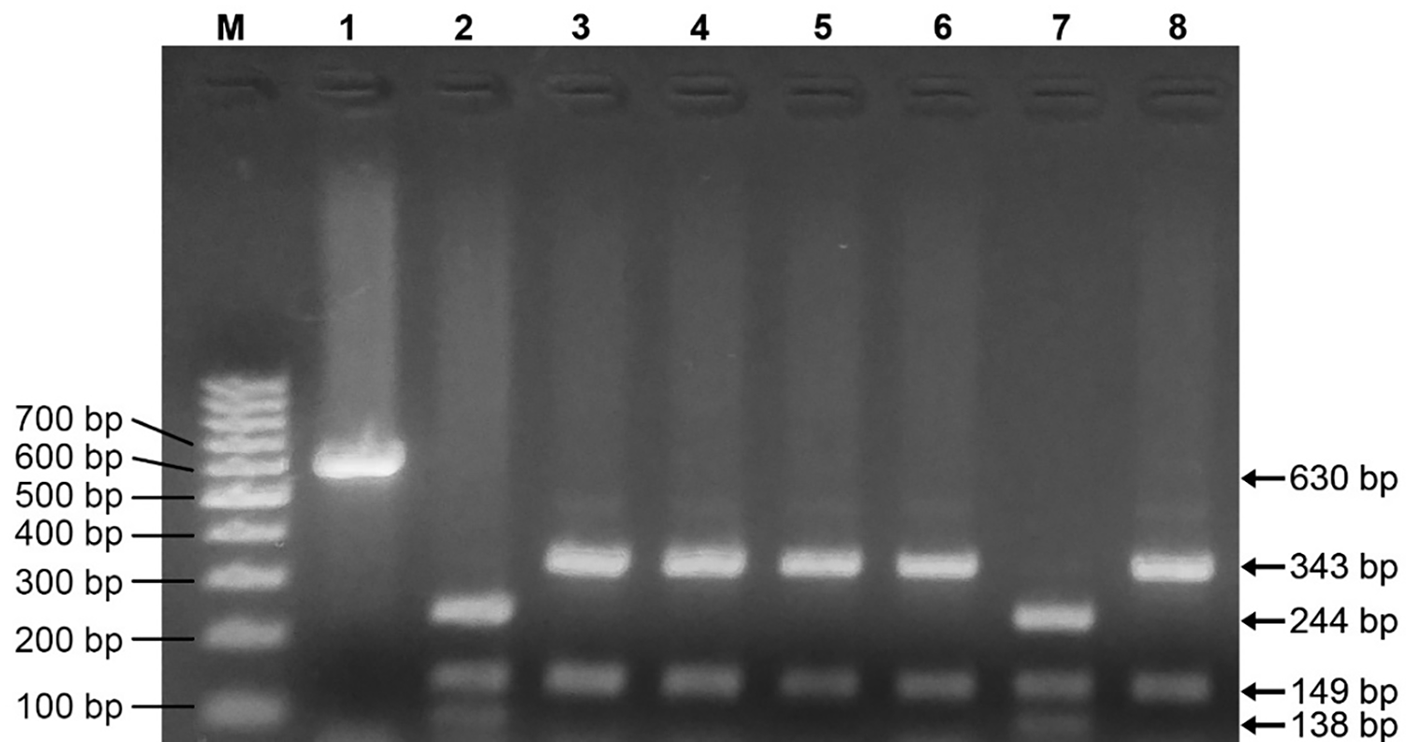
HinfI digested gyrA fragments. (Lane M: 100 bp molecular weight DNA ladder; Lane 1: PCR amplified 630 bp gyrA; Lanes 3-6 and 8-11: HinfI digested gyrA revealing 2 fragments [343 and 149 bp], ie, pattern A; Lanes 2 and 7: HinfI digested gyrA revealing 3 fragments [244, 149, and 138 bp] ie, pattern B. The gel image was cropped from the original image file for conciseness).
Distribution of gyrA HinfI restriction pattern based on fluoroquinolone resistance pattern.
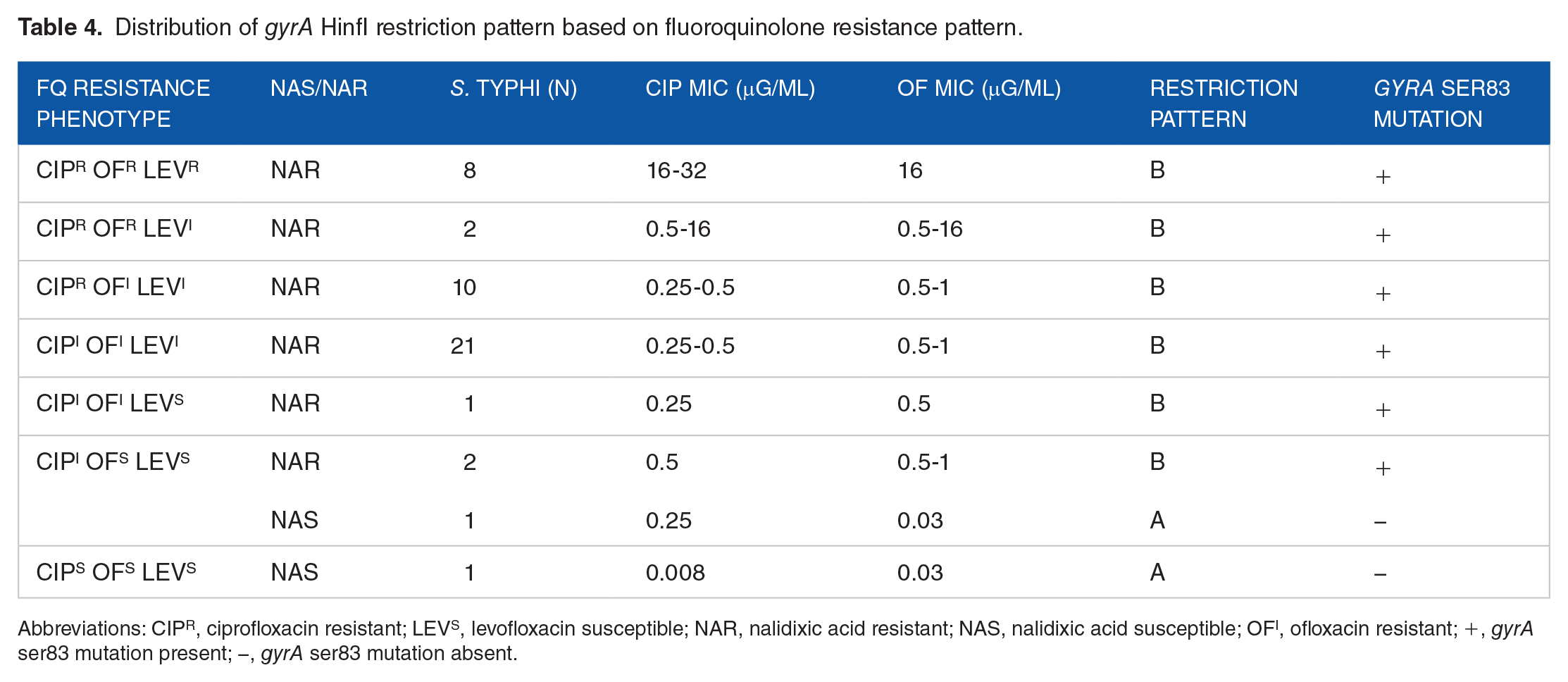
Abbreviations: CIPR, ciprofloxacin resistant; LEVS, levofloxacin susceptible; NAR, nalidixic acid resistant; NAS, nalidixic acid susceptible; OFI, ofloxacin resistant; +, gyrA ser83 mutation present; −, gyrA ser83 mutation absent.
Discussion
This study shows that the majority of S. Typhi isolates in Kathmandu, Nepal, have reduced susceptibility to second- and third-generation fluoroquinolones (FQs). The mutation at ser83 of gyrA was most frequently associated with these cases of reduced susceptibility to FQs. Additionally, our study found increasing levels of azithromycin MIC among S. Typhi isolates.
In comparison to our study, a relatively higher rate (6.94% vs 13.38%) of culture positive enteric fever cases was reported back in 2012 from the same hospital. 33 A 23-year (1992-2014) retrospective study from a different tertiary hospital of Kathmandu Valley has reported an overall decline in the absolute number of enteric fever cases over time. 6 The detected S. Typhi isolates were in the normal range in this setting and there was no any outbreak at this time. The decline in typhoid fever incidence can be attributed to increased access to safe drinking water, sanitation facilities, access to health facilities, and improved food handling practices. 34
In this study, it was found that majority of typhoid fever cases accounted to the age-group of 11 to 30 years as revealed by various other studies from Nepal.35-38 Even though more than half of the blood cultures were obtained from patients >30 years of age, S. Typhi was not found to be culture positive from this age-group. Persons of age group 15 to 30 are often engaged in outdoor activities and are more inclined to consumption of foods from street or local vendors which can significantly contribute to higher incidence of typhoid fever. Poor hygiene practice in preparation and handling of foods is common in Kathmandu valley. Persons consuming such prepared street foods are at high risk of enteric fever. 39 Similarly, the lack of safe drinking water is more prominent in public spaces and among local food vendors. In fact, study has shown that infection with S. Typhi than with S. Paratyphi is more likely associated with unsafe drinking water. 39
Fluoroquinolones (FQ) were the antibiotics of choice following the emergence of classical MDR S. Typhi, but indiscriminate use of FQs to treat typhoid fever has led to the emergence of S. Typhi isolates with reduced susceptibility to FQs. 13 Majority of the isolates in our study had either reduced susceptibility to FQs or were resistant to these FQs as revealed by both disk diffusion and MIC determination. The third generation FQ (levofloxacin) was comparatively effective than the second generation FQs (ciprofloxacin and ofloxacin) in vitro. Due to the structural difference in the carbon chain, levofloxacin (C-8-methoxy FQ) has enhanced activity against both topoisomerase IV and DNA gyrase, while ciprofloxacin and ofloxacin (C-8-H FQs) are effective against DNA gyrase only. Thus, third-generation FQs are less inhibited by common mutations of gyrA than are second-generation FQs. 40 Patients infected with S. Typhi with intermediate susceptibility to FQs may respond to increased dose or longer duration of treatment with the newer generations of FQs. 41 Reduced susceptibility to FQs is often due to the single point mutation at either the ser83 or asp87 position of gyrA 42 with gyrA ser83 being the most common mutation.32,43 All of the S. Typhi isolates with either reduced susceptibility or complete resistance to FQs in our study were found to harbor the gyrA ser83 mutation. However, 2 or more mutations in the quinolone resistance determining region (QRDR) of genes encoding gyrase or topoisomerase may be required for an isolate to show complete resistance to FQs. 42 Thus, isolates of our study with high-level resistance to FQs may have also harbored additional mutations at either of gyrA, gyrB, parC, or parE as well as plasmid encoded qnr genes besides the gyrA ser83 mutation. A study from Kathmandu valley has reported the introduction of a “triple mutant” phenotype of S. Typhi, which was found to be associated with longer fever clearance times and treatment failure with FQ. 44 Since we were unable to sequence the S. Typhi genome to assess other possible mutations, our study cannot provide a complete picture for genetic basis of FQ resistance in these S. Typhi isolates.
Majority of isolates in our study were susceptible to azithromycin by both disk diffusion and MIC determination tests. Isolates that were resistant to azithromycin by disk diffusion had elevated MIC but were within the range of susceptibility. The overall range of azithromycin MIC observed in our study (2-8 µg/mL) is more than double the range reported by the previous study (0.125-2 µg/mL) from Nepal. 45 This shows a worrying situation of increasing azithromycin MIC in recent years. As FQs have been rendered almost ineffective to treat typhoid fever, azithromycin is increasingly used, which might explain the increase of azithromycin MIC among S. Typhi isolates. Nevertheless, all isolates in our study still had azithromycin MIC within the susceptible range and it could be an option to treat typhoid fever.
A lower proportion of classical MDR S. Typhi (resistant to ampicillin, chloramphenicol, and co-trimoxazole) isolates as observed in this study is a common finding in recent years as reported by various studies from Nepal.6,37,45 The decline in incidence of classical MDR S. Typhi can be accounted to discontinuation in the use of conventional first-line antibiotics.6,46 The reemergence of Salmonella isolates susceptible to conventional first-line antibiotics has opened discussion about reintroduction of these antibiotics for the clinical management of enteric fever.33,45,47 However, the reintroduction and use of conventional first-line antibiotics must follow rational practice to avoid immediate reversal of Salmonella isolates with the classical MDR phenotype.
All Salmonella Typhi isolates in this study were found to be susceptible to cephalosporins: cefixime, cefotaxime, and ceftriaxone. Although the efficacy of ceftriaxone and cefotaxime are comparable, notably 4 of the isolates in our study were only intermediately susceptible to cefotaxime but were susceptible to ceftriaxone. A randomized control trial from Nepal has shown poor performance of cefixime in vivo even when the isolates were susceptible in vitro. 48 Thus, cefixime may not be a better drug of choice even when full susceptibility is observed in vitro. In this study, no resistance to cephalosporins were detected, which suggests that at least parenteral cephalosporins can be considered as effective therapeutic alternatives to treat complicated typhoid fever caused by MDR S. Typhi. Broad-spectrum cephalosporins are currently in wide use to treat various bacterial infections. Hence, the possible acquisition of extended-spectrum beta-lactamase (ESBL) genes by S. Typhi is a looming threat to the treatment of enteric fever as suggested by an outbreak in Pakistan associated with extensively drug resistant S. Typhi (ceftriaxone-resistant). 24 Similar reports of S. Typhi harboring β-lactamase genes have been reported from India (blaCTA-15, blaCMY-2)49,50 and Philippines (blaSHV-12), 51 as well. Therefore, we suggest that the use of cephalosporins to manage typhoid fever cases should be limited to only multi-drug resistant and complicated cases, and must be followed by antibiotic susceptibility tests so that cephalosporins will remain effective for a longer period of time.
Conclusion
Our study has reaffirmed that Salmonella Typhi with reduced susceptibility to fluoroquinolones is still a prevalent phenotype in Kathmandu, Nepal. Thus, the empirical use of fluoroquinolones to manage typhoid fever should be avoided. Even the use of newer generations of fluoroquinolones such as levofloxacin must follow the antibiotic susceptibility test results. The increase in azithromycin MIC among S. Typhi isolates in recent years as evident from our study indicates a possible widespread emergence of azithromycin resistant Salmonella isolates in the near future. As the proportion of classical MDR S. Typhi has continued to decline, reintroduction of conventional first-line antibiotics should be considered to broaden the array of effective antibiotics available for the treatment of typhoid fever.
Supplemental Material
sj-docx-1-mbi-10.1177_11786361211056350 – Supplemental material for Antimicrobial Resistance in Salmonella Typhi Isolated From a Referral Hospital of Kathmandu, Nepal
Supplemental material, sj-docx-1-mbi-10.1177_11786361211056350 for Antimicrobial Resistance in Salmonella Typhi Isolated From a Referral Hospital of Kathmandu, Nepal by Saroj Khadka, Basudha Shrestha, Anil Pokhrel, Sachin Khadka, Rajesh Dhoj Joshi and Megha Raj Banjara in Microbiology Insights
Footnotes
Acknowledgements
The authors would like to express their gratitude toward all study participants. The authors are also thankful to Mr. Anil Kunwar and Ms. Anupama Gurung for their support during laboratory work.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors Contributions
SK, BS and MRB designed this study; SK performed the experiments, AP supported the laboratory work; MRB and BS supervised the study. SK and MRB analyzed the data; SK, MRB and BS wrote the original draft. SK, RDJ examined the patients. SK, BS, AP, SK, RDJ and MRB reviewed the draft manuscript several times. All authors read and approved the final manuscript.
Disclosure
The abstract of this paper was presented at the ASM Microbe Online 2020 as a virtual poster presentation with interim findings.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
