Abstract
Neutrophils move from the blood into tissues, migrate under the guidance of chemical gradients, and accumulate at sites of infection, where they phagocytose and kill microbes within minutes. These processes have been considered the keystone of innate immune responses for more than five decades. However, it appears that these processes accurately describe only the situation when neutrophils encounter small numbers of disparate microbes. Recent observations revealed that when neutrophils encounter large fungi or clusters of bacteria that are too large for individual neutrophils to kill, one more process, known as swarming, is needed. During this process, the first neutrophils that reach the microbial target release leukotrienes and other molecules that stimulate more neutrophils in the vicinity to converge on the same target. A chain reaction ensues between the leukotrienes guiding the neutrophils toward the swarm and the attracted neutrophils releasing more leukotrienes. This chain reaction empowers multiple neutrophils to coordinate their activities, drives the explosively-fast accumulation of neutrophils, and enables neutrophils to neutralize large microbes and clusters of microbes, which would otherwise be outside the neutrophils reach. The molecular and cellular details of this chain reaction and the mechanisms that stop the chain reaction from damaging healthy tissues are just beginning to emerge, enabled by innovative, engineered tools.
Keywords
Neutrophils are critical immune cells that mount fast and powerful responses against microbes entering tissues. Neutrophils can respond to chemicals released from microbes and migrate directionally following chemical gradients towards these chemicals’ source. Neutrophils can also phagocytose the microbes and have a potent arsenal of reactive chemicals and enzymes to kill these microbes. The transmigration, directional migration, and phagocytosis are regarded as crucial processes of neutrophil anti-microbial responses. Today, these three processes are depicted in cartoons in every immunology book. Plenty of references to cell migration and phagocytosis mechanisms accompany these figures that are familiar to every medical student and immunologist. However, the traditional depiction of these processes is accurate only when small microbes, like bacteria, are present in a small number. A recently uncovered process, known as neutrophil swarming, is essential in situations when neutrophils encounter clusters of bacteria and elongated fungi. Individual neutrophils cannot handle these large microbial targets. Swarming is the result of communication and activation of large number of neutrophils against these targets. The transition from phagocytosis to swarming can be captured in quantitative detail in vitro using novel microfluidic assays and imaging at single-cell resolution. 1 These new findings expand our understanding of how neutrophils accumulate at sites of infections.
Neutrophil swarming provides a significant boost to the accumulation of neutrophils at sites of injury. 2 Swarming is essential for engulfing microbes and clusters of microbes that are too large for individual neutrophils to kill. 1 Swarming is a key process for the activation of the acquired immunity after vaccines. 3 The swarming process also seals off tissue damage sites during infections, mechanical or thermal wounds.2,4 Significant advances in the in vivo imaging technologies captured the swarming dynamics in tissues.2,5-7 Moreover, new animal models, like transparent zebrafish, are becoming essential tools for studying swarming in the complex environment of tissues. 4 For human neutrophils, new tools for ex vivo studies are essential. These tools could reveal altered swarming abilities for neutrophils from patients recovering after trauma, chronic liver failure, and chronic granulomatous disease.1,8 Together, the in vivo models and ex vivo assays are essential toward understanding the contribution of neutrophil swarming to human health and disease. In addition to the identification of key signaling molecules during swarming, at the mechanistic level, a positive feedback loop, also known as a chain reaction, gives swarming several characteristics that are unique among all the processes of innate immunity.
Chain reactions are self-sustaining processes in which the output increases the input (positive feedback loop). Once started, chain reactions continue autonomously and progress with exponentially increasing intensity. Chain reactions play essential roles in chemical, physical, social, and physiological processes that, once started, run to completion. A pile of dry wood burning is an example of a chemical chain reaction. Each piece of burning wood releases enough heat to kindle the surrounding pieces. These, in turn, ignite more wood that burns until all wood is turned into ashes. The release of neutrons after the collision of neutrons with uranium atoms is an example of a physical chain reaction. Each neutron released from the fission of a heavy uranium atom can release three more neutrons after the collision of other uranium atoms. The process repeats again and again, with an increasingly larger number of neutrons after each atomic fission. A viral video spreading in a social network through multiple sharing and re-distributions from each participant are examples of social chain reactions. Similarly, alarm or panic can spread by positive feedback among a herd of animals to cause a stampede that deepens the panic. When enough people panic about their bank deposits, their actions could create real liquidity shortages, and scenes of angry crowds around banks may, in turn, increase the panic in a classic case of bank run. The neuro-endocrine reaction during childbirth is an example of a physiologic chain reaction. When the head of the fetus pushes up against the cervix, it stimulates a nerve impulse from the cervix to the brain, which signals the pituitary gland to release a hormone called Oxytocin. Oxytocin is then carried via the bloodstream to the uterus causing more contractions and pushing the fetus toward the cervix, eventually inducing childbirth. Overall, these examples demonstrate the tremendous power of chain reactions to trigger massive changes in a short amount of time.
For the swarming neutrophils, the chain reaction is sustained by molecules that are both produced by the neutrophils and for which neutrophils possess receptors on their surface. Upon encountering a large target, neutrophils release large amounts of leukotriene B4 (LTB4) that diffuse in the vicinity of the target and generate attractant gradients centered on the target (Figure 1). These gradients serve as a guide for neutrophils that join the swarm, produce more LTB4, strengthening the gradient and triggering the attraction of more neutrophils. LTB4 is both the input—directing neutrophils toward the target—and the output—being release by neutrophils on the target, fitting all the elements of a chain reaction. The most substantial evidence for a positive feedback loop during swarming so far is for leukotriene B4 (LTB4). The results of swarming experiments using neutrophils that lack the high-affinity receptor for LTB4, 2 antagonists of the BLT1 and two receptors for LTB4, 8 and chemical inhibitors of LTB4 synthesis, 1 all converge to support the essential role of LTB4 in the chain reaction during swarming. The chain reaction assures robust neutrophil responses that are impervious to external perturbations.
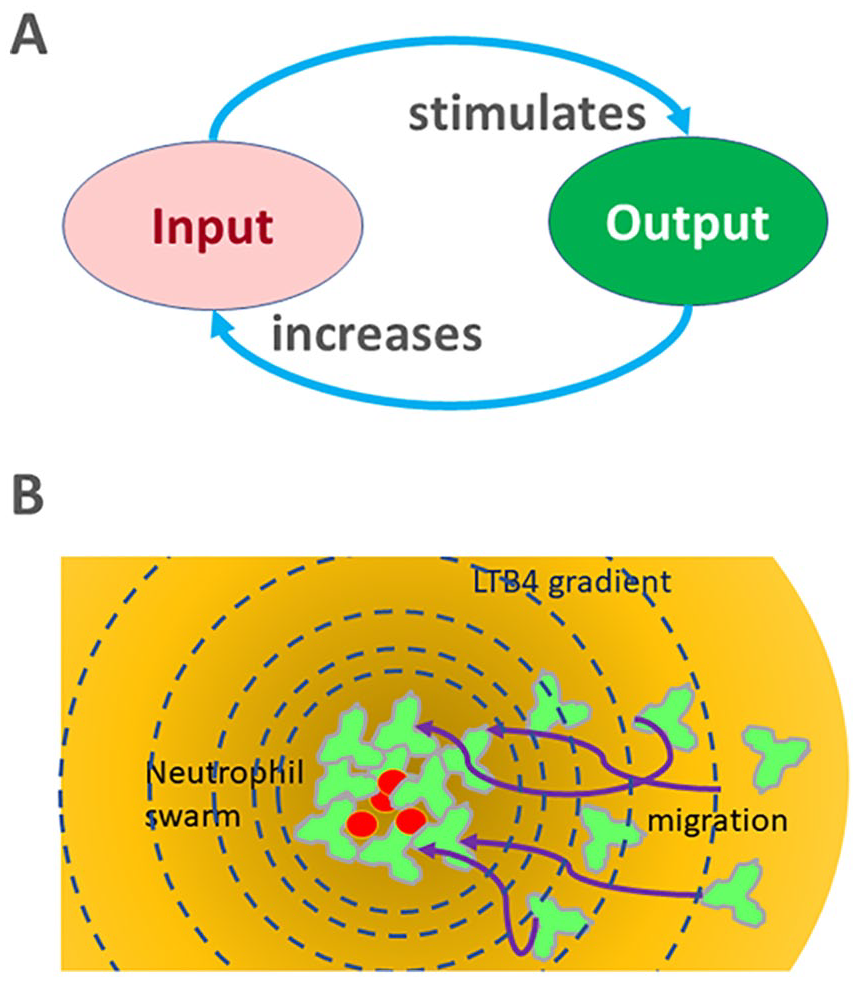
Chain reactions: (A) chain reactions are series of amplification loops in which the output increases the input which in turn stimulates the output, and (B) swarming is a chain reaction between the neutrophils joining the swarm, which release LTB4 and attract more neutrophils toward the swarm. The chain reaction accelerates the neutrophil convergence on the microbe target and makes it impervious from perturbation by the microbes.
One of the most intriguing discoveries related to the chain reaction during neutrophil swarming is that LTB4 is not the only signaling molecule involved. Blocking the LTB4 chain reaction disrupts swarming but does not stop it completely.2,8 Blocking the LTB4 receptors reveals several other molecules are contributing to swarming. A hierarchical system that includes IL8, complement factors, and maybe other signaling molecules emerge from in vivo and in vitro studies.2,8,9 Shorter range communication between swarming neutrophils has also been recently suggested, involving Ca2+, ATP, connexin 43, 4 and integrin receptors. 10 A constellation of mediators are also being released from the neutrophil swarm, potentially to coordinate activities between neutrophils, monocytes, lymphocytes, vascular, and tissue cells. 8 The significance of this robust system has yet to be elucidated. So far, the range of mediators appears to contribute to the effectiveness of the swarming process in various situations. Moreover, when a seal is formed around microbes, enzymes and reactive oxygen released inside the sealed compartment could reach higher concentrations than when these could diffuse in the tissue. Swarming also seals off healthy tissues from the release of both reactive chemicals and enzymes as well as microbe-released toxins. Most recently, it appears that swarming and the release of NETs inside the swarms provides a mechanical barrier for growing microbes and seals off from healthy tissue by layers of neutrophils. 1
One feature of chain-reactions is that their course is terminated only when all the resources are exhausted. The fire in a pile of wood ends when all the wood in a pile is consumed; a nuclear reaction ends when all uranium atoms are split; a viral video ends (being viral) when everyone on social media has seen it. Similarly, the end of the chain-reaction accumulation of neutrophils accumulation to a microbial infection site would happen when all neutrophils from the body become part of the swarm, and there are no responsive neutrophils left in the body. For a small papercut that gets infected, triggering a neutrophil swarming reaction would mean that all the 25 billion neutrophils would accumulate at that site, for a ~125 mL total volume of pus. Obviously, this is not what happens in vivo. Instead, experiments in mice have shown that swarming stops after approximately 2 hours and that only a fraction of the neutrophils in the body contributes to swarming.2,7 Most neutrophils remain in the circulation and are available to respond when a second infection takes place. Human neutrophils in vivo accumulate to a swarm for approximately 2 hours before the size of the swarm reaches a plateau, even when plenty of neutrophils are available around the swarm. 8 The natural evolution of neutrophil swarms suggests that additional mechanisms tempering the chain reaction are active. These mechanisms may be akin to the control of fire in modern furnaces and the control of nuclear fission in nuclear power plants that assure that the chain reactions deliver consistent, predictable results.
In vitro experiments using synchronized swarms enabled the collection and analysis of supernatants and revealed the identity of stop signals for neutrophil swarming. 8 These studies identified lipoxin A4 (LXA4) and resolvin E3 (RvE3) as the signals that stop the swarming. Not only LXA4 is released with a maximum at approximately 3 hours after the initiation of swarming, but pretreating the neutrophils with LXA4 reduced the size of the swarms. 8 Interestingly, no protein mediators to inhibit neutrophil swarming were identified in this study. 8 Moreover, both the initiation and plateau phases of swarming are controlled by lipid mediators, which share arachidonic acid as a common precursor. Also surprising, the synchronized-swarms experiments revealed that the neutrophils in swarms release a constellation of mediators that are known activators of lymphocytes, monocytes, endothelial cells, fibroblasts, and adipocytes. These findings place neutrophil swarming at the epicenter of inflammatory and restorative processes in health and disease.
Overall, neutrophil swarming is a robust emergent behavior, which relies heavily on the communication between cells. The chain reaction between neutrophils triggered by the large microbial target assures robust and accelerated accumulation of the neutrophils on the target. The chain reaction also makes swarming less vulnerable to various toxins that microbes possess, many interfering with neutrophil activities. Understating the key processes of neutrophil swarming requires new tools for probing inter-cellular communication. In combination with the current molecular biology tools, new treatments are coming to light that enhance neutrophils’ ability to contain microbes during infections. 11 These treatments may also protect healthy tissues from damage after infections and inflammation and trigger long-term anti-microbial protection in cooperation with the acquired immunity.
Footnotes
Declaration of conflicting interests:
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institute of General Medical Sciences (GM092804) and the Shriners Hospitals for Children.
