Abstract
Objective:
The aim of this study is to assess whether choice of test for tuberculosis (TB) infection affects decisions to accept and complete treatment among contacts to TB cases.
Methods:
Retrospective study is conducted in which TB contacts, ⩾15 years old during 2005 and 2009, were tested for infection with either a tuberculin skin test (TST) or an interferon-gamma release assay test, the QuantiFERON-TB Gold In-Tube (QFT-GIT).
Results:
Of 658 persons with valid test results, 185 (28%) had positive results, including 128 of 406 (32%) who had TST and 57 of 252 (23%) who received QFT-GIT. Treatment acceptance was 43 of 57 (75%) among QFT-GIT-positive and 97 of 128 (76%) among TST-positive persons (risk ratio [RR] = 1.0, 95% confidence interval [CI], 0.83-1.2). Treatment completion was 56% among QFT-GIT-positive (32 of 57) and 59% (75 of 128) among TST-positive persons (RR = 0.96, 95% CI, 0.73-1.26).
Discussion:
Our study showed no difference in proportions of TB contacts ⩾15 years old with positive TST results who accepted or completed LTBI treatment compared with those with positive QFT-GIT results. Future studies should include high-risk persons with no known TB exposure, who constitute the main reservoir for TB cases in the United States.
Treatment of latent tuberculosis infection (LTBI), the precursor for most tuberculosis (TB) cases, reduces the risk of progression to TB disease by 60% to 90% and is critical to the goal of TB elimination in the United States.1,2 Recent contacts to TB cases are at high risk of progression to TB disease, especially within the first 1 to 2 years after infection. Despite the abundant evidence for the effectiveness of LTBI treatment, half or more of infected persons often decline or fail to complete LTBI treatment3-6; 1 possible reason is skepticism by patients or physicians about the century-old tuberculin skin test (TST).3,7 The TST can be associated with false positive results, particularly in non-US-born persons, because it shares proteins with some nontuberculous mycobacteria8,9 and with the Bacille Calmette-Guérin (BCG) vaccine for TB,10,11 which is used in an estimated 90% of newborns in countries outside the United States.10,12
In 2001, the Food and Drug Administration approved the first interferon-gamma release assay (IGRA), a blood test for LTBI that is less likely to cross-react with nontuberculous mycobacteria.9,13 The availability of IGRAs raises the possibility that use of a blood test may improve confidence in LTBI test results and convince more contacts to accept and complete LTBI treatment. The purpose of this study was to determine if use of IGRA instead of TST increases the likelihood of acceptance and completion of LTBI treatment among contacts to TB cases.
Methods
The Tuberculosis Epidemiologic Studies Consortium–I (TBESC-I) was funded by the Centers for Disease Control and Prevention (CDC; Atlanta, GA, USA) to conduct research on TB prevention and care at 21 sites across the United States and Canada. 14 We conducted a retrospective study in 2 US sites to assess the performance of an IGRA, QuantiFERON-TB Gold In-Tube (QFT-GIT), compared with the TST, under program conditions and its impact on LTBI treatment acceptance and completion.
The sites were 2 urban areas, 1 in the Midwest and 1 on the West Coast, selected by a working group based on their applications and willingness to complete the study. The time periods used for comparison of the TST- and QFT-GIT-tested cohorts depended on program rollout of QFT-GIT and availability of data, thus differed by site. At site A, the TST cohort included all eligible contacts tested in 2005, when the most data were available; the QFT-GIT comparison group included all contacts tested with QFT-GIT in 2009, the year after site A introduced QFT-GIT testing. Site B included contacts tested with either TST or QFT-GIT from September 1, 2005, through August 31, 2006.
Both sites excluded contacts from the study who had a documented prior positive TST or QFT-GIT result, or prior treatment for LTBI or TB. Because treatment of LTBI by directly observed therapy (DOT) was legally mandated for contacts <15 years old at site A, contacts in this age group from both sites (n = 106) were excluded from the analysis.
Data collection and definitions
For contact investigations, both sites provided CDC with data extracted from their standard data collection forms; the forms requested age, gender, race, country of birth, history of BCG vaccination, HIV status, and history of homelessness, incarceration, and illegal drug use. Each site followed its standard procedures for risk counseling of patients recommended for treatment. A positive TST result was defined as ⩾5 mm for contacts, and QFT-GIT results were interpreted as negative, positive, or indeterminate, per manufacturer’s instructions. 13 A US-born person was defined as someone born in the United States or one of its territories. For study contacts with positive TST or QFT-GIT results, treatment acceptance was defined as receipt of the first month’s supply of LTBI medication (6 or 9 months of isoniazid [6H or 9H], or 4 months of rifampin [4R]); treatment completion was defined as receipt of the last refill within 9 months of initiation for 6H, within 12 months for 9H, and within 6 months for 4R. For study purposes, the sites reported only treatment results, not type of treatment offered. Contacts were followed for 1 year through telephone calls and TB registry matches to identify new TB cases.
De-identified data were combined for analysis using SAS 9.2 (Cary, NC, USA). Risk ratios (RRs) and 95% confidence intervals (CIs) assessed demographic differences between sites and differences in proportions of those who started and completed LTBI therapy by TST and QFT-GIT results. Because of small numbers and missing data, extensive multivariate analyses were not possible. This TBESC study was approved by the CDC’s Institutional Review Board (IRB) and relevant local IRBs. Underlying research materials may be provided by the corresponding author.
Results
Demographics and test results
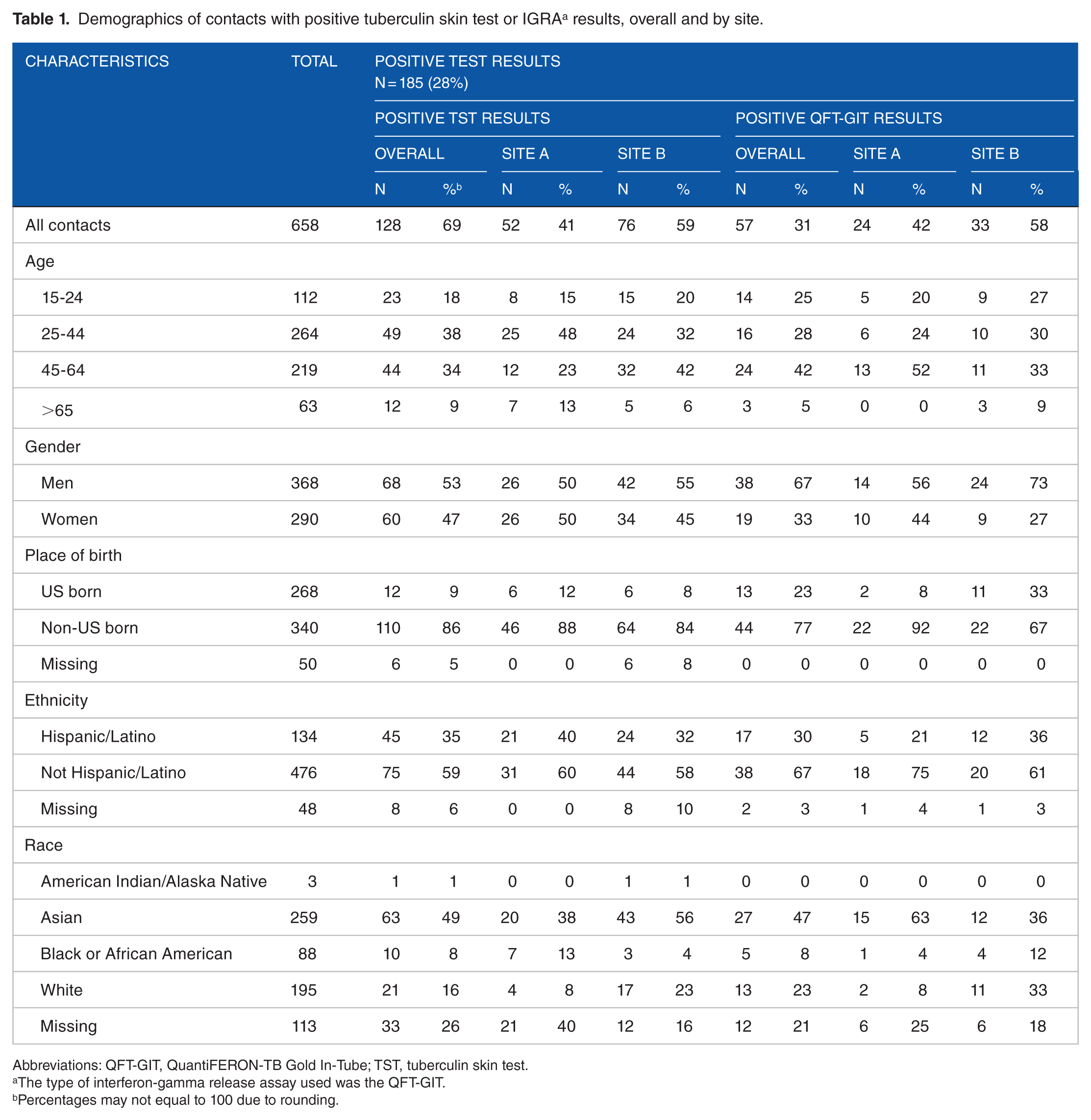
A total of 739 contacts were tested for LTBI; 81 (11%) did not return for their TST results to be read and were excluded from the analysis. Of the 658 contacts with test results, 406 (62%) received TST and 252 (38%) received QFT-GIT. Test results varied by site and characteristics (Table 1). Of the 658, 185 (28%) were positive: 128 (69%) by TST and 57 (31%) by QFT-GIT. Contacts’ mean age was 42 years old overall (range, 15-94): At site A, mean age was 40 (±18 standard deviation [SD]) and at site B, mean age was 43 (±15.5 SD) (P ⩽ .05 for Student t test comparing average age of participants at the 2 sites). Contacts at Site B were more likely to be men (P ⩽ .05) and less likely to be US born (P < .001).
Demographics of contacts with positive tuberculin skin test or IGRA a results, overall and by site.
Abbreviations: QFT-GIT, QuantiFERON-TB Gold In-Tube; TST, tuberculin skin test.
The type of interferon-gamma release assay used was the QFT-GIT.
Percentages may not equal to 100 due to rounding.
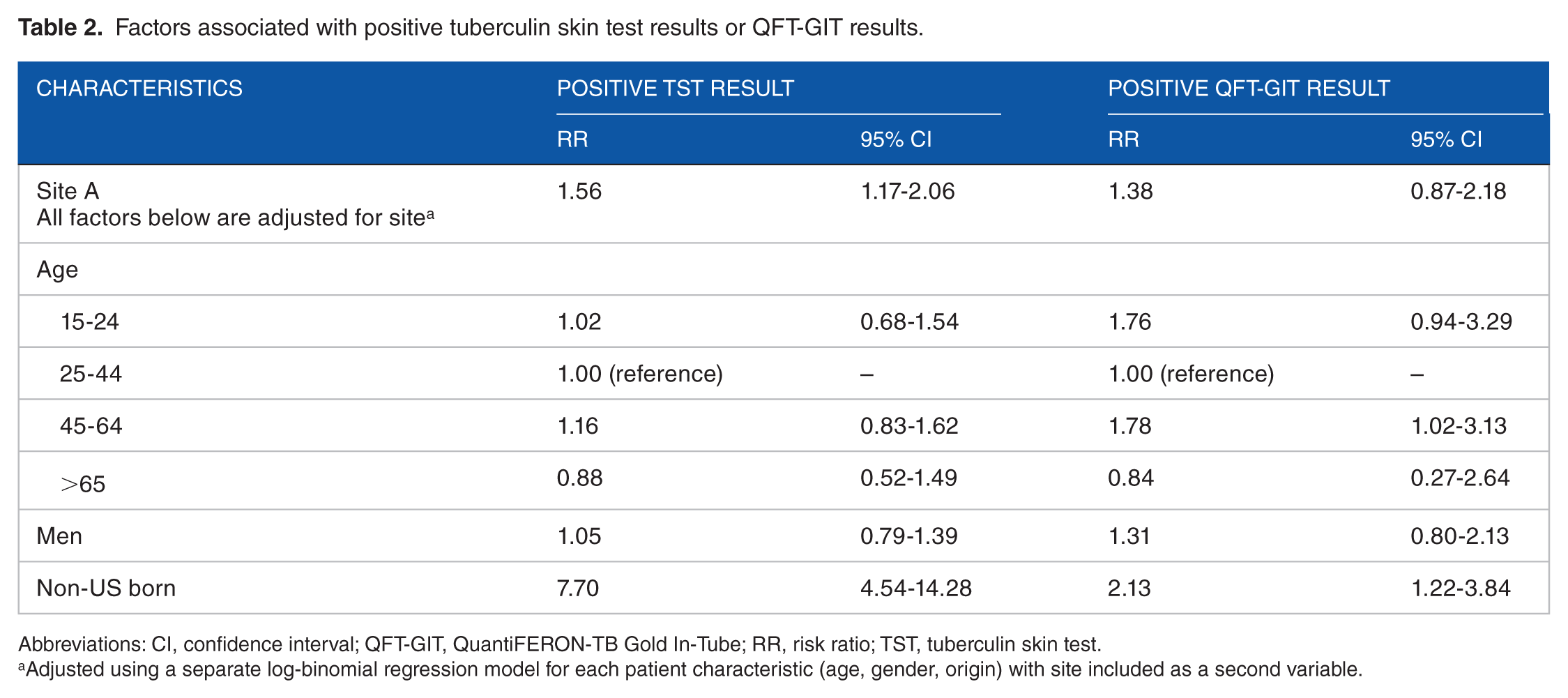
When test results from both sites were combined, a positive QFT-GIT result was associated with birth outside the United States and age 45 to 64 years; a positive TST result was associated with birth outside the United States and evaluation at site A (Table 2). Data on other variables were missing for more than 70% of contacts at both sites, including HIV and immunocompromised status, homelessness, incarceration, alcohol and illegal drug use, and BCG vaccination.
Factors associated with positive tuberculin skin test results or QFT-GIT results.
Abbreviations: CI, confidence interval; QFT-GIT, QuantiFERON-TB Gold In-Tube; RR, risk ratio; TST, tuberculin skin test.
Adjusted using a separate log-binomial regression model for each patient characteristic (age, gender, origin) with site included as a second variable.
Treatment initiation and completion
Overall, of the 185 contacts with a positive test result (TST or QFT-GIT) for LTBI, 140 (76%) accepted treatment and 107 (58%) completed treatment. Treatment acceptance and completion differed by site (Figure 1). At site A, treatment acceptance was 96% for those with positive TSTs and 83% for those with positive QFT-GITs (P = .05); treatment completion was 77% and 63%, respectively (P = .19). At site B, treatment acceptance was 62% for those with positive TSTs and 70% for those with QFT-GIT results (P = .43); treatment completion was 46% and 52%, respectively (P = .60). At site A, 72% (55 of 76) of contacts with LTBI completed treatment compared with 48% (52 of 109) at site B (P < .001 for comparison of completion probabilities).
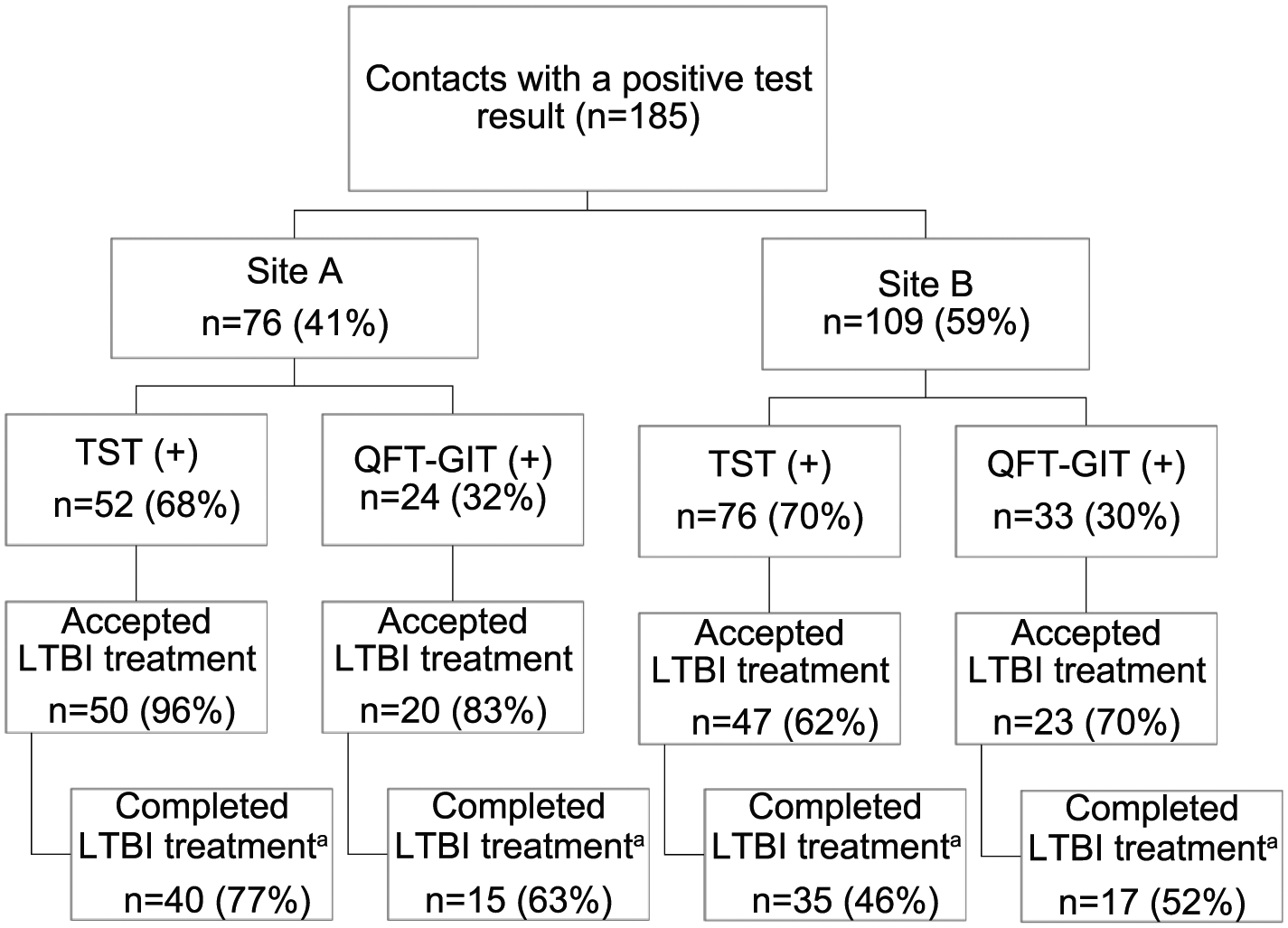
Proportions of LTBI for tuberculosis contacts for acceptance and completion of LTBI treatment, by site and test. LTBI indicates latent tuberculosis infection; QFT-GIT, QuantiFERON-TB Gold In-Tube; TST, tuberculin skin test.
Thirty-three of 185 contacts (18%) accepted, but did not complete, treatment for LTBI. Of the 33, 9 (27%) chose to stop, 5 (15%) were lost to follow up, 4 (12%) moved, 2 (6%) had adverse events, and 13 (39%) contacts had “Other” reasons why LTBI treatment was not completed. None of the 658 contacts were diagnosed with TB disease within 1 year after testing.
Analysis of the combined data for both sites showed no difference in LTBI treatment acceptance or completion by test type. Overall, 75% (43 of 57) of contacts with a positive QFT-GIT result accepted treatment compared with 76% (97 of 128) with a positive TST result (RR = 1.0, 95% CI, 0.83-1.2). Treatment completion for all contacts and for non-US-born contacts did not differ by type of test (Table 3).
Comparison of treatment completion probabilities among contacts who received TST and those who received QFT-GIT at 2 sites.

Abbreviations: CI, confidence interval; LTBI, latent tuberculosis infection; QFT-GIT, QuantiFERON-TB Gold In-Tube; RR, risk ratio; TST, tuberculin skin test.
Referent.
Discussion
Multiple studies have shown that screening and treatment for LTBI can have a significant impact on TB incidence if a high proportion of people successfully complete an adequate course of LTBI therapy.1,13,15-17 While it is clear that the 12-week dose regimen of isoniazid and rifapentine18-20 and DOT15,18 can increase adherence and improve treatment completion, the impact of other factors is less clear.3,5,6 This study compared the effect of a positive TST result with a positive QFT-GIT result on treatment acceptance and completion among contacts to TB cases who are ⩾15 years old, based on the hypothesis that a blood test result not affected by previous BCG vaccination would be more persuasive to patients to accept and complete LTBI treatment, particularly those born in countries where BCG vaccination is common. However, this study did not find a measurable difference between treatment acceptance and completion among patients based on whether they were diagnosed by a positive TST or a positive QFT-GIT result, regardless of whether the patient was born in the United States or elsewhere.
One reason this study found no difference may be that close contacts are more motivated to accept and complete LTBI treatment because they know that they have been recently exposed to an infectious TB case. Therefore, differences based on the type of LTBI test in our study population may be smaller than among patients with no known TB exposure. In addition, TB programs may focus more educational resources and personnel time on close contacts because they are at higher risk of progression to TB disease. The 58% completion proportion reported overall in this study is higher than completion proportions reported for populations that include persons who are not contacts (~35%, 50%, and 39% completion) in other studies.3,5,6 It is also possible that some of the 81 persons who did not return for reading of their TST had LTBI and might have benefited from LTBI treatment.
This study had limitations. The sample size was small, which restricted statistical power and the use of multivariate analyses. Data were collected for different time periods. Programmatic differences between the 2 sites (eg, staffing, infrastructure) may have affected acceptance and completion of LTBI treatment. Because 1 site legally mandated LTBI treatment for children <15 years old, we could not assess the impact of LTBI test type in this important population. Because sites did not report treatment type, we could not assess the effect of shorter regimens on treatment completion. Finally, because DOT is not generally used for LTBI treatment, we know only whether the medication was picked up, not whether it was actually taken. To address many of these limitations, CDC’s Tuberculosis Epidemiologic Studies Consortium–II (TBESC-II), which started in 2012, is evaluating strategies to improve LTBI treatment acceptance and completion.
In conclusion, our study in 2 urban health departments showed no difference in proportions of contacts ⩾15 years old with positive TST results who completed LTBI therapy compared with those with positive QFT-GIT results. Future studies should include high-risk persons with no known TB exposure, who constitute the main reservoir for TB cases in the United States.3,21
Footnotes
Acknowledgements
The authors are grateful to Vicki Ingle (Chicago, IL, USA), and Jennifer Grinsdale, Leah Jarlsberg, and Dr Lisa Pascopella (San Francisco, CA, USA) for data collection and review of the manuscript; to Melissa Pagaoa and Liping Zhu (Centers for Disease Control and Prevention [CDC]/Atlanta, GA, USA) for merging and analyzing data; and to Dr John Bernardo (Boston, MA, USA) for study design.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Note
Baby M Djojonegoro and Elizabeth Fair is also affilated to Curry International Tuberculosis Center, University of California San Francisco.
Author Contributions
LL, DK, and CH drafted the main portions of the manuscript and analyzed data. PJF merged data sources, performed multivariate analyses to interpret data, and verified the analytical methods. BMD, EF, SJ, SM contributed to the design of the study, collected and verified data. CRH was responsible for the original conception of the study. All authors reviewed and approved the final manuscript.
