Abstract
Objective
To investigate the effects of a daily multinutrient supplement on plasma indicators of glycemic and lipemic control and psychological wellbeing in type 2 diabetic patients.
Design
Double-blind, randomised, cross-over pilot intervention study.
Subjects
Twenty-nine subjects (15 males and 14 females) with non-insulin-dependent type 2 diabetes.
Intervention
Either a multinutrient supplement or placebo were provided daily during two intervention periods of 3 months separated by a 4 week washout.
Results
There were no significant changes of multinutrient treatment compared with placebo in HbA1c, fasting or postprandial plasma glucose and insulin concentrations and fasting plasma lipid concentrations. Using a validated wellbeing questionnaire (W-BQ 22) designed for diabetic subjects, the multinutrient supplement resulted in improvements in the secondary outcome of wellbeing of the volunteers in terms of anxiety (p = 0.020), vitality (p = 0.013) and general wellbeing (p = 0.021), relative to placebo.
Conclusions
Findings from this pilot study suggest that a multinutrient supplement may enhance the wellbeing of diabetic patients, even in the absence of a significant improvement in clinical parameters. If substantiated in a full clinical study the results would have important implications for the prevention of late complications of diabetes, as psychological factors can hinder successful management of the condition and adversely affect metabolic control.
Keywords
Introduction
Interest in and use of dietary supplements has increased in recent years. In industrialised countries, surveys indicate that nearly a quarter of the general population uses some form of dietary supplementation (e.g. Kaartinen et al. 1997). The proportion of supplement users among people with diabetes has been shown to be even higher. A Canadian study (Ryan et al. 2001) reported that more than 40% of people with diabetes were taking over-the-counter vitamin and mineral supplements, with multivitamins and individual antioxidant vitamins being most popular. A recent review of the National Health and Nutrition Examination Survey (NHANES) in the U.S.A., found that 34% of people with diabetes took dietary supplements (The Lewin Group, 2006).
An increasing body of evidence suggests that the body status of a number of key nutrients involved in glucose homeostasis may be reduced in diabetes, consequent on enhanced urinary excretion induced by hyperglycaemia (Marakis and Walker, 1999). In particular, magnesium, zinc, chromium and the B-vitamins may be affected. In addition, a mounting body of evidence indicates that the antioxidant defences (e.g. status of vitamins E and C) are compromised in diabetes. Indeed, Czech scientists (Holecek et al. 1995), in a preliminary study, observed decreased glycosylated haemoglobin levels in patients with type 2 diabetes after supplementation with multinutrients rich in antioxidants. However, improved glycaemic control alone may not address other abnormalities of type 2 diabetes such as hypertension, dyslipidaemia or mood. Since insulin resistance is an underlying mechanism of the condition, Belgian workers (Guillaume et al. 1995) over a decade ago, stressed the importance of a global approach to the treatment of diabetes, rather than use separate treatments for its various manifestations, but little has been done to follow this up.
The need for daily attention and vigilance to diet and lifestyle in the management of diabetes, as well as the anxiety of possible future complications (Eiser and Tooke, 1995) can compromise wellbeing. In reports from U.K. (Lloyd et al. 2000) and U.S.A. (Peyrot and Rubin, 1977; Donie, 2004) it has been estimated that anxiety and depression affects between 28–50% of diabetes sufferers–- about two to three times that estimated for the general population. Improvements in wellbeing may lead to better diabetic control.
Although cognitive therapy has been used to improve psychological wellbeing in diabetes, the role of diet and nutrition has been less well researched. However, an increasing number of studies have demonstrated that dietary enhancement with nutrients, such as folic acid and thiamin, can have beneficial mood-modulating action. Indeed, a common metabolic derangement in diabetes and depression has been suggested (Peyrot and Rubin, 1999) and needs to be further investigated.
In the absence of overt evidence of deficiency, most diabetes-support groups (DNSG-EASD, 1999; ADA, 2000) do not recommend the routine use of dietary supplements by diabetic patients. Nevertheless, numerous studies in the literature point to the benefits of individual vitamin and mineral supplements for type 2 diabetes and its complications. There have been far fewer studies on multi-nutrient combinations. However, Holecek et al. (1995) and Farvid et al. (2004) reported that a multi-antioxidant supplement of vitamin and trace elements induced favourable reductions in oxidative stress, blood lipid profiles and susceptibility to heart disease among people with diabetes. Furthermore, Barringer et al. (2003) showed that a multivitamin and mineral supplement taken for a year reduced infective illness and infection-related absenteeism among diabetic subjects. However, Ballinger et al. (2003) were not able to show improvements in general wellbeing using non-condition-specific 12-Item Short Form (Ware et al. 1996). Although the primary aim or our study was to investigate effects of a daily multinutrient supplemention on glycaemic and lipaemic parameters in patients with type 2 diabetes, a secondary aim was to assess its effects on wellbeing, using a questionnaire specifically designed and validated for use in diabetes.
Methods
Patients
Twenty-nine volunteers with type 2 diabetes were recruited by local advertising into this pilot study. All gave written, informed consent to participate. The study was approved by The University of Reading Ethics and Research Committee and by the West Berkshire Local Research Ethics Committee. Participation required prior agreement from the volunteer's General Medical Practitioner. Patients using insulin, taking nutrient supplements, pregnant women or those women intending to become pregnant were excluded, as well as those with clinically-significant abnormal blood constituent profiles. Information on medication was collected, together with a full medical history. Participants were advised to avoid changes in their habitual diet or exercise levels during their participation in the study.
Protocol
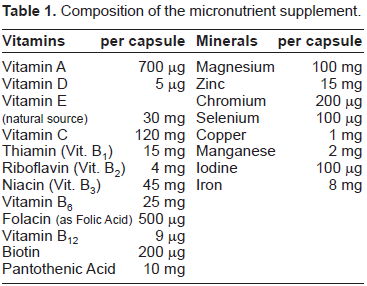
Each participant was issued with a unique code number after successful screening. Following recruitment, volunteers were allocated, by a third party not otherwise associated with trial, individually and alternately to active or matched placebo treatment, on an accession basis according to first clinic attendance. Blinding of the treatment codes was maintained during the study, until all analyses were completed. The study was of crossover design with a 1-month washout period between the two study arms. Volunteers were required to take one capsule a day of either the multinutrient supplement or matched placebo (cellulose) for 3 months on each arm of the study. The composition of the multinutrient supplement (Diabetone®, Vitabiotics Ltd, Vitabiotics House, 1, Apsley Way, London, NW2 7HF, U.K.) is presented in Table 1. Participants were instructed not to change their lifestyle (i.e. exercise and smoking habits) or their dietary habits and not take any other dietary supplements during the study.
Composition of the micronutrient supplement.
Volunteers were invited to attend a clinic at the Hugh Sinclair Unit of Human Nutrition on three occasions: before the commencement of the study (baseline) and at the end of each treatment (months 3 and 7). There was no clinic attendance at the end of the wash-out period. The supplements were sent out to the participants before the commencement of the second arm of the study and confirmation of delivery was done by telephone. Liver and kidney function tests and analysis of electrolytes were performed on plasma from blood samples drawn at baseline. On each clinic visit, height and weight was recorded. Blood pressure was taken at rest using an Omron 703CP automatic monitor (Omron Terminals Ltd, Chessington, U.K.) and recorded as the mean of the second and third readings. Dietary intake was estimated at baseline by means of a validated food frequency questionnaire (DietQ Version 3, TINUVIEL Software, Warrington, U.K.).
At each clinic visit, fasting blood was drawn after a 12-hour overnight fast into appropriate Vacutainer® tubes (containing either Lithium heparin, EDTA or Clot Activator SST™ Gel). Then the volunteers consumed a ‘test’ breakfast containing 75 g of carbohydrate. One hour after the breakfast, another blood sample was taken.
Plasma glucose and lipid concentrations were determined using the Monarch centrifugal analyser (Instrumentation Laboratories UK, Ltd–Kelvin Close, Birchwood Science Park, Warrington, U.K.) which was equipped with an appropriate IL Glucose (Hexokinase), Cholesterol or Triacylglycerol kit. HDL-cholesterol was measured after dextran-Mg precipitation of very low and low density lipoproteins according to the method by McNamara et al. (1994), and then analysed with enzymatic method as for total cholesterol. LDL-cholesterol was calculated using the Friedwald equation (Friedwald et al. 1972). Fasting and postprandial insulin concentrations were determined by an immunoreactive assay (DAKO Diagnostics Ltd, Denmark House, Angel Drove, Ely, U.K.). Glycated haemoglobin (HbA1c) as a measure of glycaemic control, was determined by fast performance liquid chromatography (HA-8121, Menarini Diagnostics Ltd, Wokingham, U.K.) at the Pathology Laboratory of the Royal Berkshire Hospital, Reading, U.K.
A timed 2-hour urine sample was collected after the first void urine. The volume was recorded and an aliquot taken for analysis of magnesium and zinc to provide a marker of compliance. Compliance to treatment was also monitored by counting returned supplement containers at the end of each study arm. Urinary Mg was analysed using the Monarch analyser (Instrumentation Laboratories UK, Ltd) which was equipped with an appropriate IL Magnesium kit. Urinary zinc concentration was determined by a colorimetric assay (Alpha Laboratories–-40 Parham Drive, Eastleigh, Hampshire, U.K.).
General wellbeing was evaluated using a Well-being Questionnaire (W-BQ 22) validated for use in diabetes (Bradley and Gamsu, 1994). It consists of 22-items, grouped into the following subscales: ‘Depression’ (6 items), ‘Anxiety’ (6 items), ‘Vitality or Energy’ (4 items) and ‘Positive Well-Being’ (6 items). Each item was assessed semiquantitatively and scored on a 0 to 3 Likert scale (3 equals to “all the time” and 0 “not at all”). Ratings on items on each subscale were summed after reversing scores where appropriate according to Table 2.
Formulae for scoring the items in the Bradley W-BQ 22.

Statistical Analysis
The number of volunteers required for this pilot study was determined by practical considerations and all analyses were carried out on an intention-to-treat basis using analysis of variance (ANOVA). Treatment comparisons were made within participants, and an adjustment was made for period, irrespective of its statistical significance. The assumptions underlying the ANOVA were examined using residual plots, and any potential influential observations were identified using Cooks D score. If an outlier was discovered, it was removed from the dataset, and the analyses repeated to check the robustness of the results obtained. A 95% confidence interval for the treatment difference was calculated. All analyses were carried out using PROC GLM in SAS, version 8 (SAS Institute Inc. Cary, NC, U.S.A).
Results
The number of volunteers who entered the study was 29 (15 men, 14 women). Two dropped out after baseline (one due to a diagnosis of cancer and one due to protocol violation). Another volunteer completed only the first arm of the study (active treatment) because, while on placebo, he had a stroke. The personal characteristics of the intention-to-treat group were as follows: average age (± sem) was 61 (± 7.5) years; mean BMI (± sem) at baseline was 30.9 (± 1.0), with no significant change throughout the study; 14/29 reported engaging in moderate or heavy exercise; 2/29 were smokers, 13/29 were past smokers; 20/29 reported being under moderate or great stress; 5/29 were on weight-reducing diets. Management of diabetes was through diet alone (13/29), diet + drugs (10/29) or drugs alone (6/29).
A pill count, performed at the end of each study arm, showed that compliance was satisfactory. This was further confirmed by increases in urinary concentrations of both magnesium (27% increase) and zinc (58% increase) after the active treatment, compared to their urinary excretion at baseline; values returned to near pre-treatment levels after the placebo period.
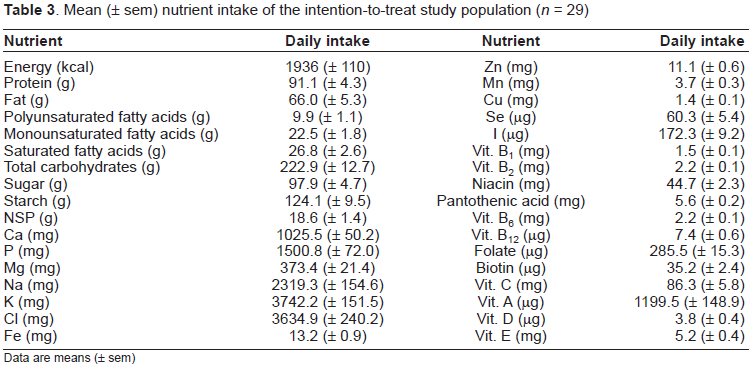
Nutrient intake was assessed by a validated Food Frequency Questionnaire at baseline. For most of the nutrients as shown in Table 3, mean (± sem) intakes were close or slightly higher than the Reference Nutrient Intake (Department of Health, 1991) except for selenium and vitamins E and D.
Mean (± sem) nutrient intake of the intention-to-treat study population (n = 29)
Data are means (± sem)
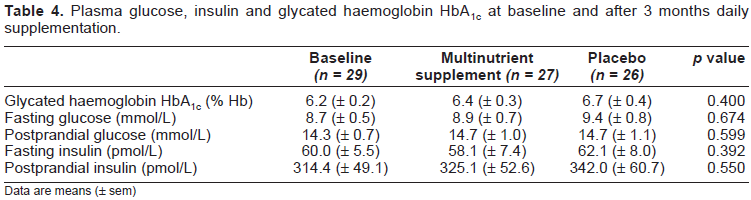
Fasting plasma glucose and insulin concentrations, glucose and insulin response to a test meal and fasting blood lipid profile (total, LDL- and HDL-cholesterol and triacylglycerol concentrations) are shown in Table 4 and Figure 1. ANOVA failed to show a significant difference between the treatments for any of these parameters. However, mean values after the placebo period appeared to be less favourable after baseline or compared to those after the active treatment. Specifically, HbA1c was 6.4% after the multinutrient treatment and 6.7% after the placebo period (while the baseline value was 6.2%). However, the mean values of HbA1c indicate that the study group, on the whole, had their diabetes well controlled according to the UKPDS report (Stratton et al. 2000), which gives a range of HbA1c for people without diabetes of 4.5–6.2%.
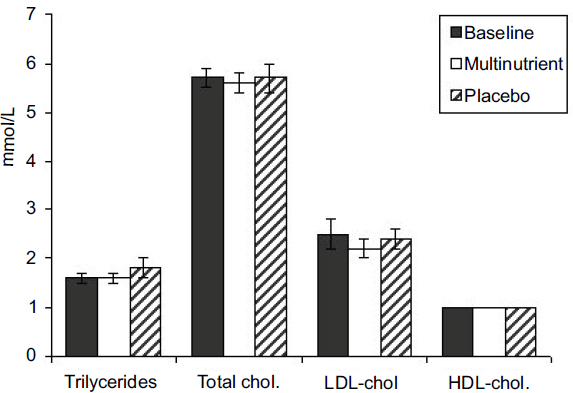
Fasting plasma lipids at baseline (n = 29) and after 3 months of daily supplementation (for Multinutrient: n = 27; for Placebo: n = 26). Significance of difference between treatments at end of study: triglycerides, p = 0.218; total cholesterol, p = 0.256; LDL cholesterol, p = 0.223; HDL cholesterol, p = 0.500. Bars indicate sem (too small to represent for HDL cholesterol).
Plasma glucose, insulin and glycated haemoglobin HbA1c at baseline and after 3 months daily supplementation.
Data are means (± sem)
Mean (± sem) systolic and diastolic blood pressure was 140.9 (± 3.0) and 80.0 (± 1.6) mm Hg, respectively at baseline and showed no significant differences after treatment. No treatment changes in pulse rate (mean 77.6 ± 2.1 at baseline) were observed either. Blood pressure appeared to be well controlled for the group as a whole according to the UKPDS report (Adler et al. 2000).
The mean (± sem) General Wellbeing score after the multinutrient treatment (55.2 ± 1.4) was significantly greater (p = 0.021) than after placebo (50.1 ± 2.5), with an adjusted treatment difference of 3.083 with 95% CI of (0.517, 5.649). Mean (± sem) scores for three subscales of the W-BQ 22 are presented in Figure 2. The mean depression and anxiety scores (both with max 18) were reduced after the active treatment compared with placebo, although, of these, only the anxiety score was significant at <5% level of probability. For the anxiety subscale the p value was 0.020, with an adjusted treatment difference of –1.042 with 95% CI of (–1.900, –0.183). The vitality subscale (max 12) was increased significantly after active treatment compared with placebo. The p value for the vitality subscale was 0.013, with an adjusted treatment difference of 1.077 with 95% CI of (0.268, 1.885). The treatment difference in positive wellbeing subscale was not statistically significant (p = 0.170).
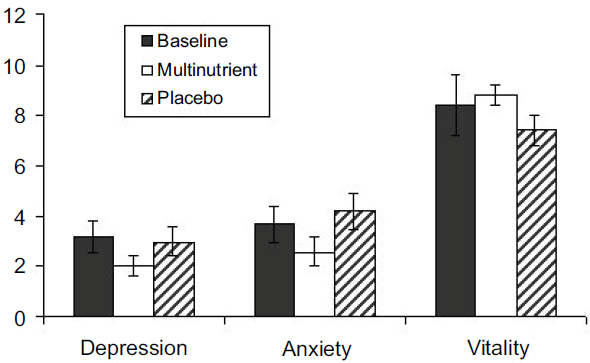
Well-being subscale scores at baseline (n = 29) and after 3 months of daily supplementation (for Multinutrient: n = 27; for Placebo: n = 26). Significance of differences between treatments at end of study: depression, p = 0.086; anxiety, p = 0.020; vitality, p = 0.013. Bars indicate sem.
Discussion
In our pilot study, a multinutrient supplement failed to significantly improve the glycemic and lipemic profile of patients with type 2 diabetes with relatively good baseline glycemic and blood pressure control. In most studies which have demonstrated such improvements in diabetes, nutrients were administered at higher doses than in our study. Pharmacological doses of single antioxidant vitamins such as vitamin C (Paolisso et al. 1995) and vitamin E (Paolisso et al. 1993) have shown improved insulin effectiveness and glucose disposal, as well as reduced oxidative stress in type II diabetic sufferers. In another study, modest doses (but higher than those used in the intervention in our study) of a combination of Mg, Zn, Vitamins C and E increased serum HDL cholesterol and LDL Apo A in type 2 diabetes after 3 months (Farvid et al. 2004).
In accordance with other studies carried out in young (Carroll et al. 2000) and elderly (Krondl et al. 1999) non-diabetic subjects, our study demonstrated that a multinutrient supplement taken daily for 3 months could improve the wellbeing of patients with type 2 diabetes. Our results must, however, be treated with caution as not only was this a pilot study carried out without prior estimation of study volunteer numbers required to reach statistical signifance using a power calculation, but the measurement of wellbeing was a secondary outcome. Furthermore, and on account of these uncertainty factors, no corrections to the level of significance were applied for errors incurred in multiple testing.
Other studies on non-diabetic patients with potential nutrient deficit have also shown positive health benefits of enhanced nutrition. In the study by Krondl et al. (1999), elderly subjects eating less than 4 portions of fruits and vegetables per day, had significantly improved general health perception and vitality after consuming a liquid nutrient supplement for 16 weeks. In our study, the nutrient intake of the subjects appeared to be, on average, satisfactory. However, diabetes can accelerate the turnover of a number of micronutrients in the body (e.g. Chausmer, 1998) and, therefore, even in the absence of signs of overt deficiency, suboptimal body nutrient status may exist. Although researchers have not traditionally employed psychological measures to monitor nutritional status, it has been argued (Benton and Donohoe, 1999) that they are particularly appropriate in cases of subclinical nutrient deficiencies, in which the impairment of psychological wellbeing can be one of the first indications.
Several studies have investigated the association between wellbeing and glycemic control with conflicting results. While some studies (e.g. Peyrot and Rubin, 1997) have found no association between glycemic control and levels of depression or anxiety, other studies (e.g. Mazze et al. 1984; Van Der Does et al. 1996) have shown that depression, anxiety and poor quality of life are related to inadequate metabolic control in diabetes. In general, psychological morbidity and impaired well-being may be more prevalent in people with diabetes and may be dependent on the presence and number of metabolic complications and secondary illnesses (Rose et al. 2002). The exact reasons for this remain unclear. Pouwer and Snoek (2001) discussed how both physiologic and behavioural mechanisms may underlie a reciprocal relationship between symptoms of depression and poor glycemic control in diabetes. In our study, we were unable to demonstrate that changes in well-being parallel changes in metabolic control. Hence, the improvements in depression, anxiety and fatigue in this study were more likely to be due to the effects of individual nutrients, such as the B complex vitamins, magnesium and zinc, acting directly on neurological tissues. There are numerous studies demonstrating that suboptimal levels of thiamin, riboflavin, folic acid and vitamin B12 are associated with low mood, low vitality, psychiatric disorders and poor cognitive function (Benton and Donohoe, 1999). In many cases, these disorders appear to respond positively to vitamin supplementation.
Preliminary evidence suggests that vitamin D, which was typically low in the diets of our participants, may also have mood-enhancing effects (Lansdowne and Provost, 1998). The contributory role of antioxidant nutrients such as vitamins C and E and selenium cannot be excluded either, as they have potential to prevent oxidative damage to brain cells, nerves and muscles. From our study we suggest that nonspecific psychological symptoms of diabetes (such as depression and anxiety) are not solely influenced by hyperglycaemia, but may be influenced by suboptimal body status of micronutrients, which can, at least partly, be corrected by multiple nutrient enhancement of the diet.
Conclusion
This small pilot cross-over study demonstrated that administration of a daily multinutrient supplement for three months can lead to significant improvements in the wellbeing of patients with type 2 diabetes, even in the absence of significant improvements in metabolic control. However, a full clinical study needs be carried out before robust conclusions can be drawn. Nevertheless, these findings should inspire further investigation into the use of micronutrient supplements for diabetes. A good quality of life in diabetes is now recognised as important for adherence to treatment regimes, so its assessment as outcome of future nutritional intervention trials for diabetes is warranted.
Footnotes
Acknowledgements
We are grateful to NATO for a scholarship (GM) and to Vitabiotics Ltd for the donation of supplements and a modest ‘top-up’ grant for clinical analyses. We are also grateful to Clare Bradley for her permission to use the W-BQ 22.
