Abstract
Background:
Currently, most Pulmonary Rehabilitation (PR) programs work with conventional physical training, using resources that are not available in public health in Brazil. Multicomponent physical training is a strategy that uses few resources and can reach a larger portion of the population.
Objective:
To investigate the effectiveness and safety of multicomponent physical training on physical-functional performance in patients with COPD.
Design:
Protocol for a randomized clinical trial with 2 groups in parallel (1:1).
Setting:
University-based, outpatient, physiotherapy clinic.
Participants:
64 patients aged ⩾50 years, clinical-functional diagnosis of COPD, GOLD II and III criteria will participate in the study.
Intervention:
Participants will be randomly allocated into 2 groups: Multicomponent Physical Training—MPT (n = 32): aerobic, strength, balance and flexibility exercises performed in a circuit training format, or Conventional Physical Training—CPT (n = 32): aerobic and strength training. The interventions will be carried out twice a week, for 8 weeks and supervised by the same physiotherapist.
Measurements:
The 3 primary outcomes are the 6-Minute Walk Test (6MWT), the 6-Minute Step Test (6MST) and VO2 consumption as measured during the 6MWT. Secondary outcomes will be exercise capacity, level of physical activity in daily life, peripheral muscle strength, functional status, dyspnea, fatigue and quality of life. Safety will be assessed by recording adverse effects. These outcomes will be evaluated before and after the intervention and the evaluator will be blind.
Limitations:
It will not be possible to perform the blinding of the physiotherapist who will supervise the interventions.
Conclusions:
This study is expected to demonstrate that MPT using simple resources is an effective and safe intervention for the improvement of the aforementioned outcomes and, in addition, to broaden the horizon of research in relation to new methods of physical rehabilitation for patients with COPD.
Introduction
Chronic Obstructive Pulmonary Disease (COPD) is characterized by a combination of pulmonary and extra-pulmonary manifestations that directly contribute to the increase in exercise intolerance and dyspnea, especially in more advanced stages of the disease, leading to physical inactivity and consequent worsening of these signs and symptoms. 1 In addition, COPD patients have a high cardiovascular risk, caused by systemic inflammatory changes in the arterial vasculature, favoring oxidative stress and the development of arterial stiffness, which in turn is associated with the development of target organ lesions (kidneys, brain and heart) predisposing the patient to a higher risk of morbidity, hospital admission and death. 2
The economic impact caused by the disease is significant worldwide, both in relation to the direct costs of managing the disease and its exacerbations, as well as the indirect costs, which mainly result from the difficulty of patients affected by the disease to be inserted in the labor market, and their family members, who often need to dedicate themselves exclusively to the care of the patient. 3
Solid evidence shows that physical training, the most important component of a Pulmonary Rehabilitation program (PR), reduces dyspnea and fatigue, increases exercise performance, and improves peripheral muscle strength, functional capacity, control of emotional function and quality of life of these patients’ lives. 4 Currently, most PR programs work with conventional physical training, using resources little available in public health, such as treadmills and exercise bikes and weight training equipment, in addition to using specific spaces only for this purpose. This creates a financial burden on the health system and patients, and limits the implementation of PR services. 5 In addition, most PR programs involve a combination of both aerobic and strength exercises. Multicomponent physical training (MPT) proposes the performance of 3 or more components or physical training modalities within the same session, and has demonstrated its benefits in several studies involving the elderly population. 6
This study hypothesizes that physical training through functional exercises, which use few resources and can serve a larger portion of the population, is safe and effective in improving the main outcomes related to physical health and quality of life of patients with COPD. In addition, this study is expected to demonstrate that MPT with simple resources is an effective and safe intervention for improving the aforementioned outcomes. It is also expected that the results can broaden the horizon of research in relation to new methods of physical rehabilitation for these patients.
This randomized clinical trial aims to investigate the effectiveness and safety of MPT on exercise capacity, level of physical activity in daily life, peripheral muscle strength, functional status, dyspnea, fatigue and quality of life in patients with COPD. The publication of this protocol aims to provide transparency about the research, in addition to contributing to the state of the art and avoiding duplication of research, avoiding the unnecessary use of resources.
Methods
Trial design
It is a randomized clinical trial protocol with 2 groups, where the participants will be randomly allocated in parallel (1:1). This protocol was developed according to the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) guidelines. 7 The study was approved by the Research Ethics Committee of Setor de Ciências da Saúde da Universidade Federal do Paraná (10/30/19 - CAAE: 20227019.0.0000.0102). All items in the Test Record Data Set (World Health Organization) were recorded in the database of the Brazilian Clinical Trials Registry (Registro Brasileiro de Ensaios Clínicos—REBEC). Any changes made to the protocol will be communicated to the aforementioned bodies.
Setting and participants
The study will be carried out in a physiotherapy clinic of a university hospital in the city of Curitiba (Brazil). The rehabilitation program will have a physiotherapist who will carry out the pre- and post-intervention assessments, and a physiotherapist who will conduct the physical training programs. Participants will be referred by 2 pulmonologists responsible for the clinic where they will be recruited. All the materials needed to carry out the study are available in the sector where the evaluation and physical training will be carried out, such as an ergometric treadmill, dumbbells and elastic bands. Participants will not receive a financial incentive to participate in the training, their participation in the study will be considered a component of the regular pathway of care.
Patients with a clinical-functional diagnosis of COPD (criteria GOLD II and III) who are undergoing medical follow-up at the Pulmonology Outpatient Clinic or at the Pulmonary Function Service will be referred by their respective services to the research team. During the initial approach, participants will be informed in detail about their voluntary participation, objectives, methodology, risks and benefits of the study, and if they agree, they will sign the Informed Consent Form and data collection will begin.
The study will include individuals of both sexes, aged ⩾50 years, with a clinical-functional diagnosis of COPD criteria GOLD II and III, 3 clinically stable (outside the period of exacerbation of the disease for at least 3 months), who do not present with a cognitive impairment, as assessed by the Mini Mental State Examination—MMSE (>18 points for schooled individuals and>13 for illiterate individuals)8,9, who are independently able to perform instrumental activities of daily living, as assessed by the Pfeffer et al functional activities questionnaire,10,11 and who have no medical contraindications for physical exercise. Patients with pulmonary diseases such as asthma, pulmonary fibrosis, pneumonia and other non-pulmonary diseases considered disabling, severe or difficult to control (such as heart disease or sequelae of acute or chronic orthopedic and/or neurological diseases), or those who use walking assist devices that could influence the application of the tests will be excluded. Patients who are enrolled in a physical training program within the last 3 months at baseline will also be excluded.
Sample size
The sample size was calculated for the 3 primary outcomes (6MWT, 6MST, VO2 consumption as measured during the 6MWT), and the 6MWT had the largest sample size among the 3 calculations, so it was chosen as the determining variable for the sample size.
Considering the results of the study by Marrara et al 12 for the 2 outcomes, an effect size of 0.80, the probability of Error type 1 (α) of .05 and type 2 (β) of .20, a sample of 27 individuals in each group was calculated. In order to avoid possible losses during the follow-up that could compromise the power of the study, a 20% increase in the number of participants was performed, thus resulting in a sample of 32 patients/group. The sample calculation was performed using the G* Power 3.1® statistical program. 13
Randomization and allocation concealment
Participant allocation will be conducted using sequentially numbered, opaque and sealed envelopes, that will be opened only at the time of participant’s inclusion in the study. Randomization and allocation procedures will be conducted by an independent physical therapist not involved with recruitment, assessment or interventions.
Interventions
After the assessments, the selected patients will be randomly allocated into 2 groups in parallel: MPT or CPT (Figure 1 and Table 3). The interventions will be carried out twice a week, for 8 weeks, in a physiotherapy clinic under the supervision of a physiotherapist who will guide the participants in the development of all the proposed activities, avoiding falls or inappropriate movements during the sessions.

Flowchart of recruitment, interventions and evaluations of participants.
Multicomponent physical training
The choice of exercises was based on the main objective: to involve the main muscle groups that patients with COPD use in their activities of daily living, and also in protocols described in recent studies.14-16
The MPT group will perform the exercises based in a circuit with 8 stations that will be distributed in a circular format, with a distance of 1 m between each station. Circuit training refers to the patient’s repeated training under moderate or low load strengths, more repetitions, and shorter training intervals to gain more balance ability. Also, it can provide some fun and challenging variety. 17 The frequency will be twice a week, for 8 weeks, totaling 16 sessions. This intervention will compose of an initial warm-up, aerobic, strength, balance and flexibility exercises (Table 1).
Multicomponent physical training.

Abbreviations: UL, Upper limbs; LL, Lower limbs; MNR, maximum number of repetitions.
The progression of the exercises will be carried out by increasing the volume of the exercises every 8 training sessions. From the first to the eighth session, the circuit will be performed twice, with each station lasting 1 minute, followed by 1 minute of active rest, thus totaling approximately 21 minutes each session (5 minutes of warm-up plus 2 times the 8-minute circuit). From the 9th to the 16th session, the circuit will be performed 2 times, with each station lasting 2 minutes, followed by 1 minute of active rest, thus totaling approximately 53 minutes each session (5 minutes of warm-up plus 2× the circuit 24 minutes).
The intensity of cardiorespiratory conditioning will be determined between 40% and 60% of the reserve heart rate (HR reserve) and calculated using the formula described by Karvonen et al 18 and the modified BORG scale (mBORG). 19 To reach the target heart rate, the participant will be instructed to increase the speed of performing aerobic exercises, and will be familiarized with frequency control in a session prior to the start of training.
Muscle strength and endurance exercises will be based on the Maximum Number of Repetitions (MNR) defined before the beginning of the protocol, in a session where the participants will become familiar with the exercises. For the calculation of the MNR, the participant will be instructed to perform the upper and lower limb strength exercises within a period of 1 minute, the maximum number of repetitions that they can perform in this period of time will be considered as the MNR. In the first 8 sessions, the muscle resistance training will be performed with 60% of the MNR, from the 9th to the 16th sessions with 70% of the MNR. All stations will have a determined performance time, as described above, however, if a participant completes the MNR before moving on to the next station, they will be instructed to perform a stationary march, as an active rest, maintaining a perception of effort below 4 on the mBORG scale.
Conventional physical training
Participants in the CPT group will participate in 16 training sessions, which will be divided into 3 sequential steps (Table 2): Warm-up, conditioning and cooling down. The exercise prescription was based on the recommendations of the American College of Sports Medicine for patients with chronic respiratory diseases. 20
Conventional physical training.

Abbreviations: RM, repetition maximum.
Safety assessment and adverse effects
All patients will have their vital signs (heart rate, respiratory rate, blood pressure and peripheral oxygen saturation) monitored at the beginning, during and at the end of each intervention using a frequency meter (Geonaute Manual Heart Monitor, Onrhythm 50, USA), aneroid sphygmomanometer (Premium, Brazil) and oximeter (Geratherm, Oxy Control, Swanley, United Kingdom). Their perception of effort will be assessed using the mBORG scale.
In addition, after each intervention, the responsible physiotherapist will record the presence of pain, fatigue, self-reported dizziness, or any other adverse events. The clinic where the intervention will take place has oxygen cylinders and a team prepared to perform basic life support.
Outcome measures
The primary outcome will be exercise capacity, as it is an essential variable in assessing prognosis and response to COPD interventions, and the secondary outcomes will be physical activity in daily life, peripheral muscle strength, functional status, dyspnea, fatigue and quality of life.
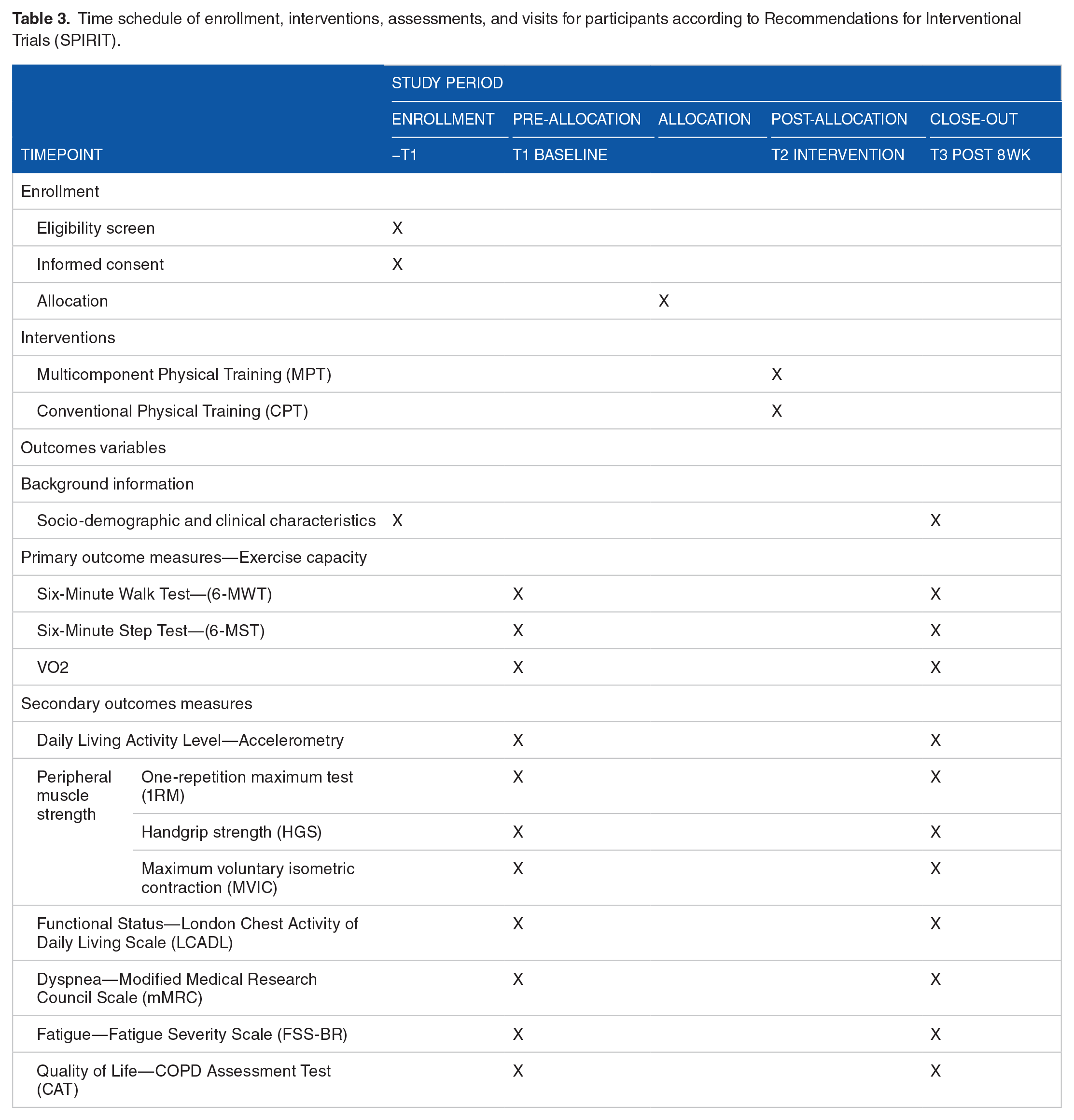
Patients will be evaluated at baseline (pre-randomization) and after 8 weeks, and all assessments will be carried out by 3 physiotherapists previously trained to apply the proposed tests. Table 3 shows the study variables, methods used and moments of the evaluations.
Time schedule of enrollment, interventions, assessments, and visits for participants according to Recommendations for Interventional Trials (SPIRIT).
Primary outcome
Exercise capacity
It will be assessed using the Six-Minute Walk Test (6MWT) and the Six-Minute Step Test (6MST), both following the recommendations of the American Thoracic Society. 21 The 6MWT will be performed in a 30 m corridor where patients will be instructed to walk for 6 minutes as far as they can, without running. At the end of the test, the total distance, in meters, covered by the participant will be recorded. For this outcome, we will consider an increase of 25 m or 14% in the distance walked as a Minimal Clinically Important Difference (MCID). 22
The 6MST will be performed on a 20 cm high step, where the participant will be instructed to go up and down the step without the support of the upper limbs, which will remain stationary along the body. The test will be performed in a free cadence. 23 The total number of climbs on the step with both feet will be recorded. For this outcome, we will consider an increase of 20 steps as a minimal clinically important difference. 24
In addition, energy consumption during the 6MWT will be assessed using a portable K5 ergospirometer (COSMED S.r.l., Italy). The following parameters will be evaluated: oxygen consumption in L/min (VO2), total ventilation in L/min (VE), VE/VO2 ratio, carbon dioxide production in L/min (VCO2), VE/VCO2 ratio, respiratory coefficient (RQ) and energy expenditure (Metabolic Equivalent of Tasks—METs). For each test, the K5 will be calibrated according to the manufacturer’s instructions, and the analyzes will be carried out breath by breath. 25
Secondary outcomes
Level of physical activity in daily life
The level of Physical Activity in Daily Life (PADL) will be assessed using the electronic accelerometer GT3X-BT (Actigraph, LLC Engineering/Marketing, Pensacola, Florida, USA), which should be used on the right hip, attached to an elastic belt. The participants will be instructed to use the accelerometer for 7 consecutive days, for the whole day, except when showering or swimming. In previous studies, a valid day has been defined as having a minimum of 70% of the use of each day in waking hours (from 8 am to 10 pm). 25 The following variables will be obtained: the daily step count (steps/day), energy expenditure, time spent on activities (lying, sitting, standing and walking), and intensity of activities. For this outcome, we will consider an increase of 600 steps/day as a MCID. 26
Peripheral muscle strength
It will be assessed using the handgrip strength (HGS) and the Maximal Voluntary Isometric Contraction (MVIC) of the quadriceps muscle. The handgrip strength (HGS) will be assessed using a hydraulic Saehan dynamometer (Saehan Corporation, 973, Yangdeok-Dong, Masan 630-728, Korea). The participant will be positioned seated with feet on the floor, hips and knees at 90° of flexion and without armrest. The shoulders will be positioned in adduction and neutral rotation, the elbow will be positioned at 90° of flexion, with the forearm and wrist in a neutral position. Three maximum movements will be performed with a 1-minute rest between them and the average of the values will be recorded.27,28 The Maximal Voluntary Isometric Contraction (MVIC) of the quadriceps muscle will be evaluated using a spring gage attached to the leg by a strap with Velcro, positioned in the distal third of the dominant lower limb, with the individual sitting in a chair, with 90° hip flexion and 90° knee flexion. The participant will be asked to perform the knee extension, followed by a maximum and rapid contraction, maintaining it for 3 seconds. Three repetitions will be performed, with an interval of 1 minute between them and the average of the values will be recorded. 29
Functional state
The London Chest Activity of Daily Living (LCADL) scale, composed of 15 activities of daily living performed in real life, will be used, divided into 4 domains: personal, domestic care, physical activity and leisure, with higher values on the scale indicating a greater limitation in activities of daily living. 30 For this outcome, we will consider an increase of 3 points as a MCID. 31
Dyspnea and fatigue
Dyspnea during activities of daily living will be assessed using the Modified Medical Research Council (mMRC) scale, whose score ranges from 0 to 4, with a high score reflecting a worse sensation of dyspnea. 32 We will consider an increase of 1 point as a MCID for mMRC. 33
Fatigue will be assessed using the Brazilian version of the Fatigue Severity Scale (FSS-BR), consisting of 9 items with statements about symptoms and the severity of fatigue in daily life, with the score for each statement ranging from 1 to 7, number 7 being the maximum level of agreement and 0 no agreement. We will consider values >4 as the presence of fatigue. 34
Quality of life
The COPD Assessment Test (CAT) questionnaire verifies the impact of the disease on quality of life, and consists of 8 items—cough, discharge, chest tightness, shortness of breath, limitation in daily activities, confidence to leave home, sleep and disposition, being that for each item the score varies from 0 to 5, and the sum goes from zero to 40 points, where ⩾31 points indicate the impact of the disease on the quality of life. 35 For this outcome, we will consider a 2 point increase as a MCID. 36
Statistical methods
The collected data will be organized in a spreadsheet and summarized using descriptive statistical analysis techniques. Qualitative variables will be summarized through the construction of frequency tables and quantitative variables, through the calculation of descriptive measures (mean, median, 25-75 percentiles and standard deviation). Data analysis will be performed using the Statistical Package for the Social Sciences (SPSS) software, version 22.0 for Windows. Normality will be assessed by the Shapiro-Wilk test. To compare the difference between the groups, the difference between the post and pre-intervention results will be calculated (Δ = post - pre). For the inter-group analysis (pre × pre; post × post and Δ × Δ), independent Test-T or Mann-Whitney will be performed. For intra-group comparison (pre and post-protocol) the T-test in pairs or Wilcoxon Signed rank test and the chi-square test will be used for categorical variables. The level of statistical significance established will be P < .05.
Discussion
According to an extensive literature review, there are a few randomized clinical trials that investigate the possible benefits and safety of a multicomponent physical training program on clinical and physical-functional outcomes in patients with COPD.
Following the growth line of population aging, there will likely be an increase in COPD cases worldwide. Since physical training is a fundamental part of a PR program, 3 it is necessary to search for alternatives that guarantee a greater offer of rehabilitation services for patients, given the high demand and the benefits achieved through PR. Most of the candidates for a PR program do not have access to this service, or even any follow-up with a physiotherapist, a fact that is even more accentuated in developing or underdeveloped countries.
This study is expected to demonstrate that multicomponent physical training using simple and low-cost resources is a safe and effective intervention for the improvement of physical and functional outcomes. In addition, it is hoped that this study can broaden the horizon of research in relation to new methods of physical rehabilitation for these patients, facilitating access to therapies with physical exercises.
Our study has some limitations and weaknesses, including the fact that patients diagnosed with mild or very severe COPD were excluded from the research to homogenize the data analysis, as these patients may represent 2 extremes—not having symptoms or being very dependent due to severity of the disease. Another limitation would be the non-blinding of the participants and physiotherapists who will conduct the exercise sessions, however, in the case of interventions with exercises, this fact is impossible.
Physiotherapy is the science that studies, diagnoses, prevents and treats dysfunctions of human body systems, and the professional physiotherapist is fundamental to conduct a successful PR program, however, there is a great demand for work on these professionals in this area. If the desired benefits are found with MPT, our study may serve as a guide for physiotherapists who work with this population. Taking into account the scientific evidence of physical training today, the physiotherapist needs to make significant investments to offer the training recommended by the guidelines. Therefore, if effective and safe, our results may simplify and expand the offer of treatment through physiotherapy for patients with COPD. On the other hand, if our intervention is not proven to be effective, our study can guide future researchers to seek effective alternatives that involve few resources and manage to reach patients who have difficult access to conventional rehabilitation programs.
In addition to the main objective of our study, our efforts are also aimed at awakening other researchers to seek viable, safe and effective interventions for clinical practice, bringing science closer to the daily lives of health professionals and users of the system.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Camila Monteiro Mazzarin: writing and revision of the article; Bruna Roberta Silveira: writing and revision of the article; Ana Cristina Lamezon: article review; Bruna Cavon Luna: article review; and Silvia Valderramas: writing and revision of the article.
