Abstract
Urban rivers in rapidly urbanizing low-income countries frequently receive untreated wastewater, converting downstream irrigation systems into persistent exposure interfaces within a wastewater-agriculture-health nexus. This study quantified Escherichia coli and total coliform concentrations in irrigation water from 16 sites along the Akaki and Dongora Rivers in Gelan, Ethiopia, over 7 weeks (n = 112) using membrane filtration on CHROMagar™ ECC to assess spatiotemporal fecal contamination loads. Indicator bacteria were detected at all locations, with concentrations routinely surpassing internationally accepted limits for irrigation water by several orders of magnitude. The Escherichia coli concentrations ranged from 1.10 × 105 to 2.82 × 106 CFU/100 mL and total coliforms from 1.13 × 105 to 2.95 × 107 CFU/100 mL, with Akaki River loads substantially higher than the Dongora River (P < .05), suggesting spatial variance from distinct contamination sources. Conceptually, exceedances position the irrigation corridor in a high-risk exposure domain, where regular agricultural activities may enhance occupational and foodborne transmission. While indicator bacteria do not directly quantify pathogen risk, their magnitude indicates elevated public health vulnerability. The site-specific evidence emphasizes the need for risk for health-based monitoring, including continuous microbial surveillance, and farm-level exposure reduction strategies to safeguard livelihood and downstream environmental health.

Plain Language Summary
This research examined the quality of river water used for irrigation in Gelan agricultural fields, located near Addis Ababa, Ethiopia. We examined the microbial pollution load in two rivers: Akaki and Dongora. The findings revealed that both rivers had significantly higher concentrations of Escherichia coli (E. coli) and total coliform bacteria, indicating the rivers’ water had severe pollution from anthropogenic interferences. The presence of indicator bacteria indicates the presence of other disease-causing bacteria in the water. Therefore, there may have been elevated concentrations of pathogens, including Salmonella, E. coli O157:H7, Cryptosporidium, Campylobacter, rotavirus, and Hepatitis A, which are associated with gastrointestinal diseases in Gelan farm irrigation water. However, the farmworkers regularly use this contaminated river water for irrigation, cooking, handwashing, and bathing. More than 87% of farmworkers reported accidentally ingesting the water during irrigation, and approximately 78% ate uncooked crops irrigated with the river. Furthermore, 77% did not use any personal protective equipment (PPE). Moreover, 23% of the farmworkers reported experiencing gastrointestinal disease symptoms within two weeks of the research, which might have been caused by this water. This public health risk could extend beyond farmworkers to consumers. In particular, vegetables irrigated with this water and eaten raw can spread the risk into the consumer’s communities. To mitigate these risks, immediate public health actions, including awareness campaigns for farmers and legislation that encourages safer irrigation techniques, should be implemented. Future studies should also investigate long-term patterns and employ mapping technologies to identify high-risk locations for focused intervention.
Background of the Study
Globally, lacking sanitation infrastructure, rapid population growth, and unregulated waste disposal are contributing to widespread microbial contamination in urban rivers, thereby creating significant exposure pathways within the wastewater-agriculture-health nexus. 1 Consequently, polluted these rivers waters serve as primary pathways for the spreading of waterborne illnesses, which pose considerable risks to occupational safety and public health. 2
In Ethiopia, the Akaki River exhibits these kinds of challenges because it receives untreated household, industrial, and agricultural effluents from Addis Ababa City, making it highly vulnerable to microbial contamination. 3 Moreover, several previous investigations have consistently reported microbial concentrations in the Akaki River exceeding permissible standards for safe irrigation,4 -6 showing persistent deterioration of the water quality and a high probability of human exposure through routine agricultural and livelihood activities. 7 Despite significant evidence of contamination, the river continues to be extensively utilized downstream in the Gelan area for irrigation, washing, bathing, fishing, swimming, and other livelihood activities. As a result, multiple pathways for occupational and foodborne infection exposure have developed for local populations.
Furthermore, seasonal dynamics exacerbate the risks: during the dry season, the irrigation demand peaks while river volume decreases, resulting in higher bacterial concentrations 2 and thereby heightening the exposure probability among farmworkers. 4 Nevertheless, data on contaminated water-agriculture- health nexus remains limited in the farm region. This gap is especially critical given that residents in Gelan rely substantially on agriculture irrigated with Akaki River. To address these knowledge gaps, this work applies an indicator bacteria-based assessment, assessing Escherichia coli and total coliform concentrations across multi-sampling locations of Gelan Farm sites. By merging spatiotemporal sampling with exposure consideration, it establishes a scientifically plausible baseline of contamination. Importantly, it situates microbiological data within a broader wastewater–agriculture–health conceptual framework, pushing beyond basic descriptive reporting. These site-specific insights give critical knowledge to enable targeted risk reduction strategies, inform irrigation water management, increase wastewater monitoring, and strengthen public health protection in water-constrained, fast-urbanizing circumstances.
Likewise, our study links local observations with worldwide evidence from low- and middle-income nations, where untreated or partially treated wastewater is commonly reused for agriculture, and high indicator bacterial loads are connected to both occupational exposure and foodborne transmission. Therefore, the findings not only cover a significant knowledge vacuum for Gelan but also contribute to a larger understanding of microbial contamination and health hazards in peri-urban irrigation systems worldwide. By situating indicator bacteria measurements within a conceptual risk-oriented lens, this study ultimately provides a solid foundation for evidence-based interventions, regulatory oversight, and sustainable water security strategies in settings facing competing demands on limited freshwater resources.
Materials and Methods
Study Area Description
The study was conducted in the Gelan Farm Area, located in southeastern Sheger City, central Ethiopia, a region noted for vast irrigation farming that relies mostly on surface water from the Akaki River. Geographically, the area is located between 8°43′N and 8°55′N latitudes and 38°46′E and 38°56′E longitudes, with elevations ranging from 1800 to 2300 m above sea level. It experiences a tropical highland climate, a mean annual temperature of approximately 19°C, and rainfall of about 861 mm, most of which occurs between mid-June and mid-September. 8
Four kebeles, Gemeda, Hechu, Marino, and Abba Samuel, were selected for sampling because of their strong dependence on the Akaki River for irrigation of agricultural production (Figure 1). The communities in the area primarily sustain their livelihoods through smallholder farming, which increasingly relies on the river water for crop irrigation. The Sidamo Awash agricultural site was also included for comparison. The Sidamo Awash site is irrigated by the Dongora River, which is not directly affected by wastewater from Addis Ababa because it originates from the Oromia Region. The irrigation period typically extends from November to June, during the dry season when river discharge declines. Nevertheless, yearly variations in the rainfall can shift this irrigation time range depending on the length of the season.

Study area map showing spatial distribution of water sampling points across Gelan farm sites, along Akaki and Dongora Rivers.
Study Design and Sampling Strategies
The sampling locations were purposefully selected based on factors such as irrigation intensity, proximity to the river channels, accessibility to river systems, and their relevance to farmworker exposure routes (Figure 2). Totally, 16 sampling sites were selected, comprising 13 sites along the Akaki River and 3 sites along the Dongora River.
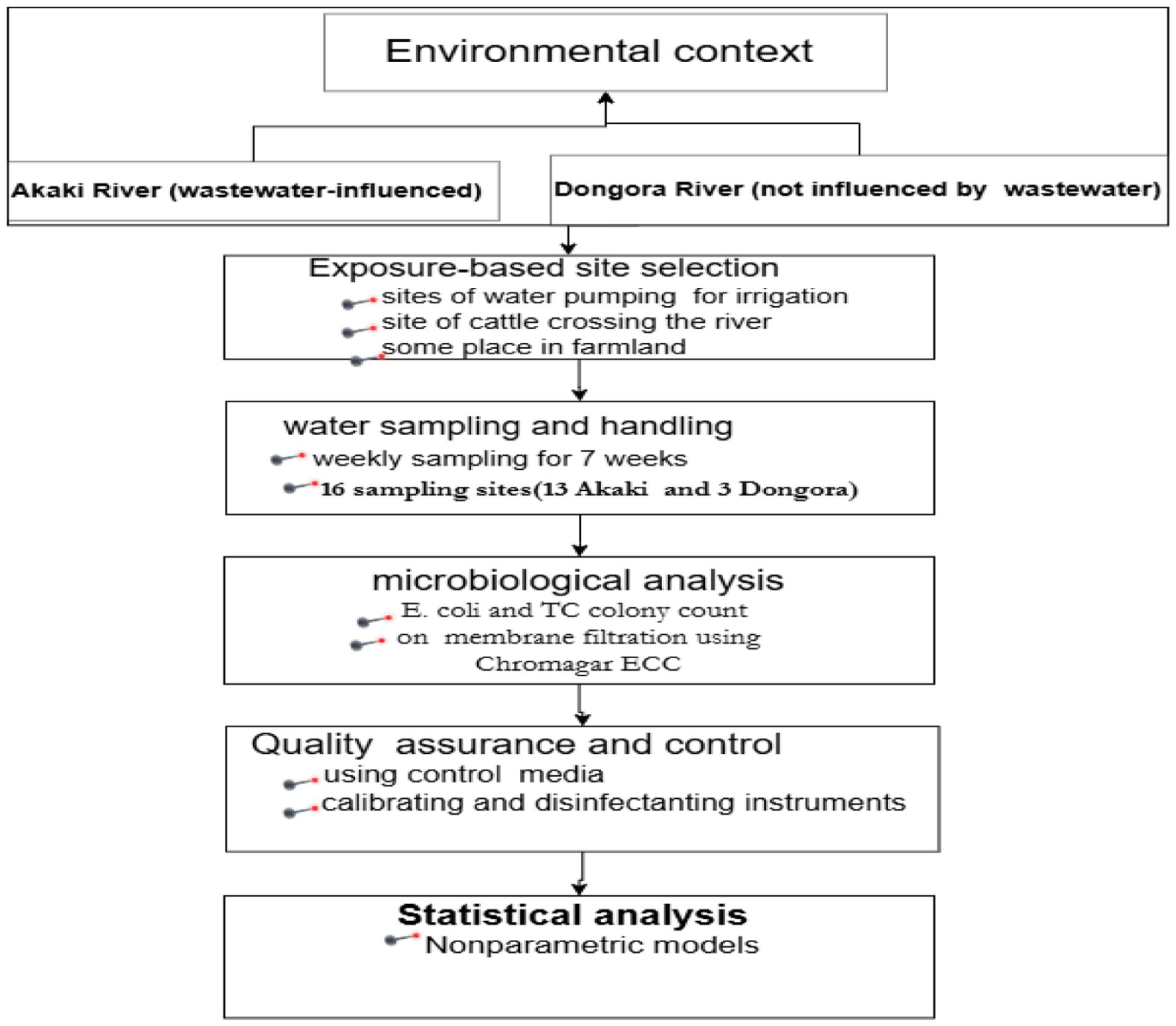
Conceptual framework of the indicator – bacteria based methodology integrating environmental context, exposure-driven sampling, microbiological analysis, and statistical evaluation within wastewater-agriculture- health nexus across Akaki and Dongora Rivers irrigation systems, Gelan, Ethiopia (2024).
Water Sample Collection and Handling
The water sampling followed internationally established protocols for the microbiological testing of surface water. The samples were collected in sterile 500 mL polypropylene bottles at approximately 20 to 30 cm below surface water to eliminate surface coatings and debris. Bottles were angled upstream during collecting to minimize contamination from the surroundings. Immediately after collection, samples were stored in iceboxes and taken to the microbiology laboratory, the Aklilu Lemma Institute of Pathobiology, for processing within 6 hours, according to suggested times for bacteriological analysis. The data collectors followed the aseptic protocols, including the use of sterile gloves and minimal sample-contaminating handling.
Microbiological Analysis
Then the samples were serially diluted to obtain countable colony ranges and analyzed using the membrane filtration techniques, a standard approach for quantifying fecal indicator bacteria in water. The filtered membranes were cultured on chromogenic (CHROMagar™ ECC), a selective chromogenic medium that enables simultaneous differentiation of Escherichia coli (E. coli) and total coliform bacteria. Duplicate plating was done for each sample to enhance analytical reliability. Following incubation at 37°C for 24 hours, colonies were enumerated using appropriate dilution factors and reported as colony-forming units (CFU) per 100 mL of water.
Quality Assurance and Quality Control
Detailed Quality Assurance and Quality Control (QA/QC) procedures were implemented throughout sampling and laboratory analysis to ensure data integrity. The culture media were sterilized prior to use, and controls were processed along with samples to detect procedural contamination. Incubators were routinely calibrated to preserve temperature stability, and all filtration equipment was sanitized before each analysis batch.
Data Analysis
All statistical analyses were executed using R software (version 4.4.1). Explanatory data assessment indicated a non-normal distribution of bacterial counts; therefore, nonparametric tests were employed. The Kruskal-Wallis test was used to evaluate the differences in microbial concentrations across irrigation sites. The Wilcoxon rank sum test was employed to compare bacterial concentrations between the Akaki and the Dongora Rivers. Statistical significance was set at alpha .05, and 95% confidence intervals were computed to quantify estimation uncertainty. The graphical summaries included were generated to visualize the distribution patterns, site-level variability, and central tendencies of microbial concentrations.
Results
Indicator Bacteria Concentrations in Irrigation Water
The study generated quantitative evidence of substantial microbial contamination across irrigation sites in the Gelan farming area. Concentrations of both total coliform (TC) and E. coli consistently exceeded recommended irrigation water quality thresholds (Table 1).
Statistical Summary of Total Coliform (TC) and Escherichia coli (E. coli) Concentrations (CFU/100 mL) Across 16 Irrigation Water Sampling Sites in the Akaki and Dongora River Systems, Gelan Farm, Ethiopia, 2024.

Values are presented in minimum, mean, maximum, and 95% confidence interval (CI).
Total coliform counts ranged from 1.13 × 105 CFU/100 mL at Andode to 2.95 × 107 CFU/100 mL at Water Development 01 and Water Development 5, with mean concentrations varying from 8.00 × 105 CFU/100 mL to 9.24 × 106 CFU/100 mL. Likewise, E. coli concentrations ranged from 1.1 × 105 CFU/100 mL (Water Development 10 farm site) to 2.82 × 106 CFU/100 mL (Sidamo laundry site), while the mean value varied between 1.79 × 105 and 9.09 × 105 CFU/100 mL. The estimated 95% confidence intervals (CIs) revealed substantial variability in both the indicator bacteria across sampling locations. Escherichia coli concentration levels at all sites exceeded the World Health Organization (WHO) recommended thresholds for unrestricted irrigation (⩽103 CFU/100 mL) by 2 to 4 orders of magnitude. Overall, concentrations of indicator bacteria in the irrigation of Gelan Farm exceeded safe irrigation water standards and showed spatial variability across the sampling sites. Collectively these results indicate persistent and spatially heterogeneous fecal contamination throughout the irrigation network of Gelan Farm sites.
Spatiotemporal Trends of Microbial Contamination in the Study
The following multifaceted line graph reveals both site and over-time variations of the bacterial concentrations in the irrigation water. Throughout the 7-week study period, bacterial concentrations revealed a steadily growing tendency. The graph reveals that both total coliform and E. coli concentrations were consistently high across all sampling sites and during the whole investigation period (Figure 3).
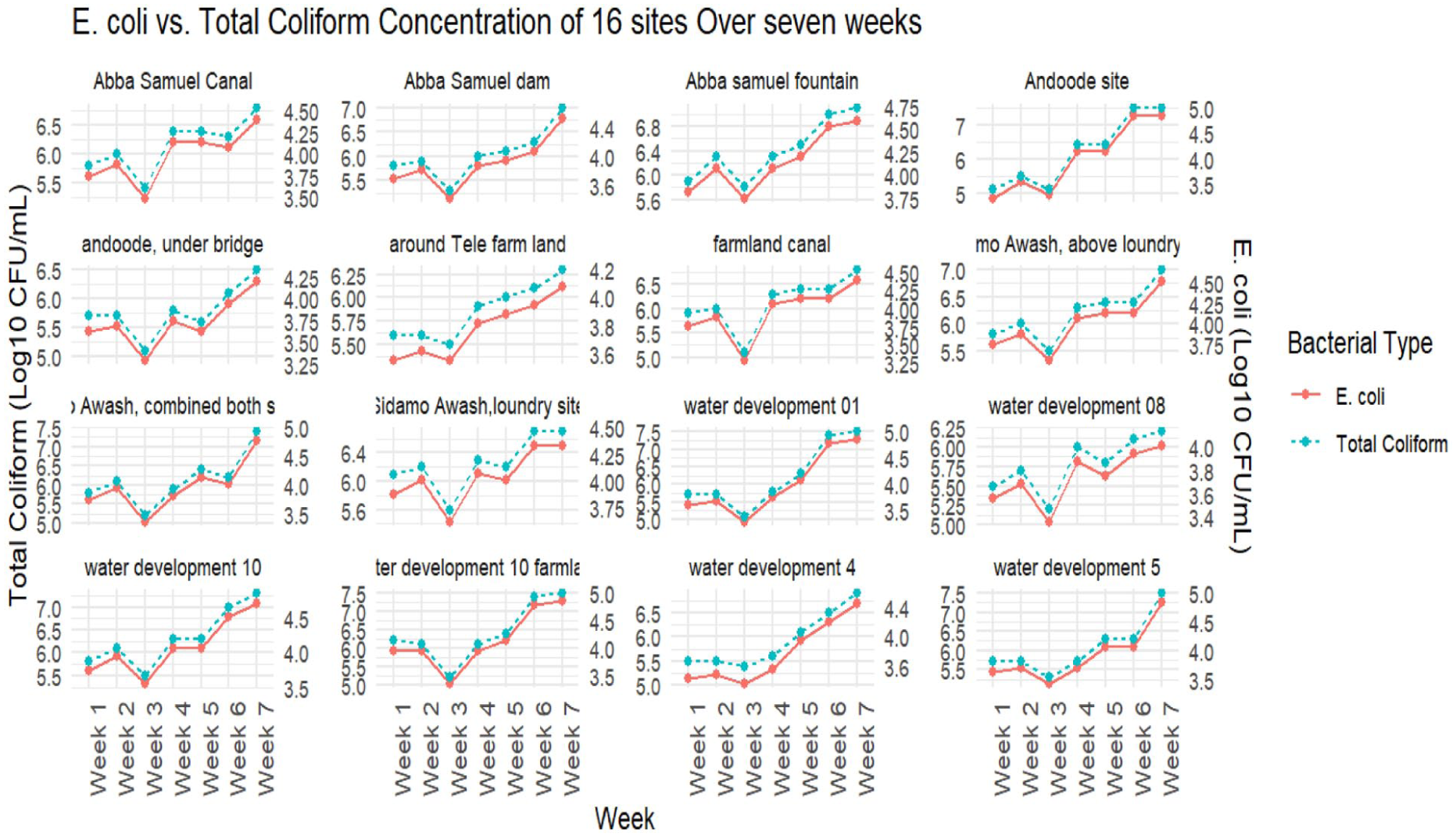
Faceted line graph with overlaid scatter points illustrating the temporal variations in Escherichia coli and total coliform (TC) concentrations across 16 sites in Akaki River, and Dongora Rivers over 7 weeks sampling periods at Gelan farm, 2024.
Additionally, the distribution of the bacteria concentrations across sampling time is presented in the following boxplot graphs (Figures 4 and 5). The graphs summarize weekly variations of total coliform and Escherichia coli concentrations throughout the study period. Elevated median values and wider interquartile ranges were observed during the final 2 weeks for both indicators. The weekly distributions of E. coli displayed a gradual upward shift in central tendency across successive sampling rounds, while TC concentration showed a relative reduction in median values at week 3.

Boxplot graph illustrating the distribution of Escherichia coli counts over 7 weeks study periods (middle April to early June, 2024).

Boxplot graph illustrating the distribution of total coliform counts over 7-week study periods (middle April to early June, 2024).
Furthermore, substantial variation in the bacterial concentrations was detected among sampling sites (Figures 6 and 7). For instance, the Sidamo Awash laundry site, Andoode water development 10 field, Abba Samuel fountain, and water development 5 consistently recorded the highest quantities of E. coli (Figure 5). Similarly, the Andoode site, water development 01, water development 10, and the Abba Samuel fountain recorded the high concentrations for TC (Figure 6) compared to other sites (P < .05). Nevertheless, sites such as Abba Samuel Reservoir, the farmland near Tele, and the farmland canal sites exhibited relatively lower bacterial concentrations. Moreover, the Abba Samuel Dam and Sidamo Awash sites showed a consistently increasing trend in bacterial concentrations over time, whereas other sites maintained uniform concentration levels.

Boxplot graph illustrating distribution of Escherichia coli (E. coli) counts across 16 sites over 7-weeks (middle April to early June, 2024) at Gelan farm sites.
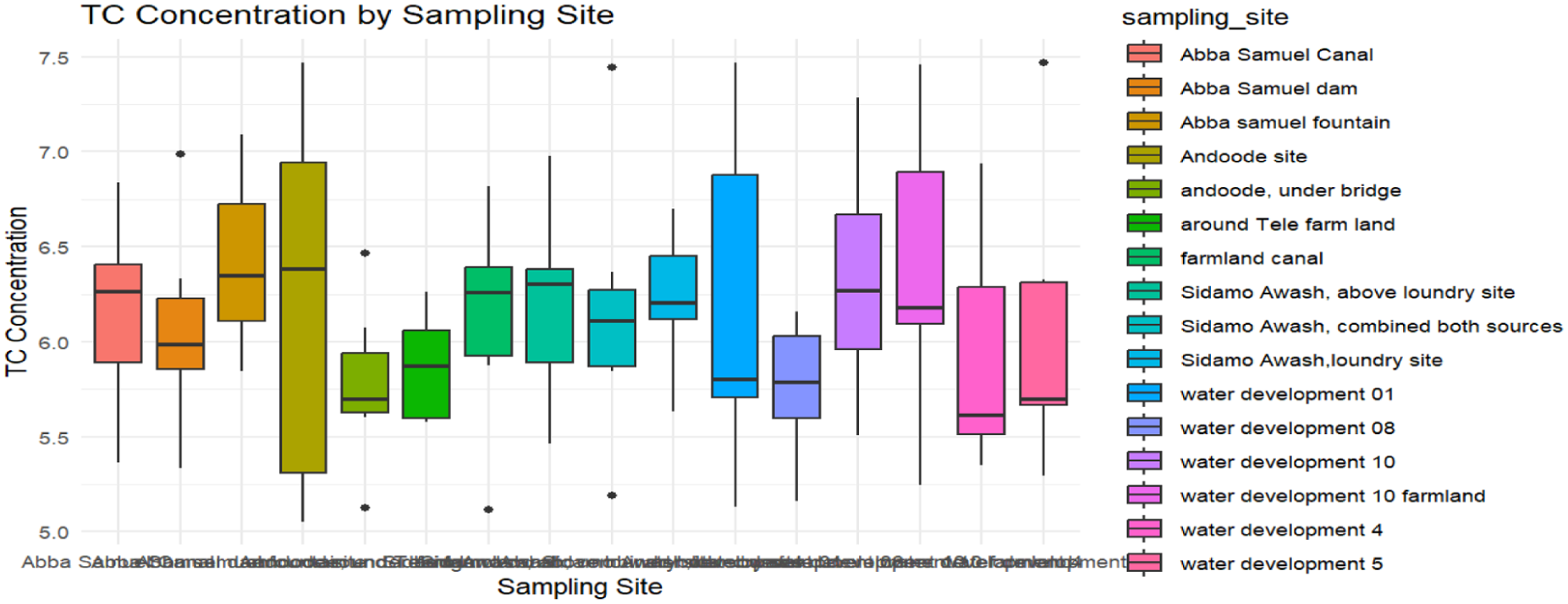
Boxplot graph illustrating distribution of total coliform (TC) counts across 16 sites over 7-weeks (middle April to early June, 2024) at Gelan farm sites.
The Kruskal-Wallis test indicated considerable temporal variability in E. coli and total TC across the farm sites over the 7-week study period (P = .01). Additionally, pairwise Wilcoxon comparison tests showed a statistically significant discrepancy in E. coli concentrations between the Akaki and Dongora Rivers irrigation sites (P = .02), with consistently higher levels observed in the Akaki River. Conversely, TC concentrations showed no significant variation between the 2 rivers (P = .54). Regarding hygiene practices and health concerns of farmworkers, consistent with microbiological findings, survey data (Table 2) revealed broad exposures to irrigation water among farmworkers. The majority of the workers reported consuming raw produce irrigated with river water (78%, n = 231) and unintentionally ingesting the irrigation water while working on the farm (87%, n = 259). Using river water for domestic purposes, handwashing (63.2%, n = 187), cooking (38%, n = 112), and bathing (20.6%, n = 61) was also common. Only a small proportion of farmworkers reported using river water for drinking (8%, n = 23), and the majority did not employ personal protective equipment (77.4%, n = 22). In alignment with these patterns, 23% of farmworkers (68 out of 296) reported gastrointestinal illness within 2 weeks prior to data collection.
Sanitation Practices Among the Farmworkers in Gelan Farm Sites, Along Akaki River and Sidamo Awash Farms (n = 296), 2024.

Discussion
The present indicator bacteria-based assessment shows empirical evidence that irrigation water in the Gelan agricultural area is extensively contaminated. The observed bacterial counts are substantially above WHO recommended 1000 CFU/100 mL for irrigation water quality, indicating a chronic microbial contamination burden within the irrigation system. 9 Within the wastewater–agriculture–health nexus, such high-order magnitude concentrations consequently indicate both water quality deteriorations and a high possibility of exposure risk for populations in daily contact with irrigation water.5,10
These findings are consistent with previous investigations conducted along the Akaki River system, which reported that E. coli concentrations in irrigation water, soils, and vegetables substantially exceeded international safety guidelines.6,11,12 This demonstrates the widespread of fecal contamination within peri-urban agricultural zones of Addis Ababa. Similar patterns of high microbial contamination have been documented in wastewater-irrigated farming systems in Sub-Saharan Africa and other low- and middle-income countries where wastewater discharge into surface water bodies occurs with limited treatment or regulatory oversight.13,14 Additionally, the finding agrees with recent studies from other places, peri-urban agricultural systems where untreated or partially treated wastewater is routinely reused for irrigation.15,16 Importantly, fecal indicator bacteria serve as a well-established indicator for the prevalence of enteric pathogens in ambient waters. Therefore, elevated concentrations of E. coli and total coliforms clearly indicate a possible threat of waterborne disease transmission through agricultural exposure pathways. 17
Spatial Variations of Bacterial Contamination
The spatial variation of bacterial contamination among irrigation locations is another key observation arising from this investigation. Specifically, microbial concentrations differed significantly between sampling locations, demonstrating that pollution levels are influenced by local environmental factors and anthropogenic activity. For example, sites located near heavily inhabited communities, wastewater discharge points, or agricultural runoff areas tended to exhibit elevated bacterial burdens. The regional contamination variability of our study is comparable with findings reported in other wastewater-irrigated systems, where microbial contamination trends often reflect specific pollution sources and hydrological characteristics.4,18 Moreover, river flow dynamics, sediment resuspension, and microbial persistence processes may contribute to site-specific alterations in bacterial contamination loads. 19 Therefore, the observed regional variability patterns indicate the need for site-level monitoring for assessing irrigation water safety.
The Akaki and Dongora Rivers Comparison
The comparative analysis between the Akaki and Dongora Rivers further shows variations in microbial contamination patterns. Despite elevated quantities of indicator bacteria reported in both rivers, statistical comparisons indicated differences in the concentrations across the 2 systems. These disparities may be due to differing land-use patterns, wastewater discharge intensities, and hydrological factors between the rivers’ catchments.
The Akaki River, which flows through densely urbanized areas, is known to absorb large untreated municipal and industrial wastewater inputs before reaching downstream agricultural zones. 20 As a result, irrigation sites supplied by this river are particularly influenced by fecal pollution. In contrast, the Dongora River flows through more less populated areas, which may partially explain the considerably lower bacterial counts recorded in these sites. In this farm area, possible pollution sources include poultry farm wastewater, residuals from farmland, laundry discharge, and open defecation practiced near irrigation canals. The poultry waste disposal to irrigation streams surely raises microbial contamination, since concentrated animal production systems are acknowledged reservoirs of fecal indicator bacteria and zoonotic pathogens. 21 Similarly, animals crossing over a river may contribute to contamination by direct fecal deposition and sediment disruption. Particularly, under low-flow conditions, livestock activity can resuspend sediment-bound microorganisms, thereby increasing bacterial densities in the water column. 22 Animal waste inputs can bring considerable nutritional loads that increase microbial survival and persistence in aquatic systems. 23
Attribution of Hydrological Variability
The fluctuations may arise from hydrological changes factors. Hydrological seasonality additionally influences microbial dynamics by regulating dilution, transport, and accumulation processes. 24 During dry seasons, lower river discharge declines dilution capacity while irrigation demand rises. Under these conditions, fecal bacteria may concentrate within irrigation water, increasing the chance of exposure for farmworkers. Similar seasonal contaminant concentrations have been reported by Hiruy et al 4 in the same region, where the lower flow during dry season increases pollutant buildup. Conversely, several other findings argue that rainfall events may contribute extra pollutants through surface runoff while concurrently increasing downstream transport and partial dilution.25,26 These periodic mechanisms therefore moderate, rather than eradicate, microbial contamination pressures inside the irrigation system.
Public Health and Food Safety Implications
Beyond environmental contamination, the findings of this study reveal 2 serious public health consequences. Irrigation with microbiologically polluted water provides several exposure paths that may affect both farmworkers and the irrigation farm produce consumers. The actions like direct skin contact with irrigation water, consuming produce irrigated with contaminated water and accidental ingestion during agricultural work are common in irrigation farm. 27
From a quantitative microbial risk assessment (QMRA) approach, these regular exposure to pathogenic can considerably increase infection probability.28 -30 Although indicator organisms cannot precisely test individual diseases, their number is universally acknowledged as directly connected with microbial risk in the systems. 14 Therefore, within the wastewater–agriculture–health nexus framework, irrigation water acts as a critical transmission interface connecting environmental contamination with human health outcomes. Several studies have shown that exposure to fecal-contaminated irrigation water increases the prevalence of gastrointestinal infections among farmworkers and communities living near wastewater-irrigated farming areas.16,31
The microbiological health concerns in the irrigation water extend beyond farmworker health to broader food safety problems. Irrigation with untreated wastewater could facilitate microbial translocation from unclean water to fresh food surfaces, particularly green crops that are normally consumed raw. Evidence from similar wastewater-irrigated agricultural systems suggests that fecal bacteria can be transmitted from irrigation water to vegetables and subsequently to consumers within urban food distribution networks. 5
Implications for Irrigation Water Safety and Management
From a policy perspective, the findings underline the urgent need for improved irrigation water safety management in wastewater systems. The associated microbiological risks require targeted risk-reduction strategies, although wastewater irrigation supports agricultural productivity and provides livelihood opportunities for farmers.
The World Health Organization recommends a multi-barrier approach for safe wastewater reuse in agriculture, which includes wastewater treatment, safer irrigation practices, crop restrictions, post-harvest hygiene measures, and farmworker health protection interventions. 9 Implementing such approaches in the Gelan irrigation system could significantly minimize workers’ exposure likelihood to microbial risks while sustaining the economic benefits of wastewater reuse. Additionally, improving water quality monitoring systems is essential for identifying contamination hotspots and supporting evidence-based decision-making. Further, indicator bacteria based regular microbiological monitoring provides a viable and cost-effective technique to assess irrigation water safety, particularly in places with limited resources. This monitoring approaches can enable the development of adaptive water safety management policies when linked with geographical risk assessment and public health surveillance.
Contribution and Novelty of the Study
This study presents new scientific information on microbial contamination in wastewater-dependent irrigation systems within agricultural environment. The primary role is to generate baseline data on fecal indicator bacteria (Escherichia coli and total coliforms) at various irrigation sites along the Akaki and Dongora Rivers. The study methodologically employs chromogenic differential culture media (CHROMagar™ ECC) alongside membrane filtration techniques to facilitate the concurrent visualization and differentiation of E. coli and total coliform colonies. Furthermore, the investigation utilizes robust non-parametric statistical analyses to assess spatial variations in microbial contamination levels across the irrigation sites and associated irrigation rivers. A further key contribution lies in the comparative analysis of the Akaki River, which receives wastewater discharges from Addis Ababa, and the Dongora River. This comparison provides a useful environmental reference to identify contamination trends associated with wastewater influence from those emanating from local agriculture or environmental sources.
Moreover, the research elucidates the potential occupational and food safety hazards linked to wastewater-irrigated agriculture by correlating microbial contamination levels with documented health problems experienced by exposed farmworkers. These combined contributions offer methodological improvements, empirically grounded data, and a nexus-based interpretation, thereby facilitating monitoring, risk assessment, and irrigation water safety management in peri-urban agricultural systems characterized by rapid urbanization and escalating wastewater reuse.
Synthesis
Collectively, the findings demonstrate that microbial contamination in the Gelan irrigation system is persistent, spatially structured, seasonally controlled, and mechanistically promoted by both anthropogenic inputs and ecological interactions. By embedding indicator bacteria measurements within a wastewater–agriculture–health nexus framework, this study provides a rigorous, risk-oriented foundation for improving irrigation water safety in peri-urban environments.
Conclusions
This study provides a robust baseline assessment of microbiological water quality within the Gelan irrigation area. The empirical evidence from the Akaki and Dongora river systems demonstrates chronically elevated loads of total coliforms and Escherichia coli that substantially exceed internationally recommended safety thresholds for irrigation water. Within the conceptual wastewater–agriculture–health framework, these contamination levels indicate a high probability of infection among farmworkers and consumers in the absence of protective interventions. In addition to local significance, the study provides statistically validated spatiotemporal evidence to improve risk-based irrigation monitoring in Ethiopia and other low- and middle-income countries. The findings stress the urgent need for targeted risk-reduction efforts aimed at enhancing irrigation water safety and behavioral interventions.
Limitations
Although the study provides novel contributions, it also acknowledges several limitations. First, the single seasonal sampling approach may not capture all hydrological events that may alter contamination dynamics. The analysis consequently captures conditions during the irrigation season but does not represent the larger raining season hydrological variability of the river system. Second, the use of an indicator bacteria-based assessment delivers indirect signs of contamination but does not directly measure pathogen presence or infectivity. Additionally, culture-based detection techniques may also underestimate living non-culturable species. Finally, the absence of microbiological source tracking hinders the ability to distinguish between human and animal fecal contamination sources.
Future Research Directions
Therefore, future investigations should integrate molecular pathogen detection, microbial source tracking, and quantitative microbial risk assessment (QMRA) modeling to convert environmental contamination data into quantitative estimates of infection risk. Longitudinal monitoring across multiple hydrological seasons would also improve understanding of temporal contamination patterns.
Footnotes
Acknowledgements
I would like to express my sincere appreciation to the Aklilu Lemma Institute of Pathobiology for their exceptional laboratory support, which was instrumental in successfully completing the laboratory of this study. My heartfelt gratitude goes to my dear friend Mr. Merga Lebeta for his unwavering support throughout the entire sample-collecting process, starting from the initial stage to the final phase. This study would not have been thoughtful without his devotional engagement and commitment.
Ethical Considerations
The study’s ethical approval was obtained from the Ethics Review Committee of the Oromia Emergency Research Bureau. The committee thoroughly assessed the study’s goals, procedures, and possible dangers to ensure consistency with ethical standards and to protect participants’ rights and well-being. Additionally, all study techniques were created in line with appropriate ethical criteria, guaranteeing adherence to ethics of secrecy, voluntary participation, and informed consent.
Author Contributions
Iyasu Gudisa: developed the concepts of study and designed the study, conducted data collection and statistical analysis, and wrote the original manuscript.
Bezatu Mengistie: Contributed to the development and refinement of the study methodology and provided overall scientific guidance throughout the research process. Adey Feleke Desta: Provided guidance for laboratory work and assisted in the execution of experimental procedures.
Haile Alemayehu supervised technical supervision and laboratory activities and supported the implementation of experimental tasks. Sirak Robele Gari: Supervised the development of the study concepts, prepared manuscripts, provided critical revisions to the contents, and edited the manuscript to enhance clarity, coherence, and overall quality.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used analyzed during the current study available from the corresponding author on the reasonable request.
