Abstract
Environmental exposure to heavy metals during pregnancy may adversely impact fetal development, particularly in low- and middle-income countries (LMICs) with limited environmental regulation. This study aimed to assess the relationship between maternal exposure to lead (Pb), cadmium (Cd), and mercury (Hg) and fetal growth parameters in an Indonesian population. A cross-sectional study of 127 pregnant women in their second trimester was conducted. Maternal characteristics and exposure sources were obtained via structured questionnaires. Blood concentrations of Pb, Cd, and Hg were measured using inductively coupled plasma mass spectrometry (ICP-MS). Fetal biometry was assessed by ultrasonography, and z-scores were calculated. Generalized additive models (GAMs) and linear mixed models (LMMs) with restricted cubic splines (RCS) were used to evaluate non-linear associations, adjusting for maternal age and secondhand smoke exposure. Among the 127 pregnant women, the median age was 30 years (range: 20-40 years), and the median blood concentrations of Pb, Cd, and Hg were 6.6, 0.022, and 0.021 µg/L, respectively. Fetal growth Z-scores were as follows: BPD_z = −0.48 (range: −2.5 to 2.5), HC_z = −0.38 (range: −2.0 to 2.0), AC_z = −0.56 (range: −2.5 to 2.0), FL_z = −0.62 (range: −2.5 to 2.5), and EFW_z = −0.75 (range: −3.0 to 2.0). Using linear mixed models with restricted cubic splines (RCS), Cd and Hg exposures were significantly associated with increased fetal Z-scores (Cd: β = 1.48, P < .001; Hg: β = .99, P < .001), while Pb showed a negative association (β = −.93, P < .001). Non-smokers exhibited more favorable fetal growth responses compared to those exposed to secondhand smoke. Maternal exposure to heavy metals showed non-linear effects on fetal growth. The findings underscore the need for environmental health monitoring and preventive measures during pregnancy.
Introduction
Prenatal exposure to heavy metals such as lead (Pb), cadmium (Cd), and mercury (Hg) has been associated with adverse pregnancy outcomes, including impaired fetal growth.1,2 Nutritional deficiency is a determinant factor often referred to as the cause of low birth weight. 3 Environmental factors also play an important role. Pregnant women and their fetuses are vulnerable to the effects of exposure to various environmental toxins, including Pb, Hg, and Cd.4,5 Exposure to heavy metals during pregnancy can have serious impacts on the health of the mother and development of the fetus.6,7 Previous studies have shown that heavy metals can enter the placenta and affect fetal growth and development,8 -10 leading to small for gestational age (SGA) and low birth weight (LBW) infants.11,12
In Indonesia, exposure to these metals is primarily attributed to industrial pollution, agricultural practices, and food contamination. In Makassar, a growing urban center, exposure is exacerbated by industrial emissions, vehicular exhaust, contaminated water sources, and seafood consumption. Previous studies have reported heavy metal contamination in several commodities commonly used in Makassar. Chromium (Cr) in fish from the Tallo River in Makassar City reached a concentration of 16.9091 mg/kg. 13 The contents of Cd and Pb in the Anadara granosa clams were 0.7 and 5.9 mg/kg. In addition, 20 whitening cosmetics circulating in Makassar contain Hg 0.12 to 78 µg/g. 14
An increasing number of studies have documented correlations between maternal exposure to toxic metals and diminished fetal development.9,10,15 The studies initiated an examination of the correlation between mixtures of metals and fetal ultrasound metrics. The non-linear dose-response relationship has been investigated for various metals; however, the majority of these metals exhibit a linear correlation with fetal size. 15 Various methods have been implemented to explore the relationship, such as principal component analysis (PCA), 10 Bayesian kernel regression (BKMR), 9 linear regression,10,15 and weighted quantile sum (WQS) regression. 15 However, more methods are necessary to explore this relationship and to complete each others.
While the dose-response relationships between maternal metal exposure and fetal growth have been well -studied in high-income countries, there is a dearth of research on these associations in Southeast Asian populations, including Indonesia.16,17 Environmental exposure levels, dietary habits, and industrial practices in Indonesia may differ from those in more industrialized countries, 18 and these factors could influence the degree and nature of fetal exposure to toxic metals.19,20 In Indonesia, there were 11.3 neonatal deaths/1000 live births were reported in 2021. In Makassar, according to data from the Makassar City Health Office, 21 there was 717, 347, and 816 cases of low birth weight (LBW) in 2021, 2022, and 2023, respectively.
However, similar studies have rarely been conducted in Indonesia.5,17,22,23 Therefore, there is still limited data available across Indonesia to gain an understanding of the extent to which pregnant women are exposed to chemical substances in the environment and their effects on both mothers and children. Such data can serve as a foundation for the development of programs and policies aimed at protecting mothers and children from environmental exposure. 24
To contribute to the existing studies, we examined the relationship between maternal metal exposure and fetal growth in an Indonesian population by applying generalized additive models (GAMs), This study represents one of the few analyses focusing on prenatal metal exposure and fetal growth outcomes in the Indonesian population, offering novel insights into environmental exposure risks in this region. Given the potential for non-linear associations between metal exposure and fetal growth, we hypothesized that exposure to higher levels of Cd and Pb would disproportionately impact fetal outcomes. Furthermore, aggregated metal exposure reflects the combined burden of multiple environmental toxicants, which may amplify their impact on fetal development.
Methods
Data Collection
This study was a biomonitoring survey as part of an ongoing cohort study conducted at health centers in Makassar City, South Sulawesi, Indonesia (Figure 1). A total of 200 mothers participated in the data collection in the health Centers. Mothers who were in trimester 2 (pregnancy weeks: 13-24), living in Makassar for more than 1 year, planned to have a birth delivery in Makassar, and willing to participate in the survey were included in the study. Pregnant women with chronic diseases or who were unable to provide informed consent were excluded from the study. In the current study, 132 mothers were included.

Map of health centers location in South Sulawesi Province, Indonesia.
Face-to-face interviews were conducted using a questionnaire that included questions regarding their characteristics and possible exposure sources, such as environmental factors and dietary habits. The questionnaire was pre-tested in a pilot study involving a small sample of pregnant women to ensure clarity and reduce bias in the responses.
Heavy Metals Analysis
Venous blood samples (10 mL) were collected from each participant in tubes containing ethylenediaminetetraacetic acid (EDTA) by the analyst and stored at a temperature of 2 °C to 8 °C prior to analysis. In brief, blood samples were extracted from the EDTA tube with a 0.5 mL micropipette and then transferred to a test tube. Each sample was treated with 10 mL of 98% nitric acid (HNO3). The samples were deposited on a hotplate at a temperature range of 200 °C to 300 °C for destruction until the vapor at the edge of the tube dissipated. Each sample was treated with 49 mL of distilled water (Amidis). The sample was homogenized following the mixture, transferred to a 50 mL measuring glass and sealed with parafilm. The sample was filtered through 0.45 µm membrane filters prior to analysis.
Hg, Pb, and Cd were analyzed using inductively coupled plasma mass spectrometry (ICP-MS) (Thermo Fisher Scientific, Bremen, Germany) and performed in µg/L. The ICP-MS is a highly sensitive technique capable of detecting low concentrations of heavy metals. However, it is recognized that mercury, in particular, can present challenges in terms of sensitivity and measurement accuracy. In this study, a low method detection limit (MDL) of 0.03 µg/L was established for Hg, addressing sensitivity concerns, particularly for low-concentration samples.
To ensure analytical accuracy and method validation, we used Certified Reference Materials (CRM BCR®-636, lyophilized human blood, Joint Research Center – JRC, European Commission) as a quality control standard. For the quality assurance and quality control (QA/QC), calibration curves were generated using multi-element standard solutions of known concentrations. Blank samples were included in each analytical batch to monitor contamination. The recovery rates were within the acceptable limits (85%-115%). The method detection limit (MDL) for Hg and Cd was 0.03 and 0.1 µg/L for Pb. There were 83 and 68 subjects had blood Cd and Hg levels below the MDL, respectively. For statistical analysis, we imputed all undetected cases to MDL/√2 values.
Fetal Growth Assessment
Qualified sonographers conducted fetal ultrasound assessments in the second and third trimesters. These professionals were certified healthcare practitioners with expertise in the operational use of equipment. Measurements included biparietal diameter (BPD), head circumference (HC), abdominal circumference (AC), femur length (FL), and estimated fetal weight (EFW).
Covariates
In the current study, we included 2 maternal factors as covariates: secondhand smoke exposure (SSE) and maternal age. Information was obtained from the questionnaires. SSE was categorized as “everyday,” “often,” and “never.”
Statistical Analysis
Outliers in heavy metal concentrations were identified and excluded to improve data integrity. Specifically, 5 extreme values from the dataset of 132 pregnant women were removed, resulting in a final sample size of 127.
In this analysis, we encountered missing data for fetal measurements and pregnancy age because of the absence of mothers in the second or third measurements. Given the importance of these variables in our analysis, we followed a systematic approach to handle the missing data. The percentage of missing data was relatively low; therefore, a complete case analysis would have led to loss of valuable information. For missing values, we used multiple imputation by chained equations (MICE) to fill in the missing data. We selected the predictive mean matching (PMM) method for continuous variables, which ensured that the imputed values were within the range of the observed data. Missing values in fetal measurements were imputed based on relationships with other fetal measurements and pregnancy age variables. Missing values in pregnancy ages were imputed based on fetal measurements for the respective trimesters.
To account for variability in fetal growth measurements across gestational ages, Z-scores were calculated for each fetal measurement. These Z-scores represent standardized values relative to gestational age, allowing comparisons across subjects and trimesters. These standardized Z-scores represent the deviation of an individual’s measurement from the mean for a specific gestational age, scaled by the standard deviation. Z-scores were derived for each fetal growth measurement in both the second and third trimesters using reference growth charts to account for fetal size variation by gestational age.
Our preliminary analyses indicated that the relationship between metal exposure and fetal growth outcomes was not strictly linear. Thus, given the non-normal distribution of the data and the potential non-linear relationships between maternal metal exposure and fetal growth outcomes, GAM were used for the primary analysis. GAMs allow the application of smooth functions to continuous variables, enabling the detection of non-linear trends in data.
To address the potential skewness of the distribution of heavy metal concentrations, log-transformation was applied to normalize the data. Log-transformed concentrations were used in all subsequent analyses to enhance the model robustness and interpretability.
While the sample size of 127 participants is deemed adequate for the analyses conducted, it is important to note that smaller sample sizes may limit the power to detect certain interaction effects. Future studies with larger sample sizes and power analyses are needed to more definitively explore these relationships, especially for non-linear or complex interactions between metal exposure and fetal growth outcomes.
For each fetal outcome (Z-score for BPD, HC, AC, FL, and EFW), separate GAMs were fitted with smooth terms for continuous exposure variables (log-transformed Pb, Cd, and Hg concentrations) and maternal age. The model formula for each outcome is as follows:
Where:
s (⋅) represents the smooth terms applied to the continuous variables to account for the potential non-linear effects.
Smoking exposure was categorized as “never,” “often,” and “everyday.”
To capture the combined exposure effect of all metals on fetal outcomes, instead of analyzing each metal separately, we applied an aggregated approach by taking all metal concentrations and combining them into a single measure. Aggregation was performed using an additive combination by summing the log Pb, Cd, and Hg values.
All analyses were conducted using R software (version 4.3.2) with the mgcv package used for fitting the GAM models, and smoothness parameters were selected via restricted maximum likelihood (REML). The statistical significance of each smooth term was evaluated using approximate P values, and the degree of non-linearity in the relationships was captured by the estimated degrees of freedom (EDF). A higher EDF suggests a more complex, non-linear relationship between the predictor and the outcome.
Human Subjects Approval
Ethical approval for this study was granted by the Ethical Commission of the Faculty of Public Health at Hasanuddin University (reference number: 1289/UN4.14.1/TP.01.02/2024).
Results
Characteristics of Mother and Descriptive of Metal Concentrations and Fetal Growth Measurement
In total, 127 mothers were included in the final analysis (Table 1). The median age of the mothers was 28 years old (Table 1). More than 50% of the mothers had at least a high school education (Table 1). Most mothers were not working and gave birth to at least 1 child (Table 1). Table 2 presents the concentrations of blood heavy metals and the corresponding fetal growth measurements across different health centers. The results revealed substantial variability in heavy metal exposure across the surveyed health centers. Pb levels were highest in Health Centre of Sudiang (39.60 ± 13.29 µg/L) and Bira (28.8 ± 16.0 µg/L), whereas Cd levels were notably elevated in Dahlia Health Centre (1.48 ± 1.60 µg/L) and Tamalanrea (1.28 ± 0.64 µg/L). Hg concentrations were particularly high in the health center of Bira (31.91 ± 123.49 µg/L), with much lower levels observed in other health centers.
Characteristic of the Pregnant Women (N = 127).
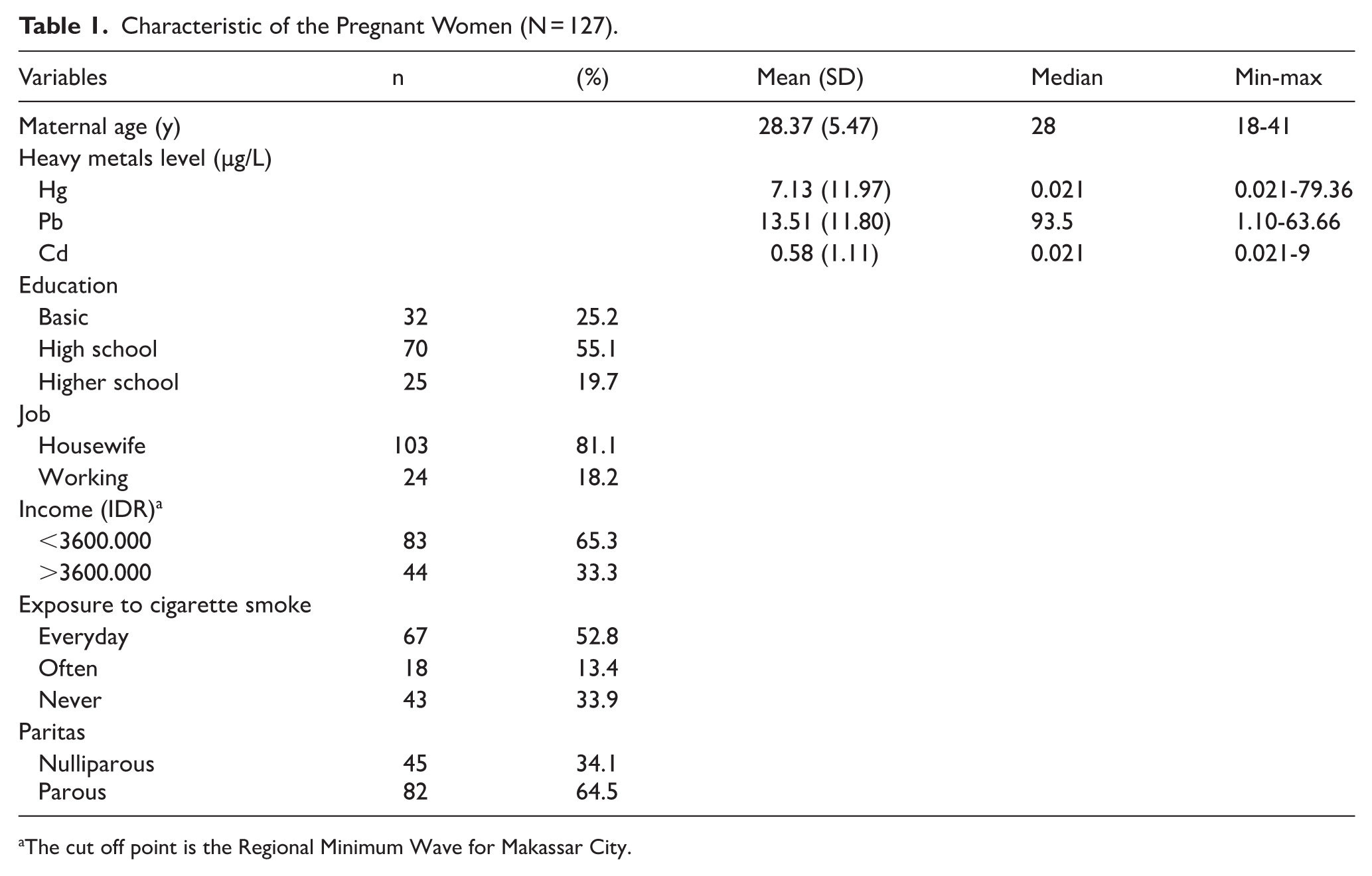
The cut off point is the Regional Minimum Wave for Makassar City.
Blood Heavy Metals Concentration and Fetal Measurement.

Mean ± SD.
Centimeter for BPD, HC, AC, FL and gram for EFW.
Fetal growth measurements also vary widely among health centers. Mothers visiting Health Centre of Makkasau exhibited the highest mean fetal growth metrics (BPD: 7.47 ± 0.31 cm, HC: 25.3 ± 0.37 cm, AC: 24.6 ± 3.64 cm, FL: 5.3 ± 0.35 cm, and EFW: 1328 ± 445 g), indicating favorable growth conditions. In contrast, Health Centre of Dahlia reported the lowest fetal growth measurements (BPD: 4.90 ± 1.52 cm, HC: 17.4 ± 5.27 cm, AC: 15.4 ± 5.15 cm, FL: 3.15 ± 1.19 cm, and EFW: 474 ± 351 g), potentially reflecting poorer maternal or environmental conditions.
Maternal Metals Exposures and Fetal Measurement
Figure 2 illustrates the correlations between metal exposures and fetal growth parameters expressed as Z-scores. The scatterplot matrix shows the strength and direction of the correlations between these variables, with density plots along the diagonal representing the distribution of each variable. The correlation between Pb and fetal measurements was generally low, with the strongest association observed for FL_z (R = 0.245, P < .01). Other correlations were positive but not statistically significant. Cd exhibited weak and non-significant correlations with most fetal measurements, except for a marginally negative correlation with BPD_z (R = −0.211, P < .05). Hg showed a significant positive correlation with FL_z (R = 0.674, P < .001), suggesting a notable association. However, the correlations with other measurements were weaker and generally non-significant.

Correlation of metal exposures and fetal measurement.
Interrelations between fetal measurements (Figure 2). Fetal measurements showed strong positive correlations. For instance, BPD and HC were highly correlated (R = 0.982, P < .001), reflecting the expected interdependence of fetal biometric indices.
Interrelations between metals (Figure 2). Log-transformed concentrations of the metals showed varied relationships. Hg had a moderately positive correlation with Cd (R = 0.674, P < .001), whereas Pb did not show significant correlations with other metals.
Metal Exposure and Smoking Exposure
To highlight any trends suggesting that smoking exposure might lead to higher metal exposure, we analyzed the distribution of metal concentrations according to smoking exposure status (Figure 3). Smoking exposure is refers to secondary hand smoke (SHS) or passive smoker. This result highlights the potential association between maternal smoking exposure and heavy metal concentrations. This indicated a strong link between frequent smoking and Hg exposure. The Pb concentrations displayed a broader range across all smoking categories. Interestingly, “often” smokers appear to have higher Pb concentrations than the other groups, but there is substantial overlap between the categories. The elevated Hg levels in “everyday” exposure may indicate that secondary hand smoking is a significant source of Hg exposure, which aligns with known research on the contribution of smoking to toxic metal exposure. However, the patterns for Cd and Pb suggest subtle or complex relationships which warrant further statistical testing.
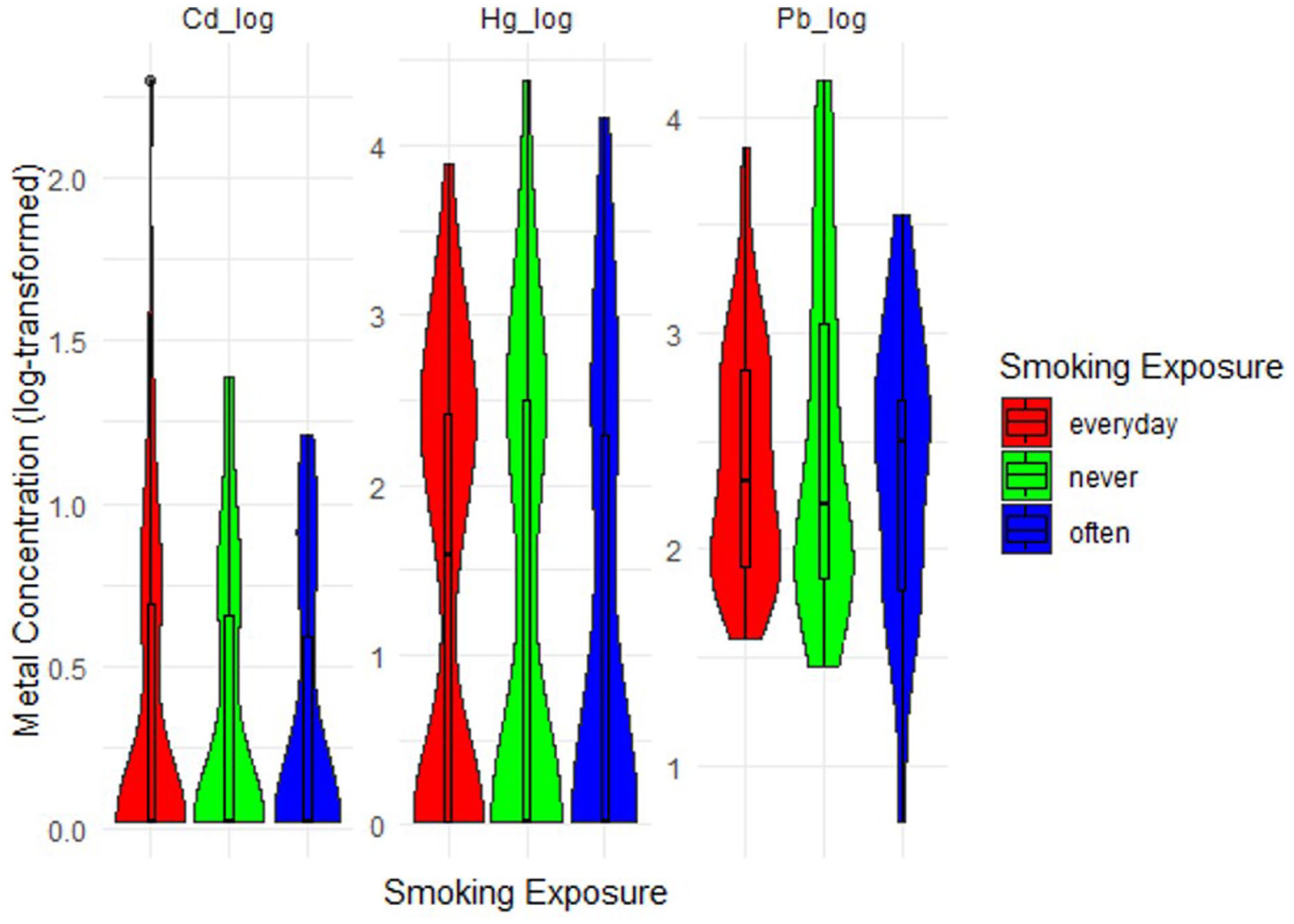
Maternal metal concentration by secondhand smoking exposure.
Secondary Hand Smoking Exposure and Fetal Measurement
Figure 4 illustrates the distribution of fetal measurements across different smoking exposure categories. For most fetal measurements (eg, HC and AC), the “never” exposed group suggesting lower variability in fetal growth metrics compared to the “everyday” or “often” smoking groups. For EFW, there was greater variability in the “everyday” smoking group, possibly indicating an influence of maternal SSE on fetal growth trajectories.

Fetal growth by secondhand smoking exposure.
Effect of Maternal Metal Exposure and Fetal Growth
The GAM highlights the trends and uncertainties (confidence intervals) for the effect of each metal concentration on various fetal measurements (Figure 5).

Relationship between metal concentration and fetal measurement.
Some nonlinear relationships were observed for certain outcomes, such as BPD_z and HC_z, suggesting potential threshold effects or diminishing returns at higher concentrations. The relationship for Cd shows a linear-like increase in most fetal measurements, particularly AC_z and FL_z. Nonlinear trends are evident, especially with AC_z and EFW_z, where higher Hg concentrations seem to correlate with increased variability in outcomes. Maternal age showed distinct nonlinear effects, with some fetal measurements (eg, HC_z and EFW_z) peaking at intermediate ages.
To examine the combined impact of all metals on fetal measurements, we aggregated the concentrations (Figure 6). For all measurements, weak or nonsignificant associations were observed within the observed range of metal concentrations. The relatively flat trends suggest that within the studied range of metal exposure, there may not be substantial effects on fetal growth measures, at least in terms of z-scores.

Nonlinear relationship between metal concentration and fetal measurement.
We assessed the nonlinear relationships between maternal exposure to metals and fetal measurements, while adjusting for maternal age and smoking exposure (Figure 7). Cd generally increases across most fetal outcomes, with the confidence intervals broadening as the concentrations increase. Hg showed a more stable pattern with smaller deviations, but noticeable divergence between smoking exposure groups at higher concentrations, and Pb was steeper at higher concentrations, especially for categories with regular smoking exposure. Furthermore, the effects of smoking exposure introduce variability in fetal measurements, with noticeable differences in the slopes or patterns of each exposure group. In some cases (eg, Pb_log with EFW_z), exposure seems to amplify the effect of the metal.

GAM smooth plots of metal concentration on fetal measurement.
In Table 3, we summarize the results of the generalized additive models (GAMs) evaluating the associations between heavy metal exposure and fetal measurements, adjusted for maternal age and smoking exposure, to provide a quantitative perspective of the modeled relationships. The results highlight that most relationships between metal concentrations and fetal measurements exhibit non-significant associations, suggesting limited evidence of strong nonlinear effects. Notably, Pb showed borderline significant effects on femur length (P = .071) and EFW_z (P = .256), indicating its potential for further exploration. Smoking exposure and maternal age were included as covariates; however, their effects were modest and did not reach statistical significance.
Generalized Additive Model for Maternal Metal Exposure and Fetal Growth Outcomes.

Overall, the GAM results emphasize the complexity of interpreting the relationship between heavy metal exposure and fetal development, particularly when controlling for key maternal factors. This analysis underscores the importance of further research to elucidate these relationships.
Furthermore, we assessed the cumulative environmental exposures for comparison with individual toxicants, as shown in Figure 8. For measurements such as EFW_z and AC_z, the aggregated metals exhibit positive associations, particularly in the never smoke exposed group. In contrast, FL_z and HC_z showed more subtle or flat associations across smoking exposure groups. Smoking exposure modified the observed relationships. Aggregated metal concentrations may have a more pronounced effect on fetal growth in non-smokers. Everyday and often groups showed flatter or negative trends, possibly suggesting a mitigating or suppressive effect of smoking on the relationship between metal exposure and fetal development. The wide confidence intervals in some categories reflect the variability in the data, emphasizing the need for cautious interpretation of trends.

The effect of aggregated metals on fetal measurements.
Discussion
This study investigated the effect of prenatal exposure to Pb, Cd, and Hg on fetal growth outcomes in a population of pregnant Indonesian women. The findings provide important insights into environmental exposure in low- and middle-income countries (LMICs) and their potential implications for fetal development.
Heavy Metal Concentrations
The blood concentrations of Pb, Cd, and Hg in this study fell within the ranges reported for other LMICs, reflecting shared environmental challenges, such as industrial pollution, dietary contamination, and inadequate waste management systems.16,19,25 However, levels varied across metals and between regions, underscoring the complex interplay between dietary, environmental, and socioeconomic factors.
Lead
The Pb concentrations in this study align with findings from pregnant women in Indonesia, where exposure is often attributed to vehicle emissions, paint residues, and industrial activities.17,26 For instance, the levels in this study were similar to those reported in Brebes, Central Java, where the mean Pb levels ranged between 5 and 10 µg/L. 17 Comparatively, the Pb concentrations in this study were within the range observed in pregnant women in Japan as part of the Japan Environment and Children’s Study (JECS), which reported blood Pb levels averaging below 10 µg/L. 6 However, levels observed in areas with high industrial activity, such as Shanghai, China, reported significantly higher Pb concentrations (41 μg/L) among the exposed mothers. 27 The Pb levels observed in this study suggest moderate environmental exposure, which may be linked to vehicular emissions, contaminated water sources, and dietary intake.
Cadmium
In this study, blood Cd levels were comparable to those in Southeast Asian populations where contaminated rice and tobacco farming were identified as significant sources of exposure.19,27 Similar levels were observed in pregnant women in Central Java, Indonesia, as reported by Wibowo et al, reflecting the dietary contributions to Cd exposure. 28 In contrast, populations in Europe have reported lower Cd concentrations, likely due to stricter environmental controls and reduced reliance on Cd-contaminated crops. 11 In Japan, research has highlighted higher levels (up to 1.5 µg/L) of Cd in pregnant populations consuming Cd-contaminated rice. 12 The association between Cd exposure and fetal growth restrictions observed in previous studies 2 highlights the need for public health strategies that target dietary and environmental sources.
Mercury
Hg concentrations were consistent with the findings from coastal communities and other fish-consuming populations. In Indonesia, high fish consumption in coastal areas is linked to elevated Hg levels, as reported in Makassar and Semarang in Central Java.5,16 The levels observed in this study were lower than those in the artisanal gold-mining regions, which represent extreme exposure hotspots, 29 and higher than those in Shanghai. 27 In Japan, mean blood Hg levels have reached 10 to 15 µg/L among fish-eating populations, reflecting similar dietary exposure patterns. 6 However, the levels were significantly lower in populations with minimal fish consumption, such as urban European and North American cohorts, where the levels often did not exceed 2 µg/L. 7 The adverse effects of Hg on fetal neurodevelopment and anthropometric outcomes, as demonstrated in studies from Japan and Scandinavian countries, emphasize the significance of moderate exposure.6,7
These findings suggest that the levels of Pb, Cd, and Hg in this study reflect the environmental and dietary factors common to LMICs, emphasizing the need for targeted public health interventions to reduce exposure risks. Efforts to improve environmental regulations, promote dietary diversification, and raise awareness about heavy metal contamination could help mitigate these risks in vulnerable populations.
Association Between Metal and Fetal Growth Outcomes
The current study found distinct associations between Pb, Cd, and Hg and fetal growth outcomes. Pb exhibited a negative association with fetal growth, consistent with its known mechanism of interfering with calcium transport and skeletal development. The results also suggest that Pb has borderline effects on FL and EFW, with the observed trends being stronger in non-smokers. These findings align with prior research, which has shown that prenatal Pb exposure may impair fetal growth by interfering with calcium transport, which is critical for skeletal development.6,30,31 However, the lack of consistent and significant results could reflect relatively low exposure levels in the study population or the buffering effect of maternal nutrition.
In contrast, Cd and Hg showed positive associations with fetal growth, which may appear counterintuitive. However, this can be explained by the non-linear dose-response relationships for these metals.
Cd exhibited a generally increasing trend across fetal measurements, with broader confidence intervals at higher concentrations. This may indicate a variability in susceptibility among individuals. Cd mimics zinc (Zn) and disrupts fetal development by interfering with nutrient transport across the placenta.12,28 The absence of statistically significant associations aligns with other studies suggesting threshold effects of Cd exposure, particularly at higher concentrations.32,33 While these positive associations may seem counterintuitive, they may reflect threshold effects where Cd only affects fetal development at higher exposure levels, consistent with studies highlighting non-linear dose-response relationships.34,35
Hg showed a more stable pattern with some divergence among smoking exposure groups at higher concentrations. Methylmercury (MeHg), the predominant form of Hg exposure, is known to cross the placenta and accumulate in fetal tissues, potentially leading to neurotoxicity and growth retardation.7,36 The divergence observed in smoking groups may suggest that the effects of Hg exposure on fetal growth could be moderated by other factors, such as secondhand smoke exposure, which can increase metal absorption.
At low levels of exposure, Cd and Hg may not show the harmful effects that are typically associated with higher concentrations. This phenomenon is supported by the hypothesis that very low levels of these metals may have neutral or even slightly positive effects, possibly due to their interactions with essential nutrients that could promote fetal development. For instance, Cd has been found to interact with micronutrients like Zn37,38 and calcium, 39 which are critical for fetal growth, and Hg may have antioxidant effects when balanced with other protective nutrients like selenium. 20 These interactions could mitigate some of the otherwise harmful effects of exposure at low concentrations, potentially explaining the positive associations observed.
The use of restricted cubic splines (RCS) in our analysis may have revealed these trends by capturing the non-linear nature of the relationship between metal exposure and fetal growth. While a linear model would suggest a direct relationship between metal exposure and fetal size, the RCS model allows for more flexibility in detecting subtle effects, particularly at different levels of exposure. This is particularly important because higher concentrations of Cd and Hg were rare in this study, and toxicity might only manifest at those higher levels, which were not represented in the majority of the population.
Although individual heavy metals did not demonstrate strong significant associations with fetal measurements, the potential interactive and cumulative effects merit attention. When considering aggregate exposure to Pb, Cd, and Hg, adjusted for maternal age and smoking exposure, distinct patterns emerged. Smoking exposure, primarily from secondhand sources, moderated the relationship between heavy metal exposure and fetal growth outcomes, with notable differences in trends across exposure groups. The inclusion of smoking exposure as a covariate revealed its potential role as a confounding or moderating factor, consistent with studies linking secondhand smoke to enhanced heavy metal absorption and associated health risks.1,19
Although individual metals showed limited associations, the aggregated exposure metrics provided a more comprehensive view of the environmental impacts on fetal growth. This approach aligns with recent studies that emphasize the need to account for cumulative exposures when assessing environmental health risks.3,36 For instance, the deviation in slopes for aggregated exposure suggests potential synergies among Pb, Cd, and Hg, a phenomenon observed in other studies analyzing toxicant mixtures.3,11 This finding underscores the importance of considering combined exposures in future epidemiological studies, as the impact of multiple toxicants may be greater than the sum of their individual effects.
Public Health Implications
The observed levels of heavy metals, while within the expected ranges, raise concerns about their long-term implications for maternal and child health in LMICs. The interplay between dietary practices, environmental pollution, and smoking exposure warrants targeted interventions to mitigate these risks. Strategies such as promoting dietary diversification, reducing reliance on contaminated food sources, and curbing secondhand smoke exposure can substantially lower the heavy metal burden in vulnerable populations.
Limitation
While the study used longitudinal fetal measurements (trimesters 2 and 3), exposure data (blood metal levels) were collected at a single time point. This limits causal inference and does not capture the temporal changes in metal exposure during pregnancy. Temporal misclassification may arise in this design because the exposure data, collected in the second trimester, may not fully reflect changes in metal exposure levels throughout the pregnancy. Metal exposure can vary over time, and fluctuations between the 2 measurements could lead to some degree of misclassification in the assessment of exposure in relation to fetal growth, especially if exposure levels changed after the second trimester. This limitation should be addressed in future studies by collecting repeated exposure measurements across different trimesters to better understand the temporal relationship between metal exposure and fetal growth outcomes.
Additionally, the reliance on 1 blood sample for each participant may not fully reflect chronic or cumulative exposure to metals, especially Hg and Cd, which can fluctuate over time and accumulate in other tissues. This limitation should be considered when interpreting the findings, especially for metals known to have variable exposure patterns.
The exposure assessment was based on a questionnaire, which is susceptible to recall bias, particularly in relation to dietary intake and lifestyle factors. The questionnaire was pre-tested to reduce errors, but the accuracy of self-reported data might still be influenced by memory recall or subjective interpretation of exposure sources. While other environmental data, such as air quality or water contamination levels, were not collected, future studies should integrate such environmental measurements to improve exposure assessment and reduce the reliance on self-reporting.
Additionally, the study only accounted for maternal age and smoking exposure as confounders, but other potential factors like nutritional status, occupation, and environmental pollutants were not controlled for. These factors could influence both maternal metal exposure and fetal growth, and their exclusion from the analysis may introduce bias.
Furthermore, we used log-transformed concentrations of metals and aggregated exposure, which simplifies complex exposure patterns but may not capture the full variability in individual metal exposure. The relatively low variation in Hg and Cd levels, with many values falling below the detection limit, likely reduced the sensitivity of the model.
Given that the study involved 127 participants from an urban region in Indonesia, the results may not be generalizable to rural populations or regions with different environmental and nutritional contexts. Although the sample size was adequate for the analyses conducted, it may limit the power to detect certain interaction effects, particularly those involving complex or non-linear relationships between environmental exposures and fetal growth outcomes. Larger sample sizes would improve the robustness of the findings and facilitate the detection of more subtle effects. Future studies should perform power analyses to ensure adequate statistical power for exploring these relationships.
Finally, while fetal biometry was measured by trained personnel using ultrasonography, some inherent inter- and intra-observer variability could still exist, potentially introducing noise into the fetal growth outcome variables.
Conclusion
Pb exhibited a U-shaped relationship with fetal measurements, suggesting a potential risk at moderate levels. Cd and Hg showed positive associations with fetal growth, particularly at higher exposure levels, although the biological plausibility of these findings may reflect the underlying dietary or environmental complexities. Smoking exposure was significantly associated with reduced fetal growth, independent of metal exposure. These findings highlight the importance of considering nonlinear dynamics and environmental co-factors when evaluating prenatal exposure and fetal development. Ongoing biomonitoring and public health interventions are required to mitigate the potential risks associated with heavy metal exposure during pregnancy.
Footnotes
Acknowledgements
The Institute for Research and Community Services at Hasanuddin University facilitated financial support for this study.
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Directorate of Research, Technology, and Community Service of the Ministry of Education, Culture, Research, and Technology, Indonesia (number 02035/UN4.22.2/PT.01.03/2024).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

