Abstract
Heavy metal pollution in freshwater ecosystems threatens food safety and public health, particularly in industrializing regions. This study evaluated the bioaccumulation of heavy metals in edible tissues of Cyprinus carpio from Ethiopia’s Koka Reservoir, assessing potential health risks for communities dependent on this fish as a dietary staple. Water, algae, and fish samples were collected from 2 sites influenced by river inflow and tannery effluents. Heavy metal concentrations were analyzed alongside bioaccumulation factors (BAFs), estimated weekly intake (EWI), target hazard quotients (THQ), and carcinogenic risk (CR). Although water quality parameters (pH: 8.57-8.62; dissolved oxygen: 3.60-6.30 mg/L) complied with guidelines, heavy metals in fish tissues exceeded safe levels: Pb (<0.034-3.29 mg/kg), Cd (<0.001-0.36 mg/kg), Fe (1.55-74.98 mg/kg), and Zn (0.18-135.32 mg/kg). Elevated BAFs for Zn (756.00) and Cr (110.00) indicated significant bioaccumulation and potential biomagnification. While THQ values were <1 (suggesting minimal non-carcinogenic risk), CR for Cr6+ and Pb2+ exceeded safety thresholds, signaling long-term carcinogenic hazards. These findings demonstrate that water quality monitoring alone is insufficient to safeguard food safety, as metals accumulate disproportionately in aquatic biota. Thus, the study recommends stricter controls on industrial effluents, sustainable agricultural practices, systematic biomonitoring, and community education to mitigate dietary heavy metals exposure, contributing to SDGs on health, food security, and sustainable resource management.
Introduction
The developmental activities such as industries, agriculture and mining are being observed polluting the natural resources in nations. Surface waters are among the most vulnerable resources to contamination as they are often used as convenient recipients for wastewaters, and runoff unlike groundwater which is relatively less accessible. These water bodies accumulate a complex mix of organic and inorganic pollutants that can harm both the environment and human health. 1 While natural processes can influence water quality, rapid urbanization and industrialization have greatly intensified surface water pollution in recent decades. 2 In many developing countries, industries frequently discharge untreated effluents directly into rivers and open land, exacerbating contamination, violating the WHO and EPA standards. 3
In Ethiopia, the Awash River Basin, the Modjo River and Koka Reservoir have become a hotspot of such anthropogenic pollution, receiving significant industrial discharges (that contain heavy metals) from tanneries and other factories.4-6 The entry of heavy metals into these waters threatens aquatic biodiversity and disrupts ecological balance. 7 Although trace metals like Fe, Cr, Cd, Cu, Pb, Ni, and Zn are essential micronutrients at low concentrations, they become toxic when accumulated beyond physiological thresholds, leading to bioaccumulation in phytoplankton and zooplankton and biomagnification up the food web.8,9 This contamination ultimately alters aquatic community structure and poses serious health risks to human consumers.10,11 Algae and Fish are used as bio-indicators of contamination, especially as top consumers in the systems; fishes are recognized bio-indicators of heavy metal pollution. 12
Specific to Lake Koka, studies has shown concerning levels of heavy metals such as Fe, Zn, Pb, and Cd in both the water and organisms, posing potential health risks to nearby communities.13-15 Other prior research16,17 in Rift Valley Lakes, including Koka, further reported bioaccumulation of various metals and mercury in edible fish species, signaling an overlooked but significant threat to food safety and public health. Additional studies have also detected biological contaminant (microcystins) in the muscle and liver tissues of fish from Koka Reservoir, emphasizing the complexity of pollution beyond metals alone.18,19
While these studies provide valuable data on contamination, there remains a critical gap in systematically linking water quality, heavy metal concentrations in algae, and bioaccumulation in economically important fish species like common carp (Cyprinus carpio). Existing research often focuses separately on water, fish, or plankton, limiting holistic risk assessments. Moreover, there is no sufficient recent data on the concentration of heavy metals in the reservoir, algae and edible fish tissues and the associated health risks for consumers though the increase in industrial and agricultural activities is considerable. As the reservoir supports significant fisheries and livelihoods for communities in Modjo and Adama, reliable, timely and site-specific information about the extent of bioaccumulation and potential human exposure is highly essential. Therefore, this study addresses this gap by assessing the physicochemical characteristics of the reservoir water, quantifying heavy metal loads (Zn, Cr, Cd, Fe, Cu, Ni, and Pb), and evaluating their bioaccumulation in representative algae (Chlamydomonas) and fish (Cyprinus carpio). The findings also aim to inform food safety, public health protection, and sustainable water resource management.
Materials and Methods
Description of Study Area
The Koka Reservoir is situated in the Awash River Basin within the East Shewa Zone of the Oromia Regional State, approximately 90 km southeast of Addis Ababa. The reservoir was created in 1960 following the construction of the Koka Dam, primarily for hydroelectric power generation. As shown in Figure 1, the surrounding landscape is dominated by agricultural land, with scattered industrial activities, including tanneries, located upstream. These industries discharge effluents directly into the tributary rivers, while urban runoff from Modjo Town also flows into the reservoir via the Modjo River. Beyond its role in electricity production, the reservoir supports local livelihoods through fishery activities. The map (Figure 1) provides a clear spatial representation of the reservoir, river networks, agricultural zones, and sampling locations, offering valuable context for understanding the study area’s hydrological and ecological dynamics.

Location map of the study area.
Koka Reservoir is located in the Central Ethiopian Rift Valley, with an elevation ranging between 1589 and 1592 m above sea level. Its geographic coordinates extend from 8°24′407″N to 8°25′746″N latitude and 39°35′0″E to 39°01′649″E longitude (1). The reservoir spans a surface area of 255 km², with a maximum depth of 14 m and a minimum depth of 9 m. It extends 20 km in length and 15 km in width, featuring a shoreline of 195 to 205 km (2).
The primary inflows to the reservoir are the Awash and Modjo Rivers, while the Awash River serves as its sole outflow. The region experiences a bimodal rainfall pattern, with the main rainy season occurring from June to September and a shorter wet period from March to May. The mean annual air temperature in the area is approximately 30.4°C.
Research Designs
This study employed field measurements and standard laboratory procedures. A field-based experimental test was designed to analyze heavy metals, including Cd, Cr, Fe, Cu, Zn, Ni, and Pb, in water as well as in the edible organs of Common Carps (Cyprinus carpio) from Koka Reservoir. The study also computed the associated human health risks due to fish consumption. Samples were purposively collected from Koka Reservoir near the inlet of the Mojo River around the tanneries and near the Awash River inlet around the flower farms. These inlets were observed to contain high concentrations of algal growth and fish production. Physicochemical analyses of the samples were carried out in an ex-situ laboratory.
Methods of Sample Collection
A total of 1000 mL water samples were collected from each sampling site using pre-cleaned polyethylene bottles to prevent contamination. Concurrently, key physicochemical parameters, including temperature (°C), electrical conductivity (µS/cm), total dissolved solids (mg/L), salinity, pH, dissolved oxygen (mg/L), and turbidity (measured using a WGZ-200 turbidimeter) were recorded in situ to ensure accurate environmental characterization. Algae samples (1000 mL) were similarly collected in pre-cleaned polyethylene bottles to assess bioindicator responses to heavy metal exposure. Besides, 3 adult specimens of Cyprinus carpio were obtained from each site using standardized fishing methods. Morphometric data, including total length (cm) and body weight (g), were measured immediately after capture to minimize stress-induced variability. All samples (water, algae, and fish tissues) were carefully sealed, labeled, and transported under controlled conditions to the central laboratory for heavy metal analysis (Cd, Cr, Fe, Cu, Zn, Ni, and Pb) using accredited analytical protocols. This systematic approach ensured data integrity while minimizing pre-analytical errors during sample handling and preservation.
Digestion of Water Samples and Heavy Metals Analysis
Water samples were first filtered through Whatman No. 42 filter paper to remove suspended particulates. For analysis, 100 mL aliquots were acidified with concentrated HNO₃ and carefully evaporated on a hot plate to reduce the volume to 10-15 mL. The concentrated samples were then transferred to 50 mL volumetric flasks and diluted with deionized water. All prepared samples were stored at 4°C in acid-washed polyethylene bottles to prevent contamination prior to analysis. Sample digestion was performed using a mixture of 50 mL water, 4 mL HNO₃, and 1 mL HCl, with digestion proceeding for 3 hours to ensure complete dissolution of metal constituents. Following digestion, samples were cooled, filtered, and analyzed for heavy metal content (Cd, Cr, Fe, Cu, Zn, Ni, and Pb) using inductively coupled plasma optical emission spectrometry (ICP-OES; Spectro ARCOS FSH 12, Germany). 20 This rigorous preparation protocol ensured accurate quantification of trace metal concentrations while minimizing matrix interference and maintaining sample integrity throughout the analytical process.
Digestion of Algal Biomass and Fish Samples, and Heavy Metals Analysis
100 mL biomass containing water samples were filtered by a Whitman filter paper of No. 42. The solid algal biomass on the filter paper was dried. Whereas, the fish samples (wet weight) were taken and dissected using a stainless knife and edible components were taken for analysis. The samples were rinsed in distilled water and dried in a 105°C oven for 24 hours to achieve a constant weight. A mortar and pestle were used to grind the dried fish and algal biomass samples. 1 g of the powder samples were transferred in to digestive tubes mixed with 5 mL conc. HNO3 and 3 mL conc. HClO4. The mixtures were heated at 110°C for 1 hour in a fume hood followed by cooling at room temperature. About 20 mL of distilled water was added in each digestion tubes and the mixtures were filtered. Consequently, the filtrates were transferred in to 100 mL volumetric flasks, distilled water was filled to the marks and the mixtures were left for 15 hours. Finally, the heavy metals contents of both filtrates were analyzed using ICP-OES instrument (Spectro ARCOS FSH 12 MODEL, Germany) 20 with detection limits of the metals reported. 21
Bioaccumulation Factor (BAF)
The bioaccumulation factor (BAF) for heavy metals was calculated following the methodology established. 22 The BAF represents the ratio of heavy metal concentration in the fish tissue (mg/kg wet weight) to the corresponding metal concentration in the ambient water (mg/L), as expressed by equation (1).
Where, MCF and MCW are the heavy metal concentration in edible parts of the Fish and water, respectively. The BAF values were interpreted according to established ecological risk assessment frameworks, where values >1 indicate significant bioaccumulation potential.
Estimation of Health-Risk Assessment
The Estimated Weekly Intake (EWI) of heavy metals through fish consumption was calculated using the standardized equation employed, 12 as expressed in equation (2).
Where, MC is the metal concentration in the fish muscle (mg/kg dry weight) and AC is the average weekly consumption of fish in the area from Koka Reservoir.
The local population surrounding Koka Reservoir consumes fish at an average rate exceeding 10 kg per person annually, equivalent to approximately 0.19 kg per week. Given this substantial dietary intake, the study focused on quantifying concentrations of 6 potentially toxic heavy metals such as Pb, Cr, Ni, Cu, Zn, and Fe in water, algae, and edible tissues of Common Carp (Cyprinus carpio). To assess associated health risks, a dual-parameter approach was employed, incorporating (1) the Cancer Slope Factor (CSF) to estimate carcinogenic risk potential (equations (3) and (2) the Reference Dose (RfD) to evaluate non-carcinogenic risk thresholds (equation (4)). This framework integrated site-specific exposure data with internationally recognized toxicity parameters (WHO/USEPA) to characterize both long-term carcinogenic probabilities and systemic health hazards.
To comprehensively evaluate potential health impacts, all collected data on heavy metal concentrations in water, algae, and fish tissues were systematically analyzed following the risk assessment framework established. 23 For non-carcinogenic risk assessment, the Target Hazard Quotient (THQ) was calculated using equation (3), which compares the estimated exposure level of each heavy metal with its corresponding reference dose (RfD). The formula was also used.24,25
Where, IR represents the weekly fish ingestion rate (0.275 kg/week), ER is the exposure rate, and ED is the exposure duration set at 30 years. The average body weight (BW) considered was 48 kg for women and 56 kg for men. MC refers to the measured concentration of each heavy metal in the fish samples (mg/L). AT denotes the averaging time for non-carcinogenic risk, this was 92 weeks for carcinogenic risk, it was calculated based on total exposure days over 30 years (365 days/year× 30 years) and adjusted to 92 × 18 weeks. The chronic weekly ingestion of fish (CWI) was assumed as 10−3 mg/kg/week. The non-carcinogenic hazard is termed as hazard quotient (HQ), which was a unit less number that expresses the probability of an individual suffering an adverse effect. It is a quotient of CWI (Chronic weekly Intake) divided by RfDo (chronic oral reference dose): For the number of heavy metals, the total non-carcinogenic risk is termed as hazard index, which is the summation of the hazard quotient of each heavy metal. 23
The Hazard Index (HI) was calculated as the sum of individual heavy metal Target Hazard Quotients (THQs) using equation (4), where n represents the concentration of each heavy metal. According to the US National Research Council, 23 risk assessment framework, HI < 1 indicates that the exposed population is unlikely to experience significant adverse health effects. HI ⩾ 1 suggests potential non-carcinogenic health risks or possible carcinogenic effects requiring further investigation.
For carcinogenic risk assessment, the probability of cancer development was estimated using equation (5), which calculates the incremental lifetime cancer risk (ILCR) from chronic exposure to carcinogenic heavy metals.
Where, CSF is the cancer slope factor (mg /kg−1 day−1) and CR: cancer risk.
Statistical Analysis
The data collected using the ICP-OES model was analyzed for statistical differences in the mean value of heavy metals concentration in water, algae, accumulation of heavy metals in organs of fish, and physical parameters between the 2 sample sites. Linear regression analysis was done to assess the linear relationship of heavy metal concentration in the water with the concentration of the fish tissue. The difference in mean value was accepted as being statistically significant if P < .05.
Results
Physico-Chemical parameters of the Koka Reservoir
Table 1 presents the measured physico-chemical characteristics that showed notable variation between Site-1 and Site-2. The pH values were fairly similar across both sites, with an overall mean of 8.60, indicating slightly alkaline conditions. Temperature ranged from 24.06 ± 0.23°C at Site-1 to 26.53 ± 0.13°C at Site-2, with an average of 25.30°C, suggesting modest spatial variation, and possibly due to lower shading or higher inflow from warmer tributaries at site 2 than site 1. Site-1 had higher total dissolved solids (TDS) and dissolved oxygen (DO), suggesting better water mixing or less organic pollution.
The Physico-Chemical Parameters of Water Samples in the Study Area (mean ± SD).
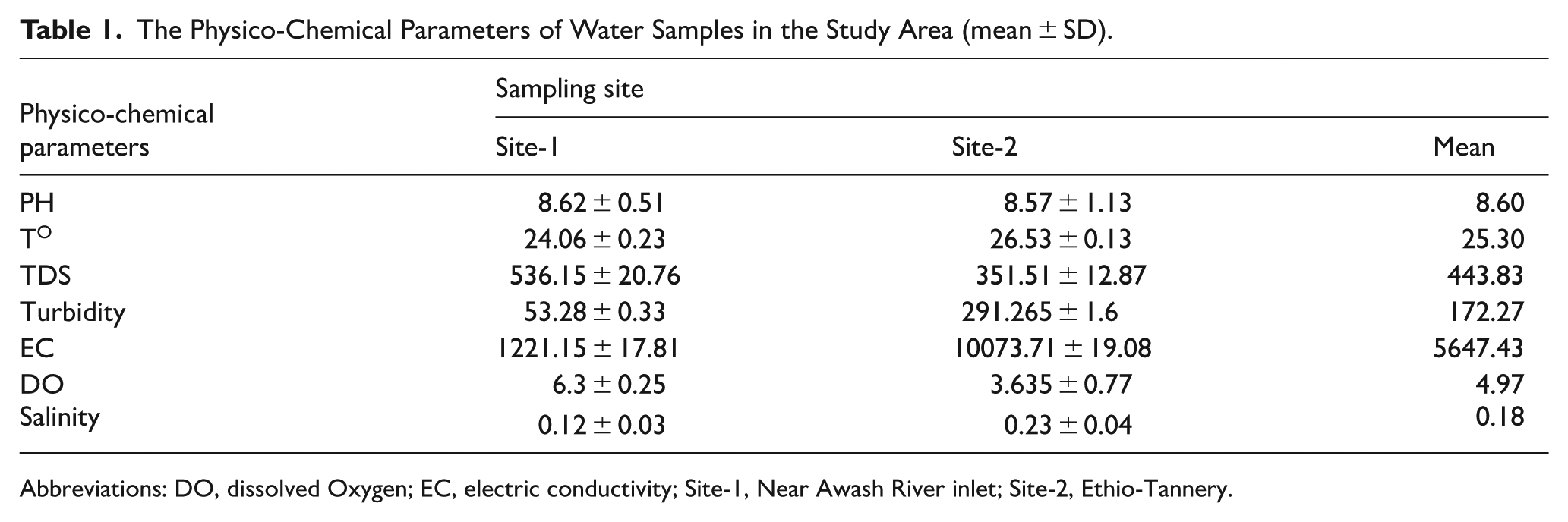
Abbreviations: DO, dissolved Oxygen; EC, electric conductivity; Site-1, Near Awash River inlet; Site-2, Ethio-Tannery.
Turbidity was substantially higher at Site-2 (291.27 ± 1.6 NTU) than Site-1 (53.28 ± 0.33 NTU), resulting in a mean turbidity of about 172.27 NTU, likely indicating greater particulate matter at Site-2. Electrical conductivity (EC) differed markedly, with Site-2 recording an exceptionally higher mean of 10 073.71 ± 19.08 µS/cm, in contrast to 1221.15 ± 17.81 µS/cm at Site-1, likely linked to runoff carrying more sediments, salts, and pollutants from surrounding areas at site 2. Dissolved oxygen (DO) levels were higher at Site-1 (6.3 ± 0.25 mg/L) compared to Site-2 (3.64 ± 0.77 mg/L), averaging 4.97 mg/L, may be due to more organic matter decomposition and reduced aeration at site 2. With regard to salinity, it was relatively higher at Site-2 (0.23 ± 0.04 ppm) than Site-1 (0.12 ± 0.03 ppm), with an average of 0.18 ppm.
Heavy Metal Concentration in Water Samples
The availability of the 7 heavy metals in water, algae, and fish samples from Koka Reservoir was investigated. The concentration of heavy metals in the water, followed the descending order pattern of Fe > Zn > Cu > Cr > Ni > Pb > Cd (Table 2). The result showed that all selected heavy metals were detected in measurable amounts across the water samples from both study sites. Overall, the concentration of heavy metals in the reservoir across all matrices ranged between <0.001 and 74.98 mg/L during the winter/dry season.
Mean Concentrations of Metals in Water Samples at Each Site in This Study (mg/L).

Table 2 also indicates that the mean concentrations of Fe, Pb, and Cr in water samples from the study sites notably exceeded WHO and EPA guideline limits, especially iron at Site-2 (17.68 mg/L) and lead (mean = 0.058 mg/L). These elevated levels are likely linked to industrial effluents, particularly from facilities such as the Ethio-tannery, and urban runoff carrying untreated or partially treated wastewater into the reservoir. Notably, the highest concentration of iron in fish (74.98 mg/L) was recorded at the Ethio-tannery entry site during the dry season, while the lowest concentration (1.56 mg/L) was observed near the awash inlet. This spatial variation indicates localized pollution hotspots driven by industrial discharges and seasonal accumulation. On the contrary, zinc, copper, nickel, and cadmium concentrations remained below or near guideline limits, although even low levels of cadmium and lead pose significant health risks due to their toxicity and bioaccumulation potential. Generally, the higher metal loads observed in Site-2 compared to Site-1 suggest that direct discharges and proximity to the major industrial activities significantly influence contamination patterns in the Koka Reservoir.
Table 3 shows strong positive correlations among most heavy metals in Koka Reservoir water, particularly between Zn, Cu, Ni, Cr, and Cd, indicating they likely share common sources or similar environmental behavior. Fe has moderate positive correlations with some metals, suggesting partial association. In contrast, Pb exhibits negative correlations with several metals, implying it may originate from different sources or respond differently in the environment.
Pearson’s Correlation Matrix Between Heavy Metals in Water From Koka Reservoir.

Correlation is significant at the .01 level (2-tailed).
**Correlation is significant at the .05 level (2-tailed).
Heavy Metal Concentration in Algae Samples
Table 4 shows that algae samples from the Koka Reservoir accumulated heavy metals in the general order of Fe > Zn > Cr >Ni > Pb > Cu > Cd. Fe had by far the highest mean concentration (39.42 mg/kg), especially at Site-2 near industrial activity, where it reached 69.98 mg/kg notably above WHO and EPA guidelines. This pattern suggests algae’s high capacity to take up and store Fe from the water, likely driven by effluent from the Ethio-tannery and related runoff. Zn also showed notable accumulation (mean of 0.25 mg/kg), exceeding EPA limits at Site-2, while Cr and Ni levels were elevated compared to guideline values, pointing to industrial and possibly agricultural runoff as contributing sources. Pb concentrations in algae (mean of 0.094 mg/kg) were higher than the WHO recommended level of 0.01 mg/kg, signaling significant contamination risk. The lowest concentrations were found for cadmium, which remained near or just above detection limits except at Site-2 (0.01 mg/kg).
Mean Concentrations of Metals in Algae Samples at Each Site in This Study (mg/kg).
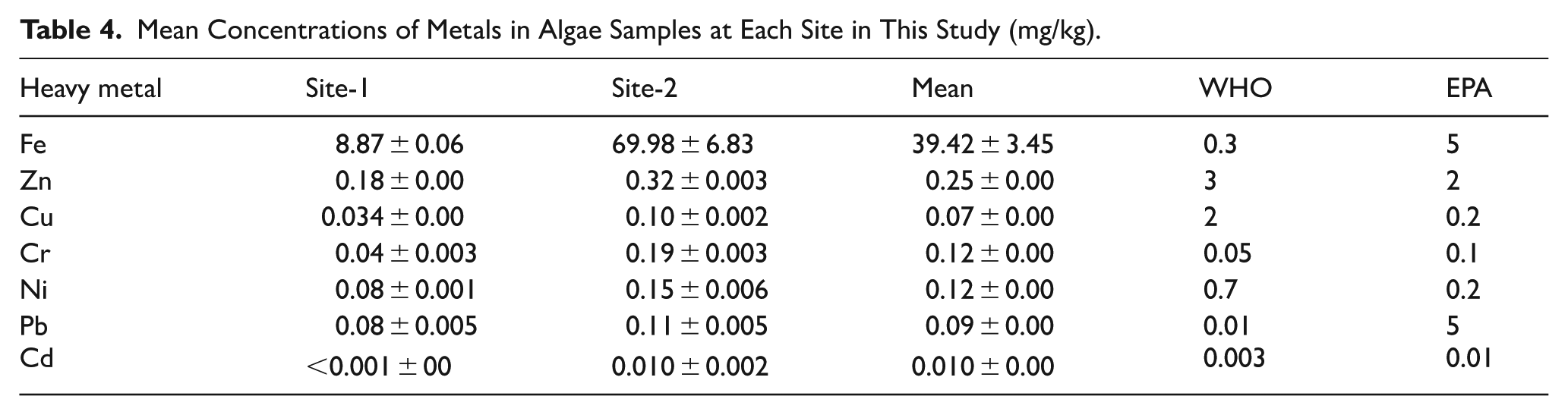
Heavy Metal Concentration in Fish Samples
As indicated in Table 5, the pattern of heavy metal accumulation in fish samples followed the order Zn > Fe > Cr > Pb > Cu > Ni > Cd. Zinc had the highest mean concentration (135.32 mg/kg), over 3 times higher than WHO and EPA recommended limits (40 mg/kg), reflecting significant bioaccumulation likely influenced by high zinc levels observed in algae and water, particularly near industrial discharge sites. Fe was the next most abundant metal (74.98 mg/kg), exceeding guideline values and aligning with its elevated presence in algae and water.
Mean Concentrations of Metals in Fish Samples at Each Site in This Study (mg/kg).

Cr and Pb concentrations in fish (3.74 and 3.29 mg/kg, respectively) also exceeded recommended limits, underlining health risks from fish consumption. Cu and Ni were detected at moderate levels (2.58 and 1.57 mg/kg, respectively), remaining below guideline values. Cadmium, though found at the lowest concentration (0.36 mg/kg), still exceeded WHO’s maximum allowable level of 0.05 mg/kg, indicating contamination concern as well.
Bioaccumulation Factors and Human health Risks
The bioaccumulation factors (BAFs) presented in Table 6 highlight substantial differences in heavy metal uptake by fish across sites. Overall, fish near the Awash inlet (Site-1) exhibited higher BAFs than those near the Ethio tannery (Site-2), except for slight differences in specific metals. Zn showed the highest BAF (755.97), indicating a strong tendency to accumulate in fish tissues despite its lower concentrations in water. Similarly, Cd (363.00), Pb (96.84), and Cr (110.00) had notably high BAFs, demonstrating considerable bioaccumulation potential. Fe, while abundant in water, had a moderate BAF (48.15), suggesting less efficient transfer to fish tissues relative to other metals. Cu also showed moderate BAFs (35.88). The variation in BAFs reflects differences in metal availability, fish feeding habits, and metabolic processes. The overall pattern of bioaccumulation in descending order was observed as Zn > Cd > Pb > Cr > Fe > Cu > Ni, with Zn and Cd clearly dominating.
Bioaccumulation Factors for Heavy Metals in Edible Parts of Fish to Water in the Study Area.

Site-1, Near Awash inlet; Site-2, Ethio-tannery.
Notably, these high BAF values, particularly for Zn, and Cd align with earlier observations of elevated concentrations of these metals in fish compared to water and algae, highlighting significant biomagnification along the aquatic food web. These findings point to the urgent need for strengthened monitoring, pollution control, and risk mitigation strategies to limit heavy metal transfer from the environment to consumers through fish.
Table 7 presents the estimated weekly intake (EWI), target hazard quotient (THQ), hazard index (HI), and carcinogenic risk (CR) calculated from fish consumption reveal important insights into potential human health risks. Among the metals, zinc had the highest EWI (25.71 mg/kg), followed by iron (14.25 mg/kg) and copper (0.49 mg/kg), reflecting their higher concentrations in fish tissues. The calculated THQ values for all metals were below 1, indicating no significant non-carcinogenic risk from individual metal exposure; specifically, THQs ranged from 2.1 × 10−4 (Cu in males) to 4.7 × 10−3 (Cr6+ in females).
The EWI, THQ, HI, and CR for Individual Metals From Fish Consumption.

Abbreviations: CR, carcinogenic risk; EWI, estimate weekly intake; HI, hazard index; THQ, target hazard quotation.
The HI, which accounts for cumulative risk from multiple metals, was also low for males (1.0 × 10−2) and females (1.2 × 10−2), suggesting overall exposure remains within acceptable limits. However, the carcinogenic risk (CR) values showed Cr6+ presented the highest cancer risk (3.55 × 10−1), followed by lead (1.0 × 10−1) and nickel (5.1 × 10−2). These CR values exceed the commonly accepted threshold of 10−4, indicating potential long-term carcinogenic concern, particularly for Cr6+, which remains considerably higher than safe limits. Collectively, these results suggest that while non-carcinogenic risks from consuming fish in the study area appear minimal, certain heavy metals, especially Cr6+, Pb, and Ni pose notable carcinogenic risks that require attention, regular monitoring, and mitigation strategies to protect public health.
Discussion
Physico-Chemical Parameters of Koka Reservoir
The spatial variation observed in physico-chemical parameters, with notably higher turbidity, electrical conductivity (EC), and lower dissolved oxygen (DO) at Site-2, reflects pronounced anthropogenic pressures, consistent with findings, 26 who highlighted that industrial effluents increase EC and turbidity, subsequently impacting aquatic biota by limiting light penetration and oxygen availability. Similar study 27 emphasize how reduced DO levels caused by organic pollution impair fish respiration and disrupt aquatic ecosystems. The slight alkalinity indicated by pH values aligns with observations in tropical reservoirs, where alkaline conditions may buffer some metal toxicity but also influence metal speciation and bioavailability. 28 These alterations jeopardize the reservoir’s ecological balance and threaten fisheries, an important livelihood source. The elevated EC and turbidity at Site-2 signal increased sediment and pollutant loads likely originating from tannery discharge and urban runoff, echoing findings 29 in industrial hotspots. Thus, it requires enforcement of effluent treatment regulations, catchment management to reduce sediment influx, and continuous physico-chemical monitoring to detect early signs of degradation and guide remediation efforts.
Heavy Metal Concentrations in Water
The detection of elevated Fe, Pb, and Cr concentrations exceeding WHO and EPA guidelines corroborates earlier reports for Lake Tana 30 ; Lake Ziway 31 ; and for Awash, 32 which identified these metals as primary pollutants in industrially influenced aquatic systems. The high levels of Fe pose ecological risks through oxidative stress on aquatic organisms, 33 while the toxicity of Pb and Cr (VI) is well-documented, with chronic exposure linked to neurological, renal, and carcinogenic outcomes in humans. 34 Similar studies that were reported in northeastern bay of Bengal has shown the exceeded Pb concentration 3 times beyond the allowable limits. 35 The concentration of heavy metals (Table 8), have shown comparatively lower concentration of heavy metal pollutants reported in the Pyra river sediments in Bangladesh. 36 Despite the fact that the presence of anthropogenic pollution sources for heavy metals along Bangladesh’s southern coast, its concentration are not reported above the environmental and health risk limits. 37 Whereas, the findings of this study has shown the higher concentration of heavy metals in the studied reservoir when it is compared with the heavy metals pollutants load in the Cox’s Bazar, Bangladesh. 38 In addition, the concentration of heavy metals in tributary river of Bangladesh 39 which was reported at acceptable limits which are far less than the findings of this study. In general, the majority of findings including this study have shown an acceptable limit of individual heavy metal despite its cumulative have created a significant environmental and health effects.
Mean Concentrations of Metals in Surface Water Samples at Each Site in This Study, and Maximum Concentrations Reported for Previous Studies (mg/L).
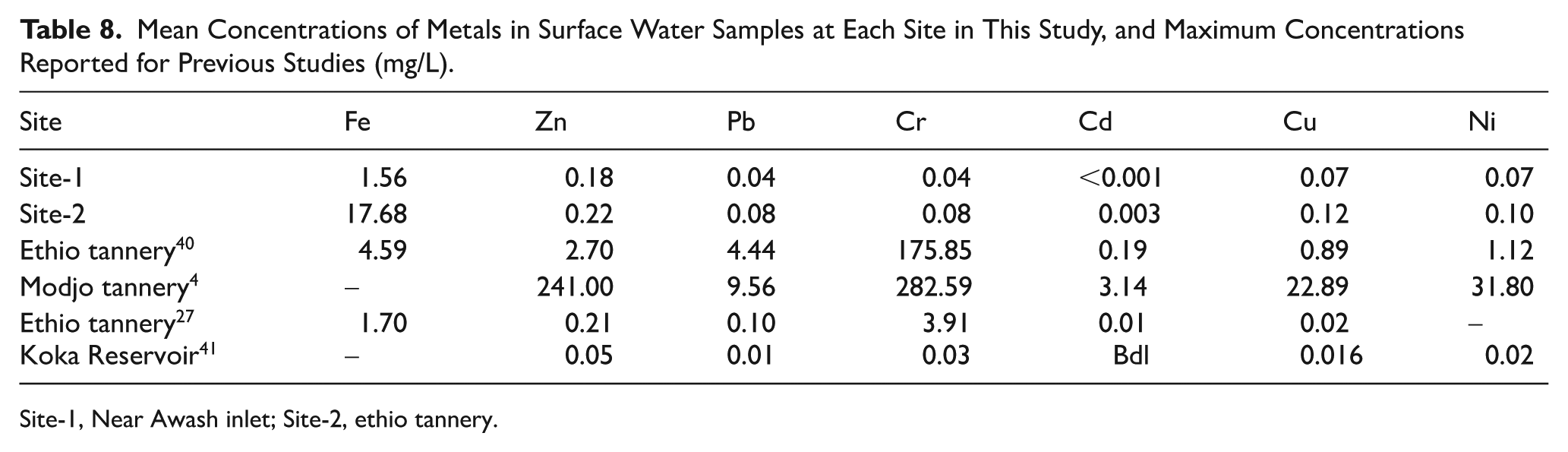
Site-1, Near Awash inlet; Site-2, ethio tannery.
The strong positive correlations among Zn, Cu, Ni, Cr, and Cd (Table 3) suggest a shared anthropogenic origin, consistent with research 42 demonstrating that mixed-metal pollution often results from combined industrial, agricultural, and urban sources. Contrasting with the relatively low concentrations of cadmium and nickel, even trace amounts pose significant long-term risks due to bioaccumulation and biomagnification, as highlighted in other study report.43,44 These findings reinforce the need for integrated watershed pollution management, including stricter wastewater treatment standards, pollution source control, and public health surveillance to mitigate human and ecological risks.
Heavy Metal Accumulation in Algae
Algae’s role as bioindicators and bioaccumulators is reaffirmed by the elevated metal concentrations recorded, particularly Fe and Pb, which surpass guideline thresholds. This aligns with the conclusions of study report, 45 who demonstrated that benthic and planktonic algae effectively concentrate metals, serving as early detectors of pollution and facilitating biomonitoring. The high accumulation of metals in algae is critical because it marks the initial step in metal transfer to higher trophic levels, threatening food web integrity. 46 The elevated Pb level is in parallel with the findings by other study 47 in similar African reservoirs impacted by tannery effluents. Importantly, the variation between sites highlights the influence of local pollution sources on metal bioavailability. From an applied perspective, these findings advocate for leveraging algae as sentinel species in routine monitoring frameworks and exploring phytoremediation technologies that harness algal metal uptake capacity to mitigate pollution. 45 Consequently, because algae occupy the foundational level of the aquatic food web, their accumulation of heavy metals directly channels these pollutants into higher trophic levels, ultimately endangering fish health and overall biodiversity. This underscores the importance of implementing more stringent industrial effluent controls alongside ecological approaches like phytoremediation or bioremediation, which harness the natural capacity of algae to absorb and sequester metals. Moreover, raising awareness among local communities about the health risks linked to consuming contaminated fish and aquatic vegetation is crucial for safeguarding both environmental integrity and human well-being.
Heavy Metal Accumulation in Fish
The substantial accumulation of Zn, Fe, Cr, and Pb in fish tissue exceeding recommended limits reflects the patterns reported in studies48,49 for industrially impacted freshwater ecosystems, where similar bioaccumulation leads to compromised fish health and elevated human exposure risks. In this regard, Study 50 reported the importance of Zn and Fe along with other metals that aligns with their ecological roles but also highlights potential toxic effects when in excess, including interference with metabolic processes and oxidative stress. Besides, the elevated Cr and Pb concentrations which surpass thresholds raise serious concerns due to their documented carcinogenic and neurotoxic effects. 51 The same literature also reflects the Cd human health risk at high concentrations. Despite its low water concentrations, the Cd presence above safety limits in fish confirms its tendency for biological concentration and long-term toxicity.
Over all, these collective evidences strengthen the concern for public health interventions such as fish consumption advisories, especially for vulnerable populations, such as children and pregnant women and stringent control of industrial discharges. Furthermore, integrating contaminant monitoring into fisheries management can safeguard food security and ecosystem health. From a policy perspective, fisheries management should incorporate contaminant monitoring and promote aquaculture practices that minimize exposure. Besides, the study advocates for environmental justice, ensuring that communities bearing the brunt of industrial pollution have access to clean food and water resources.
Bioaccumulation Factors (BAF) and Human Health Risks
Bioaccumulation factors (BAF) provide a useful metric to assess how effectively fish concentrate pollutants from their Bioaccumulation factors (BAF) are key indicators of a fish’s ability to concentrate pollutants from its environment; values above 100 signify notable accumulation. 52 The elevated BAFs for Zn and Cd in this study confirm their strong affinity for biological tissues, consistent with findings of other study, 48 who attributed such patterns to fish metabolism and metal bioavailability. The exceptionally high transfer factor for Zn (755.97) and Pb levels in edible fish tissue (3.29 mg/L) further raise potential food safety concerns. Given an average local fish consumption rate of 0.19 kg/person/week, 16 the estimated weekly intake (EWI) of Zn (25.71 mg/kg/week) surpasses the oral reference dose (RiDo) of 0.3 mg/kg/day, a threshold linked to adverse physiological effects, 53 and exceeds estimates reported from Aligarh, India. 54 Cr, present in both essential trivalent Cr(III) and toxic hexavalent Cr(VI) forms, poses additional concern; the EWI for Cr (0.71 mg/kg/week) in this study exceeded the U.S. EPA’s RiDo for Cr(VI) (0.003 mg/kg/day) though it remained below previous values from Koka Reservoir (29.87 mg/kg/week). 16 Cd levels (0.07 mg/kg/week) were lower than prior reports from the same site (0.19 mg/kg/week) 16 and from other study 16 (0.2 mg/kg/week) but still exceeded the EPA RiDo (0.001 mg/kg/day). Despite these moderate concentrations, the results underscore a potential chronic exposure risk.
Non-carcinogenic risk analysis showed target hazard quotients (THQ) followed the order Cd > Zn > Cu > Ni > Pb > Fe > Cr, consistent with study finding in Weldegebriel et al. 55 Cd contributed most to potential risk (1.2 × 10−3), followed by Zn (1.5 × 10−3) and Cu (2.1 × 10−4). All THQ values were below 1, indicating negligible non-carcinogenic risk from fish consumption. 56 Similarly, the hazard index (HI), 1.0 × 10−2 for males and 1.2 × 10−2 for females—remained well below the safety threshold, confirming limited cumulative risk. However, carcinogenic risk assessment revealed slightly higher Cr-related values in males (4.1 × 10−5) than females (5.5 × 10−5), reflecting physiological differences in body weight and exposure duration. 56 Although these lie within the generally acceptable risk range (10−6-10−4), Cr remains the primary carcinogenic concern associated with consumption of Cyprinus carpio from Koka Reservoir. While THQ values indicate minimal immediate threat, the elevated carcinogenic risk (CR) for hexavalent Cr, Pb, and Ni aligns with study finding, 57 highlighting long-term health implications from chronic exposure. These findings support the adoption of a precautionary environmental health policy that integrates chronic risk assessments with acute toxicity evaluation. Gender-based differences observed here also align with studies linking physiological and lifestyle factors to metal uptake and susceptibility. 58
Overall, the results call for continuous biomonitoring, community awareness on safe fish consumption, stricter industrial discharge control, and public health surveillance for metal-related diseases. Policymakers should also promote alternative livelihoods to reduce dependence on contaminated aquatic resources and safeguard food security in affected communities.
Implications
This study conveys significant implications that directly align with Ethiopia’s search of its national green development plan, the African initiatives in Agenda 2063, and the global Sustainable Development Goals (SDGs). The study has a focus on multiple SDGs such as, SDG 2 (Zero Hunger), SDG 3 (Good Health and Well-being), SDG 6 (Clean Water and Sanitation), SDG 12 (Responsible Consumption and Production), and SDG 14 (Life below Water). The evidence of heavy metal accumulation (Fe, Zn, and Pb) in the edible tissues of Common Carp from Koka Reservoir, exceeding WHO/FAO safety thresholds, exposes an often overlooked dietary risk to local communities that depend on these fish for affordable animal protein. This directly threatens food security and nutrition (SDG 2) while posing chronic health risks (SDG 3), highlighting the vulnerability of marginalized populations in rapidly urbanizing and industrializing areas.
These results thus, emphasize the urgent need for integrated monitoring strategies that combine routine water quality testing with systematic biomonitoring of the species like fish and algae, providing a clearer picture of both ecological and public health threats. Policymakers should act by enforcing stricter regulations on industrial discharges, particularly from high-impact sectors like tanneries, and reducing agrochemical runoff through better land-use planning and more sustainable agricultural practices, thereby supporting SDG 12 and SDG 6. Besides, targeted public health education campaigns are vital to raise community awareness about the potential hazards of consuming contaminated fish, enabling households to make safer dietary choices and reducing long-term exposure risks.
In addition to the study area, this research offers a scalable model for other lakes in Ethiopia, and for low and middle-income countries facing similar challenges of rapid industrialization and limited environmental concern, directly contributing to global efforts under SDG 14 to protect aquatic ecosystems. Generally, these findings call for coordinated, evidence-based policies that link environmental sustainability and food safety, strengthening the resilience of freshwater resources, supporting sustainable agriculture, and safeguarding the health of present and future generations.
Conclusion
This study demonstrates that some heavy metal contamination in Koka Reservoir poses significant health risks to communities relying on its fish as a dietary staple. The accumulation of metals such as iron, zinc, and lead in edible fish tissues exceeds international safety guidelines, raising particular concern over the carcinogenic potential of metals. The high bioaccumulation factors observed further reveal how effectively these metals move through the aquatic food web, highlighting that traditional water quality measurements alone are insufficient to capture the true ecological and human health risks. These findings call for immediate and coordinated interventions: stricter regulation of industrial effluents, especially from high-impact sectors like tanneries. Integrating food chain analysis into routine environmental monitoring is essential to ensure the long-term sustainability of freshwater ecosystems, protect biodiversity, and safeguard human health for current and future generations.
Footnotes
Acknowledgements
We would like to thank the Ethiopian Civil Service University for sponsoring this research.
Ethical Considerations
There is no clinical studies conducted in his studies, so that ethical consideration is not applicable.
Author Contributions
Mekonnen Maschal Tarekegn and Abu Regassa Melka have contributed in study design, data collection, and analysis and manuscript preparation. Yitayal Addis Alemayehu contributed for manuscript draft preparation and reviewing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Ethiopian Public Service University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data is available and will be shared up on request.





