Abstract
Background:
Despite the adoption of rotavirus vaccines, sporadic outbreaks of the virus have been reported in many parts of the world. These outbreaks are facilitated by several factors including the ease of transmission of rotavirus through water environments. This systematic review aimed to determine the global prevalence of rotavirus in water environments.
Methodology:
Comprehensive Boolean searches were conducted in PubMed, SCOPUS, and Web of Science. A total of 75 eligible studies were included in the study, from which data was extracted for both systematic review and meta-analysis. Extracted prevalence data was grouped according to six water categories: drinking water, untreated sewage, treated sewage, surface water, groundwater, and others. A single-group prevalence meta-analysis was conducted in RStudio version 4.3.3 subjecting the data to the random-effects model.
Results:
The included studies were conducted in 32 countries that span 5 continents: Africa, Asia, Europe, North America, and South America. The pooled prevalence of rotavirus in water environments was 40.86%. Among the individual water environments, untreated sewage had the highest prevalence (68.27%), followed by treated sewage (53.07%), surface water (33.40%), groundwater (25.64%) and drinking water (9.46%). Continental stratification of the prevalence data was as follows: Africa (51.75%), Asia (32.48%), Europe (55.90%), North America (41.80%), and South America (28.51%).
Conclusion:
There is a high prevalence of rotavirus in water environments, especially in untreated sewage, and in Europe. Further research is needed to find more efficient methods that can effectively eliminate rotavirus to insignificant levels in water environments.
Introduction
Rotaviruses are enteric viruses belonging to the Sedoreoviridae family. 1 Their genome consists of eleven segments made up of ten double-stranded mono-cistronic RNA molecules and one poly-cistronic RNA molecule that encode for structural and non-structural proteins. 2 The International Committee on Taxonomy of Viruses (ICTV) has grouped rotaviruses into 10 species: Rotavirus A, B, C, D, E, F, G, H, I, and J. Among these, rotavirus A and C have been identified as causative agents of diarrheal diseases in humans (mostly affecting children below 5 years) and animals. Rotavirus A is the most extensively studied while much is not known of rotavirus C infections in humans even in the present. 3 The two outer capsid proteins of rotavirus A, namely, VP4 (which is cleaved into VP6 and VP8 by trypsin) and VP7 are usually used in the molecular detection of the virus. Other proteins such as NSP2, NSP3, VP1, and VP3 are also used for detection. Molecular characterization of the outer capsid proteins of rotavirus A has led to the identification of several G and P genotypes. Currently, there are 42 G genotypes and 58 P genotypes identified by the Rotavirus Classification Working Group (RCWG). 4
According to the World Health Organization (WHO), rotaviruses stand as the predominant cause of severe diarrheal illness in infants and children worldwide, leading to over 2 million hospitalizations annually, and resulting in an estimated 527 000 fatalities in 2004. 5 While rotaviruses primarily cause severe gastroenteritis in children, which can lead to fatalities, adults can also experience milder forms of the illness. However, these adult cases typically do not result in death, largely due to the partial immunity acquired during childhood. The introduction of four (4) oral rotavirus vaccines, namely, Rotarix, RotaTeq, RotaSiil and Rotavac, within the past two decades, has drastically reduced the burden of rotavirus infections and mortality globally.6-8 In 2013, the WHO reported that 215 000 deaths representing 37% of global deaths among children aged <5 years were due to rotavirus infections. 9 This represents a significant reduction in the global death toll among children due to rotavirus and can be attributed to the effectiveness of rotavirus vaccines. A systematic review further provided evidence of the effectiveness of rotavirus vaccines by reporting a 59% reduction in rotavirus hospitalizations and a 36% decrease in rotavirus mortality among children below 5 years from data analyzed from 49 countries. 6 By the end of 2019, a total of 100 countries across the world had included at least one (1) of the four (4) rotavirus vaccines in their vaccination programs with Rotarix and RotaTeq being the most widely used.10,11 Despite this achievement, outbreaks of rotavirus infections have been reported in the post-vaccine era in Germany, the United States, Singapore, and Kiribati.12-15 These reports highlight the persistent threat rotavirus poses to public health, even with the availability of rotavirus vaccines.
Transmission of the virus is primarily via the fecal-oral route which makes water environments very important reservoirs for both human and zoonotic strains of rotaviruses. Several studies have detected rotavirus in both natural and artificial water sources that are used for various purposes including domestic, agricultural, and industrial uses, highlighting the need for a wider look at global prevalences of the virus in different water environments.16-18 Moreover, co-infection with different strains of rotavirus A enhances their ability to undergo genetic reassortment owing to their highly segmented genome leading to the emergence of new strains that are not covered by existing vaccines, some of which have been detected in water environments. 19 To the best of our knowledge, there has been no systematic review on the global prevalence of rotavirus in water environments, though a recent systematic review to determine the epidemiology of rotavirus in humans, animals, and the environment in Africa provided some limited information on environmental water sources. 20 This has necessitated the need for this comprehensive systematic review and meta-analysis of the global prevalence of rotavirus in water environments to help provide policy-related information needed to control and prevent transmission of the virus via the water route.
Methodology
Search Strategy and Data Sources
This systematic review and meta-analysis study adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines of 2020. 21 A comprehensive search was conducted for the study in the following databases: PubMed, SCOPUS, and Web of Science. The search was conducted using Boolean searches, which employ logical operators such as AND, OR, and NOT to combine or exclude keywords or phrases. The specific Boolean phrases used in the search are as follows:
PubMed: (“Rotavirus”[Mesh]) AND (“Rotavirus/classification”[Mesh] OR “Rotavirus/isolation and purification”[Mesh] OR “Rotavirus/pathogenicity”[Mesh] OR “Rotavirus/physiology”[Mesh]) AND (“X”[Mesh]) NOT REVIEW [Publication type]
SCOPUS: (“Rotavirus”) AND (“X”) not AND review [publication AND type]
Web of Science: (“Rotavirus”) AND (“X”) NOT (“Review”[Publication type])
“X” in the phrases above represents a Boolean phrase for a defined water environment. Supplemental Table 1. shows the water environments selected for the study and their corresponding Boolean phrases used for the searches.
Study Selection
Research articles published from January 2000 to February 2024 were scrutinized according to our set inclusion and exclusion criteria. Two investigators independently conducted data screening and together resolved any discrepancies. In instances where conflicting decisions arose on the inclusion or exclusion of a paper, both investigators came to a consensus. The parameters for the inclusion criteria were:
(1) Quantitative primary study;
(2) Studies in English; and
(3) Studies that contained prevalence data of rotavirus in a defined water environment.
On the other hand, the parameters for the exclusion criteria were:
(1) Qualitative studies;
(2) Systematic reviews, meta-analyses, case series, and case reports;
(3) Quantitative primary studies with less than or equal to (⩽)10 samples;
(4) Studies with no rotavirus detected; and
(5) Studies with no prevalence data on rotavirus.
A total of 1244 studies were obtained from the databases. Following the removal of duplicates, 770 studies remained for screening, while 650 studies were excluded based on inappropriate titles or abstracts. Among the remaining 120 studies, 75 were included based on the predefined inclusion and exclusion criteria (Figure 1).2,18,22-94
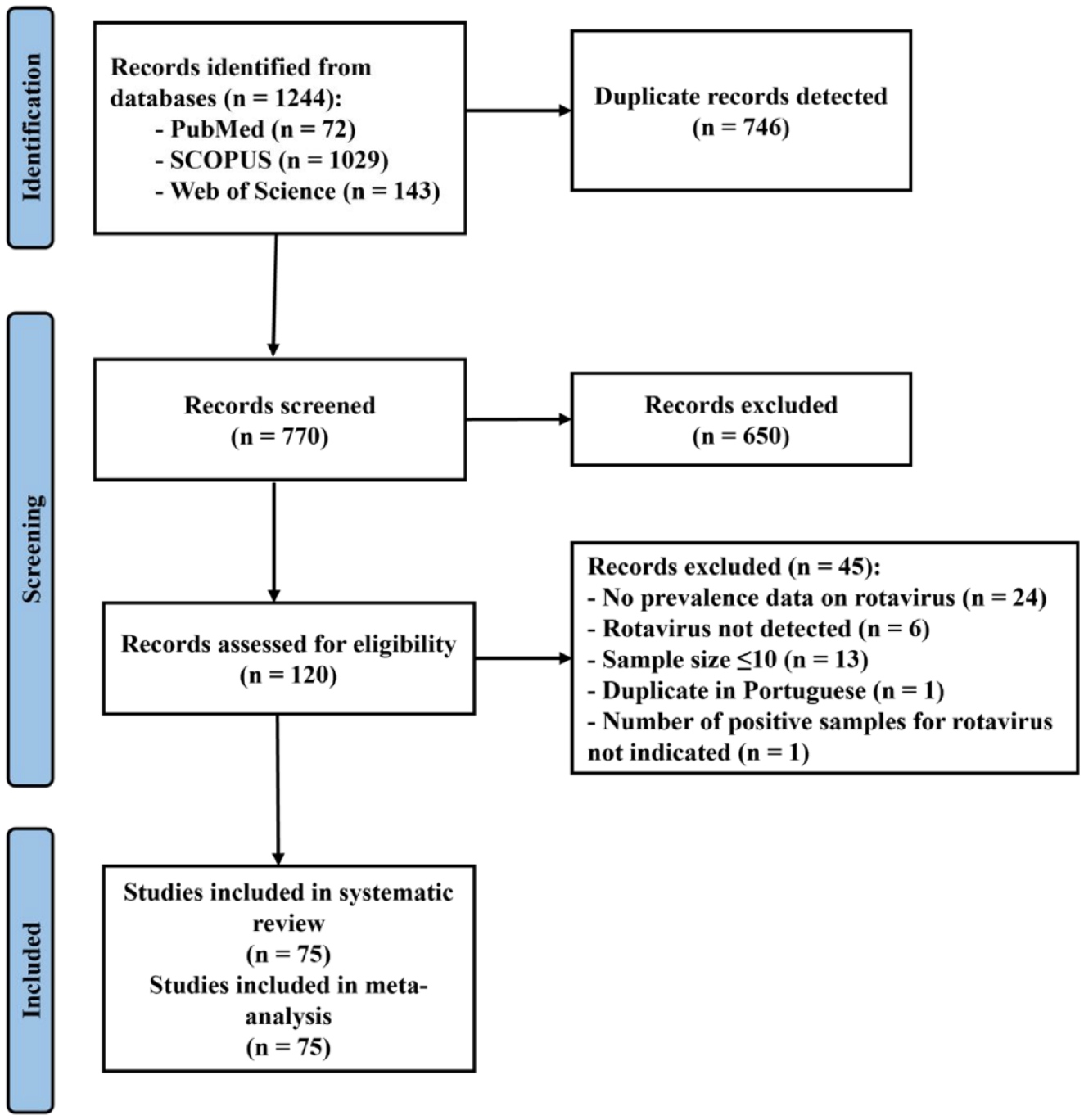
A PRISMA flow diagram detailing the screening done in this study.
Data Extraction
Data extracted from the studies included in this systematic review were; the name of the first author, country, study duration, water environments used for rotavirus detection, concentration method, detection method, rotavirus group detected, and rotavirus genotypes identified (Supplemental Table 3). For the meta-analysis, the number of positive samples and the total number of samples were extracted for defined water environments that met the total sample inclusion criterion of more than ten (10) samples.
Quality Assessment
External and internal validity assessment tools for prevalence studies modified from Hoy et al. 95 were used to determine the quality of the included studies. The external validity tools included the target population representation, reasonability of study samples use, adaptation of random selection and sample size calculation. The internal validity tools were the definition of the water environment, detection assay used, data collection modes, length of study and reporting of prevalence calculation parameters. By subjecting each of the included studies to these tools, study quality (risk of bias) was determined as either low risk (score = 7-9), moderate risk (score = 4-6) or high risk (score = 0-3; Supplemental Table 2).
Data Analysis
Prevalence data on rotavirus extracted from the 75 included studies were analyzed using RStudio version 4.3.3. The meta package was utilized for deriving pooled prevalence through the DerSimonian-Laird method. To ensure uniformity in variances among studies and facilitate pooled prevalence calculation, the Freeman-Tukey double arcsine transformation was employed. For individual studies, 95% confidence intervals were determined using the Clopper-Pearson method. Heterogeneity was assessed using the I2 statistic, where values of 25%, 50% and ≥75% were indicative of low, moderate and high heterogeneity, respectively. The Jackson method was applied to establish 95% confidence intervals for the I2 statistics. Publication bias was examined using funnel plots and Egger’s test. Subgroup analysis was conducted based on the water environment, continent, World Bank country income level, and country. A p-value of <.05 was considered statistically significant.
Results
Description of Study Characteristics
The 75 studies included in this systematic review were conducted from 1994 to 2022, with publication dates ranging from 2000 to 2024. They were conducted in 32 countries distributed across 5 continents including Africa (8 countries; 15 studies), Asia (8 countries; 20 studies), Europe (10 countries; 15 studies), North America (2 countries; 5 studies), and South America (4 countries; 19 studies). A total of 108 prevalence data of defined water environments were extracted from the 75 studies. The water environments were categorised as drinking water (7 studies; 9 prevalence data), untreated sewage (27 studies; 28 prevalence data), treated sewage (14 studies; 14 prevalence data), surface water (23 studies; 27 prevalence data), and groundwater (6 studies; 7 prevalence data); water environments that could not be included in any of the aforementioned categories were designated as others (20 studies; 23 prevalence data). The prevalence data was subgrouped into continents with Asia (32 prevalence data) having the most data, followed by South America (27 prevalence data), Africa (21 prevalence data), Europe (21 prevalence data) and North America (7 prevalence data). Out of the 75 studies, 46 of them explicitly detected rotavirus group A, (one) 1 study detected rotavirus group C and 1 (one) study detected both rotavirus groups A and C. The common G and P genotypes reported in the studies were G1 (7.7%-75%), G2 (7.2%-33.3%), G3 (2.9%-50%), G4 (6.1%-25%), P[4] (5.9%-67.5%), P[6] (0.6%-67%), and P[8] (10.8%-100%). Three (3) studies detected rotavirus vaccine genotypes in water environments: two (2) studies detected both Rotarix and Rota Teq genotypes and one (1) study detected only RotaTeq genotypes (Supplementary Table 3). The most commonly used rotavirus concentration method reported in the studies was the adsorption-elution method (20 studies), followed by filtration (19 studies), polyethylene glycol precipitation (17 studies) and centrifugation (8 studies). The most commonly used rotavirus detection method was One-step real-time quantitative polymerase chain reaction (RT-qPCR) (63 studies), followed by the enzyme-linked immunosorbent assay (ELISA) (3 studies).
Prevalence of Rotavirus in Water Environments
The global prevalence of rotavirus in water environments ranged from 9.46% to 68.27% in the different water environments with a pooled prevalence of 40.86% (95% confidence interval (CI) [34.04; 47.85]. In descending order, the prevalence was 68.27% (95% CI [58.43; 77.39]) in untreated sewage, 53.07% (95% CI [37.18; 68.66]) in treated sewage, 33.40% (95% CI [24.10; 43.35]) in surface water, 26.76% (95% CI [18.68; 35.62]) in others, 25.64% (95% CI [6.67; 50.72]) in groundwater, and 9.46% (95% CI [3.71; 17.18%]) in drinking water (Figure 2). Generally, prevalence of rotavirus in the various water environments was higher during the pre-vaccination era compared to the post-vaccination era (Table 1).

Global prevalence of Rotavirus in water environments.
Summary of global meta-analysis results for the prevalence of rotavirus in water environments stratified by rotavirus vaccination era.

Abbreviations: 95% CI, 95% confidence interval; NA, not applicable.
H is a measure of the extent of heterogeneity; a value of H > 1 indicates a potential heterogeneity of the prevalence of rotavirus.
I2 describes the proportion of total variation in prevalence of rotavirus that is due to heterogeneity; a value >50% indicates the presence of heterogeneity.
Heterogeneity and Publication Bias
The funnel plot (Supplemental Figure 1) illustrated a publication bias which was confirmed by Egger’s regression test (p = .016) in the different water environments. Estimated prevalences had significant heterogeneity across the different water environments (H > 1 and I2 ⩾ 75%). The overall prevalence also showed significant heterogeneity (H = 5.88 [5.60; 6.18] and I = 97.1% [96.8%; 97.4%]).
Subgroup Analyses
There was a statistically significant difference (p = 0) in the overall prevalence of rotavirus among the five continents that were represented in this study (Supplemental Figure 2). The highest prevalence was in Europe (55.90%; 21 prevalence data), followed by Africa (51.75%; 21 prevalence data) and North America (41.80%; 7 prevalence data). Based on the World Bank country income level (Supplemental Figure 3), the highest prevalence was recorded in low-income economies (65.00%; 1 prevalence data), followed by lower-middle-income economies (48.72%; 25 prevalence data) and high-income economies (48.27%; 40 prevalence data). For countries (Supplemental Figure 4), the highest prevalence was in the Netherlands (91.67%, 1 prevalence data) followed by Tunisia (88.52%; 4 prevalence data) and Sweden (88.46%; 1 prevalence data).
Discussion
The world has made commendable efforts in reducing the immense threat rotavirus poses to human health, especially, to children below the age of 5years. Despite these efforts, a significant threat to public health persists with the occurrence of sporadic waterborne outbreaks of rotavirus in many countries. The rapid transmission of the virus through water sources that are utilized for various purposes including domestic, industrial, and agricultural purposes highlights the importance of water in the effective control and prevention of transmission of rotavirus.75,96 To the best of our knowledge, no comprehensive review and meta-analysis on the global prevalence of rotavirus in water environments have been conducted. Hence, this systematic review and meta-analysis sought to bridge that gap by providing a clear overview of the global prevalence of rotavirus in water environments, reporting globally circulating rotavirus genotypes, and ultimately providing empirical evidence to guide future research, vaccine production, and efforts to effectively control and prevent the transmission of rotavirus in communities.
The prevalence of rotavirus in water environments in this study was 40.86% which is alarming taking into consideration the significant threat rotavirus infection poses to the lives of children globally. In assessing the prevalences among the various water categories used in this study, untreated sewage had the highest prevalence of 68.27%. This high prevalence is unsurprising as untreated sewage serves as a reservoir for enteric viruses including rotavirus and also mirrors the circulating pathogens in a community as both carriers and infected persons can equally pass out enteric viruses through feces. 34 However, this prevalence also indicates a significant shortfall in global efforts to effectively control and prevent the transmission of rotavirus.
Interestingly, the prevalence of rotavirus in treated sewage was 53.07%, which is not significantly different from that found in untreated sewage. This finding could be explained by reports of the ineffectiveness of biological and tertiary raw sewage treatment processes such as chlorination, ultraviolet disinfection, activated sludge, and ultrafiltration in destroying rotaviruses, specifically, those of rotavirus A.97-99
The prevalences of rotavirus in surface water and groundwater were 33.40% and 25.64% respectively. The relatively high prevalence in surface water could stem from the high prevalence of rotavirus in treated sewage which is discharged into surface waters and also contamination from animal feces. Then again, these results indicate a significant leakage of rotavirus into groundwater owing to their significantly high presence in surface waters. Other potential sources of contamination for groundwater and surface water include the machinery and tools used in groundwater well drilling, as well as the movement of rotavirus through soil pores into surface water. 25 The ability of viruses to move rapidly through soil was demonstrated by Paul and colleagues. 100 Although these factors remain consistent across urban and rural areas, it is expected that the prevalence of rotavirus in groundwater and surface water will be higher in rural areas, where these sources are more commonly used for water supply. 25
Drinking water had a 9.46% prevalence of rotavirus which is considerably high and calls for immediate attention. The relatively low immunity in children below the age of 5years increases their risk of getting infected from drinking water. A study that investigated a rotavirus outbreak in the Philippines in 2016 linked the outbreak incidence to a drinking water source. 101 This outbreak resulted in 22 deaths out of 2936 suspected cases, with children below 5 years of age being the most affected (65%; 1093 cases). It comes as no surprise that in 2018, the United Nations (UN) reported that the world was considerably distant from reaching the targets outlined in SDG 6 which is related to clean water and sanitation. 102 Furthermore, in a report from 2022, it was revealed that 2.2 billion people worldwide still did not have access to safely managed drinking water. 103 The SDGs Report 2023 highlights the urgency of speeding up progress sixfold to attain the target of SDG 6. 104 Implementing safer treatment and disposal of human excreta, restricting access to water bodies by farm animals, and developing more robust water treatment methods that can effectively eliminate rotavirus are measures that can help reduce the levels and transmission of rotavirus through drinking water.
The high prevalence of rotavirus in irrigation waters increases the risk of contamination of fruits and vegetables through irrigation and processing activities. This remains a great challenge to breaking the transmission route as there have been reports of contamination of fruits and vegetables which are usually eaten raw or undercooked.105,106 There have also been reports of some farmers using water from raw sewage tanks for irrigation of their crops due to the unavailability of appropriate water sources to be used for such purposes. 107 This practice directly leads to contamination of farm produce which can be a major vehicle of rotavirus transmission.
Continental subgroup analysis revealed very interesting patterns. Five continents; Asia, Africa, Europe, North America, and South America were represented in this study Figure 3. Notably, the prevalence of rotavirus in Europe (55.90%) was higher than in Africa (51.75%) and Asia (32.48%). Similarly, in the Americas, the rotavirus prevalence was higher in North America (41.80%) than in South America (28.51%). These findings could be explained by the presence of high vaccine hesitancy in Europe and North America, especially in the United States, which is largely influenced by negative perceptions in the general population.108,109 Parents often question the necessity of certain vaccines for their babies, which sometimes leads them to refuse the administration of these vaccines by health professionals. 109 This occurrence is less frequently experienced in Africa, Asia, and South America despite the relatively low literacy rates.110,111 The high vaccine hesitancy in high-income countries puts their children below the age of 5years at a higher risk of rotavirus infection as opposed to same-aged children in lesser economically developed countries. Country-specific health policies and rotavirus vaccination programs could also be implicated. For instance, Italy’s national policy and recommendations regarding the rotavirus vaccine consider it non-essential. Whiles in France, 41% of French respondents believed that vaccines were unsafe compared to 12% from other countries. 108 Nonetheless, the rotavirus prevalence in Africa is significantly high and may be explained by reports of low rotavirus vaccine efficacy in African children. 112 Contrary to the findings in Africa from this systematic review, the prevalence of rotavirus in water environments in a systematic review and meta-analysis of the epidemiology of rotavirus in humans, animals, and the environment in Africa was lower at 31.4% (18 studies; 24 prevalence data; 95% CI: 17.7%–46.9%). 20 The disparity in the prevalence of rotavirus in the 2 studies could be explained by the different parameters adopted in study selections. Additionally, the observed high prevalences across the 5 continents contradict the notion that rotaviruses thrive in cooler temperatures and disintegrate in warmer temperatures above 20°C, as these continents have wide variations in temperature.113-115 This indicates a potential evolutionary adaptation of rotavirus to the ongoing changes in global climate, ensuring its survival and transmission in diverse environmental conditions. It is also worth noting that other factors such as human behaviors, quality of hygiene, and sanitation arrangements in different geographical locations could account for our findings. 116
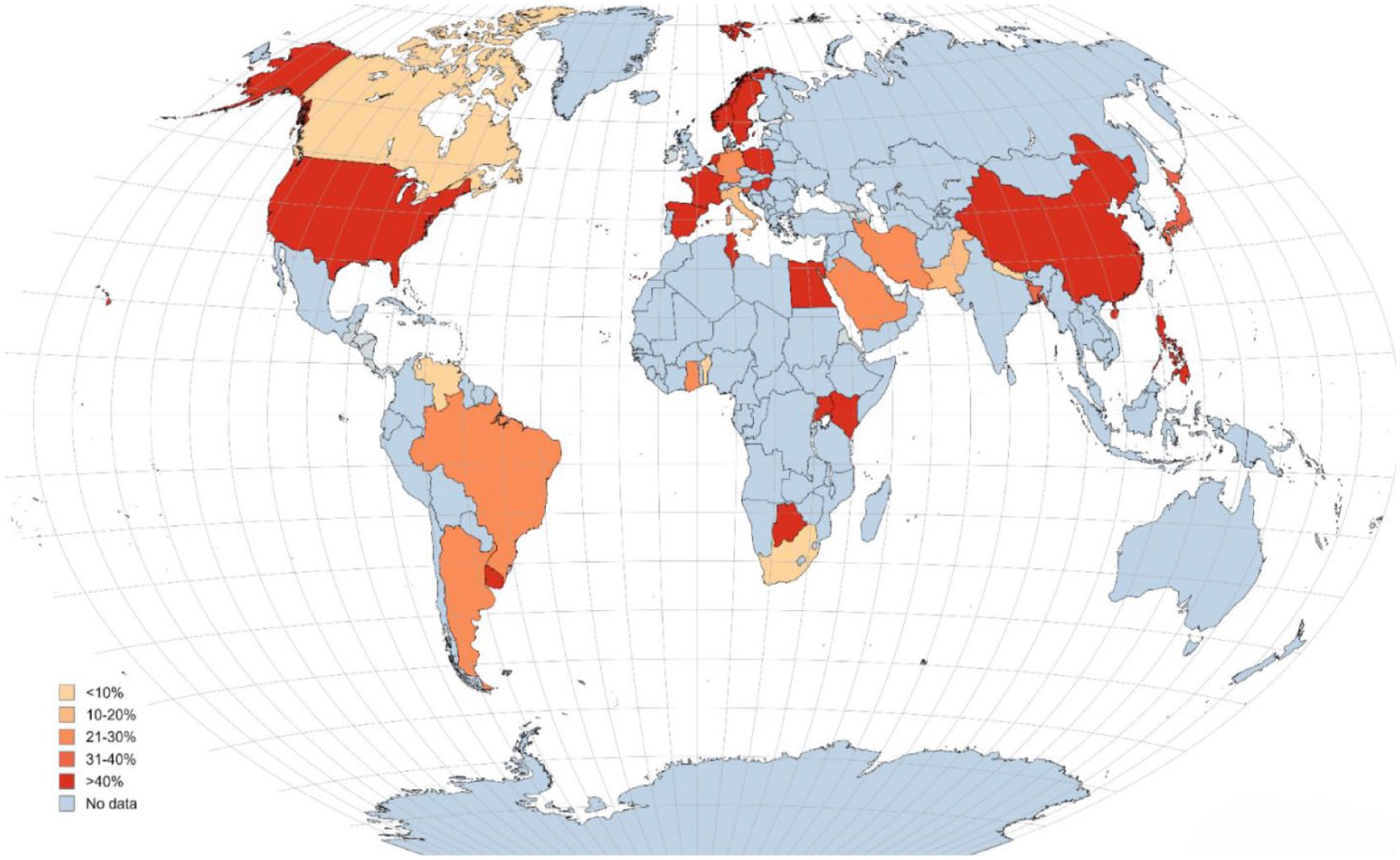
Global repartition of rotavirus prevalence in various water environments.
The economic strengths of countries directly affect all sectors of their economies including health. Stratifying rotavirus prevalence according to the World Bank country level of income revealed that high-income economies (France, United States, Spain, Italy, Slovenia, Japan, Germany, Hungary, Norway, Saudi Arabia, Netherlands, Uruguay, Poland, Sweden, Canada) had a higher prevalence of 48.27% compared to 29.50% of upper-middle-income economies (Brazil, South Africa, China, Argentina, Botswana). The prevalence in lower-middle-income countries (Pakistan, Iran, Egypt, Bangladesh, Tunisia, Philippines, Kenya, Ghana, Nepal, Benin) was 48.72% which was slightly higher than the high-income economies (48.27%). These findings suggest that upper-middle-income economies are adopting and implementing recommendations from the WHO for rotavirus control and prevention of transmission more effectively compared to high-income economies which are no different from lower-middle-income economies in terms of efforts. The prevalences in low-income economies (Uganda) and Venezuela (unclassified in the recent categorization by the World Bank) were 65.00% and 5.49% respectively. These findings could be highly influenced by the presence of only one prevalence data in each category and hence cannot be largely relied upon. We therefore recommend that more studies on rotavirus detection in water environments be conducted in low-income countries to provide ample information on rotavirus in their surroundings.
The country-based analysis of the prevalence revealed staggeringly high prevalences in the Netherlands (91.67%), Sweden (88.46%), Tunisia (88.52%), the Philippines (85.71%), and Botswana (84.44%). Conversely, low prevalences were recorded in Canada (4.0%), Venezuela (5.49%), South Africa (7.48%), and Nepal (8.12%).
Li and his colleagues detected rotavirus using both RT-qPCR and ICC-RT-qPCR. 55 Interestingly, the detection rates of the ICC-RT-qPCR method were higher compared to the RT-qPCR method, indicating its greater efficacy. Prevalence rates were 69%, 47%, and 14% for primary, secondary, and tertiary effluents, respectively, using the ICC-RT-qPCR method, compared to 44%, 22%, and 6% for the same effluents using the RT-qPCR method. The majority of the studies included in this systematic review employed RT-qPCR, indicating that the actual prevalence of rotavirus in water environments may be higher than what is reported. The ICC-RT-qPCR method involves infecting host cells with rotavirus concentrates from water samples and quantifying infectious rotavirus after 2 days. An additional advantage of this method over RT-qPCR is the reduction of PCR inhibitors, which are a major challenge in the RT-qPCR method. The sampling approach of water samples for the detection of rotavirus could also affect the detection rates. One study that employed both passive and active sampling methods found the former to give higher viral yields but only for small-quantity samples. 60 Recently, a review by Hayes and Gagnon also highlighted the efficacy of passive sampling methods over conventional active sampling methods. 117 Hence the use of passive sampling methods for future rotavirus detection works is recommended.
Our study revealed the global prevalence of common human G and P rotavirus genotypes as follows: G1 (7.7%-75%), G2 (7.2%-33.3%), G3 (2.9%-50%), G4 (6.1%-25%), P[4] (5.9%-67.5%), P[6] (0.6%-67%), and P[8] (10.8%-100%). With regards to the common G/P genotypes combinations, G1P[8], G2P[4], G3P[8], G4P[8], G9P[8], and G12P[8] were all identified. Notably, the G9P[4] strain that was reported to have emerged in Ghana and some Latin American countries after the introduction of rotavirus vaccination, has been identified in Kenya, South Africa, and Venezuela. 7 The prevalences of untypeable G and untypeable P genotypes were 4.2-50.0% and 12.5-66.7% respectively, in this study. These findings are suggestive of the high emergence of new strains through genetic reassortment that cannot be identified by current sequencing methods. Moreover, G/P combinations consisting of untypeable G and untypeable P genotypes have also been reported in water environments. These include G12P[?], G1P[?], G9P[?], G?P[6], G?P[8], and G?P[4].
Interestingly, studies conducted in Egypt (1 study) and Japan (2 studies) detected Rotarix and RotaTeq vaccine genotypes. In Egypt, both vaccine strains were detected, while in Japan, one (1) study detected only the RotaTeq strain, and the other study detected both strains. Rotarix and RotaTeq vaccines are administered twice before 24 weeks and 3 times before 32 weeks, respectively. Given that these vaccines are among the most commonly used globally, their detection in water environments could be attributed to their widespread usage. Additionally, research indicates that infants are typically weaned before 6 months (24 weeks) and 20 to 24 weeks in Egypt and Japan, respectively, after which their feces become part of sewage.65,118 This increases the likelihood of these vaccine strains reaching environmental waters. Further, this finding threatens the efficacy of the two (2) vaccines with the possibility of the emergence of vaccine-resistant strains looming.
The detection of rotavirus group C in 2 studies conducted in wells and sewage influent and effluent poses a threat to both adult and infant populations. While rotavirus group C typically causes mild diarrheal disease in adults, it has also been reported as a causative agent in diarrheal illness among infants. 3
The prevalence of rotavirus in untreated sewage, treated sewage, surface water, and groundwater environments decreased during the post-vaccination era (Table 1). Conversely, that of drinking water and the others category increased in the post-vaccination era. The approximately 9.5% increase in the prevalence of rotavirus in drinking water in the post-vaccination era is particularly worrying as it potentially points to an increase in rotavirus infections and outbreaks in the foreseeable future if drastic control measures are not taken.
One major limitation of this study pertains to heterogeneity. Specifically, 86.67% of the studies were conducted during the post-vaccination era, contributing to significant diversity in sample sizes across the 6 water environments, continents, and World Bank country income levels. The observation of very wide prediction intervals also reflects the high heterogeneity in the included studies. Other significant limitations were the use of different methods for the detection of rotavirus and varying conditions of sampling across continents. Additionally, the majority of studies (93.33%) exhibited a moderate risk of bias, whereas only 4% demonstrated a low risk of bias. Some of the potential sources of bias include differences in sampling frequency, sampling methods, analytical methods, and atmospheric conditions. Even though this study followed the PRISMA guidelines, it was not registered prior to its conduct.
Conclusion
There is a high prevalence of rotavirus in water environments, especially in untreated sewage, and in Europe. The high prevalence of rotavirus in water environments observed in this study calls for drastic control measures to be implemented. Moreover, the detection of new rotavirus genotypes that are not covered by the existing rotavirus vaccines suggests that plans for the development of new vaccines should be accelerated and the establishment of a rotavirus surveillance network similar in magnitude to that of other virus surveillance networks needs to be considered. Additionally, more research is needed to find more efficient methods to effectively eliminate rotavirus to insignificant levels in water environments.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302241276667 – Supplemental material for Rotavirus in Water Environments: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-ehi-10.1177_11786302241276667 for Rotavirus in Water Environments: A Systematic Review and Meta-Analysis by Aaron Awere-Duodu and Eric S Donkor in Environmental Health Insights
Footnotes
Correction (December 2024):
The article has been updated with some textual changes.
Author Contribution
Conceptualisation, ESD and AA-D; methodology, ESD and AA-D; validation, ESD and AA-D; formal analysis, ESD and AA-D; resources, ESD and AD-D; data curation, ESD and AA-D; writing—original draft preparation, AA-D; writing—review and editing, ESD and AA-D; visualisation, ESD and AA-D; supervision, ESD; funding acquisition, ESD.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review paper was supported by the Fogarty International Center of the National Institutes of Health through the Application of Data Science to Build Research Capacity in Zoonoses and Foodborne Infections in West Africa Training Program hosted at the Department of Medical Microbiology, University of Ghana Medical School (Award Number: UE5TW012566). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
