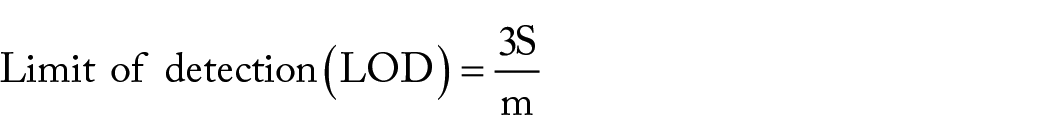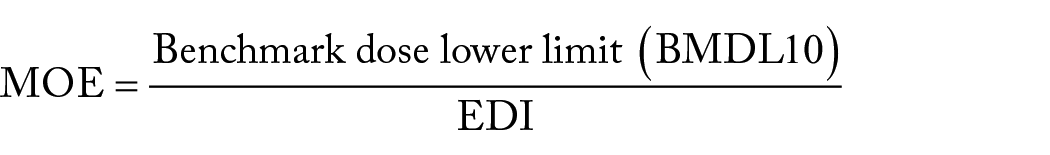Abstract
Aflatoxins (AFs) are secondary metabolites mainly produced by Aspergillus flavus and A. parasiticus and their contamination of red peppers can cause hepatocellular carcinoma, growth retardation in children, immune suppression, and death. In addition, their presence in the red peppers can affect international trade and cause significant economic burdens. Thus, the objective of this study was to assess the level of AFs contamination in packed powder (from supermarkets) and raw red pepper samples commercially available in the towns of Fiche and Mukaturi. Furthermore, this study aimed to determine the potential health and cancer risks associated with the consumption of red pepper contaminated with AFs. Red pepper samples (raw and packed powder) were collected randomly from the Fiche and Mukaturi open markets. Then AFs in the samples were extracted using methanol: water (80:20, v/v). These extract samples were then cleaned up using an immunoaffinity column (IAC) and determined with a high-performance liquid chromatography-fluorescence detector (HPLC-FLD). The finding showed that the amount of AFB1, AFB2, and AFG1 in raw red pepper was found to be 3.19 ± 0.01, 0.19 ± 0.001, and 4.07 ± 0.01 μg kg−1, respectively. The raw red pepper samples had a total of 7.66 ± 0.01 μg kg−1 of AFs. On the other hand, the amount of AFB1, AFB2, and AFG1 in Afiya-packed red pepper was found to be 7.04 ± 0.03, 2.15 ± 0.06, and 0.50 ± 0.01 μg kg−1, while Mudayi packed red pepper contained 31.60 ± 0.22, 24.40 ± 0.17, 3.37 ± 0.02 and 2.48 ± 0.004 μg kg−1 of aflatoxins, respectively. Afiya and Mudayi packed powder peppers had a total AFs content of 10.4 ± 0.07 and 61.90 ± 0.28 μg kg−1, respectively. The total AFs concentrations in packed pepper powder samples were higher than maximum toleratable limits (MTLs) set by the European Commission Regulation (EU) 2023/915 (5.00 μg kg−1 for AFB1 and 10 μg kg−1 for total AFs). AFB1 (31.60 ± 0.22 μg kg−1) had the highest level of contamination, followed by AFB2 (24.40 ± 0.17 μg kg−1) in packed pepper powder. In the adult population, the estimated daily intake (EDI) of AFB1, AFB2, AFG1, and AFG2 ranged from 0.80 to 7.90, 0.04 to 6.10, 0.02 to 1.02, and 0.05 to 0.62
Introduction
In Ethiopia, hot red pepper (Capsicum annuum L.) is a highly valued spice that is cultivated in various regions. 1 It is the second most widely consumed spice in the world and plays a crucial role in generating income for smallholder farmers. 2 Ethiopia produces 14 types of spices, including pepper, korarima, paprika, ginger, turmeric, fenugreek, garlic, coriander, capsicum, black cumin, cardamom, basil, and white cumin.3,4 Red pepper, a widely produced spice, enhances food taste and supplies vital vitamins and minerals. 5 It also contributes to economic development by boosting income for smallholder farmers, creating employment opportunities, and generating foreign exchange,3,5 thus reducing poverty and ensuring food security. In daily cooking, pepper is a crucial component at the home level. It is frequently used as a paste or sauce to alter the color, flavor, and aroma of nearly every type of food.
Despite the importance of hot red pepper for home consumption, household income, and the national economy, mycotoxin contamination caused by fungi during the stages of growth, harvesting, transporting, storing, and processing is a severe issue for global food safety. 6 A noteworthy illustration of a trade impact is the European market’s rejection of spicy peppers from Ethiopia, valued at over $10 million, due to hazardous levels of AFs and ochratoxin. In line with this, the EU Rapid Alert System for Food and Feed (RASFF) reports 7 that border rejections on spice items from Ethiopia were made between 2017 and 2019 due to elevated levels of AFs and ochratoxins (AFs or OTA) and a lack of verified analytical results.
Mycotoxins are secondary metabolites produced by Fungi, commonly called molds. 7 They are among the microbial toxins that pose the greatest threat to public health and hinder more extensive international trade in agri-food goods. 8 Deoxynivalenol (DON), AFs, ochratoxins, zearalenone (ZEA), T-2 toxins, and other mycotoxins with toxicological priority have been found in foods. 9 AFs are extremely toxic and are mainly produced by various strains of Aspergillus, including Aspergillus flavus, Aspergillus parasiticus, and Aspergillus nomius.10 -12 These secondary metabolites are known for their harmful effects. These AFs have the potential to contaminate various agricultural food products, including cereals, soybeans, grapes, tree nuts, groundnuts, coffee, chocolate, and spices, throughout the entire production and consumption process when environmental conditions are favorable.
A recent review by Eskola et al 13 suggests that about 60% to 80% of the global food crops are contaminated with mycotoxins. Additionally, nearly 4.5 billion people are potentially exposed to these harmful substances through their daily consumption of contaminated food.12,13 Among the 4 types of AFs (AFB1, AFB2, AFG1, and AFG2), AFB1 is considered the most toxic. It poses a significant risk for human health issues globally because it causes carcinogenic, teratogenic, hepatotoxic, mutagenic, and immunosuppressive effects on humans and animals.12,14 -17 AFs exposure at high levels has occasionally resulted in the deaths of both humans and animals. For instance, in 1981, the first occurrence of human deaths from consumption of aflatoxin-contaminated maize was reported 12 fatalities 9 in Machako district of Kenya. The second episode of the aflatoxicosis outbreak that occurred in Kenya in 2004 was the most significant worldwide, as it caused 317 cases with 125 deaths. 8 Therefore, it is necessary to analyze hot red peppers sold in commercial markets that came from other potentially produced areas to ensure the levels of these contaminants meet agreed international requirements to protect human health.
Recently only a few studies have been conducted to determine aflatoxins in hot red pepper cultivated in Ethiopia. For instance, earlier research on Ethiopian hot red pepper revealed that powdered red peppers from Addis Ababa had AFs contents ranging from 0.4 to 52.3 g/kg.
18
Similarly, a recent study by Tolera et al
19
revealed that only AFB1 and AFG1 were detected in packed pepper powder (43.61 AFG1 and 22.18 AFB1), followed by unpacked pepper powder (30.53 AFG1 and 13.50 AFB1). Another study by Fuffa and Urga
20
reported that the average contamination of Ethiopian red pepper containing AFB1 and AFG1 ranged from 26 to 75 and 32 to 120
Several countries have set regulatory limits for AFs in food and feed to protect humans against adverse effects from AFs exposure. 21 The European Commission (EC) has established the most stringent regulations for mycotoxins, including those found in Capsicum fruits. These regulations include MTLs for total AFs (AFB1 + AFB2 + AFG1 + AFG2) at 10 μgkg−1 and for AFB1 at 5.0 μgkg−1. 22 Therefore, regular monitoring of AFs occurrence in raw red pepper and packed powder pepper spices is needed to control compliance with food standards and protect the population from the risks associated with its proven toxicity and carcinogenicity. 23
Hence, the objectives of this study were to determine the levels AFs in raw red pepper and packed powder red peppers sold in the local markets of Mukaturi and Fiche town via HPLC-FLD (Figure 1). Furthermore, this study aimed to determine the potential health and cancer risks associated with the consumption of red pepper contaminated with AFs. Finally, the levels of investigated AFs were compared with recommended levels by European Commission Regulation (EU) 2023/915.

Schematic diagram of the analytical methods used for determining AFs levels in red pepper.
Materials And Methods
Description of the study areas
The study was conducted in Mukaturi and Fitche town of the North Shew Zone Oromia regional state, Ethiopia. The powder and raw red pepper used in this study were collected from available local markets in Mukaturi and Fiche town in the North Shoa Zone, Oromia regional state, Ethiopia. The distance of Fiche town from Addis Ababa, the capital city of Ethiopia, is 112 km, with a latitude of 9°47'59.99"N and a longitude of 38°43'59.99"E. It has an elevation between 2738 and 2782 m above sea level. Additionally, raw red pepper samples were collected from the available market in Mukaturi town in Wuchale Woreda, North Shoa Zone, Oromia Regional State, Ethiopia. The distance from Mukaturi to Addis Ababa, the capital city of Ethiopia, is 78 km, with a latitude of 9°33'0"N and a longitude of 38°52'0"E. Mukaturi is located at an elevation of 2652 m above sea level, as shown in Figure 2.

Sampling and sample preparation
According to EU 401/2006 regulations, a minimum sample weight of 500 g will be collected for the determination of AFs in spices. 26 The sampling process involved selecting 4 random sites from the Fiche and Mukaturi open markets, where red peppers are commonly sold. Then, 250 g of red peppers were collected from each location and mixed well to obtain 1 kg (1 kg) of sample from Fiche and 1 kg of sample from Mukaturi. The samples were carefully packed in polyethylene plastic bags, labeled, and transported to the Laboratory of Ethiopia Conformity Assessment Enterprise, Addis Ababa, Ethiopia. Similarly, 0.25 kg of Mudayi and Afiya packed powder red pepper (the most widely available and consumed powder red pepper) were randomly purchased from supermarkets (1 kg each). The raw hot red pepper samples were dried, ground, sieved through a 0.425 mm mesh, and then homogenized using a mortar and pestle. The dried, sieved, and homogenized samples were carefully stored in clean polyethylene bags and placed in desiccators until extraction and purification.
Apparatus and instruments
Analytical balance-IV BCTL/040, graduated pipettes (1, 5, 10, 25, and 50 mL), volumetric flasks (10, 25, 50, 100, 500, and 1000 mL), measuring cylinders (50 and 100 mL), beakers (50, 100, and 500 mL), conical flasks (250, 500 and 1000 mL), micropipettes, micropipette tips, syringe filters (0.45 µm pore size), disposable syringes (needle size: 22 × 11/4), paraffin, sample labels, vials with screw caps, and immunoaffinity columns (IACs) were purchased from Libios (Pontcharra-sur-Turdine, France) and contained antibodies against AFB1, AFB2, AFG1, and AFG2. The glassware was decontaminated by treating it with a sodium hypochlorite solution and then rinsing it with distilled water until it reached a neutral pH. The HPLC system setup included an autosampler, a column (C18 column 250 L × 4.6 mm, 5 μm), a degasser, and a fluorescence detector set to an excitation wavelength of 365 nm and an emission wavelength of 435 nm. Data collection was carried out using a desktop computer with Shimadzu LC software.
Chemical and reagents
Acetonitrile of high-performance liquid chromatography grade (manufactured by Fisher Scientific), n-hexane, methanol (>99.0%, Sigma Aldrich), sodium chloride (37% purity from Fisher Scientific), sodium phosphate dibasic and sodium dihydrogen phosphate, nitrogen gas (purity > 99.8%), and analytical grades of AFs standards (AFB1, AFB2, AFG1, and AFG2) used in the experiments were purchased from Sigma‒Aldrich (Burlington, Germany). The mobile phase consisted of a mixture of water: methanol: and acetonitrile (60:15:25) and was filtered using a Millipore filtration apparatus. Pure standard solutions of all four AFs were prepared in acetonitrile and methanol (50:50) at 1 mg mL−1 and stored in the dark at 4°C before use to prevent AFs degradation, which occurs when stored at room temperatures and exposed to sunlight.
Method validation
According to Raposo and Ibelli-Bianco,
27
method validation is an essential process that ensures the accuracy, reliability, and suitability of analytical methods for a particular task. The figures of merit are the standards used to evaluate the analytical techniques. The following figures of merit were evaluated as part of the technique validation strategy to identify AFB1, AFB2, AFG1, and AFG2 in raw and packaged red pepper samples: linearity, recovery, limit of detection (LOD), limit of quantification (LOQ), and precision.
28
A 7-point calibration curve with concentrations of 0.50, 1.00, 2.00, 3.00, 5.00, 7.00, and 10.0
Precision
According to Araki and Flynt, 30 the magnitude of random (indeterminate) errors associated with the use of an analytical method is referred to as precision. The range of conditions over which the data are collected determines the sources of random error that are analyzed. The relative standard deviation of the sample was calculated using
Limit of detection (LOD) and limit of quantification (LOQ)
According to Miller and Miller 31 (2010), the limit of detection is the lowest concentration of the analyte that results in an instrument signal (y) that differs significantly from the “blank” or “background” signal. The LOD, the most crucial validation measure, can be calculated in several ways. Among these techniques, visual evaluation, signal-to-noise ratio, standard deviation of the blank, and calibration curve procedures are among the most common. When an actual blank is unavailable or when it is impossible to measure the blank’s signal, such as when using instruments that automatically remove background noise from responses, 32 the LOD and LOQ are calculated using the residual standard deviation of a regression line or the standard deviation of the y-intercepts of regression lines. As a result, the slope of the calibration curve and the residual standard deviation of the regression line were used to calculate the LOD for the AFs. Based upon this, the LOD was obtained by multiplying the standard deviation of a regression line (S) by 3 and dividing it by the slope of the calibration line, as given by the equation below.
On the other hand, the limit of quantification (LOQ), or the smallest amount that can be measured with reasonable accuracy and precision, was calculated as
Sample extraction and clean-up
The procedure used for AFs extraction and cleanup was based on the instructions of the Ethiopian Confirmative Assessment Enterprise Agency (ECAE). Briefly, 25 g of sample and 2.5 g of sodium chloride were placed in a 250 mL beaker and mixed using a polytron homogenizer for 10 minutes at 6000 rpm. Then, 150 mL of methanol:water (80:20, v/v) was added. The mixture was filtered through fluted filter paper. Then, 5 mL of clear filtrate was transferred to a beaker, and 30 mL of phosphate-buffered saline (PBS) was added to bring the final filtrate to 35 mL and mixed well.
The aflatoxins were purified using an immunoaffinity column (IAC)
The immunoaffinity column (IAC) was prewashed with 10 mL of deionized water before the extracts were purified. After that, 35 mL of the filtrate was passed through a conditioned IAC Afla test (Libios). The 10 mL of deionized water was added, and the mixture was air-dried. AF was then eluted using 3 mL of methanol in a 4 mL vial. Then, the cleaned extract was evaporated using nitrogen to dryness at 40°C. After drying, derivatized using 200
Determination of aflatoxins in red pepper samples
The concentrations (µg kg−1) of 4 types of AFs (AFB1, AFB2, AFG1, and AFG2) in raw red pepper and packed powder red pepper were determined using HPLC‒FLD. The actual amount of AFs in
where:
A = µg L−1 Aflatoxins as elated
T = volume of sample solution (mL)
df = dilution factor
W = mass (kg) of the commodity represented by the final extract
Assessment of AFs exposure
The estimated daily intake (EDI) due to the consumption of AFs -AFs-contaminated red pepper was assessed for AFB1, AFB2, AFG1, and AFG2. The dietary exposure was calculated as the ratio of the product of the AFs concentration (
The margin of exposure characterization for AFS
To estimate carcinogenic risk, the margin of exposure (MOE) was calculated by dividing the benchmark dose lower limit (BMDL10) of AFs by the EDI.
The BMDL10 (benchmark dose level confidence limit of 10%), which is an estimation of the lowest dose that is 95% certain to cause no more than 10% cancer incidence, is recommended for use when calculating the MOE.34,35 The BMDL10 value for AFB1 was 0.170 µg kg−1bw d−1, and those for AFB2, AFG1, and AFG2 were 0.250 µg kg−1bw d−1 (Wang et al). It has been reported that when the MOE is less than 10 000, AFs are a potential risk to public health.
Cumulative risk assessment of the aflatoxins analyzed
The cumulative risk of carcinogenic and genotoxic mycotoxins was calculated using the combined margin of exposure (MoET) which was used for cumulative risk assessment in this study.36,37
Statistical analysis
The results are reported using descriptive statistics (mean, standard deviation, range) as the mean ± SD and percentage. To assess mycotoxin dietary exposure, left-censored data (those reported below the limit of detection (LOD) and not detected) were assigned a value equal to half the limit of detection (LOD/2) based on the standards outlined by the European Food Safety Authority. While other possibilities, including treating left-censored data as zero, could underestimate the actual description, this is thought to be the worst-case scenario that could exaggerate contamination and exposure levels. 36
Results and Discussion
Method validation
Calibration curves, limit of detection, and limit of quantitation
The standard and sample chromatograms showed good resolution and revealed that all 4 types of AFs (AFB1, AFB2, AFG1, and AFG2) could be clearly distinguished from 1 another. AFB1, AFB2, AFG1, and AFG2 were eluted in that order on the chromatogram, with retention durations of 8.11, 9.36, 9.92, and 11.7 minutes, respectively. Working standard concentrations, regression equations, and correlation coefficients (R2), for the determination of AFs using HPLC-FLD in both raw and packed powder pepper are shown in Tables 1 and 2. The correlation coefficient (R), which ranges from .992 to .9998, revealed that there was a good linear association between the analytical signal and the AFs concentration for all analytes. All analytes exhibited correlation coefficients greater than >0.992, which was considered proof that the data fit the regression line reasonably well. 38
Working standard concentrations, regression equations, and correlation coefficients for the determination of AFs using HPLC-FLD in raw red pepper.

Working standard concentrations, regression equations, and correlation coefficients for the determination of AFs using HPLC-FLD in packed red pepper.

Due to the lack of standardized, verified reference materials, the validity (accuracy) of the analytical methodologies employed for sample analysis in this work was established by spiking experiments. To determine recovery, standard AFs solutions were added to the raw and packed powder red pepper samples, as shown in Table 3. The recoveries of AFB1, AFG1, AFB2, and AFG2 from raw red pepper were 83.90%, 80.25%, 90.30%, and 89.40%, respectively. These values fall well within the range of 75.80%−90.30%, which is acceptable according to Gilbert and Anklam. 39 Additionally, according to Commission Regulation (EC) No. 401/2006,
Recovery results (% mean ± SD, μg kg−1, n = 3) were obtained for validation of the procedure for raw and packed red pepper samples.

The recoveries were adequate (between 70% and 110% for 1−10 μg kg−1 AFs, with RSD <20%) (European Commission, 2006/401).26 Additionally, the recoveries of AFB1, AFB2, AFG1, and AFG2 from packed powder red pepper were 93.10%, 83.10%, 98.20%, and 88.50%, respectively. These values fall between 83.33% and 98.15%, which is also acceptable according to Gilbert and Anklam. 39
The LODs and LOQs of the method are presented in Table 4, together with the mean values of AFs in raw and packed powder red pepper. The limits of detection (LODs) of AFB1, AFB2, AFG1, and AFG2 in raw red pepper were found to be 0.14, 0.17, 0.25, 0.23 μg kg−1, respectively. The limits of quantitation (LOQs) of AFB1, AFB2 AFG1, and AFG2 in raw red pepper were found to and 0.46, 0.56, 0.82, 0.76 μg kg−1, respectively (Table 4). The limits of detection (LODs) of AFB1, AFB2, AFG1, and AFG2 in packed red pepper were found to be 0.87, 0.99, 0.35, and 0.32 μg kg−1, respectively (Table 6). On the other hand, the limits of quantification (LOQs) of AFB1, AFB2, AFG1, and AFG2 in packed red pepper were 2.91, 3.32, 1.16, and 1.05 μg kg−1, respectively. With good precision, the RSD values for AFs ranged from 0.13% to 2.92% and 0.17% to 7.60% for packed powder and raw red pepper, respectively.
AFs concentration (mean ± SD, μg kg−1, n = 3), range, and percent relative standard deviation (% RSD) in raw red pepper and packed red pepper from Fiche and Mukaturi Town.

ND: not detected.
Aflatoxins concentrations in red pepper samples
The mean concentrations of AFs in raw red pepper from Fitche were AFB1 (3.19 ± 0.01), AFB2 (0.19 ± 0.001), and AFG1 (4.07 ± 0.01) in
AFs are food pollutants that cannot be avoided and are harmful to both human and animal health. These harmful chemicals also harm the economy and pose a threat to food safety. In this study, as indicated in Table 4, the levels of various types of AFs in raw and packed powder red pepper spices purchased from open markets (Fiche and Mukaturi) and supermarkets, respectively, were compared with those reported in the literature. The mean concentrations of AFs in raw red pepper from Fitche were AFB1 (3.19 ± 0.01), AFB2 (0.19 ± 0.001), AFG1 (4.07 ± 0.01) in
According to Fofana-Diomande and Kouakou,
40
low-level mycotoxin contamination of spices is associated with improved drying and storing conditions. However, the mean concentration of AFs in raw red pepper purchased from both markets is less than the EU limit of 5
The average AFs concentrations of the Afiya and Mudayi-packed pepper powders were found to be AFB1 (7.04 ± 0.03, 31.60 ± 0.22) AFB2 (2.15 ± 0.07, 24.40 ± 0.17), AFG1 (0.50 ± 0.01, 3.37 ± 0.02), and AFG2 (<LOD, 2.48 ± 0.004),
Risk assessment
The EDIs of AFs resulting from red pepper consumption were determined during the experiment. As shown in Table 5, the EDI values for AFB1, AFB2, AFG1, and AFG2 ranged from 0.05 to 0.620.05 to 0.62, 0.035 to 6.10, 0.023 to 1.02 and 0.80 to 7.90, respectively. The highest EDIs were obtained for AFB1 and AFB2 in packed red peppers from Mudayi, while the lowest EDIs were obtained for AFB1, AFB2, AFG1, and AFG2 in packed pepper from Afiya. However, the highest EDIs of AFB1 were recorded for packed pepper from Mudayi.
Estimated daily intake (EDI) for adults via consumption of raw and packed red pepper.

Abbreviations: EDI, estimated daily intake (
The average body weight of an adult in Ethiopia = 60 kg. 33
All AFs had MOE values smaller than 10 000 (Table 6), indicating that there are adverse health effects as a result of consuming red pepper obtained from sampling locations for adults. 44 As a result of MoET values lower than 10 000, cumulative exposure to AFs via the consumption of both raw and packed red peppers poses a potential health concern for adults. 45 .
MOE for adults via consumption of raw and packed red pepper.

BDML10 was used for AFB1 (0.170 µg kg−1bw d−1), AFG1 = AFB2 = AFG2 (0.250 µg kg−1bw d−1),36 and MoET (combined margin of exposure) = 1/[(1/MoEAFB1) + (1/MoEAFB2) + (1/MoEAFG1) + (1/MoEAFG2)]37
Conclusion
In this study, the total AFs contamination in raw red peppers and packed pepper powder collected from open markets (Fiche and Mukaturi) and supermarkets was determined. The levels of AFB1 and total AFs determined in the raw red pepper samples were found to be below the MTLs. However, the levels of AFB1 and total AFs in Afiya and Mudayi packed powder peppers were higher than the EU limits; indicating inadequate care during harvesting, production, and storage conditions and relatively unfavorable humidity. The MOE and MoET values obtained after the consumption of red pepper indicated that consumers are at greater risk of toxicity. Since packed red peppers contain more AFs than raw pepper, their long-term consumption may pose a potential hazard to public health, as indicated by the MOE and MoET values. Therefore, continuous monitoring of the levels of AFB1 and total AFs in red pepper should ensure the safety of AFs in food and minimize consumer exposure.
Footnotes
Acknowledgements
The authors would like to thank the Department of Chemistry, Salale University, for financial support for this research. Additionally, we would like to express our thanks to the Ethiopian Confirmative Assessment Enterprise Agency (ECAE), Addis Ababa, for providing laboratory facilities during the study period.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Department of Chemistry, Salale University
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions Statement
This research was conducted by Hundessa Alemu. Girma Salale wrote the manuscript, prepared the figures and tables. All the authors have read and agreed to the published version of the manuscript.
Availability of Data and Materials
All the data generated in the study are included in the manuscript.
Ethical Approval
All the authors have read, understood, and complied with the statement on “Ethical responsibilities of Authors,” as found in the Instructions for Authors.