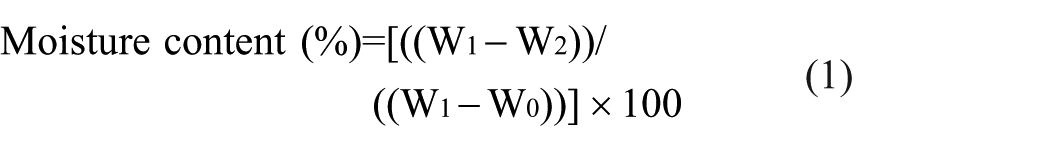Abstract
Valorisation of cashew apples (Anacardium occidentale L.) offers an opportunity to address food waste in Ghana, where approximately 90% of these edible fruits remain unutilised despite the country’s position among global leaders in cashew production. This study examined the development of mycotoxins in cashew apple-wheat composite flour systems during ambient tropical storage (25°C ± 2°C, 65 ± 5% RH) over 14 days. Three composite flour formulations were prepared for subsequent bread production, with cashew apple flour substituting wheat flour at 5%, 10%, and 15% (w/w) levels. The storage stability of these pre-mixed flour blends was evaluated, as they represent the critical phase when processors store prepared formulations prior to baking. Monitoring of moisture content, fungal growth, and aflatoxin B1 levels was conducted using standardised AOAC and ISO methods. An increase in moisture was observed across all formulations, with the 5% substitution reaching a moisture content of 13.21% by day 14. Fungal populations increased exponentially (102-104 CFU/g) and showed a strong positive correlation with moisture content (r = .87, P < .001). Aflatoxin B1 was detected on day 7, rising to 8.7 μg/kg in the 5% formulation by day 14, exceeding European Union safety limits (2 μg/kg) for cereal-based products. However, higher cashew apple substitution (15%) demonstrated dose-dependent antimicrobial effects, resulting in significantly lower fungal counts (6.8 × 103 CFU/g) and aflatoxin levels (4.1 μg/kg), likely due to naturally occurring phenolic compounds and other bioactive substances. While incorporating cashew apple flour offers considerable potential to reduce food waste and enhance nutrition, the current formulations pose unacceptable food safety risks under uncontrolled tropical storage conditions. For commercial viability, strict moisture control (⩽13%) and temperature regulation are essential to meet international food safety standards.
Plain Language Summary
This study evaluated microbial and aflatoxin contamination in suya powder, a popular spice blend used by khebab vendors across Sunyani Municipality, Ghana. Analysis of 85 samples revealed variable contamination levels, with total coliform counts ranging from undetectable to 3.8 log CFU/g, though Escherichia coli was absent. Detectable aflatoxin levels (predominantly G-type variants) remained below EU regulatory thresholds, likely due to thermal degradation during traditional peanut roasting. However, the persistent contamination poses health risks, particularly for children, as evidenced by elevated hazard indices. Spatial contamination patterns correlated with environmental factors including high temperatures (26-30°C), humidity fluctuations, and suboptimal storage conditions near drainage systems. These findings highlight the need for improved handling practices, moisture-controlled storage solutions, and regular monitoring to ensure food safety in Ghana’s informal street food sector. The study provides critical baseline data for developing targeted interventions while suggesting future research directions including seasonal variation assessments and evaluation of cost-effective preservation methods suitable for local contexts.
Introduction
Despite producing a substantial amount of agricultural produce, Africa experiences widespread food insecurity. 1 Ghana produces a large quantity of metric tons of cashew apples annually2,3; however, nearly 90% of these apples go to waste due to inadequate processing infrastructure. 3
Food waste refers to food suitable for human consumption that is discarded, lost, or allowed to deteriorate at various points along the food value chain. 4 This encompasses both active disposal and passive deterioration through inadequate handling or storage. The magnitude of this waste is a significant concern within Ghana’s food security landscape. 5 A study revealed that more than 90% of the cashew apples go to waste, while only about 10% of the cashew apples are utilised as fruits for domestic consumption in Africa. 6 This underutilisation persists despite the documented nutritional richness of cashew apples, which are rich in vitamin C, antioxidants, and bioactive compounds. 7 After juice extraction from cashew apples, the remaining fibrous material, termed cashew pomace, retains significant nutritional value, including dietary fibre, phenolic compounds, and antioxidants. When properly dried and processed into flour, this cashew pomace offers opportunities for food system integration.
Despite being the edible fleshy fruit accessory to the cashew nut, cashew apples face systematic underutilisation. Several causes contribute to this edible food waste. 8 These include various systemic failures within the agricultural value chain. 9 In rural farming communities, inadequate cold storage facilities are prevalent, which are essential for preserving the highly perishable cashew apples. 10 The predominant smallholder farming systems in Ghana lack the capital investment and infrastructure needed to establish cold storage facilities, contributing significantly to post-harvest losses of cashew apples. 2 Cashew, as a seasonal crop, also exacerbates these challenges. There is always a temporal concentration of cashew that exceeds the processing capacity available in local communities. In Tanzania, only 5% of raw cashews undergo local processing due to limited processing capacity and inefficient tools, resulting in substantial economic losses. 11 Additionally, the cashew apple’s inherent astringency presents a significant sensory barrier to consumer acceptance,8,12 while limited technological knowledge restricts value-addition opportunities at the community level.
Various studies have valorised cashew apples by incorporating them into shelf-stable products.3,10,13 -15 Cashew pomace flour can be incorporated into various food matrices, including baked goods, cereals, and functional food products.16 -18 Cashew pomace, when dried and milled into flour, is high in dietary fibre and antioxidants, addressing both food waste reduction and nutritional enhancement objectives. 7
However, the drying of cashew apples introduces food safety considerations that remain inadequately addressed in the current literature. Drying is a globally accepted method of preservation for fruits and vegetables. 19 Though drying extends the shelf life of food, the process of drying can create a conducive environment for mycotoxin production. 20 The humidity and temperature in the storage are key environmental factors that affect fungal growth and mycotoxin production, making them the primary reasons for mycotoxin problems in dried fruits. 20
Aflatoxins, produced primarily by Aspergillus species, represent the most significant mycotoxin threat in dried fruit products.21,22 These compounds are carcinogenic and are classified as Group 1 carcinogens by the International Agency for Research on Cancer (IARC). 23 The maximum levels for mycotoxins in food are very low due to their severe toxicity. 21
This study aimed to evaluate the dynamics of mycotoxin contamination in cashew-wheat composite flour formulations for bread production, incorporating dried cashew apple flour at 5%, 10%, and 15% levels during 14 days of storage under tropical conditions. The research aimed to investigate the relationship between moisture content, fungal load, and aflatoxin production, with the goal of establishing safe utilisation strategies for agricultural waste valorisation in tropical food systems.
Materials and Methods
Raw Material Acquisition
Fresh cashew apples (Anacardium occidentale L.) were sourced from a single commercial plantation located in Wenchi District, Bono East region of Ghana. The plantation was selected based on uniform cultivation practices, consistent varietal composition, and absence of recent pesticide application (minimum 30-day withdrawal period). Cashew apples were harvested at physiological maturity, identified by characteristic bright red colouration and firm texture, during the peak harvest season (March-April 2024).
A total of 50 kg of cashew apples were collected from randomly selected trees across the plantation to ensure representative sampling. Fruits showing visible signs of mechanical damage, insect infestation, or fungal contamination were excluded from collection. Harvested apples were transported to the laboratory within 6 hours of collection in ventilated plastic crates to minimise postharvest deterioration.
Cashew Apple Flour Preparation
Upon arrival at the laboratory, the cashew apples were sorted and thoroughly washed with potable water to remove field debris and surface contaminants. The cleaned apples were processed to extract juice, with the remaining cashew pomace (the fibrous residue after juice extraction) used for flour production. The wet pomace was spread uniformly (2-3 cm thickness) on perforated trays and dried using a cabinet-type solar dryer equipped with transparent polycarbonate glazing and black-painted absorber plates. Drying was conducted over 3 to 4 consecutive sunny days with ambient temperatures ranging from 28°C to 35°C and solar irradiance levels of 400 to 800 W/m2. The pomace was periodically turned every 4 hours during daylight to ensure uniform moisture removal. Drying was considered complete when moisture content reached below 10%, as determined by a portable moisture analyser (Sartorius MA35, Germany). The dried material was milled using a laboratory hammer mill (Thomas-Wiley, Model 4, USA) and then sieved through a 0.5 mm mesh to obtain a uniform particle size. Wheat flour was procured from the Sunyani Main Market and stored at 4°C until use. The cashew pomace flour and wheat flour composites (CPF:WF) were 5:95, 10:90, 15:85; there were also 100% wheat flour (WF) and 100% cashew pomace flour (CPF). The resulting composite flour blends were the final products subjected to storage testing and analysis. No further processing (such as bread making) was performed on these flour samples, as this study focussed on establishing safe storage parameters for the flour formulations that would subsequently be used in bakery applications. This approach aligns with standard industry practice in which flour composites are prepared, stored, and subsequently used in production.
Storage Conditions and Sampling Protocol
Following cooling to ambient temperature, the samples were individually wrapped in polyethene bags and stored under controlled ambient conditions (25°C ± 2°C, 65% ± 5% relative humidity) in an environmental chamber (Binder KBF 720, Germany). The selected storage environment was designed to simulate typical tropical storage conditions prevalent in Ghana and similar West African countries, where average indoor storage temperatures of 24°C to 27°C and relative humidity of 60% to 70% 24 during the cashew harvest season (March-May). These conditions represent realistic scenarios for small-scale processing facilities and market storage environments where cashew apple products would likely be stored. The 14-day storage period was selected based on: (1) preliminary studies showing detectable mycotoxin development within 7 to 10 days under tropical conditions, (2) typical shelf-life expectations for processed flour products in Ghanaian markets (7-14 days), (3) the exponential phase of fungal growth and mycotoxin production typically occurring within the first 2 weeks under favourable conditions, 20 and (4) practical considerations for small-scale producers who need rapid assessment protocols. Previous studies on similar composite flours have shown that critical mycotoxin levels develop within 10 to 15 days under comparable conditions. 25 Sampling was conducted at regular intervals: immediately after cooling (day 0), and subsequently on days 7 and 14 of storage.
Moisture Content Determination
Moisture content was determined gravimetrically using the AOAC Official Method 925.10, 26 with minor modifications. Duplicate 5 g samples from each sample replicate were weighed in pre-dried aluminium dishes using an analytical balance (Sartorius ED224S, Germany) with 0.1 mg precision. Samples were dried in a forced-air oven (Memmert UF110, Germany) at 105°C ± 2°C until constant mass was achieved, typically requiring 4 to 6 hours. Dishes were cooled in a desiccator containing silica gel before final weighing.
Moisture content was calculated using the formula:
Where
W0 = weight of empty dish
W1 = weight of dish + fresh sample
W2 = weight of dish + dried sample.
Fungal Enumeration and Isolation
Fungal load determination followed a modified version of ISO 21527-1:2008 from. 27 Sample preparation involved aseptic homogenisation of 10 g flour samples in 90 ml sterile peptone water (0.1% w/v) using a stomacher (Seward 400, UK) for 2 minutes. Serial decimal dilutions were prepared in sterile peptone water to achieve dilution factors ranging from 10−1 to 10−4. Enumeration was performed using the pour plate technique with Potato Dextrose Agar (PDA, HiMedia, India), which was acidified to a pH of 3.5 ± 0.1 using a sterile tartaric acid solution (10% w/v) and supplemented with chloramphenicol (100 mg/l) to suppress bacterial growth. Duplicate plates were prepared for each dilution level, with 1 ml inoculum mixed with approximately 15 ml tempered agar medium (45°C ± 2°C). Plates were gently swirled to ensure uniform distribution and allowed to solidify before incubation. Incubation was conducted at 25°C ± 2°C for 5 to 7 days in a temperature-controlled incubator (Memmert IN110, Germany). Colony counts were performed using a colony counter (Stuart SC6+, UK), with results expressed as colony-forming units per gram (CFU/g) of flour. Only plates containing 10 to 100 colonies were considered for counting, and final counts were calculated as the geometric mean of duplicate determinations.
Aflatoxin Extraction
Aflatoxin was extracted using QuEChERS (Quick Easy Cheap Effective Rugged Safe) methods 28 with slight modifications of acetonitrile: acetic acid v/v (9:1) as the extraction solution and additional agitation steps. Samples were homogenised using a mixer blender. A weight of 2 g of sample was transferred into a 50 ml centrifuge tube, 5 ml of deionised water was added and allowed to stand for 15 minutes. Afterwards, 5 ml of the extraction solution was added. The resultant mixture was vortexed using the Genie Vortex machine for 3 minutes and agitated using the Ohaus Orbital Shaker at 250 rpm for 15 minutes. A mixture of 2.0 g of anhydrous MgSO4 and 0.5 g of NaCl was added, vortexed for 1 minute, and agitated at 250 rpm for 5 minutes. The tube was centrifuged for 5 minutes at 4000 rpm, and the upper organic layer was filtered through a 0.45 µm nylon syringe before injection. A volume of 20 µl of the filtered extract was injected into the high-performance liquid chromatography (HPLC).
HPLC Aflatoxin Determination
HPLC analysis was performed according to AOAC Official Method 2005.08 (AOAC, 2006), as described by Asare Bediako et al. 29 A Photochemical Reactor for Enhanced Detection (PHRED) was used for post-column derivatisation. An Agilent 1200 Quaternary Pump with a fluorescence detector (Ex: 333 nm, Em: 477 nm) with a Nucleodur® plus C18 column (4.6 mm × 150 mm, 5 µm) was used to determine aflatoxin. The mobile phase used was water: methanol: acetonitrile (60:20:20, v/v) at a flow rate of 1 ml/min with column temperature maintained at 40°C. Post column derivatisation was achieved using LCTech UVE. The analytical method was validated for aflatoxin B1, B2, G1, and G2 detection. Calibration curves were constructed using certified reference standards (Sigma-Aldrich, USA) at concentrations ranging from 0.1 to 20 μg/kg, achieving linearity (R2 > 0.999) across this range. The limits of detection (LOD) and quantification (LOQ) were 0.03 and 0.1 μg/kg, respectively, for all 4 aflatoxin congeners. However, only aflatoxin B1 was detected in any samples throughout the study period; aflatoxins B2, G1, and G2 remained below the detection limits in all analyses.
Quality Control and Sample Handling
Control samples were prepared using certified reference materials for microbial and aflatoxin analyses. All samples were stored in sterile containers at 4°C and analysed within 24 hours of collection to maintain sample integrity. Method validation was performed using spiked samples with known concentrations of target organisms and aflatoxins, achieving 95% to 98% recovery rates for bacterial counts and 92% to 96% for aflatoxins. Control samples were prepared using certified reference materials (CRM-AFM-001, LGC Standards, UK) for microbial and aflatoxin analyses.
Method reproducibility was confirmed by replicate analyses of spiked samples (n = 6) at 3 concentration levels (2, 5, and 10 μg/kg), yielding relative standard deviations (RSDs) of 3.2%, 2.8%, and 2.5%, respectively, indicating acceptable precision (<5% RSD). Inter-day reproducibility (n = 3 days) showed RSD values of 4.1%, 3.7%, and 3.4% at the same concentration levels, confirming the reliability of the analytical method across different days of analysis.
Statistical Analysis and Data Visualisation
All analyses were conducted using R software (version 4.3.0). Graphs were created using the ggplot2 package, adhering to professional formatting standards. Statistical significance was determined using one-way ANOVA followed by Tukey’s HSD test for multiple comparisons. Correlation analyses were performed using Pearson correlation coefficients. All figures were prepared at 300 DPI resolution in vector format to ensure publication quality. Error bars represent standard deviation unless otherwise specified.
Results and Discussion
Moisture Content Changes During Storage
The moisture content of cashew-wheat flour composite samples showed variations across different cashew apple flour incorporation levels and storage periods (Table 1). At day 0, moisture content ranged from 7.47% (CPF) to 11.58% CPF:WF (5:95). The control wheat flour maintained a moisture content of 11.33% ± 0.82%.
Moisture Content (%) of Cashew-Wheat Flour Samples During Storage.

Abbreviations: CPF, cashew pomace flour; WF, wheat flour.
Values are mean ± standard deviation (n = 3). Different superscript letters within rows indicate significant differences (P < .05).
The incorporation of cashew apple flour at 15% concentration (CPF:WF (15:85) resulted in the lowest initial moisture content (10.14%), though with the highest variability (±0.28), suggesting potential processing inconsistencies. CPF:WF (5:95) demonstrated the most consistent moisture content with minimal standard deviation (±0.02) initially.
All samples showed progressive moisture uptake during the 14-day storage period. (CPF:WF (15:85) exhibited the most significant moisture gain (2.62% increase), rising from 10.14% to 12.76%, likely due to its initially low moisture content creating a higher driving force for moisture absorption from the storage environment (65% ± 5% RH). CPF:WF (5:95) reached the highest final moisture content (13.21%), crossing the critical 13% threshold associated with increased mycotoxin risk. The CPF sample showed substantial moisture uptake (3.4% increase) due to its very low initial moisture content (7.47%).
An increase in moisture content affects the shelf stability of many dried fruit products, 30 as it influences the rate of deterioration, susceptibility to microbial growth, and overall storage potential. In other dried fruits, moisture increases in storage and is attributed to the hygroscopic nature of cashew pomace flour, 3 which readily absorbs moisture from the surrounding environment. The hygroscopic characteristic of flour materials causes moisture content to change with temperature and humidity of the storage environment, leading to quality degradation through biochemical reactions that affect protein, starch, and lipid components. 31 Storage conditions play a crucial role, as demonstrated in dried apricot studies where temperature significantly affected moisture evolution in plastic packaging, with moisture content varying from 10.9% at 35°C to 27.9% at 5°C after 354 days of storage. 32 Inadequate packaging with poor barrier properties allows moisture migration from the storage environment into the product, particularly evident in porous packaging materials like polypropylene bags compared to hermetic alternatives. 31 The critical moisture threshold for optimal shelf stability has been established for various dried fruits, with tunnel-dried samples showing significantly lower moisture content (14.0%-15.1%) and extended shelf life of 38.2 to 41.5 months compared to cabinet-dried samples. 33 For cashew-based products specifically, gradual moisture increase during storage from 45 to 225 days has been observed, with packaging materials of lower oxygen and moisture permeability, such as PET and polyethene, proving most suitable for long-term storage. 34
Fungal Load Assessment
Fungal enumeration was conducted using the pour plate technique on acidified PDA medium, with results expressed as colony-forming units per gram (CFU/g) of flour. The fungal load showed a progressive increase throughout the storage period, with distinct patterns observed across different sample types (Table 2) and the fungal load changes over time is shown in Figure 1.
Fungal Load (CFU/g) in Cashew-Wheat Composite Flour Samples During Storage.

Progression of fungal contamination in cashew-wheat composite samples during storage at 25°C ± 2°C and 65% ± 5% RH. Data points represent mean ± SD (n = 3). Different letters indicate significant differences (P < .05) between time points for each sample type. Exponential growth patterns are evident between days 0 to 7, with growth rate stabilisation occurring thereafter.

Changes in fungal load during 14-day storage under tropical conditions.
All samples showed relatively low initial fungal counts, ranging from 0.3 × 102 CFU/g (CPF) to 1.2 × 102 CFU/g CPF:WF (5:95). The CPF sample exhibited the lowest initial contamination, likely due to more controlled processing conditions. CPF:WF (5:95), (5% cashew incorporation) showed the highest initial fungal load, which may be attributed to the lower concentration of potentially antimicrobial cashew compounds. A dramatic increase in fungal load was observed between day 0 and day 7, with all samples showing 10 to 25 fold increases. Similar observations have been reported in flour storage studies, where increased moisture content creates favourable conditions for fungal proliferation through enhanced enzyme activity and metabolic processes. 31 This exponential growth phase coincided with moisture content increases above 12% in most samples. The growth pattern observed is characteristic of the log phase of microbial growth, where optimal environmental conditions support rapid cell division and colony expansion. The growth rate appeared to stabilise somewhat between day 7 and day 14, though absolute numbers continued to increase.
The 15% cashew incorporation, CPF:WF (15:85), consistently showed the lowest fungal counts throughout the storage period, despite significant moisture uptake. By day 14, CPF:WF (15:85) had a fungal load of 6.8 × 103 CFU/g, compared to 1.4 × 104 CFU/g in CPF:WF (5:95). This suggests that higher cashew incorporation levels may provide antimicrobial benefits, possibly due to increased concentrations of phenolic compounds or other bioactive components (Figure 2).

Correlation between moisture content and fungal load across all sample types.
A strong positive correlation (r = .87, P < .001) was observed between moisture content and fungal load across all samples and time points. Samples that reached moisture contents above 13% CPF:WF (5:95) showed the highest fungal counts, while those maintaining lower moisture levels showed correspondingly reduced fungal proliferation. This relationship has been extensively documented in dried fruit storage studies, where moisture content above critical thresholds significantly accelerates microbial proliferation and subsequent quality deterioration. 6
Fungal counts exceeding 104 CFU/g were observed in CPF:WF (5:95) by day 14, approaching levels that may pose food safety concerns. Similar fungal load progressions have been reported in solar-dried mangoes, where storage under high humidity conditions (>65% RH) promotes rapid microbial growth and reduces shelf stability. 33 The fact that CPF:WF (5:95) reached 1.4 × 104 CFU/g by day 14 suggests that lower incorporation levels may not provide sufficient antimicrobial protection under tropical storage conditions.
The stabilisation of growth rates between day 7 and day 14, despite continued absolute increases in colony counts, suggests that the microbial populations may be approaching the stationary phase of growth. This plateau effect could be attributed to nutrient depletion, accumulation of metabolic waste products, or the establishment of competitive inhibition among different fungal species. Previous studies on cashew-based products have shown similar growth patterns, where reduced growth rates follow initial exponential increases as environmental conditions become less favourable. 34
The pure cashew pomace flour (CPF) sample demonstrated the most favourable microbial profile throughout the storage period, with the lowest initial contamination (0.3 × 102 CFU/g) and the most controlled growth progression (3.2 × 103 CFU/g by day 14). This superior performance can be attributed to the concentrated levels of antimicrobial compounds in the pure cashew material, as well as its lower moisture content and potentially altered pH environment that inhibits fungal proliferation.
These findings have significant implications for the practical application of cashew apple flour in commercial food production. The demonstrated antimicrobial benefits of higher incorporation levels suggest that formulations with 15% or greater cashew apple flour content may provide inherent preservation benefits, potentially reducing the need for synthetic preservatives in tropical storage environments. However, the critical importance of maintaining moisture content below 13% remains paramount, regardless of the level of natural antimicrobial compounds present.
Aflatoxin Contamination Assessment
Aflatoxin analysis revealed complete absence of detectable levels across all sample types and replicates throughout the initial assessment period (Table 3 and Figures 3–5). The complete absence of detectable aflatoxin levels at day 0 across all samples demonstrates the effectiveness of the controlled solar drying process in preventing initial mycotoxin formation. This finding is particularly significant given that dried fruit products are inherently susceptible to Aspergillus contamination due to their hygroscopic nature and susceptibility to moisture uptake during storage. 30 The achievement of target moisture levels below 10% during the drying phase created an unfavourable environment for aflatoxigenic fungi, effectively preventing the initial establishment of toxin-producing microorganisms.
Aflatoxin Concentration (μg/kg) in Composite Flour Samples During Storage.
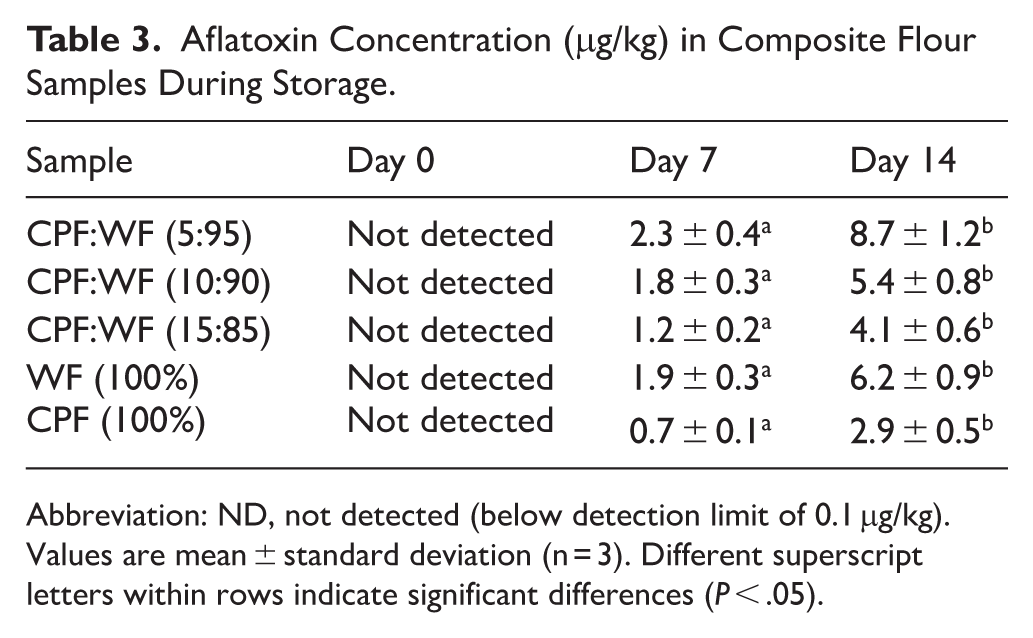
Abbreviation: ND, not detected (below detection limit of 0.1 μg/kg).
Values are mean ± standard deviation (n = 3). Different superscript letters within rows indicate significant differences (P < .05).

Correlation between moisture content and aflatoxin B1 concentration.

Comparative analysis of fungal load and aflatoxin concentration.

Aflatoxin progression during storage.
While the analytical method was validated for the detection of aflatoxins B1, B2, G1, and G2, only aflatoxin B1 was detected in the samples. Aflatoxins B2, G1, and G2 remained below the detection limit (0.03 μg/kg) throughout the storage period across all formulations. This pattern is consistent with the typical aflatoxin profile of Aspergillus flavus contamination, where AFB1 predominates and often occurs as the sole detectable congener, particularly in early-stage contamination (Kamle et al., 2019). 35 The total aflatoxin burden, therefore, corresponded to AFB1 concentrations alone. This finding has important regulatory implications, as both individual AFB1 limits and total aflatoxin limits must be considered. The European Union sets maximum limits of 2 μg/kg for AFB1 specifically and 4 μg/kg for total aflatoxins in cereals and cereal products, while the Codex Alimentarius Commission recommends 5 μg/kg for total aflatoxins in ready-to-eat cereals.
The emergence of detectable aflatoxin B1 levels by day 7 in all samples coincided with moisture content exceeding 12%, confirming the critical threshold relationship between water activity and mycotoxin production. The 5% cashew incorporation showed the highest initial contamination at 2.3 ± 0.4 μg/kg, already exceeding the EU limit of 2 μg/kg for AFB1. Pure cashew pomace flour (CPF) demonstrated the lowest levels at 0.7 ± 0.1 μg/kg, remaining below regulatory limits at this time point. This differential response suggests that the concentration of bioactive compounds in cashew apple flour plays a protective role against mycotoxin production. The phenolic compounds and tannins present in cashew apple pomace (815.3 mg GAE/100 g and 376.4-1387.8 mg/100 g, respectively) have been documented to exhibit antifungal properties that can inhibit Aspergillus growth and subsequent toxin production. 3
By day 14, aflatoxin concentrations had increased substantially, with CPF:WF (5:95) reaching 8.7 ± 1.2 μg/kg, representing a 278% increase from the levels on day 7. This exponential increase pattern is characteristic of secondary metabolite production during the stationary phase of fungal growth, where nutrient stress and environmental conditions favour mycotoxin biosynthesis over primary metabolic processes. The relationship between fungal load and aflatoxin production was not strictly linear, suggesting that factors beyond simple colony counts, including fungal species composition, environmental stress conditions, and substrate characteristics, influence toxin production.
The 15% cashew incorporation demonstrated the lowest aflatoxin profile throughout the storage period, reaching only 4.1 ± 0.6 μg/kg by day 14 despite experiencing significant moisture uptake (from 10.14% to 12.76%). This 53% reduction in aflatoxin levels compared to the 5% cashew incorporation, indicates that higher cashew incorporation provides substantial protective effects against mycotoxin production. Recent research has highlighted the potential of cashew apple pomace as a source of bioactive compounds with antimicrobial properties, which may explain the observed reduction in toxin production at higher incorporation levels. 18 The mechanism likely involves the inhibition of aflatoxin biosynthetic pathways through antioxidant activity and direct antimicrobial effects on Aspergillus species.
The food safety implications of these findings are particularly concerning when evaluated against international regulatory standards. The European Union’s maximum limit of 2 μg/kg for aflatoxin B1 was exceeded by day 7 in all samples except pure cashew pomace flour. Since only AFB1 was detected (with B2, G1, and G2 remaining below detection limits), the total aflatoxin burden corresponded to AFB1 concentrations alone. By day 14, CPF:WF (5:95) reached 8.7 μg/kg and CPF:WF (10:90) reached 6.3 μg/kg, exceeding both the EU’s individual AFB1 limit (2 μg/kg) and the Codex Alimentarius Commission’s total aflatoxin limit (5 μg/kg for ready-to-eat cereals). The 5% incorporation level resulted in concentrations more than 4 times the EU limit for AFB1 and nearly twice the total aflatoxin limit. These exceedances are consistent with patterns observed in other dried fruit products stored under high humidity conditions, in which rapid aflatoxin accumulation occurs once critical moisture thresholds are exceeded. 25
As shown in Figure 3, the strong correlation between moisture content and aflatoxin production (r = .92, P < .001) underscores the critical importance of controlling moisture in preventing mycotoxin contamination. Samples that maintained moisture levels closer to 12% showed significantly lower aflatoxin accumulation rates, while those exceeding 13% demonstrated exponential increases in toxin production. This relationship has been well-documented in flour storage studies, where moisture content above 12% to 13% creates optimal conditions for Aspergillus growth and aflatoxin biosynthesis. 31
The temporal lag between peak fungal growth rates and maximum aflatoxin production rates. While fungal populations demonstrated exponential growth primarily between day 0 and day 7, with growth rates beginning to plateau thereafter, aflatoxin production showed its most dramatic increases during the day 7 to 14 period. This pattern confirms that mycotoxin biosynthesis represents a secondary metabolic process that occurs after initial colonisation and establishment phases. Similar temporal patterns have been reported in flour storage studies, where mycotoxin production is typically triggered by nutrient depletion and environmental stress conditions that characterise the transition from exponential to stationary growth phases
The temporal progression of aflatoxin contamination revealed distinct phases: a lag period (days 0-7), where initial colonisation and establishment occurred, followed by an exponential production phase (days 7-14) characterised by rapid toxin accumulation. This pattern suggests that intervention strategies implemented within the first week of storage could be effective in preventing significant mycotoxin accumulation. The delayed onset of detectable aflatoxin levels, despite the presence of growing fungal populations, indicates that mycotoxin production requires specific physiological conditions and metabolic switches that occur after initial colonisation.
The superior performance of pure cashew pomace flour in limiting aflatoxin production (2.9 ± 0.5 μg/kg by day 14) demonstrates its potential for developing cashew-based formulations with enhanced food safety profiles. This finding suggests that optimised processing and formulation strategies could leverage the natural antimicrobial properties of cashew apple components to create products with inherent resistance to mycotoxin contamination.
Previous studies on cashew nut storage have reported similar patterns of aflatoxin development under high humidity conditions, with contamination levels directly related to storage moisture content and environmental relative humidity. 36 The current findings extend this understanding to processed cashew apple products, demonstrating that the protective effects of bioactive compounds can be maintained even after processing into flour form.
The implications for tropical storage environments are particularly significant, as the observed contamination occurred under conditions typical of many sub-Saharan African storage facilities (25°C ± 2°C, 65% ± 5% RH). The rapid progression from undetectable to regulatory-exceeding levels within 14 days highlights the critical need for improved storage infrastructure and moisture control systems in regions where cashew apple valorisation initiatives are being implemented.
Food Safety Implications
The observed aflatoxin levels in several samples exceeded regulatory limits established by various international bodies. The European Union sets maximum limits of 2 μg/kg for aflatoxin B1, while the Codex Alimentarius Commission recommends 5 μg/kg for total aflatoxins. By day 14, the 5% and 10% incorporation samples exceeded these limits, indicating potential food safety concerns.
Conclusion
This study shows that while incorporating cashew apple flour offers potential for food waste valorisation and nutritional enhancement, careful attention to food safety parameters is essential. Higher levels of cashew apple flour (15%) provide antimicrobial benefits, potentially due to bioactive compounds. Moisture content above 13% significantly increases the risk of mycotoxin production. Storage conditions typical of tropical environments (65% RH, 25°C) promote rapid fungal growth and subsequent aflatoxin production. Initial processing conditions (solar drying to <10% moisture) effectively prevent initial contamination. Future research should focus on optimising storage conditions, developing natural preservation strategies, and establishing critical control points for the safe use of cashew apple flour in food products.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302261418385 – Supplemental material for Mycotoxin Development in Cashew Apple-Wheat Flour Composite During Tropical Storage
Supplemental material, sj-docx-1-ehi-10.1177_11786302261418385 for Mycotoxin Development in Cashew Apple-Wheat Flour Composite During Tropical Storage by Afia Sakyiwaa Amponsah, Emmanuel Tetteh-Doku, Barikisu Mohammed, Moses Kwaku Golly and Belinda Agyei-Poku in Environmental Health Insights
Footnotes
Author contributions
All authors contributed equally to the conception, design, execution, analysis, and writing of this study. All authors have read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data is available on request from the author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.