Abstract
Type 1 diabetes (the pancreas producing little or no insulin) is usually diagnosed in children and young adults and was previously known as juvenile diabetes. McArdle disease is a common metabolic defect caused by an inherited deficit of myophosphorylase. These 2 diseases might have some clinical heterogeneity. Here, we discuss a McArdle disease case where insulin-dependent diabetes overshadows its early diagnosis. In this case, an insulin-dependent 22-year-old female patient with diabetes mellitus had been on diabetes treatment for 15 years. Although her blood glucose was regulated, her anamnesis showed that muscle weakness, fatigue, cramps or myalgia never healed. Based on her anamnesis, the patient was asked to take a nonischemic forearm exercise test, which revealed significant elevation in levels of creatine kinase (5968-7906 U/L), but no increase was found in lactate concentration, but a slight increase in ammonia concentration (not statistically significant) at the end of the test made us consider McArdle disease. A genetic test was done to confirm this possibility. A homozygous c.2128_2130delTTC/p.Phe710del mutation was detected in the examination of exons of the PYGM gene, which confirmed the diagnosis of McArdle disease in our patient. According to the data, this is a rare case of McArdle disease with type 1 diabetes. During treatment for diabetes, if the above-mentioned symptoms are present in a patient, and especially if the patient’s creatine kinase concentration is high, muscle diseases should be suspected. Therefore, we suggest that this case report will provide new insight to clinicians on metabolic defects in this disease and increase the patient comfort. In such cases, an early diagnosis should reduce health costs.
Learning Points
In a case of insulin-dependent diabetes with serum creatine kinase concentration, more than 1000 U/L—more than 30 fold above the normal range (22-198 U/L), muscle diseases (such as McArdle disease) should be suspected. Constant lactate values along with the increase in ammonia concentration should be suspected in McArdle disease. The diagnosis of McArdle disease may be also confirmed with the following criteria: homozygous c.2128_2130delTTC/p.Phe710del mutation or absence of myophosphorylase in a muscle biopsy.
Sucrose (37 g) is recommended as first-line therapy in the case of McArdle disease. Patients’ blood glucose should be regulated daily by using 36 IU fast-acting and 24 IU long-acting basal insulin when insulin-dependent diabetes and McArdle disease coexist.
Branched-chain amino acid supplementation, vitamin B6 and creatine were given orally to patients experiencing rapid fatigue and hyperglycemia.
Background
Diseases that occur owing to disruptions in the production or destruction of glycogen are called glycogen storage disorders. 1 Currently, approximately 15 different types of glycogen storage disorders are known. McArdle disease (myophosphorylase deficiency, glycogen storage type 5) is one of an autosomal recessive glycogenosis disease characterized by exercise-induced myalgia, weakness, myoglobinuria, and also permanent weakness after a longer disease. The disease is due to a lack of the enzyme myophosphorylase (EC 2.4.1.1), 2 which catalyses the separation of glucose-1-phosphate from the glycogen chain. There is a genetically common truncating mutation (R50X) in the myophosphorylase gene (PYGM); this common R50X mutation is >50% of the mutant alleles in Caucasian patients and rare mutations have been also reported. 3
McArdle was the first person to diagnose the disease using an ischemic forearm test, which shows little or no lactate being released in the skeletal muscle during exercise. However, excessive ammonia production has frequently been noted. Today, the diagnosis of the disease is based on the absence of phosphorylase activity in muscle biopsy, or by detection of mutations by genetic testing, which concerns autosomal recessive inheritance. The disease often causes marked muscle weakness and pain and usually manifests itself in the second or third decade of life with pain, fatigue, and cramps in the muscles developing after exercise. 4 Davies et al 5 in 1977 reported concurrent insulin-dependent diabetes and glycogen storage disease type V, and in 1996, concurrent noninsulin-dependent diabetes and glycogen storage disease type V2 was published. Other than these 2 reports, there are no others in the literature showing the concomitance of McArdle and diabetes; this therefore delays a precise early diagnosis of the disease because the clinician tends to treat diabetes first. Here, we discuss our present case which should be noted to avoid overlooking this particular concomitance.
Case Report
A 22-year-old female patient with a diagnosis of type 1 diabetes mellitus for ~15 years applied to our endocrine and metabolic diseases clinic following a blood glucose reading of 250 to 300 mg/dL (13.88-16.65 mmol/L) at home. The urine was ketone-positive, and therefore, routine clinical biochemistry tests of the patient were run. The patient did not complain of nausea, vomiting, abdominal pain, cough, chest pain, dyspnea, diarrhea, constipation, and burning urination. She stated that, in her anamnesis, she had had pain in the proximal muscles of the upper and lower extremities since her childhood, which occurred occasionally after physical effort and disappeared after resting. She felt pain when she climbed a ladder or lifted a load, but it went away after resting. In the last year, her complaints had started to increase and an orthopedic surgeon had administered anti-inflammatory and myorelaxant treatment.
In our follow-up, the patient was given appropriate fluid replacement and intensive insulin therapy. During routine examinations, the creatinine kinase (CK) and lactate dehydrogenase (LDH) concentration were 2247 and 460 U/L, respectively. Physical examination did not reveal any abnormality. The patient was examined neurologically, and electromyography (EMG) was requested with a prediagnosis of myopathy. As a result of needle EMG, motor-unit action potentials characteristic of myogenesis were recorded in the muscles locally, and myogenic conduction appeared to be normal. A nonischemic forearm exercise test was run, with a preliminary diagnosis of myopathy. Ammonia, CK and lactate concentration were measured at baseline, 1, 3, 5 and 7 min. The patient’s laboratory data are shown in Table 1. Other biochemical parameters are shown in Table 2.
Laboratory data on admission.
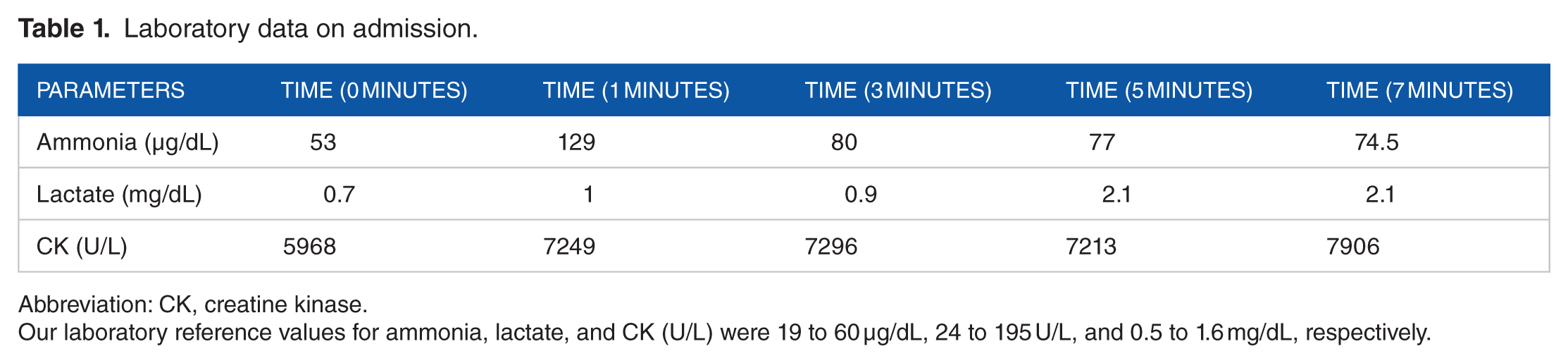
Abbreviation: CK, creatine kinase.
Our laboratory reference values for ammonia, lactate, and CK (U/L) were 19 to 60 µg/dL, 24 to 195 U/L, and 0.5 to 1.6 mg/dL, respectively.
Biochemical parameters.
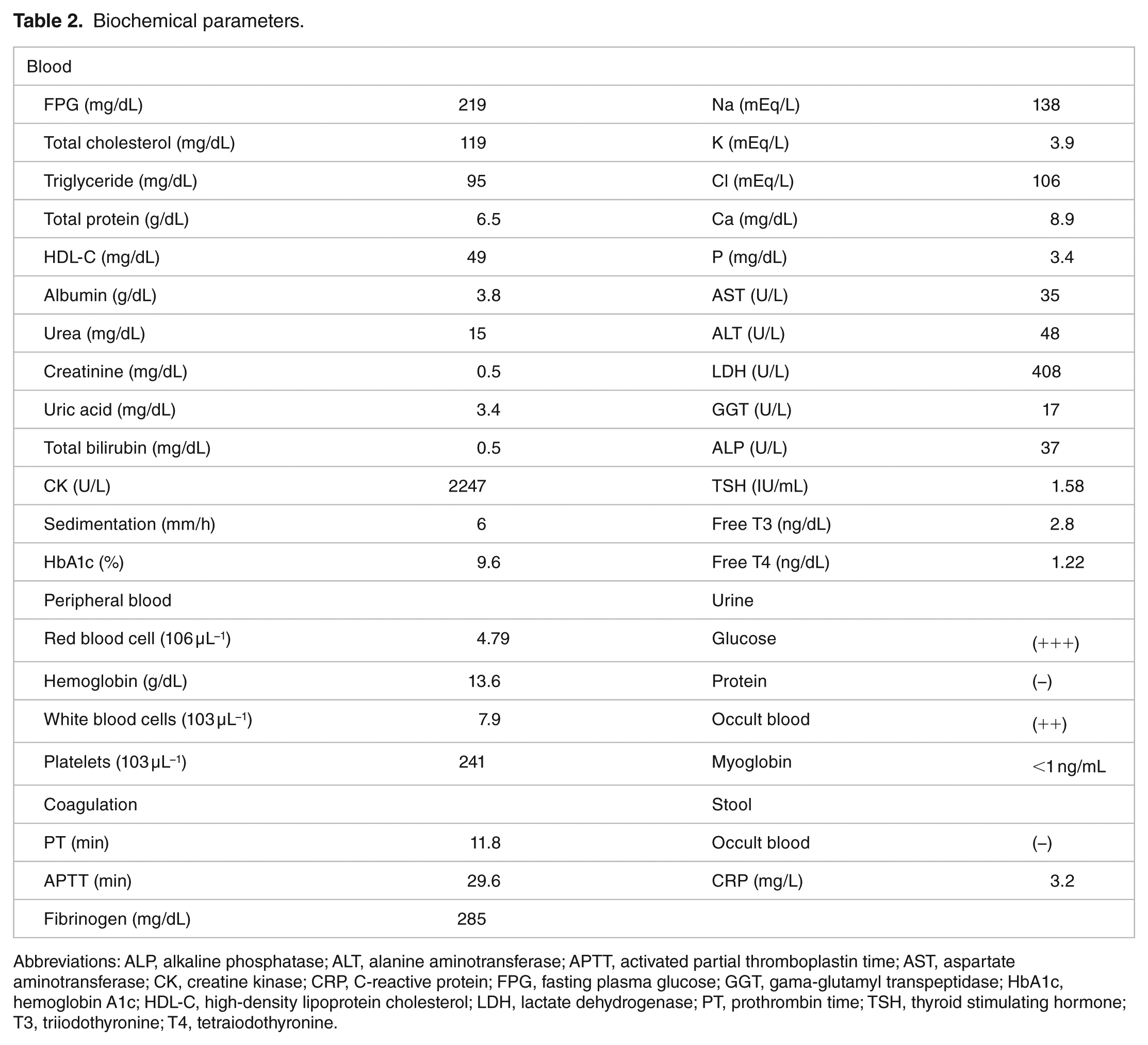
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; APTT, activated partial thromboplastin time; AST, aspartate aminotransferase; CK, creatine kinase; CRP, C-reactive protein; FPG, fasting plasma glucose; GGT, gama-glutamyl transpeptidase; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein cholesterol; LDH, lactate dehydrogenase; PT, prothrombin time; TSH, thyroid stimulating hormone; T3, triiodothyronine; T4, tetraiodothyronine.
The test results indicated that there was a statistically significant increase in CK concentration. Although there was an increase in the ammonia concentration, the difference was not statistically significant, and no significant increase was found in lactic acid concentration. McArdle disease was suspected when a statistically significant increase in the concentration of ammonia and lactic acid was not detected as a result of the nonischemic forearm exercise test. A genetic examination for McArdle syndrome was requested. A homozygous c.2128_2130delTTC/p.Phe710del mutation was detected in the exons of the PYGM gene, thus confirming the presence of glycogen storage disease type V. Following these results, we checked other family members (father, mother, parents, sister, brother, uncle, and aunt), but none had any muscle symptoms. Her mother and father was uncle children. Focusing on our patient herself, the data indicated the concomitance of McArdle disease and diabetes mellitus. The patient was followed for both insulin-dependent diabetes and glycogen storage disease. The case complied word-for-word with the Helsinki Declaration. As a single case report involving a human subject does not require approval by our Institutional Review Board (IRB) during reporting, written informed consent was obtained from patient before sharing these data. The patient confirmed that all her data could be used anonymously.
Treatment and Outcome
The patient’s blood glucose was daily regulated using 36 IU fast-acting and 24 IU long-acting basal insulin. She patient was also advised to take 37 g sucrose orally before undertaking any activity requiring effort. Branched-chain amino acid supplementation, vitamin B6 and creatine were given orally to the patient for rapid fatigue and hyperglycemia.
Discussion
From this case, we wish to discuss the diagnostic difficulties associated with McArdle disease, when it is concurrent with type 1 diabetes. The patient who was diagnosed with McArdle disease had been treated for type 1 diabetes mellitus until she applied to our clinic. When admitted, she reported in her anamnesis that, in addition to her diabetes, she had myalgia, nonhealing exercise intolerance, and periodic muscle cramps. The nonischemic forearm test indicated that the patient’s muscle and cramp pain were not healing. At the end of this test, a high ammonia concentration and a constant lactate concentration were found. A high ammonia concentration indicates that there was no defect in muscle adenosine monophosphate (AMP) deaminase; normally, the ammonia concentration increases owing to AMP deamination when a muscle becomes contracted. However, we assumed that, as suggested before, allosteric activation of adenylate kinase (AK), AMP deaminase (AMPD), or enzyme induction of AK or AMPD might be responsible for producing the high levels of ammonia in muscles of a McArdle patient due to increased substrate availability of adenosine diphosphate (ADP) and AMP and probably an elevated level of potassium. This appears to be the nature of the disorder in our case reported here. 6
The absence of lactate during exercise is a sign of a defect in the glycolysis pathway. If there had been no defect, lactate formation would have been generated from pyruvate when the muscle contracted. However, in this case, there was no change in lactate concentration. In the ischemic forearm test conducted in healthy people, the lactate concentration increases 5 to 10 mg/dL compared with the basal values and the ammonia concentration rises >100 µg/dL. Constant lactate values along with an increase in ammonia concentration primarily suggest McArdle disease with muscle glycolysis disorder. To confirm the presence of McArdle disease, myophosphorylase deficiency can be checked by muscle biopsy, but this invasive procedure is difficult. Therefore, to determine the presence of McArdle disease, screening for mutations in the exons of the PYGM gene is more widely accepted today. Based on the results, a homozygous c.2128_2130delTTC/p.Phe710del mutation was detected, confirming McArdle disease. In McArdle disease, a table of rhabdomyolysis characterized by hypersensitivity and swelling in the muscle and muscle destruction has been published. 7 Rhabdomyolysis attacks cause excessively high creatine kinase concentrations. In that report, CK concentrations were extremely high, and we conclude that high creatine kinase concentrations in our own case were also due to excessive muscle destruction (rhabdomyolysis).
Conclusions
If a patient complains of myalgia, nonhealing exercise intolerance, regular muscle cramps, and fatigue, McArdle disease, a glycogen storage disease, should be suspected. In such patients, creatine kinase levels should be considered before running expensive tests, because high values indicate muscle destruction. In this case, it should be confirmed with a nonischemic forearm test that lactate concentrations remain constant, whereas ammonia concentrations increase. For a definitive diagnosis, myophosphorylase concentrations should be measured in a muscle biopsy or a test for appropriate genetic mutations.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed equally to the care of the patient and the writing of this report.
Informed Consent
Written informed consent was obtained from the patient.
