Abstract
Pharmaceutical and dye residues are emerging contaminants of concern in various aquatic ecosystems, and they may pose risks to the health of these ecosystems. Adsorption is recognized as an excellent and versatile strategy for remediating these contaminants. In this study, a nanocomposite adsorbent of Cu-rich Ag-Cu, denoted as Ag25Cu75, was used to adsorb diclofenac sodium (DCF), a popularly used pharmaceutical compound, and crystal violet (CV), a model of a cationic dye. Ag25Cu75 nanoparticles were synthesized by the NaBH4 reduction method and characterized by X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FTIR), electron microscopy (TEM), and UV-Vis spectroscopy (UV-Vis.), and Batch adsorption experiments were conducted to investigate the effects of pH, temperature, adsorbent dosage, contact time, and initial concentrations of contaminants. The adsorption efficiency of Ag-Cu nanocomposites for DCF and CV contaminants was found to be high, with 85% adsorption efficiency for DCF within 60 min and 80% adsorption efficiency for CV within 150 min at pH 3 and 252°C. Thermodynamic calculations showed that adsorption of both contaminants by Ag-Cu nanocomposites was spontaneous and entropy-driven, with endothermic adsorption of DCF and CV. The point of zero charge (PZC = 9.0 ± 0.1) was also responsible for the favorable adsorption of CV under acidic conditions and the amphoteric adsorption behavior of DCF. The binary adsorption tests indicated minimal competitive effects, indicating the independent adsorption on heterogeneous surfaces. The fixed-bed column tests with Sand-Ag-Cu nanoparticle composites were superior to the sand-alone columns, which further demonstrated the potential of the Ag-Cu nanoparticle material for water treatment. This study demonstrated the effectiveness of Ag-Cu nanoparticles for the removal of pharmaceutical and dye contaminants.
Keywords
Introduction
The introduction of new pollutants, such as pharmaceutical compounds and synthetic dyes, is increasingly affecting all aquatic environments around the world. These pollutants pose a significant threat to the ecological balance because of their chemical stability, persistence, and biological activity even at very low concentrations, thus allowing them to build up and gradually affect the environment over a long period of time (Castiglioni et al., 2006; Sulaiman & Al-Jabari, 2021). One of the major pharmaceutical pollutants is Diclofenac sodium (DCF; Figure 1), which is a commonly used nonsteroidal anti-inflammatory drug (NSAID) that can often be found in surface waters and wastewater discharges at a concentration of up to micrograms per liter (Patil & Chaudhari, 2012; Zuccato et al., 2000). DCF is a widely detected pharmaceutical compound in aquatic environments and is frequently used as a model contaminant due to its persistence and potential ecological effects. Its presence has been associated with various problems, such as affecting the endocrine system, chronic poisoning, and the development of resistance in bacteria, thus emphasizing the necessity of having fast and reliable removal techniques. Advanced oxidation processes (AOPs) are widely investigated treatment technologies for the degradation of persistent organic contaminants in water (Sulaiman & Shahwan, 2017). Adsorption of bimetallic zero-valent iron nanoparticles (B-nZVI) is often considered an effective and versatile treatment approach due to its operational simplicity, high removal efficiency, and potential for minimal secondary by-product formation (Castiglioni et al., 2006; Patil & Chaudhari, 2012; Sulaiman & Al-Jabari, 2020; Sulaiman & Al-Jabari, 2021; Sulaiman & Shahwan, 2017; Thakur et al., 2026; Yadav et al., 2026; Zuccato et al., 2000).
Diclofenac sodium (a) and Crystal violet (b) chemical structures.
In a similar vein, synthetic dyes and specifically crystal violet (CV; Figure 1) are the most important pollutants found in water systems. CV is a triarylmethane dye that has been first used as a topical antiseptic and still finds its application in microbiology for Gram staining (Gessner & Mayer, 2002). During textile processing, about 15% to 20% of the used dye is lost and this percentage is the part of dye discharged into the effluent (Verma et al., 2020). CV is a commonly studied cationic dye known for its stability and documented toxic effects in aquatic organisms. The situations where humans could be affected are mainly ingestion and inhalation. The probable effects include renal and respiratory failing, skin and eye irritation and irreparable blindness if exposed to the dye in high concentration. Aquatic plants and animals are among the most susceptible ones, which eventually leads to an ecological imbalance (Gessner & Mayer, 2002).
Given the aforementioned risks, it is crucial to remove DCF and CV from contaminated water. A variety of treatment methods have been researched. Among them, AOPs consisting of ozonation, photocatalysis, and photo-Fenton reactions have shown great efficiency in degrading the pollutant. Nevertheless, they might face issues like partial mineralization, generation of harmful byproducts, and high operational costs (Sulaiman & Al-Jabari, 2020). On the other hand, membrane filtration technologies (ultrafiltration, nanofiltration) can achieve almost total rejection of dyes, but their effectiveness may be impacted by fouling, high power consumption, and shorter life span (M. E. A. Ali et al., 2022). Pollution control via biological methods such as microbial biofilms and enzymatic systems could be viewed as environmentally friendly alternatives but are generally slow, and may be inadequate for the removal of the stubbornest compounds like CV and DCF (Votat et al., 2024). Among all these methods, adsorption has been recognized as a cheap and very efficient way, where either engineered or bio-derived adsorbents such as carbon-based materials, clays, metal-organic frameworks, and nanomaterials are used to obtain the very high removal of pollutants (Ponder et al., 2001; Rathi et al., 2025; Sen et al., 2024; Yassin et al., 2023).
Lately, bimetallic nanoparticles (BNPs) have been the subject of interest for their synergistic properties which include increased adsorption capacity, catalytic activity and stability when compared to monometallics. The combination of two metals, for instance, silver and copper, can result in the creation of materials with enhanced surface reactivity, electrical conductivity and optical properties which are greatly beneficial for environmental remediation (Barcaro & Fortunelli, 2008; W. Li et al. 2016). The Ag-Cu nanoparticles have been produced in different forms like alloys, core-shell and multi-shell through chemical reduction, polyol synthesis, solvothermal and microwave-assisted methods (Mendes et al., 2017; Ogarev et al., 2019; Zhang et al., 2015).
To determine the adherence of pollutants to silver-copper bimetallic nanoparticles, studies on their kinetic, isotherm, and thermodynamic characteristics are carried out. These studies help in understanding the interfaces between pollutants and the surfaces of these nanoparticles. These studies are critical in developing expandable and ecologically friendly treatment technologies for pharmaceutical as well as dye pollutants in water. Yet, despite the progress achieved, several questions still linger. These include optimizing bimetallic nanoparticles, their potential for recycling, as well as their applicability in real contaminated water. This underscores the need for more research in this area (Mishra et al., 2026; Sulaiman & Al-Jabari, 2020; Sulaiman & Shahwan, 2017).
Although Ag-based, Cu-based, and Ag-Cu bimetallic nanoparticles have been widely investigated for contaminant removal, most previous studies focus on either dyes or pharmaceutical compounds under batch conditions and often emphasize material synthesis rather than comparative adsorption behavior. In this context, the present study aims to contribute by systematically evaluating a single Ag25Cu75 bimetallic composition for the removal of both a pharmaceutical contaminant DCF and a cationic dye CV under identical experimental conditions. Furthermore, the study combines batch adsorption analysis with fixed-bed column experiments and mechanistic interpretation, providing insight into adsorption behavior across different operational modes rather than proposing a new adsorbent material.
The selection of these two compounds allows evaluation of adsorption behavior for chemically distinct pollutants with different molecular structures and charge characteristics, thereby facilitating mechanistic interpretation of adsorption processes.
In this work, the adsorption performances and removal possibilities of DCF and CV by Ag-Cu nanoparticles were investigated. Ag-Cu nanoparticles were initially prepared by reduction with NaBH4 in the presence of sodium hydroxide as a rate-controlling agent and characterized for their physical and chemical properties by various analysis tools. Then, the adsorption performance of such nanoparticles for the removal of DCF and CV from aqueous solutions was investigated as a function of some experimental parameters, including pH, dose of adsorbent, temperature, adsorbate concentration, and contact time. The Langmuir isotherm and pseudo-second-order kinetic models were utilized to explain the mechanism of such adsorption; in addition, thermodynamic parameters are computed to interpret such a process. Finally, the practical performance of removal was evaluated by using a column filtration system packed with a mixture of sand and Ag-Cu nanoparticles.
Materials and Methods
Chemicals
All chemicals used in this experiment were of high purity and used directly without further processing, thus providing reliable results in our experiments. Both silver nitrate (AgNO3; ⩾99.0%) and copper (II) sulfate pentahydrate (CuSO4·5H2O; ⩾98.0%) were obtained from Sigma-Aldrich (USA), which were used as starting metal salts for preparing bimetallic silver-copper NPs. Sodium borohydride (NaBH4; ⩾98.0%), obtained from Merck (Germany), was used due to its strong reduction ability. Moreover, polyvinylpyrrolidone (PVP), with a molecular weight of approximate 40,000, was used in this experiment as a stabilizer to prevent aggregation of any formed NPs in suspension. We focused on two main substances: diclofenac sodium (DCF, C14H10Cl2NNaO2, ⩾98.0%) and crystal violet (CV, C25H30ClN3, ⩾90.0%). Both These chemicals were obtained from Sigma-Aldrich and used to produce artificial samples of wastewater, corresponding to their concentrations found in nature. For adjusting the acidity/alkalinity (pH) concentration of our solutions, hydrochloric acid (HCl; 37%) and sodium hydroxide (NaOH; 97.0%) from Fisher Scientific were used. For preparing the solutions, high-purity deionized water obtained from a Millie-Q system was used.
In adsorption studies, the presence of DCF and CV dyes in the water samples was determined before and after treatment by high-performance liquid chromatography. These results were confirmed by the use of UV-Vis spectrophotometry techniques, where the wavelengths 276 nm for DCF and 590 nm for CV dyes are applied. The proposed technique enables the quantitative determination of the efficiency of removing pollutants from adsorption materials that use nanomaterials (Kumar et al., 2021; Patel et al., 2018).
Methods
The experimental work of this study was conducted over a period of August/2024 to August/2025 in the laboratories of Chemistry department/Birzeit University.
Preparation of Ag-Cu Bimetallic Nanoparticles
Ag-Cu bimetallic nanoparticles were synthesized using a chemical reduction method based on sodium borohydride (NaBH4), adapted from previously reported procedures (Sun & Xia, 2002; Zhang et al., 2015). In a typical synthesis, aqueous solutions of AgNO3 and CuSO4 were mixed at predefined molar ratios to obtain the desired Ag:Cu composition. The mixed precursor solution was stirred continuously, followed by the dropwise addition of freshly prepared NaBH4 solution under vigorous stirring to induce metal ion reduction and nanoparticle formation. Minor modifications to the reported protocol were introduced in the present study, including adjustment of precursor concentrations and reduction conditions, in order to achieve the targeted Ag25Cu75 composition and to ensure reproducible particle formation. The resulting nanoparticles were collected, washed repeatedly with deionized water, and dried prior to characterization and adsorption experiments.
The order of addition of reactants is also a crucial issue in regulating the reduction mechanism, nucleation, and subsequent nanoparticle formation due to diverse coordination chemistry involving Ag+ and Cu2+ ions as well as competitive reduction of Ag+ with Cu2+ ions by the reducing agent (Sharma et al., 2019; Sun & Xia, 2002).
In this work, Ag-Cu nanoparticles (NPs) were prepared using a chemical reduction process with NaBH4 as a powerful reducing agent and NaOH as an accelerator to increase the reaction rate.
In a typical synthesis, water soluble precursor solutions were obtained by dissolving silver nitrate (AgNO3) and copper sulfate pentahydrate (CuSO4·5H2O) salts in 40 mL of deionized (DI) water to give the total molar concentration equal. The homogenization was conducted at 90°C for 5 min with vigorous stirring. Then, NaBH4 (0.45 g/10 mL DI water) and NaOH (0.64 g/10 mL DI water) freshly dissolved solution were fast and simultaneously injected into the precursor solution. The fast development of active effervescence clearly indicated the evolution of hydrogen gas in the process of decomposition of borohydride and the reduction of metal ions. The reaction was also permitted to continue until the effervescence was complete, which was about 1 hr.
The resulting solid was obtained using a centrifuge. It was then extensively washed five times with pure water. This was an important step for cleaning the nanoparticles. This helped remove any excess salts that were left. Unreacted substances were also eliminated. This ensured that the resulting nanoparticles were pure.
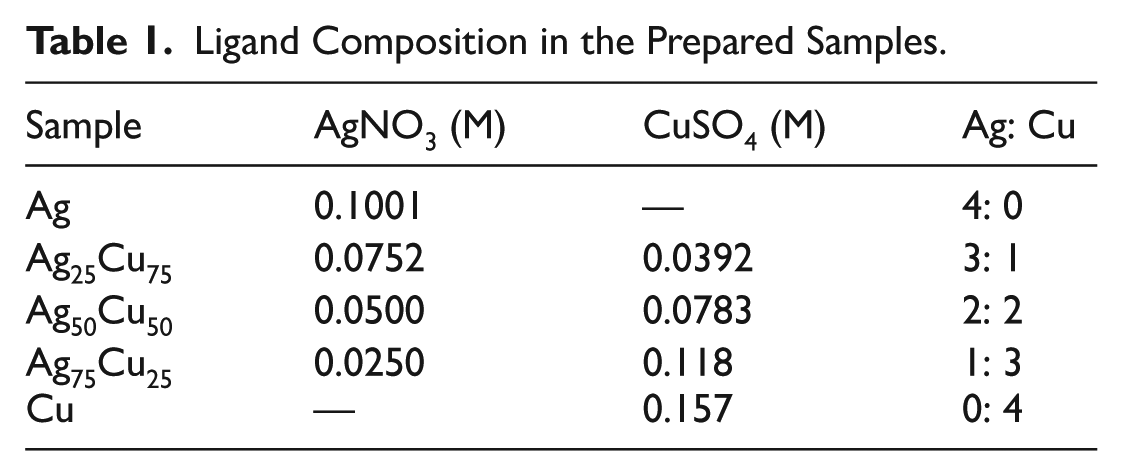
In order to accomplish our objectives, we formulated three different proportions of silver and copper. These samples consisted of 25% silver and 75% copper, 50% silver and 50% copper, and 75% silver and 25% copper. In each sample, the percentage listed above refers to the molar content. For comparison purposes, two other samples consisted of pure silver nanoparticles and pure copper nanoparticles. A clearer illustration of the compositions can be seen in Table 1.
Ligand Composition in the Prepared Samples.
This specific synthesis technique is practical to use since it provides a way to obtain stable nanoparticles that have a controlled size and perfectly adjustable ratios of metals. Bimetallic nanoparticles have been found to increase their role as a catalyst, the interaction between light, and the ability to absorb other compounds, compared to single-metal nanoparticles since they performed even better than single metal particles in those aspects (Hervés et al., 2012; S. Li et al., 2015).
Multiple Ag-Cu compositions (Ag25Cu75, Ag50Cu50, Ag75Cu25), as well as monometallic Ag and Cu nanoparticles, were synthesized for comparative purposes. Preliminary adsorption screening experiments (not shown) indicated that the Ag25Cu75 composition exhibited the highest and most consistent removal efficiency for both DCF and CV; therefore, this composition was selected for detailed adsorption, kinetic, thermodynamic, and column studies.
Characterizations
To ensure the comprehensive characterization of the produced nanoparticles, various techniques have been applied to this end; for example, the crystallinity of the prepared bimetallic nanoparticles was examined using an X-ray powder diffraction (XRD) instrument (Rigaku Ultima IV) with operating conditions set at 40 kV and 30 mA and a 2θ scan angle between 20° and 80°. To ensure the determination of the morphology of the produced nanoparticles, the FE-scanning electron microscope (JEOL JSM-6700LV) coupled with an EDS unit was deployed for the examination of the produced nanoparticles. However, the nanoparticles had to undergo gold coating using the DESK V HP TSC cold sputter coater prior to examination by the FE-scanning electron microscope. To provide further information about the produced nanoparticles, the TEM operating at 200 kV was also employed for the ensuing analysis; for raw samples to be examined by the TEM instrument, the raw nanoparticles had to be deposited after ultrasonic treatment with alcohol for 15 min. Additionally, the FTIR spectroscope served the purpose of identifying functional groups on the surface of the produced nanoparticles. Finally, UV-visible spectroscopy (Agilent HP 8453) was employed to evaluate the optical properties and adsorption behavior of the target compounds on the nanoparticle surface. Collectively, these techniques provide a reliable understanding of how the structural and surface properties of the synthesized materials govern their physicochemical behavior (Sharma et al., 2019; Zhao et al., 2020).
Elemental Characterization
The elemental composition and spatial distribution of the synthesized Ag-Cu nanoparticles were analyzed using energy-dispersive X-ray spectroscopy (EDX) coupled with elemental mapping. Complementary X-ray fluorescence (XRF) analysis was performed to confirm the bulk elemental composition.
Experiments on DCF and CV Adsorption
We conducted experiments systematically to determine the level of adsorption for two substances, DCF, a frequently used negatively charged pharmaceutical, and CV, a positively charged dye. We repeated each experiment three times to ensure accuracy in the results, as this would minimize random errors. All adsorption experiments were conducted in batch mode.
In order to ensure consistent and comparable conditions, the effect of solution pH was first investigated to determine the optimal pH for adsorption. The optimized pH value obtained from this study was then used in all subsequent adsorption experiments, including kinetic, isotherm, and thermodynamic studies. For each test, a combination of 100 mL of pollutant solution, with a deliberately set initial concentration, was mixed with a specified amount of silver-copper NPs. This blend was then stirred at a rate of 105 revolutions per min to guarantee that a proper distribution of the nanoparticles was achieved, facilitating sufficient interactions with the pollutants. For a precise estimation of concentrations, a standard set of lattice points or graphed calibration curves was established first. This required a series of dilutions from a high concentration solution of 1,000 mg L−1 toward a specified set of concentrations. DCF concentrations in their curves spanned from 1 to 100 mg L−1, while in CV, concentrations spanned from 1 to 10 mg L−1. These settings aimed to suitably span environmental concentrations likely encountered in natural systems, while assuring that consequent concentrations are within the prescribed sensitivity limits of available analytical instrumentation. After stabilization of concentrations in the system, a filtering process using specific water-safe PVDF syringe filter cartridges with a pore diameter of 0.45 µm followed. Finally, a precise assessment of remaining pollutant quantities was done using a standard UV-Vis Spectrophotometer, set at wavelengths of 276 and 590 nm, respectively, for DCF and CV compounds.
The pH effects on the adsorption mechanism were extensively studied with specific pH ranges explored: 3.0 to 10.0 for DCF and 3.0 to 11.0 for CV. It is important to highlight that pH exerts a fundamental role in determining both the charge of the nanoparticles’ surfaces and the ionization states of the adsorbable molecules; hence, it acts as a crucial parameter in the effectiveness of the adsorption mechanism. Specifically, in the case of CV, higher removal percentages are mostly apparent in alkaline solutions due to the pronounced electrostatic attraction existing among the positively charged molecules of CV and the negatively charged surfaces of the adsorbents. Subsequently, in the case of DCF removal, acid environments are most preferred, in which protonation efficiently overcomes electrostatic repulsion forces (Gupta & Suhas, 2009). To better understand the relevance of the previous observations under the different pH ranges examined, the point of zero charge (PZC) of the Ag25Cu75 NPs was determined. This was achieved through the dispersion of 0.025 g of Ag25Cu75 in 0.01 M NaCl solutions with initial pH values initially spanning from 1.0 to 12.0. Finally, the final pH was determined after a timeframe of 24 hr. The resulting PZC was used to analyze the determined points related to the adsorption mechanism under the pH range under examination.
The impact of adsorbent dose on its efficiency was analyzed in order to ascertain the least required dose for the removal of contaminants using Ag25Cu75 NPs. In this study, solutions with DCF (50 mg L−1) and CV (10 mg L−1), with an approximate pH close to 3.0, were treated with varied amounts of NPs (ranging from 0.015 to 0.200 g) at 25°C. Generally, it has been known that increasing amounts of adsorbent tend to enhance contaminant removal efficiency because of the augmentation in adsorbent binding sites (Foo & Hameed, 2010). Nonetheless, there is a point beyond which it can be assumed that a sort of equilibrium will exist, where increasing amounts will not necessarily contribute much toward contaminant removal.
For the adsorption kinetic studies and adsorption isotherms, a fixed quantity of the adsorbent (0.025 g) was added to 50 mL samples for the respective contaminants. The initial concentrations were varied, that is, from 1 to 100 mg L−1 for DCF and 1 to 10 mg L−1 for CV. This methodological consideration made it possible to identify the applicable rate-limiting steps (for example, film diffusion, intraparticle diffusion, or surface kinetics) and the total adsorption capacity for the composite material. Considering that DCF has the characteristics of an ion with a pKa value close to that under neutral to basic pH conditions (4.2) and that CV is essentially the cationic part of the respective dye, it was expected that the coulombic force between the surface properties of the Ag25Cu75 nanoparticles would play a dominant role (Foo & Hameed, 2010).
The effect of temperature, particularly between 5°C and 45°C, was also explored to identify whether the processes of adsorption are endothermic or exothermic. The key thermodynamic values (ΔG°, ΔH°, ΔS°) were determined later from the equilibrium information obtained at the two extreme temperatures (5°C & 45°C). These values offer significant understanding toward the spontaneity of adsorption, alongside its nature (Girish, 2025). It has been found that the endothermic nature is preferably documented for the adsorption of CV, mostly due to the increased mobility of adsorbate molecules with elevated temperature values. However, pharmaceuticals like DCF might vary according to the surface characteristics of the adsorbent material.
In all experiments, adsorption performance was quantified using Equation 1:

Mix Adsorption Experiments of DCF and CV
To investigate the competitive adsorption process between DCF and CV in a binary system, the batch experiments with multiple contaminants were extensively performed. For this purpose, an aqueous solution was prepared and contains 50 mg L−1 DCF and 10 mg L−1 CV. These aqueous solutions were then treated with the addition of Ag25Cu75 nanoparticles (at a molar ratio of 1Ag:3Cu and a dose of 25 mg). This aqueous solution was then agitated mechanically at a speed of 105 rpm and a temperature and pH of 25°C and 3.0, respectively. This agitation phase was carried out for a period of 2.5 hr to assure the complete interaction between the target contaminant and the surface active sites on the nanomaterial. Once equilibrium was obtained, the suspensions were filtered to measure the remaining concentrations of the DCF and CV. After filtration, the concentrations of the DCF and CV remaining in the aqueous solution were measured using a UV-Vis Spectrophotometer by considering the λmax value of each contaminant (276 and 590 nm). The removal efficiency of the contaminant was then calculated on the basis of Equation 1.
Studies in binary adsorption represent an essential aspect for understanding the capabilities of competitive, synergistic, or antagonistic interactions within aqueous streams containing various pollutants. In multi-component aqueous environments, various pollutants tend to compete for similar binding sites. However, in some instances, electrostatic and π-π interactions can provide an opportunity for increased uptake capacity for one substance in the presence of another substance (Foo & Hameed, 2010; Gupta & Suhas, 2009; Sposito, 1989). Results achieved in this study provide essential insight to shed light on the applicability potential of Ag-Cu nanocomposites for simultaneous removal of pharmaceuticals and dyes in aqueous samples.
Column Adsorption Experiments
To effectively model a fixed-bed adsorption reactor close to a real-world application, adsorption columns were prepared using precise combinations. These adsorption columns were filled with a combination consisting of equal parts of commercially available quartz sand and Ag25Cu75 nanoparticles, in two separate ratios: 49:1 (w/w) and 49.5:0.5 (w/w). These adsorption columns had a length and inner diameter of 25 and 5 cm, respectively. A support bed, 3 cm in height and consisting of specially prepared commercial-grade quartz sand, was placed at the base of each column. This was done with the purpose of assisting in a smooth flow and also to prevent any elution of the nanoparticles from happening in the column. Before its utilization, the commercial sand was thoroughly washed with deionized water to remove all dirt and then treated with thermal drying at a temperature of 110°C for a period of 24 hr to remove any remaining water from it.
For the experimentally conducted trials designed in columns using single contaminants, the DCF and CV stock solutions were prepared with initial concentrations of 50 and 10 mg L−1, respectively. Also, both were continually fed into the columns at a uniform volumetric flow rate of 2.0 mL min−1 using the peristaltic pump. The corresponding exit liquid portions were sampled based on the predefined time intervals. Afterward, the remaining concentrations of DCF and CV were determined using spectrophotometry with measurements taken at a wavelength of 276 and 590 nm, which corresponds to the maximum absorbance wavelength of spectrally distinct DCF and CV entities, respectively. Finally, breakthrough curves were generated to determine the adsorptive performance, total capacity of the bed, as well as the saturation time of the bed.
Column studies are important in ensuring that necessary understanding and knowledge are generated with regard to the dynamic adsorption characteristics of nanomaterials as adsorbents. The studies are done on a continuous flow basis. This is important in that it accurately represents the actual processes involved in wastewater treatment. Additionally, the data generated can be further analyzed through various dynamic models. These models include the Thomas Model, Yoon-Nelson Model, and Bohart-Adams Model. The data analysis enables the understanding of break-through points in addition to feasibility studies (Aksu & Gönen, 2004; Han et al., 2007). All experiments were conducted in triplicate, and Error bars have been added to the relevant figures. Control experiments were conducted to validate the adsorption results. Blank experiments without adsorbent were performed to confirm that no significant removal of DCF or CV occurred in the absence of nanoparticles. In addition, control tests using quartz sand alone were carried out in the column experiments to assess the contribution of the support material. These controls confirmed that contaminant removal was attributable primarily to the presence of Ag-Cu nanoparticles.
Results and Discussion
Based on preliminary comparative screening of all synthesized Ag-Cu compositions and monometallic nanoparticles, Ag25Cu75 demonstrated superior adsorption performance toward both DCF and CV. Consequently, the following results and discussion focus exclusively on the Ag25Cu75 nanocomposite. Error bars shown in the figures represent the standard deviation of triplicate measurements. The absence of significant contaminant removal in control experiments confirms that the observed adsorption behavior arises from interactions with the Ag-Cu nanoparticles.
Characterization of Adsorbents Particles
The synthetic Ag25Cu75 NPs were subjected to various systematic characterizations based on their physicochemical properties, which aimed at confirming their successful formation, as well as giving information about their structure, morphology, and optical characteristics. The representative characterization data from XRD, FTIR, SEM, TEM, and UV-Vis analyses are shown in Figures 2 and 3.
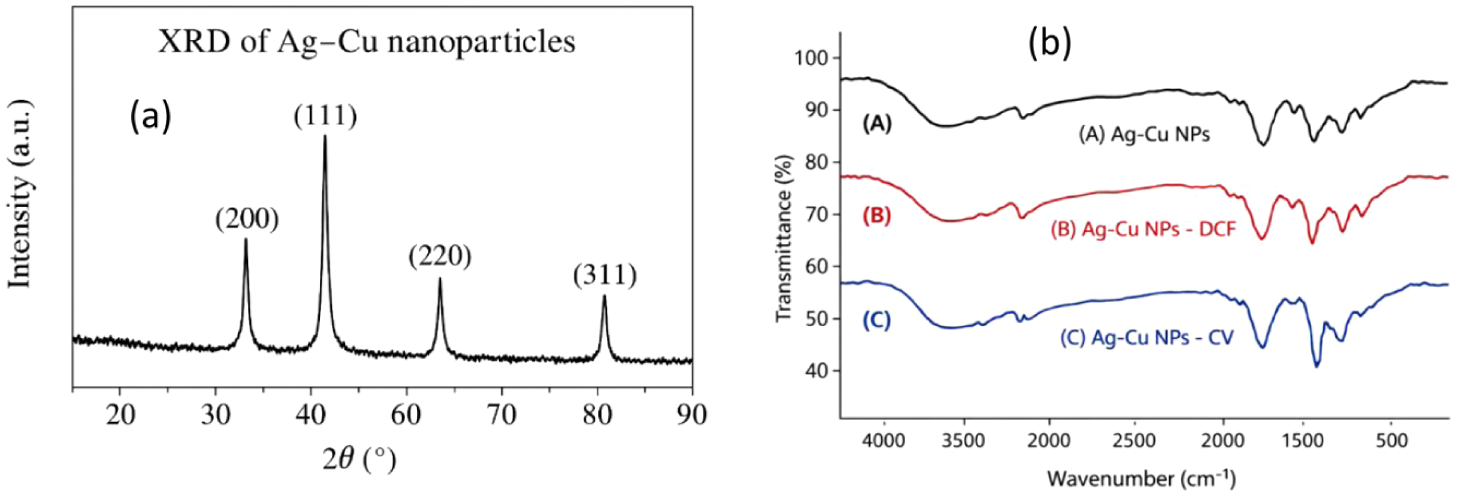
(Left (a)) X-ray diffraction (XRD) pattern of Ag-Cu bimetallic nanoparticles. (Right (b)) FTIR spectra of (A) Ag-Cu NPs, (B) Ag-Cu NPs—DCF, (C) Ag-Cu NPs—CV.
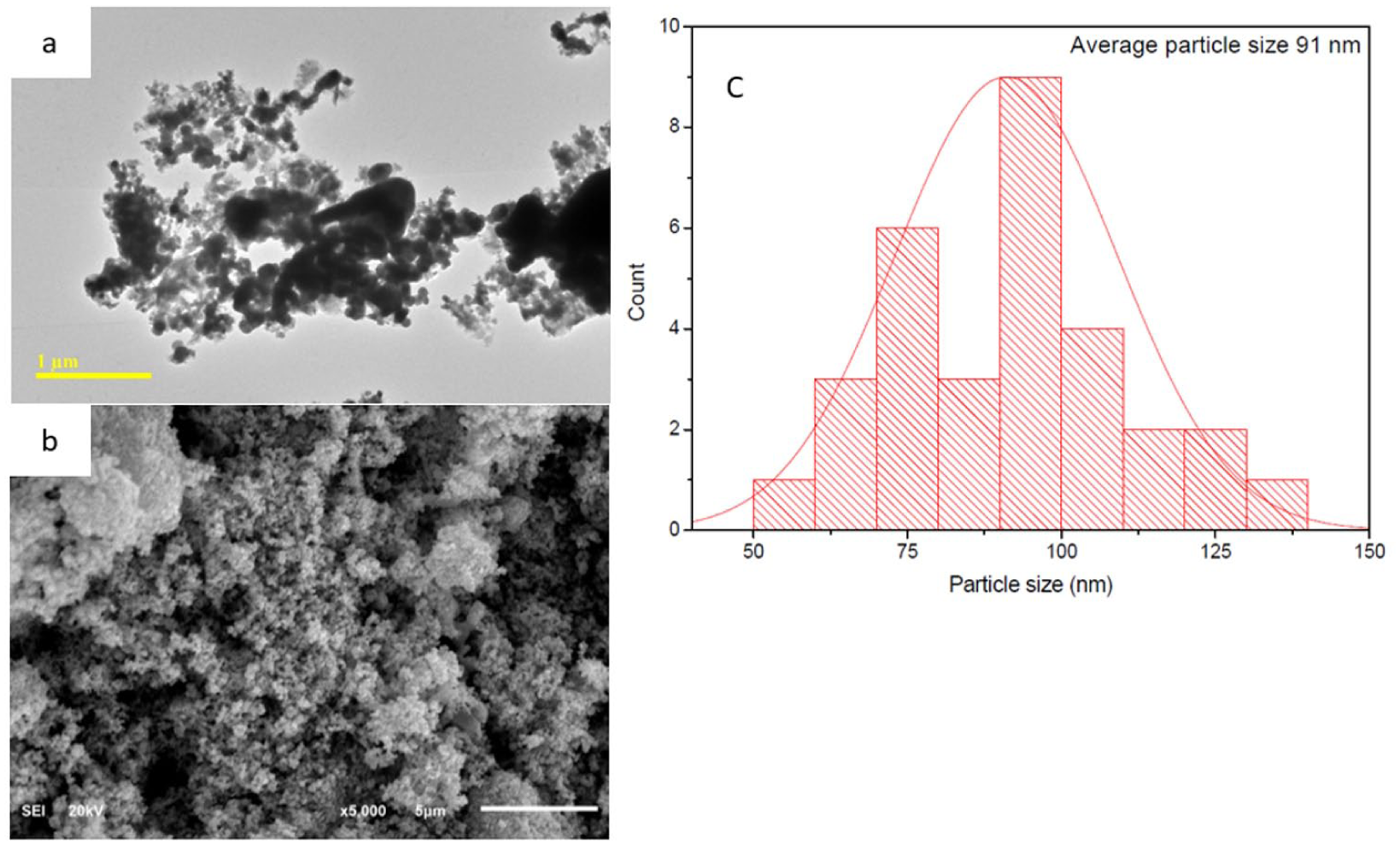
(a) TEM image showing agglomerated clusters of near-spherical Ag-Cu NPs. (b) SEM micrographs at different magnifications illustrating quasi-spherical Ag-Cu NPs forming rough-surfaced agglomerates typical of chemically reduced metallic nanoparticles. (c) Particle size distribution of Ag-Cu NPs determined from calibrated TEM image analysis using equivalent circular diameters based on the 1 µm scale bar, representing individually resolved primary particles.
X-ray Diffraction (XRD)
The XRD pattern of bimetallic nanoparticles of silver and copper (Ag-Cu) reveals clearly defined peaks of diffraction at 2θ ≈ 38.1°, 44.3°, 64.5° and 77.4°, which correspond to (111), (200), (220), and (311) planes of an fcc structure, respectively (Figure 2a). The predominance of the peak corresponding to (111) suggests that the crystals have a preferred orientation and are of high quality. No peaks linked to oxide have been clearly seen, so the nanoparticles are definitely metallic, but little peak broadening indicates very small crystal sizes and lattice distortion due to the interaction between silver and copper in a bimetallic form. These tests back up the FTIR method, which has shown the presence of surface functional groups without oxide formation, and are in line with SEM/TEM at which dispersed crystalline nanoparticles are observed, thereby confirming the successful synthesis of Ag-Cu NPs that can be used for adsorption applications (Patakfalvi et al., 2003; Sharma et al., 2019).
Fourier-Transform Infrared Spectroscopy (FTIR)
In the present study, qualitative analysis of the surface function groups presents on the surface of the Ag25Cu75 nanoparticles before and after adsorption was carried out using FTIR spectroscopy techniques to confirm the present state of the loaded species (Figure 2b). In the case of the pure Ag-Cu nanoparticles, the sharp broad peak around 3,400 cm−1 is attributed to the presence of the O-H stretching modes corresponding to the surface-bound hydroxyl groups or physically absorbed water molecules. The sharp band at 1,630 cm−1 is due to the H-O-H bending modes, which confirms the presence of surface-bound H2O species. The minor broad bands in the region below 600 cm−1 are due to the metal-oxygen stretching modes, which have been reported for various metallic particles and are ascribed to surface oxidation upon exposure to air (Chowdhury & Balasubramanian, 2014).
After adsorption of DCF and CV, the overall spectral profiles remain largely similar to that of the pristine material, indicating that no major chemical transformation of the Ag-Cu nanoparticle surface occurred during the adsorption process. Nevertheless, additional bands and subtle changes in the 1,600 to 1,400 cm−1 region can be observed, which are consistent with the presence of aromatic and functional groups associated with the adsorbed DCF and CV molecules. In particular, the appearance and/or enhancement of bands in this region is compatible with contributions from aromatic ring vibrations and, in the case of DCF, carboxylate-related modes.
It must be pointed out that these bands in the spectra, being relatively wide and sometimes overlapping, correspond to absorption near the limit of the technique, and as such, the FTIR data should be seen as indicative and supportive rather than conclusively proving the bonding or adsorption configurations. In that sense, the FTIR data serve primarily to prove the existence of the organic material on the Ag-Cu surface following adsorption, and the actual mechanisms and processes of adsorption, as well as the relevant interaction mechanisms, are more clearly outlined through the pH, kinetic models, isotherm, and thermodynamic data that follow (Table 2).
Assignments of FTIR Bands in Ag-Cu Bimetallic NPs for Both Samples Before and After the Absorption of DCF and CV Molecules. The Table Provide Information on the Characteristic Vibrations and Corresponding Functional Groups Responsible for Stabilizing Bimetallic NPs Through Absorption.

Scanning Transmission Electron Microscopy
TEM, as shown in Figure 3a, further confirmed the nano-metric size of Ag-Cu bimetallic nanoparticles, with densely packed nanostructures that show contrasting images on top of a transparent substrate. Although some signs of aggregation were noted, the images from TEM still show the existence of subnanometric domains that assemble into larger particles. Based on analysis of representative TEM images, the Ag25Cu75 nanoparticles exhibit an approximate particle size in the nanometer range, with individual particles appearing relatively uniform in morphology; however, a statistically rigorous particle size distribution was not determined.
SEM images (Figure 3b) show that Ag-Cu bimetallic nanoparticles generally featured a relatively uniform near-spherical shape, although evidence of some agglomeration was apparent. This particular tendency would be typical for metallic nanostructures prepared from chemical reduction reactions. This may be accounted for by their high surface energies, especially when particles become subnanometric in size, so inter-particle interactions become strong enough for them to easily aggregate. These morphological characteristics are in line with the findings from XRD data, showing crystalline features for these subnanometric particles, and help reinforce the theory that these nanostructures possess abundant surface active sites, which are important in improving adsorption capacity in Ag-Cu nano-composites (Wei & Wang, 2015).
In addition, quantitative particle size analysis was accomplished through the use of calibrated TEM image processing techniques. From the TEM images, the particle diameters were quantitatively determined with reference to the built-in 1 µm scale bar in the TEM images, and the size was described as the equivalent diameter. To accurately determine the size and shape of the particles, the segmented images were optimized in such a manner that the noise, edge effects, and aggregates were reduced to consider the size of individually resolved particles. From the particle size distribution results (Figure 3c), the existence of a population of particles in the nanometer range and the right-skewed size distribution indicate the presence of primary nanoparticles and minor evidence of agglomeration.
UV-Visible Spectroscopy
In the UV-Vis absorption spectrum recorded for the Ag25Cu75 bimetallic nanostructures (Figure 4), there is a strong and broadened surface plasmon resonance (SPR) peak. This typical absorption phenomenon can be seen to be centered at about 433 nm. In its spectrum that falls between the SPR peaks of pure monometallic silver (about 400–420 nm) and copper (about 560–580 nm), its position strongly verifies the successful formation of Ag-Cu bimetallic nanostructures and not just physical mixtures of the individual materials. The visible broadening with a slight redshift of the SPR peak position also symbolizes intense electronic coupling and wholesale alloying between silver and copper atoms, with moderate size variability. In particular, the lack of any sharp absorption features also strongly indicates high material homogeneity. Significantly enough, there were also no absorption features detectable that could be attributed to the presence of copper oxides; this strongly verifies the homogeneity and chemical purity of the bimetallic system.
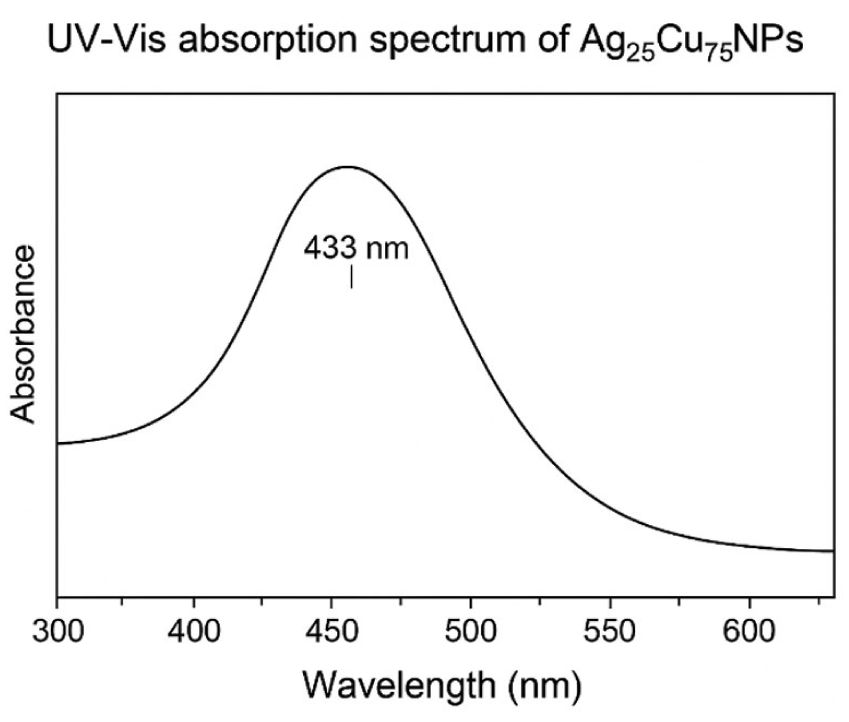
UV-Vis absorption spectrum of Ag25Cu75.
Taken together, the above findings of UV-Vis spectroscopy offer strong supporting evidence for the inference drawn from another set of analyses, namely XRD and SEM/TEM analysis. These pieces of evidence support the successful creation of stable nanosized particles of Ag25Cu75 with superior optical properties, making them highly valuable for usage in environmental applications based on adsorption.
The UV-Vis absorption spectrum obtained for the Ag25Cu75 nanocomposite is presented in Figure 4, where a strong SPR absorption band is observed at wavelengths within the visible region, which is characteristic for Ag nanoparticles. The SPR absorption band is absent for the Cu nanoparticles. This is understandable, since Cu nanoparticles have been reported to have weaker SPR bands that are very sensitive to their sizes as well as their surroundings.
In Ag-rich Ag-Cu systems, the intense Ag SPR band commonly dominates the optical response, effectively masking any contribution from Cu. Moreover, partial oxidation of Cu to Cu(I) or Cu(II) species in aqueous environments can significantly damp or eliminate Cu-related plasmonic features. Particle size dispersion and nanoparticle aggregation may further broaden and suppress weaker optical bands.
Therefore, the UV-Vis spectrum primarily reflects the Ag-dominated optical behavior of the nanocomposite and should not be interpreted as direct evidence for the absence of Cu or for definitive alloy formation. Confirmation of Cu incorporation relies on complementary compositional analyses rather than optical spectroscopy alone.
XRD analyses confirmed the crystalline nature of the silver-copper bimetallic nanoparticles and exhibited characteristic diffraction patterns characteristic of face-centered cubic crystals. The results clearly demonstrate the successful creation of an alloy at the nanolevel. Fourier transform infrared spectroscopic studies detailed the existence of surface chemical functional groups that remained intact even after the synthesis and adsorption process, thus unequivocally indicating the lack of undesirable oxide species and the evidence of interfacial interactions. Morphological studies through scanning electron microscopy analyses demonstrated near-spherical nanocrystals. However, significant clusters were observed, and such occurrences can be attributed to their higher surface energies. Further analyses through transmission electron microscopy studies clearly supported the structural nanoscale properties and high packing density of such particulate formations. Such comprehensive analyses clearly demonstrate the successful creation of crystalline silver-copper nanoparticles at the nanolevel that possess abundant adsorption sites and are responsible for their significantly higher adsorption activities toward diclofenac and crystal violet.
While the material is an Ag-Cu system for all practical purposes in this study, for clarity, the current characterization does not provide any direct evidence of discrete Cu nanoparticles or fully alloyed Ag-Cu structures. For one thing, the XRD patterns are dominated by Ag-related reflections; this may be expected due to the higher crystallinity and scattering factor of Ag relative to Cu. Similarly, TEM provides, foremost, morphological information and does not resolve Cu-rich domains.
It follows that the obtained material should more correctly be termed an Ag-Cu nanocomposite, whereby Cu is included either within or associated with Ag-rich nanoparticles. The presence of Cu is confirmed by the use of elemental analysis-EDX/XRF-while its exact structural distribution-alloyed versus surface-associated-cannot be determined by the techniques applied.
Elemental Characterization
Energy-dispersive X-ray spectroscopy coupled with elemental mapping was used to investigate the elemental composition and its spatial distribution. An EDX spectrum (Figure 5) evidenced Ag and Cu as the dominant elements, with a normalized weight percent composition consistent with the nominal Ag25Cu75 composition. Elemental mapping in Figure 6 shows a rather homogeneous distribution of Ag and Cu on the nanoparticle surface, thus confirming the formation of bimetallic nanocomposites and not phase-separated domains.
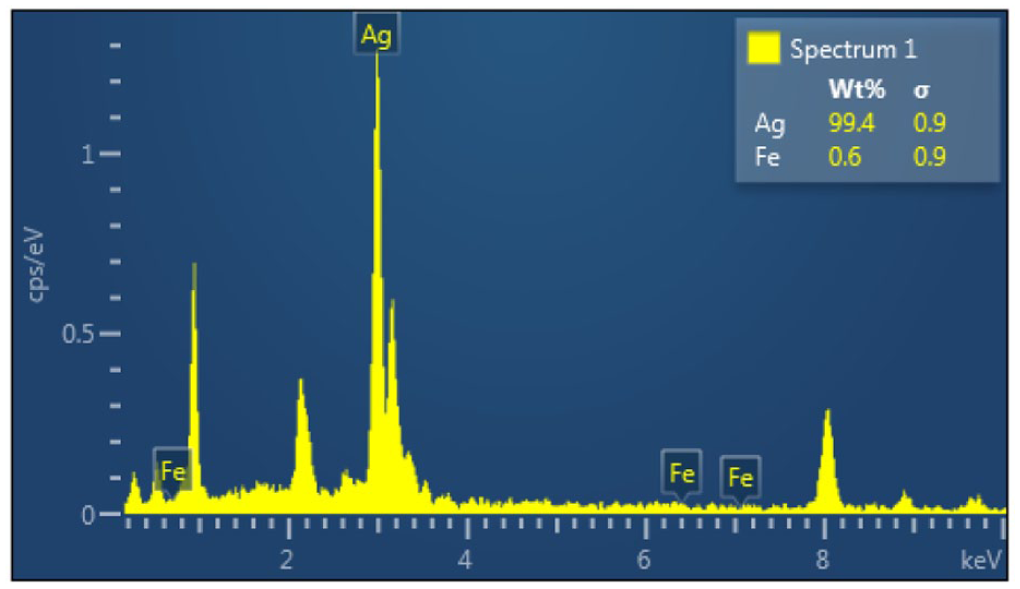
EDX spectrum of Ag25Cu75 nanoparticles.

EDX elemental mapping of Ag and Cu in Ag25Cu75 nanoparticles.
Complementary X-ray fluorescence analysis was carried out to validate the bulk elemental makeup of the synthesized material. Indeed, the XRF results presented in Figure S1 in the Supplemental Information confirmed Ag and Cu as the major components, in agreement with the EDX observations. The presence of other, weaker elemental signals can easily be assigned to background contributions or sample preparation artifacts.
The nitrogen adsorption-desorption BET analysis and thermogravimetric analysis (TGA) were not carried out in the present study; hence, the surface area, pore structure parameters, and thermal stability could not be determined quantitatively. This was explained in the discussion section, and these tests are recommended to be carried out in order to establish a relationship between the physicochemical properties and the adsorption activity.
EDX mapping and XPS are among the advanced surface-sensitive techniques that are very helpful in resolving several issues related to bimetallic nanomaterials with regard to elemental distribution and its oxidation states. However, these analyses have not been performed in the present study. Thus, the available characterization does not allow definitive confirmation of alloying at a nanoscale or the presence of discrete Cu nanoparticles.
Bulk elemental analysis through XRF and controlled composition of the synthesis support the presence of Cu in the material synthesized, whereas XRD and TEM reflect Ag-dominated crystalline and morphological features. The correct description of the material would more precisely be an Ag-Cu nanocomposite in which Cu is incorporated within or associated with Ag-rich nanoparticles.
Efficacy of DCF and CV Removal
Effect of pH
The study confirmed that the adsorption performance of Ag25Cu75 NPs for DCF and CV was highly pH dependent, as depicted in Figures 7 to 9. The variation in pH for both contaminants changed the surface charge of the adsorbent, the ionization status of the adsorbates and, in turn, the electrostatic interactions that constituted the adsorption mechanism governing the adsorption process. For DCF, the adsorption effectiveness diminished gradually with rising pH. The acidic milieu (pH ≈ 3) produced the highest removal, where the nanoparticle surface took on a positive charge and hence enhanced the electrostatic attraction directed toward the deprotonated carboxylate moiety (–COO−) of DCF. Above the pKa of DCF (≈4.2) the molecule's charge is mostly negative; concurrently, the Ag25Cu75 NPs develop a progressively larger negative surface charge beyond their point of zero charge (PZC ≈ 7). The resulting charge repulsion, combined with possible precipitation and OH− blocking the active sites, lowers the adsorption capacity at alkaline pH levels.
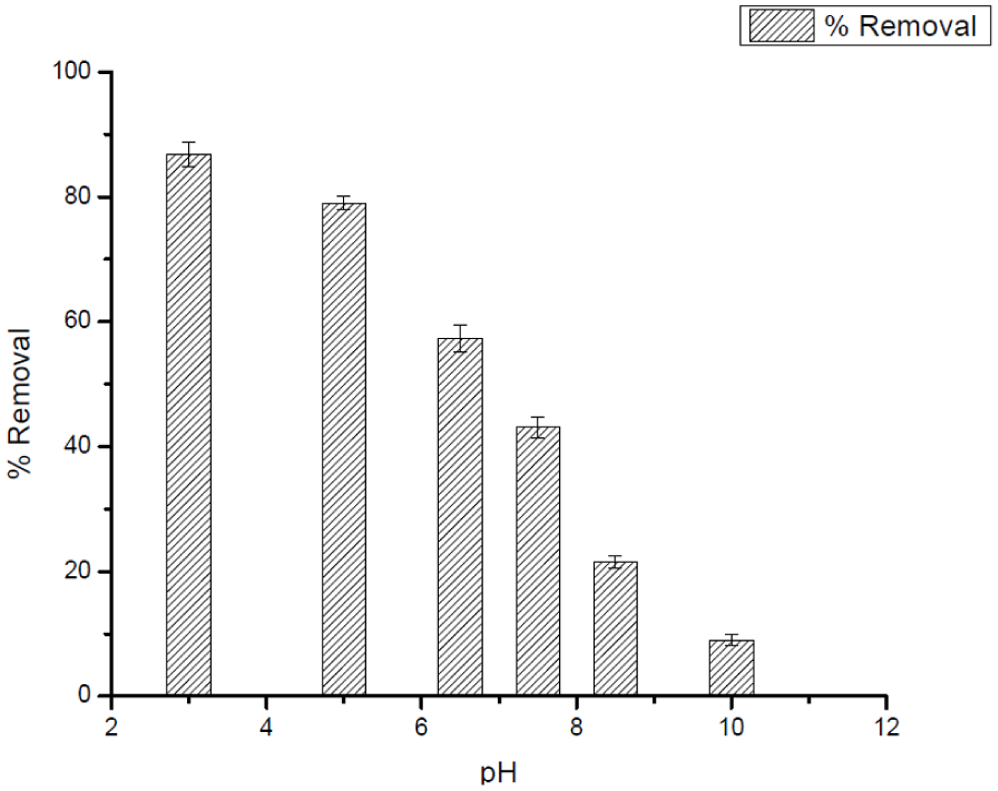
Factors affecting the adsorption efficiency of 50 mg L−1 DCF: Effect of pH using 0.025 g L−1 of Ag25Cu75 NPs.
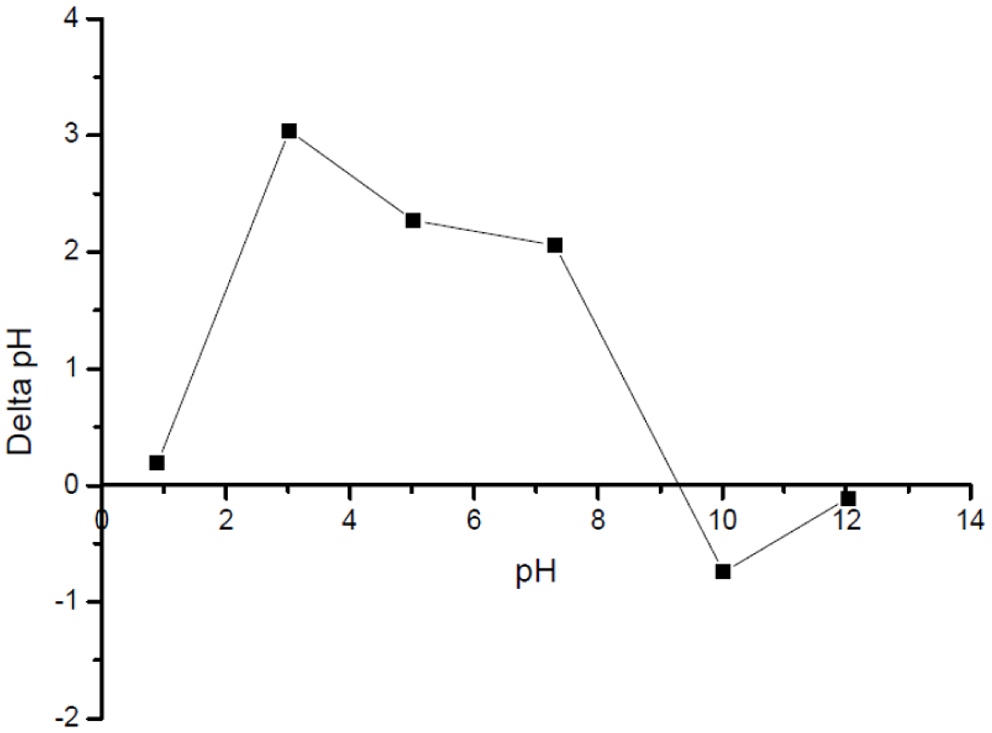
Point of zero charge of Ag25Cu75 NPs by the salt addition method (pH vs. pH initial).
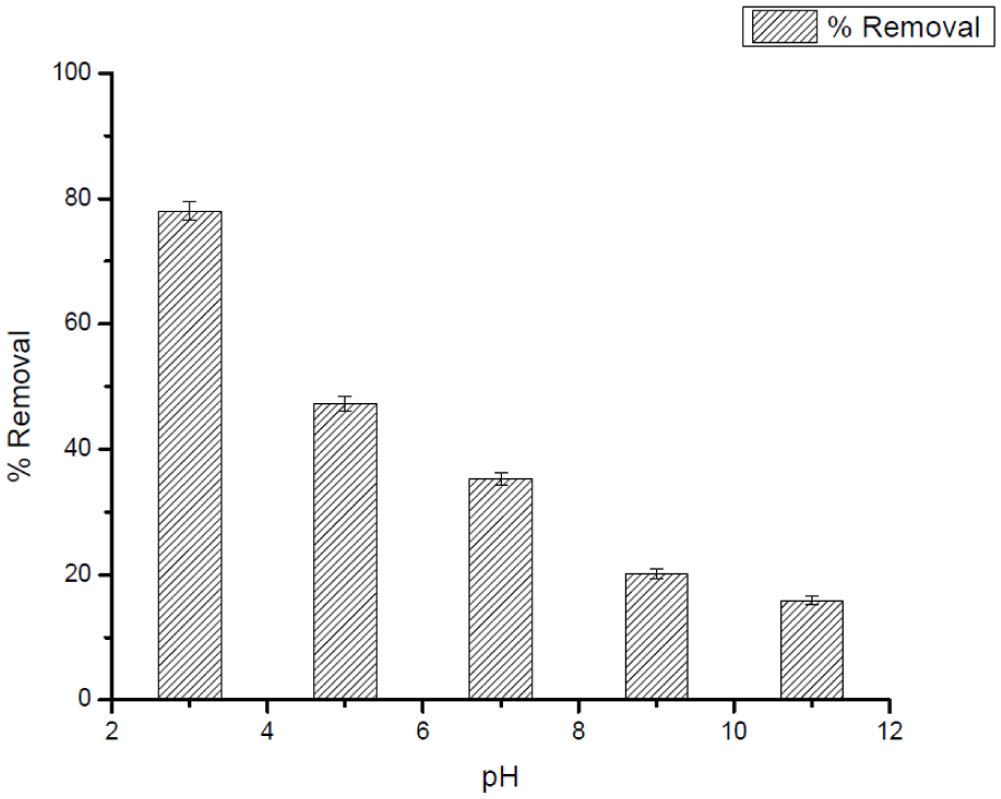
Factors affecting the adsorption efficiency of 10 mg L−1 CV: Effect of pH using 0.025 g L−1 of Ag25Cu75 NPs.
The Ag-Cu nanocomposite had a relatively high point of zero charge (PZC ≈ 9.0). This value suggests that, under acidic and near-neutral environments, the surface of the nanocomposite is predominantly positively charged. Thus, in this range, electrostatic interactions between the adsorbent surfaces and any anionic species would be relatively significant. Hence, the high removal of DCF, which predominantly has an anionic form across a wide range of pH, can be said to result mainly through this electrostatic attraction with the positively charged nanoparticle surfaces.
On the other hand, the adsorption behavior of CV, which is a cationic dye, also occurs through different mechanisms. Since the surface of Ag-Cu carries a positive charge below its PZC, electrostatic attraction between CV and CV+ species is unfavorable at acidic pH. However, the adsorption of CV onto the Ag-Cu surface at low pH occurs due to non-electrostatic force, including π-π stacking between the rings of CV and the surface-bound groups, π-cations, and van der Waals forces. The similar adsorption behavior of CV and other cationic dyes on positively charged metallic-based surfaces has been reported in the literature and can be attributed to the above interaction forces.
As the pH increases toward and above the PZC, deprotonation of surface functional groups leads to the development of negatively charged sites, which promotes strong electrostatic attraction toward CV+ molecules. Under these conditions, electrostatic interactions become the dominant adsorption mechanism, supplemented by π-cation and π-π interactions, resulting in enhanced adsorption efficiency.
Similar trends have been observed in the literature where the adsorption of drugs and dyes was heavily influenced by the pH level because of the simultaneous changes in both the ionization of adsorbate and the surface chemistry of adsorbent (I. Ali et al., 2022; Ayawei et al., 2017; Guo et al., 2011; Zhou et al., 2021).
Effect of Adsorbent Dose
A systematic investigation was conducted on how the Ag25Cu75 NPs dosage affected the DCF and CV adsorption efficiency, and the findings are shown in Figures 10 and 11. In the case of DCF, the first step was to increase NP dose from 0.015 to 0.025 g and then to 0.05 g, and the result was that removal efficiency improved considerably. The reason for this improvement is the larger surface area that is available and the number of active binding sites at higher NP dosages, thus leading to more effective interactions with DCF. The removal efficiency wasn't able to go beyond ~0.05 g, which indicates that the DCF molecules in solution have been consumed and that their number is now equal to the sorbent surface of the NP. At this point, there are still unoccupied adsorption sites and, in some cases, particle clustering may occur at high dosage which would then limit the effective surface area and accessibility of the active sites (Gupta & Suhas, 2009; Tran et al., 2017). Thus, the choice of ~0.05 g of Ag25Cu75 NPs as the optimum dosage for DCF adsorption in this system was made.
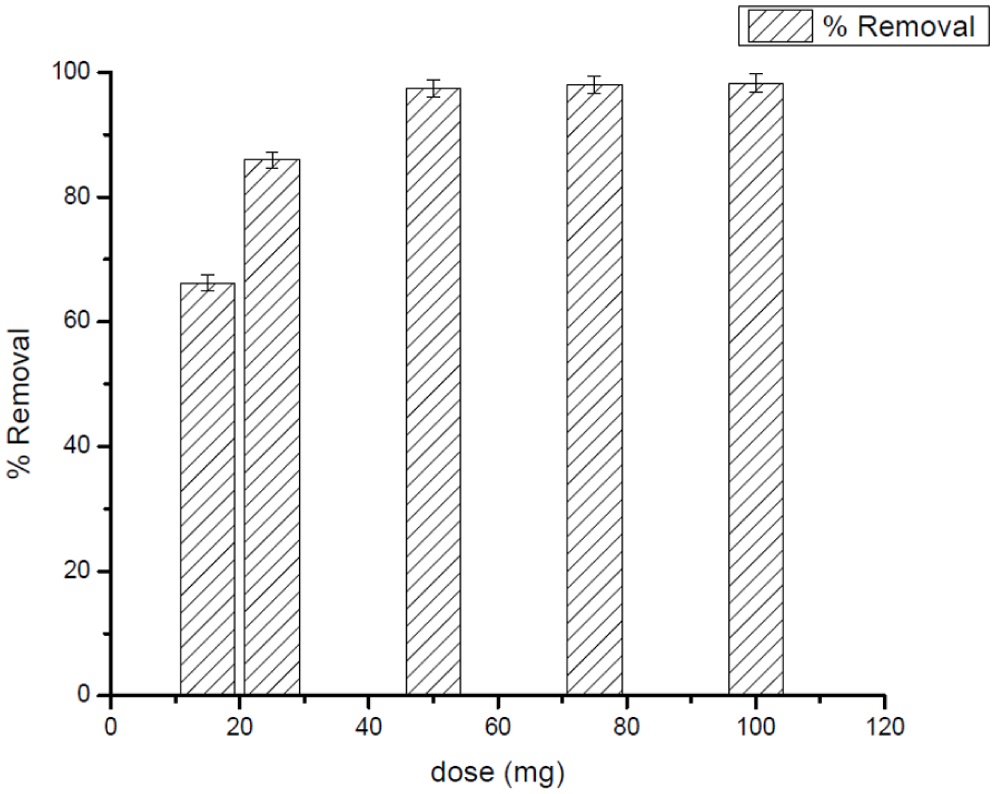
Factors affecting the adsorption efficiency of 50 mg L−1 DCF: Ag25Cu75NPs dosage at 25°C and pH = 3.
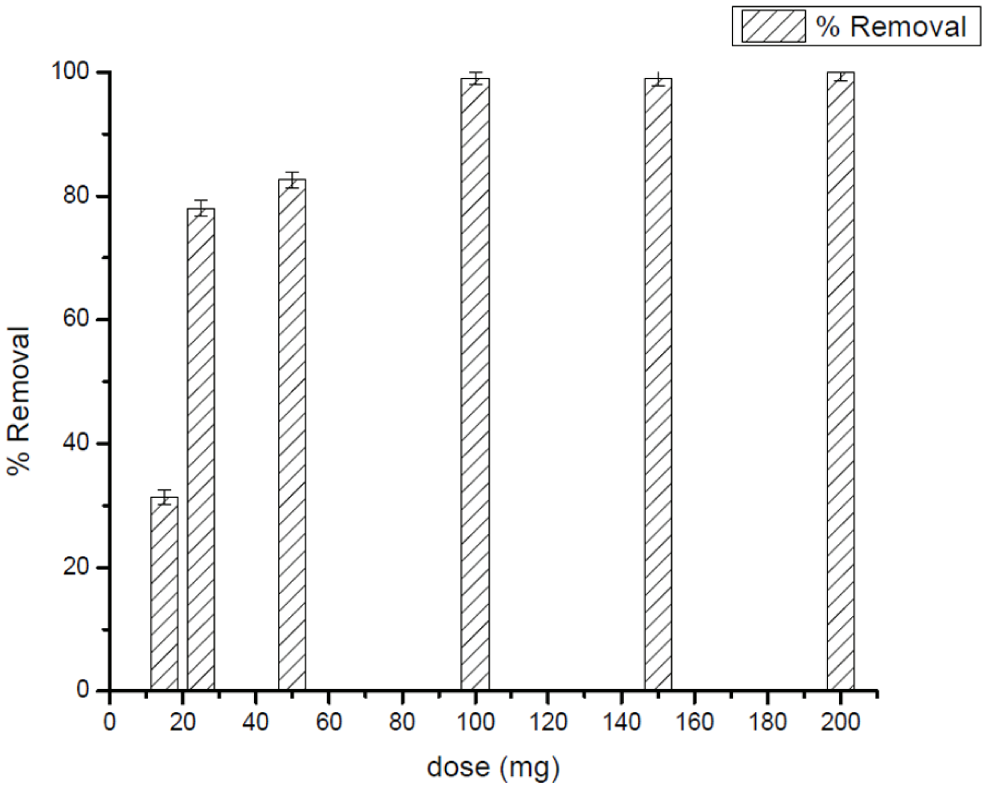
Causes affecting the adsorption efficiency of 10 mg L−1 CV: Ag25Cu75 NPs dosage at 25°C and pH = 3.
An analogous trend was observed for CV adsorption, where the removal efficacy progressively rose as the nanoparticle dosage was increased from 0.015 to 0.05 g. However, in contrast to DCF, CV adsorption continued to exhibit a slight enhancement up to approximately 0.10 g before reaching a stable maximum. These differences have been found to be a result of the distinctive chemical characteristics of CV, which are a cationic dye, enabling the formation of strong electrostatic forces and pi-cationic interactions to take place between CV and the surface of the nanoparticles. This enabled a higher number of the available adsorption sites to be occupied by CV even before the saturation point was attained. However, beyond a dose of 0.10 g, no additional improvement could be perceived.
In fact, examination of both contaminants indicates that even though a pattern of augmenting elimination with increasing rates, eventually plateauing, has been evident for both DCF and CV, the respective saturation levels are not the same. In other words, the saturation point for DCF occurred around 0.05 g, while for CV, it occurred around 0.10 g. This finding clearly supports the fact that cationic systems, such as CV, may require a higher concentration of the adsorbent to ensure optimum elimination capacity, whereas anionic pharmaceuticals, such as DCF, reach their saturation point at a lower concentration of the adsorbent. Such findings remain pivotal to ensure that the quantity of adsorbent used meets the specific requirements of the physicochemical properties of the respective contaminants to guarantee optimal elimination performance.
Effect of Temperature
The effect of temperature on the uptake of both DCF and CV to Ag25Cu75 nanoparticles was thoroughly examined, and results presented in Figures 12 and 13. In relation to the effectiveness in the removal of DCF, it can be seen that its effectiveness consistently increased, advancing from around 80% at 15°C to a point where nearly 92% effectiveness was reached at 35°C. The adsorption efficiency increased with rising temperature, indicating that higher thermal energy enhances the interaction between the adsorbent surface and the adsorbate molecules. This behavior suggests that adsorption is favored at elevated temperatures within the investigated range, consistent with an endothermic adsorption process. Under such circumstances, increased thermal energy would be expected to serve to enhance dispersal and reduce impedance within a process useful in increasing the interaction levels between dispersal molecules and surface reactive points on Ag25Cu75 nanoparticles. However, at temperature points above 35°C, there appeared to be a marked decrease in effectiveness levels. These might be suggested to be due to a number of reasons, including unbounded behavior and reduction in electrostatic forces, as well as alterations in nanomaterial surfaces (Girish, 2025; Özacar & Şengil, 2003).
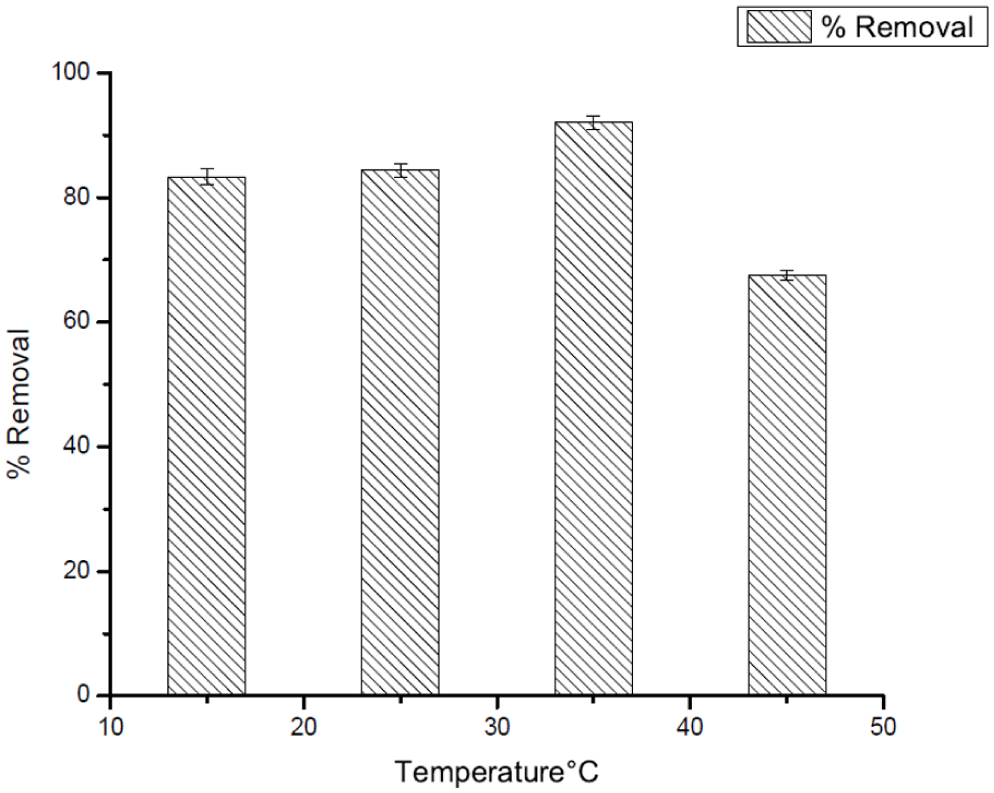
Effect of temperature on removal efficiency of DCF at pH = 3.
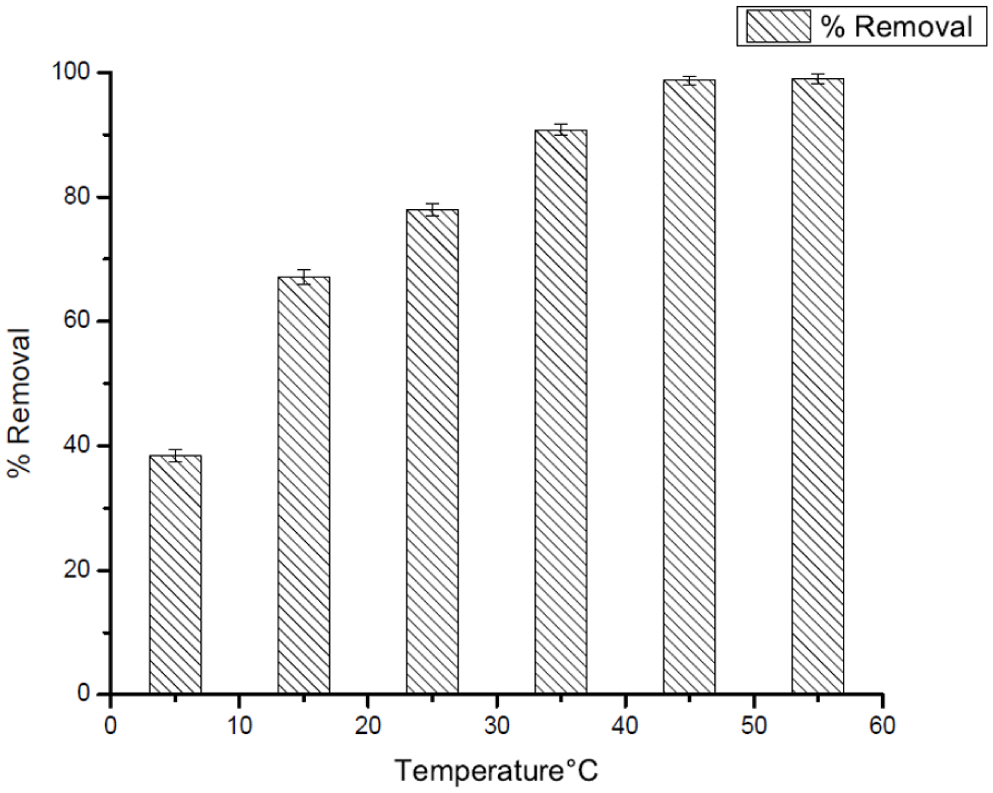
Effect of temperature on removal efficiency of CV at pH = 3.
On the contrary, the adsorption of Crystal Violet (CV) was strongly positively correlated with the increase in temperature. The efficacy of its removal became progressively better from around 40% at 5°C to close to total removal, i.e., around 100%, at 45°C. Such behavior indicates that the process of the adsorption of CV to the Ag25Cu75 NPs is largely endothermic over the entire range of the tested temperatures.
The increased intensity of the dye detected at higher temperatures can be ascribed to the increased electrostatic force of attraction between the cationic dye molecules and the negatively charged adsorption sites due to the facilitation of π-π interactions and van der Waals forces at higher temperatures. Unlike in the case of DCF, the amount of CV desorbed was negligible at higher temperatures, showing stronger adsorbate-adsorbent interaction.
Analysis of these two pollutants shows a clear difference in the behavior, in which the adsorption capability of DCF reaches its optimized efficiency at moderate temperatures, with increased temperatures later causing a reduction in efficiency, contrary to the adsorption process of CV, which increases with the enhancement of temperatures with no apparent decrease in efficiency.
This is due to the different physicochemical properties of the two compounds. While the pharmaceutical compound, DCF, with the anionic charge, is mostly prone to electrostatic and hydrogen bonding interactions, whose strength is reduced by the higher thermal energy, the cationic dye, CV, forms stronger complexes with the surface of the nanoparticles.
Such results re-emphasize the need for the adjustment of adsorption conditions to the specific nature of each pollutant. In terms of applicability, temperatures around 35°C would be ideal for the elimination of DCF, while higher temperatures would be favored for the adsorption of CV. This difference clearly illustrates the flexibility of use of Ag25Cu75 NPs in dealing with contaminants of diverse natures.
Mix Adsorption Experiments of DCF and CV
The adsorptive capacities of the Ag25Cu75 NPs have been checked for diclofenac (DCF, 50 mg L−1) and crystal violet (CV, 10 mg L−1) dye mixtures, as shown below in Figure 14, after a contact time of 150 min, wherein the removal percentages for DCF & CV reached 84% and 79%, respectively. In mixed-solute systems, the adsorption capacities and kinetic parameters for CV and DCF were comparable to those obtained in single-solute experiments within experimental uncertainty. No statistically significant decrease in adsorption performance was observed under the investigated conditions, suggesting limited competitive interaction between the two pollutants at the tested concentrations. However, given the inherent variability of batch adsorption experiments, this observation is interpreted as an indication of weak competition rather than complete independence of adsorption sites.
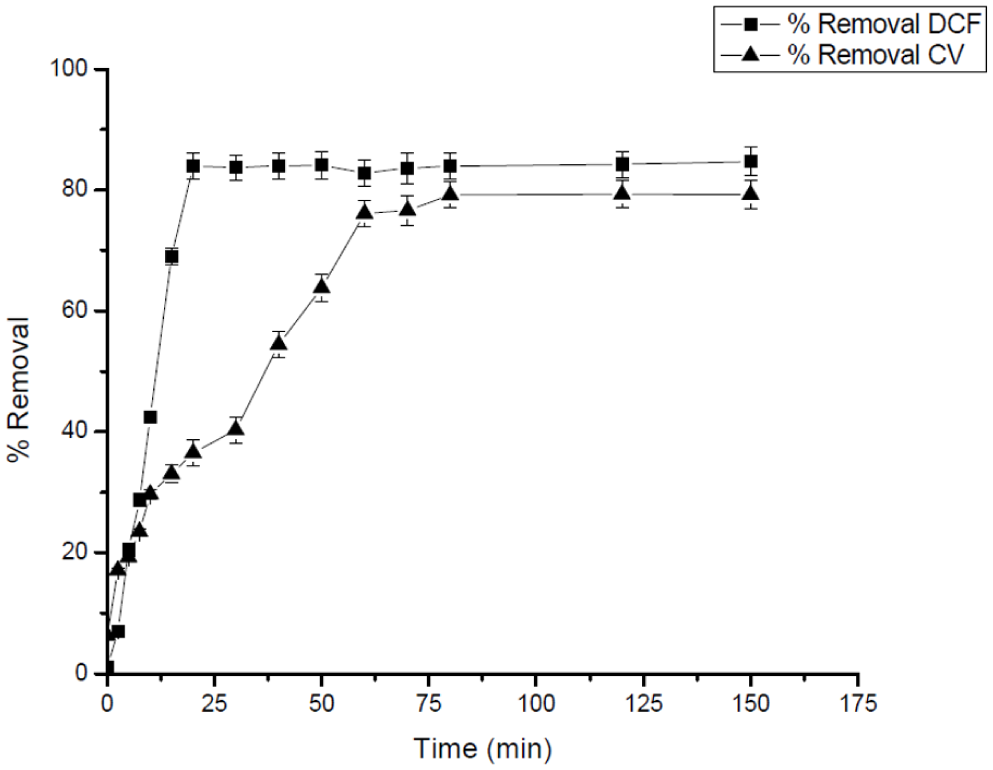
Removal of DCF (50 ppm) and CV (10 ppm) Using 25 mg of 1Ag:3Cu Nanoparticles at pH 3, 25°C, and 105 rpm.
This scant presence of antagonist effects signifies that the surface of the Ag-Cu nanoparticles maintains an adequate specific surface area with considerable densities for reactive adsorption sites, allowing for the simultaneous adsorption of both molecules. Despite the larger molecular size and higher space requirement of CV ion as against diclofenac ion, whose supposed reduction might potentially impede adsorption capacity due to the occlusion of the reactive sites, results obtained showed that steric hindrance effects were not significant. This result agrees with previous studies showing that binary metallic nanoparticles comprising different surface functional groups enable the collaborative adsorption of multiple analytes due to the combined effects of electrostatic, π-π, and H-bonding interactions (Ayawei et al., 2017; Gupta & Suhas, 2009; Tran et al., 2017).
It is pertinent to point out that though both pollutants exhibit a high removal efficacy, a slightly superior adsorption property was noticed in DCF relative to CV under equal conditions. This discrepancy can, probably, be attributed to the relatively smaller molecular structure of DCF, which tends to display better diffusion toward the inner active sites. On the contrary, the absorption of CV is more inclined toward outer surface interactions. This type of mechanistic study, hence, highlights that though Ag25Cu75 nanoparticles exhibit a non-selective, broad-spectra type of adsorption property, still properties like molecular size, charge, and functional groups play a critical role in dictating their absorption kinetic patterns and equilibrium isotherms. Finally, this study, thus, re-emphasizes the efficacy and robust nature of Ag25Cu75 nanoparticles as a superior, versatile, and very capable class of adsorbent agents targeting multi-pollutant systems, possessing a vital implication in practical-scale wastewater treatment plants, especially in treatment conditions where pharmaceutical agents and dye pollutants commonly co-exist.
It should be noted that while the bimetallic composition may influence surface chemistry and adsorption behavior, the present results do not permit conclusive distinction between synergistic and additive effects.
Plausible Adsorption Mechanism
Based on the combined experimental observations obtained from pH-dependent adsorption, thermodynamic trends, and kinetic modeling, a plausible adsorption mechanism for CV and DCF onto the Ag-Cu nanocomposite is proposed. The schematic illustration in Figure 15 qualitatively summarizes the dominant interaction pathways governing pollutant uptake under the investigated conditions.
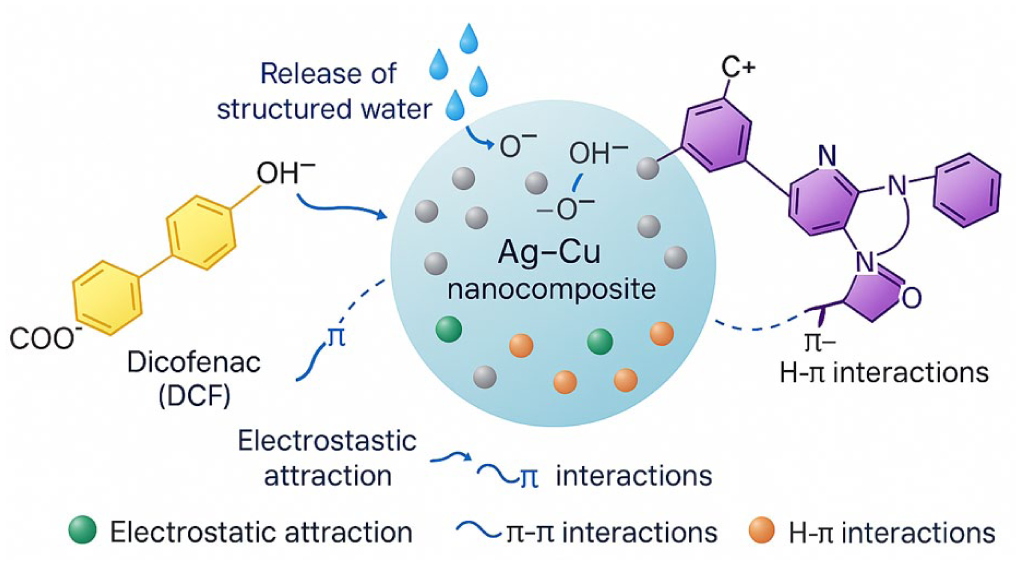
Schematic illustration of the plausible adsorption mechanisms of CV and DCF onto the Ag-Cu nanocomposite, highlighting electrostatic interactions, π-π stacking, hydrogen bonding, and desolvation effects. The schematic is intended to qualitatively support experimental observations rather than to represent a definitive molecular structure.
As illustrated in Figure 15, adsorption of CV is primarily governed by electrostatic attraction between the cationic dye molecules (CV+) and negatively charged surface sites (–O−) formed at pH values above the point of zero charge, in addition to π-π interactions between the aromatic rings of CV and Ag-rich domains on the nanocomposite surface. In contrast, adsorption of DCF involves electrostatic interactions between the anionic carboxylate group (–COO−) and protonated surface hydroxyl groups, as well as hydrogen bonding and surface complexation with Cu/CuOx-associated sites. The release of hydration water molecules during adsorption contributes to the observed positive entropy change, supporting an entropy-assisted adsorption process.
Adsorption Isotherms and Capacity Evaluation
Adsorption equilibrium data for CV and DCF adsorption onto both adsorbents was analyzed employing Langmuir and Freundlich isotherm equations to understand the adsorption mechanism and quantify adsorption capacity. Percentage removal is a preliminary measure of adsorption capacity and efficacy; however, adsorption capacity (q) in mg g−1 was used as a priority parameter for a thorough comparison with other adsorbents reported.
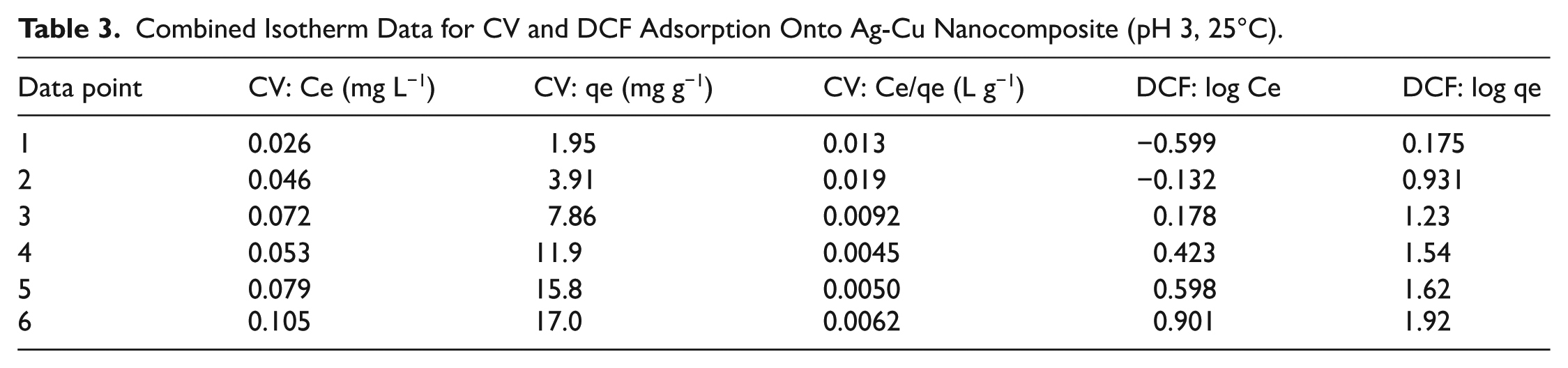
For the CV system, it is found that the Langmuir isotherm fit was satisfactory in terms of experimental data points (
Combined Isotherm Data for CV and DCF Adsorption Onto Ag-Cu Nanocomposite (pH 3, 25°C).
Isotherm Model Parameters for the Adsorption of CV and DCF Onto the Prepared Adsorbent.
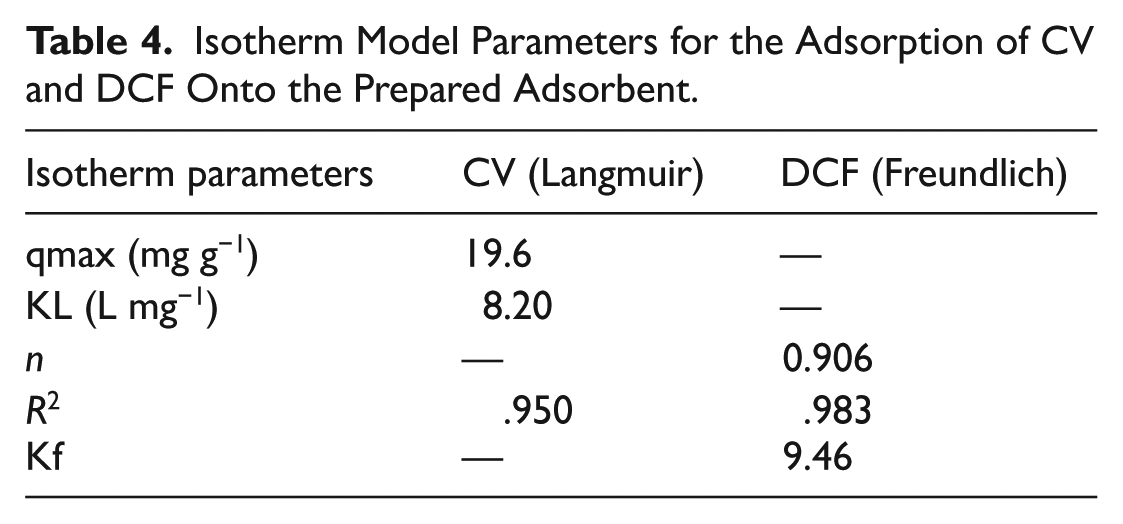
At higher CV concentrations, small deviations from Langmuir linearity were found, which could be characteristic of partial surface heterogeneities or the beginnings of a multilayer. These data points were excluded from the analysis to obtain a valid estimation of the monolayer.
In contrast, the adsorption of DCF could be better fitted using the Freundlich isotherm equation (
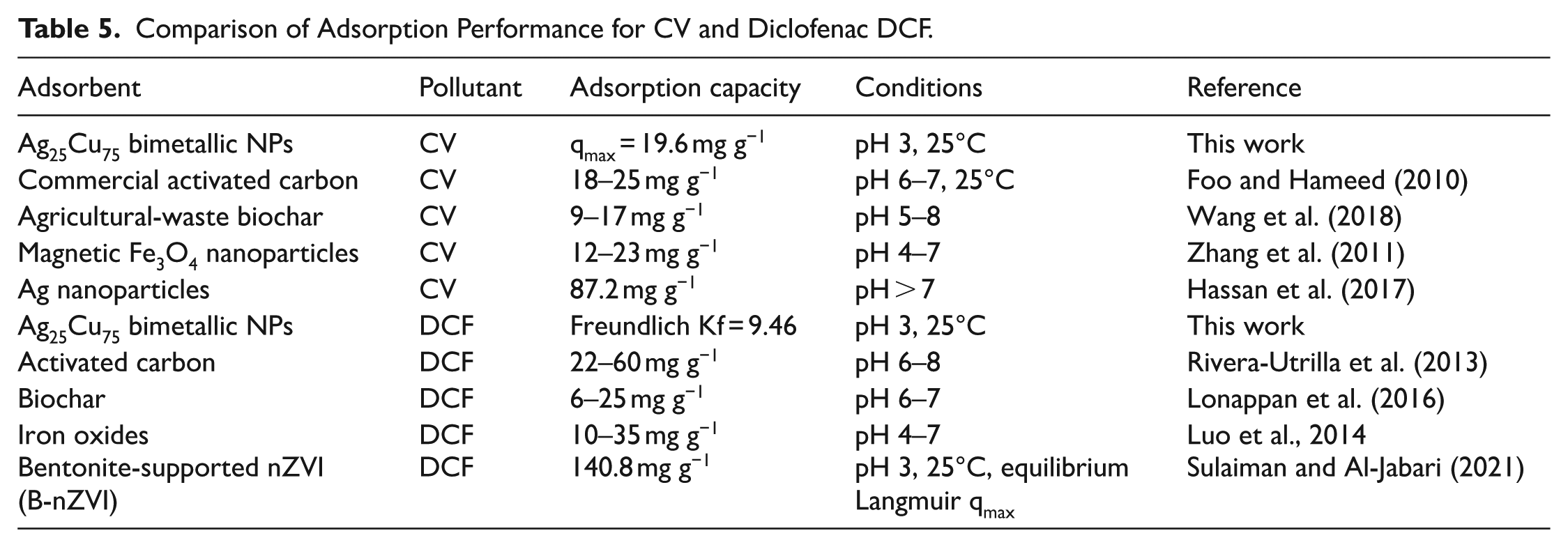
From the comparative study of the adsorption capacity of Ag25Cu75 nanoparticles toward both CV and DCF in relation to commercial adsorbent materials (as shown in Table 5), the adsorption capacity of Ag nanoparticles toward CV is analogous to other adsorbent materials encountered in previous studies. Regarding the adsorption capacity for DCF, the adsorption is within the prescriptions of other heterogeneous adsorbent materials. As opposed to striving to beat all hitherto existing adsorbents in terms of capacity, the emphasis in the present work is placed on evaluating the adsorption behavior of chemically different contaminants using the same bimetallic compound through batch equilibrium and column fixed-bed studies.
Comparison of Adsorption Performance for CV and Diclofenac DCF.
Very importantly, the study conducted on single-solute versus mixed-solute systems clearly showed that the difference in adsorption isotherm parameters was negligible, ensuring that competitive adsorption was not significant in the tested conditions. This trend indicates that the Ag-Cu nanocomposite has available diverse adsorption sites, which allows it to adsorb both DCF and CV simultaneously without any inhibitory effects on adsorption. These adsorption tendencies can be attributed to those reported in previous studies on multicomponent adsorption, in which surface heterogeneity as well as the presence of diverse functional groups make organic pollutants adsorb non-selectively on adsorbents (Ayawei et al., 2017; Gupta & Suhas, 2009; Tran et al., 2017).
The Freundlich constant (Kf), which reflects the adsorption capacity of the Ag-Cu nanocomposite, is included in Table 4 together with the corresponding n and
Thermodynamic Analysis of Adsorption
In order to gain insight into the temperature dependence of the adsorption of DCF and CV onto Ag25Cu75 nanoparticles, thermodynamic parameters including the standard Gibbs free energy change (ΔG°), enthalpy change (ΔH°), and entropy change (ΔS°) were estimated. These parameters were calculated from adsorption equilibrium constants determined at different temperatures using the van’t Hoff equation:

where
For CV, the enthalpy change was found to be positive (ΔH° = +87.3 kJ mol−1). This suggests that the adsorptive capacity increased with temperature within the range used in this investigation. Hence, it may be inferred that a thermally assisted adsorptive process is involved, which aligns well with the overall experimental observations. In this case, it was found that the CV removal efficiency increased from 38% to above 98% upon increasing temperature from 5 up to 45°C. However, it should be noted that the observed positivity for the ΔH° value may not be considered conclusive for materials, since it was found for a relatively narrow range of temperature values.
The positive entropy change obtained for CV (ΔS° = +295.1 J mol−1 K−1) suggests an increase in disorder at the solid-liquid interface during adsorption. This behavior may be attributed to the release of structured water molecules from the adsorbent surface and solvated dye molecules into the bulk solution; however, such interpretations are constrained by the limited temperature range explored.
The dependence of temperature on molecular adsorption on a surface can also be seen from the Gibbs free energy change (ΔGº). At low (e.g., 5°C) temperatures, the ΔGº was positive (+5.1 kJ mol−1); hence, molecular adsorption was disfavored at low (5°C) temperatures. However, the ΔGº is increasingly negative with increasing temperatures; for example, (−18.1 kJ mol−1) at 45°C, suggesting that the adsorption of CV at elevated temperatures will behave similarly to what has been currently published in the scientific literature for the adsorption behavior of nanomaterials/dyes, based on their temperature dependence (Ayawei et al., 2017).
For diclofenac, thermodynamic analysis revealed similar temperature-dependent behavior. The positive enthalpy change (ΔH° = +65.8 kJ mol−1) indicates that adsorption capacity increased with temperature, although the magnitude of ΔH° was lower than that obtained for CV. This observation is consistent with experimental results, which showed an increase in DCF removal efficiency from approximately 42% at 5°C to more than 95% at 45°C. The positive entropy change (ΔS° = +198.4 J mol−1 K−1) suggests increased randomness at the solid–liquid interface, which may be associated with partial dehydration of DCF molecules and surface sites during adsorption.
The ΔG° for DCF also was temperature dependent, showing a similar behavior as the ΔG° described above. The value of ΔG° at 5°C is a small positive number (+3.8 kJ mol−1), which becomes more negative as the temperature increases and therefore less so at a temperature of 45°C (ΔG° = −15.6 kJ mol−1). This suggests that the adsorption of DCF becomes thermodynamically favorable with increasing temperature; however, since the temperature range tested was very limited, caution should be exercised when drawing any conclusions from this information.
Temperature-Dependent Adsorption Behavior
Whereas in the two adsorption processes there was a clear endothermic trend and the processes were guided by entropic reasons, there were also marked differences in the temperature dependence. For the DCF adsorption process, there was a marked increase in the effectiveness of adsorption from 15 to 35°C, from a value of about 80% to a value of 92% (as indicated in Figure 12). This can be mainly ascribed to the increased ease of motion of the molecules as well as the reduced resistance in the interfacial layer and the ease of access to binding sites. On the other hand, above 35°C, there was a slight drop in the effectiveness values. This may be accounted for by the fact that the adsorbate is not fully released from the surface. This could be the effect of the high kinetic energy and the decreased intensity of the electrostatic attractive forces. This trend indicates that there must be an optimum temperature range that facilitates the adsorption process after which the adsorption becomes adversely affected by the high temperature agitations that reduce the stability on the surface of the adsorbate and the adsorbent (Ayawei et al., 2017; Özacar & Şengil, 2003).
Contrary to this, adsorption of CV exhibits greater robustness in terms of temperature variations. Rather, its effectiveness in removal gradually increased from around 40% at 5°C to almost 100% at 45°C (as reflected in Figure 13). Indeed, there has been an increasing trend in more negative standard Gibbs free energy with higher temperatures, which strongly supports its greater affinity for self-reactivity with increasing temperatures. This thermal robustness is accredited to strong π-π stacking interactions existing among the CV’s own cyclic aromatic domains and its silver-copper nanocomposite material, along with extremely strong coulombic forces of attraction among the positively charged dye molecules and negatively charged or electrically polarized reactive domains, which in turn remain unchanged even under higher thermal settings, thus accounting for continuous progress in CV’s adsorption efficiency (Gupta & Suhas, 2009; Tran et al., 2017).
Comparative Thermodynamic Discussion
The thermodynamic evaluation in general shows that the adsorption processes of both DCF and CV are spontaneous at high temperatures and mainly driven by positive entropy changes, though their mechanisms of adsorption are quite different. The adsorption of CV is very much endothermic and gets supported by temperature all the time, which implies that entropy gain and interfacial reorganization are at the forefront of the process. On the other hand, DCF adsorption is only slightly endothermic and has a temperature range of best activity, after which the effects of desorption become more pronounced. These differences point out the necessity of pollutant-specific optimization regarding the operating conditions, especially in wastewater treatment plants with a combination of pharmaceutical and dye contaminants. The relatively small standard deviations observed across triplicate experiments indicate good experimental reproducibility.
The different thermal responses observed for CV and DCF adsorption can be rationalized by considering their molecular structures and interactions with the heterogeneous Ag–Cu nanoparticle surface. Crystal violet is a bulky, cationic dye with a highly delocalized positive charge and extensive aromatic character, which promotes strong electrostatic attraction and π–metal or surface complexation interactions. The relatively high enthalpy change observed for CV adsorption suggests that elevated temperatures facilitate partial desolvation and structural rearrangement of the dye molecules, allowing more effective interaction with surface sites.
In contrast, DCF, which is an anionic and less bulky pharmaceutical compound with carboxylate and aromatic groups and fewer degrees of charge delocalization, likely has the adsorption properties determined by a combination of electrostatic, hydrogen bond, and coordination interactions, which might not demand as much reorganizational energy as is the case with CV. This is also in agreement with the lower change in enthalpy observed during the adsorption of DCF and its weaker temperature dependence.
The positive entropy changes obtained for both pollutants may be associated with the release of hydration shells and interfacial water molecules during adsorption; however, the extent of this effect is expected to differ due to variations in molecular size, charge density, and solvation behavior. These considerations provide a molecular-level framework for interpreting the observed thermodynamic trends, while acknowledging that definitive mechanistic conclusions would require complementary spectroscopic or computational analyses.
Adsorption Kinetics and Mechanistic Implications
The adsorption kinetics of CV and DCF onto the Ag-Cu nanocomposite were evaluated using pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models to ensure a non-speculative assessment of adsorption behavior. Among the tested models, the PSO model provided the most satisfactory description of the experimental data for both pollutants.
For CV adsorption, the PSO model yielded a high correlation coefficient (
Filtration Results
Continuous-flow fixed-bed column experiments were carried out to give a preliminary evaluation of the dynamic adsorption behavior of the Ag25Cu75 nanocomposite in continuous-flow operation and to complement the batch adsorption results. Quartz sand was added as an inert granular media to enable column packing and proper flow distribution, and also to act as a reference material for comparison to demonstrate the negligible removal capability of non-functional filtration media under identical operating conditions.
The experiments conducted by mixing the quartz sand with Ag25Cu75 nanoparticles at weight ratios of 49:1 and 49.5:0.5 (w/w) showed obvious increases in DCF and CV removal efficiencies compared to the sand alone. Figure 16 illustrates that the increase of nanoparticles loading within the fixed-bed column enhanced contaminant removal, thus reflecting a positive contribution by the bimetallic nanocomposite to column performance. These results reflect the effectiveness of the Ag-Cu nanomaterial under continuous flow conditions, thus possibly making its integration into the flow-through treatment configuration feasible.
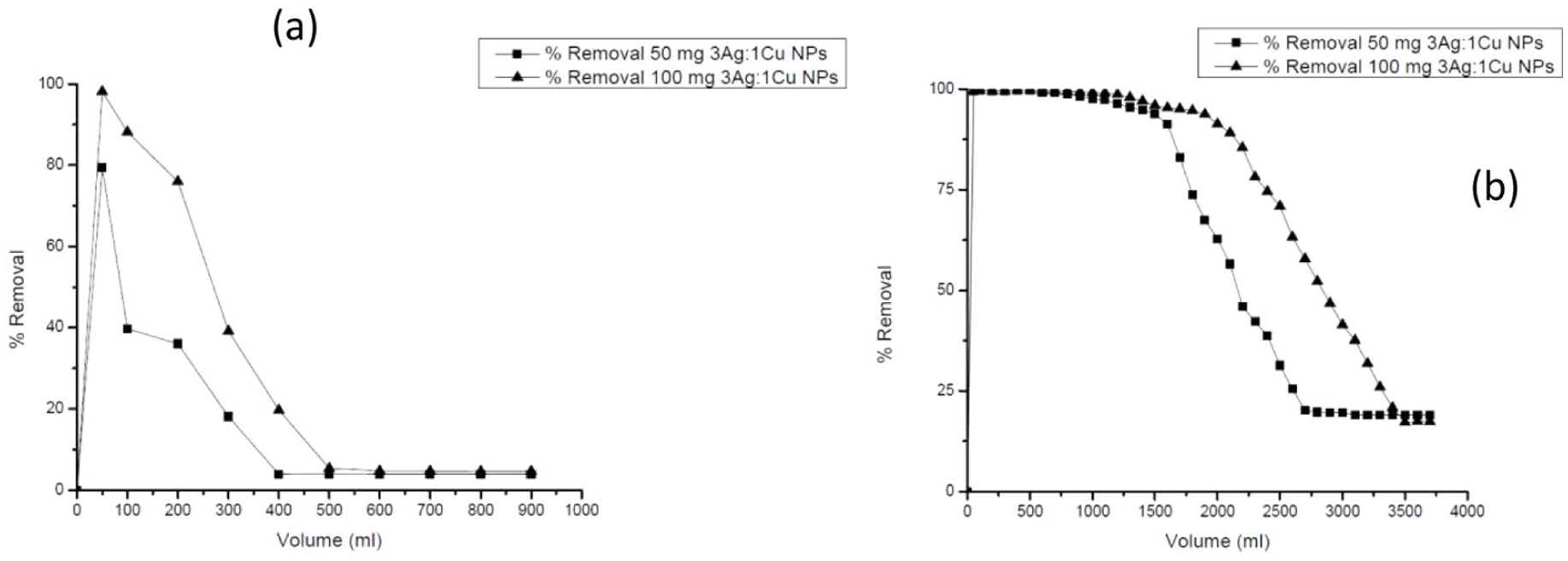
Removal efficiency of (a) DCF using 0.05 (g L−1) and (b) CV using 0.01 (g L−1) by column laboratory which included excess sand mixed with Ag25Cu75 NPs at different ratios.
It needs to be emphasized, however, that sand filtration technology, which is commonly used in water treatment, is only intended, among other uses, for the removal of turbidity and particulate matter. It is not intended or used as an adsorption process for the removal of dissolved organic compounds such as DCF and CV. The task of comparing the new material with sand, therefore, is intended to be construed only as an experimental control, and not comparable as an adsorption technology. In fact, the removal of dissolved organic matter, as explained earlier, can be readily accomplished as a water treatment technology using active materials. These materials can be, but are not limited to, activated carbon, biochar, ion-exchange resins, and metals. It must also be stated, as discussed earlier, that the water treatment using the Ag-Cu nanocomposite material, as explained and discussed earlier in the comparison on the water adsorption capacity of the new material with existing materials (see Table 5), is supportive.
The continuous adsorption behavior observed corroborates the pH-dependent interaction mechanisms and adsorption trends in batch experiments, thereby reinforcing the validity of proposed removal pathways under static and dynamic conditions. Unfortunately, however, complete breakthrough curves were not obtained due to the limited scope of experiments conducted in this work, and quantitative column models. Whereas bed depth, influent concentration, and flow rate are well known to strongly influence breakthrough behavior and service time in fixed-bed systems, these parameters are not varied systematically in the present study.
The experiments conducted in this column should therefore be regarded as proof-of-concept demonstrations of continuous-flow adsorption performance rather than as a comprehensive basis for process design or scale-up. Further studies incorporating extended operational time, full breakthrough curve analysis, and Bed Depth Service Time (BDST) modeling would thus be essential for predictive design optimization as well as the further development of the Ag-Cu nanocomposite applicability in large-scale water treatment systems.
When compared with previously reported Ag-, Cu-, and Ag-Cu-based adsorbents, the adsorption performance observed in this study falls within the range reported for similar nanomaterials under comparable laboratory conditions. Rather than aiming to outperform all existing adsorbents, the present work emphasizes consistent evaluation of adsorption behavior for two chemically distinct contaminants using a single Ag25Cu75 composition. This approach allows clearer interpretation of adsorption mechanisms, thermodynamic behavior, and column performance, which are often reported separately in the literature. The inclusion of both batch and continuous-flow experiments further distinguishes this study by providing insight into the applicability of Ag-Cu nanocomposites beyond equilibrium batch systems.
Study Limitation
Even in view of the enhanced adsorption performances presented herein, some limitations need to be recognized. All the adsorption runs were carried out under controlled laboratory conditions using synthetic aqueous solutions that are hardly representative of the chemical complexity, competing species, and variability that characterizes real wastewaters. Therefore, the transferability of the herein described adsorption behavior into practice is still to be validated. The adsorption tests were conducted over short-time scales, and stability of materials for extended time scales, regeneration ability, and reusability of Ag25Cu75 nanocomposites were not evaluated. In addition, although column tests were conducted, the ability of nanoparticles to leak from the system under extended time scales was not taken into account, making opportunity for further studies on this particular area. From a characterization point of view, although the Ag and Cu composition and spatial location were verified through EDX, elemental mapping, and XRF, the lack of BET and corresponding TGA measurements meant that the quantification of certain physicochemical characteristics was not carried out. Specifically, the involvement of the aforementioned techniques would be beneficial in future to consider the effects of the specific surface area, pore volume, size distribution, and thermal stability, among others, associated with the adsorbent materials. Furthermore, while adsorption activity of this Ag25Cu75 nanocomposite was observed, it is not within the scope of this work to conclusively determine if synergy extended into synergistic effects or was purely additive on its own, since direct quantitative comparisons were not possible against the monometallic Ag and Cu based nanoparticles using this experimental approach. Thus, adsorption activity is attributed to additive surface effects between Ag and Cu. Lastly, although various Ag:Cu combinations have been synthesized in the preliminary stages, a detailed optimization study of these different Ag:Cu combinations was not conducted. The focus was on one particular composition that is concentrated in copper to facilitate a thorough mechanistic investigation. For further work, optimization studies on the composition, as well as comparisons with monometallic materials, are essential.
Conclusions
This work investigates the adsorption efficacy of a Cu-rich Ag-Cu nanocomposite, namely Ag25Cu75, for the removal of DCF and CV from aqueous systems under both the batch system and continuous-flow conditions. In this study, it was indicated that the nanocomposite provided effective removal of contaminants while the mechanisms of adsorption are distinct but well-defined, depending on contaminant type and solution chemistry.
The pH of the solution and surface charge effects greatly influenced the adsorption of DCF, while the removal of CV was dominated by non-electrostatic interactions, including π-cation and surface complexation mechanisms. These maintained effectiveness under acidic conditions. An isotherm and thermodynamic analysis revealed heterogeneous surface adsorption and spontaneous uptake for both contaminants, substantiating the role of combined surface interactions in governing adsorption performance.
Additionally, fixed bed column experiments proved that the incorporation of these Ag25Cu75 nanoparticles into the sand media is effective in contaminant removal and delaying breakthrough in nanoparticle-assisted filtration systems for water treatments. The results, although carried out in a laboratory setting, offer useful insights about the practical application of the different mechanisms of adsorption in the removal of the pharmaceutical and dye contaminants with the Ag-Cu nanoparticles composite material.
In a general context, the work contributes to the knowledge base regarding the application of mixed metal nanocomposites in the field of water treatment and establishes a base upon which future work can be conducted to optimize the material composition.
Supplemental Material
sj-docx-1-asw-10.1177_11786221261433801 – Supplemental material for Ag-Cu Bimetallic Nanoparticles for Combined Removal of Pharmaceutical and Dye Contaminants: Studies From Batch Adsorption to Packed Column Operation
Supplemental material, sj-docx-1-asw-10.1177_11786221261433801 for Ag-Cu Bimetallic Nanoparticles for Combined Removal of Pharmaceutical and Dye Contaminants: Studies From Batch Adsorption to Packed Column Operation by Saleh M. Sulaiman, Mohammed H. Al-Jabari, Naji T. Jobran and Mazen K. Nazal in Air, Soil and Water Research
Footnotes
Acknowledgements
The authors gratefully acknowledge the support provided by the Scientific Research Committee at Birzeit University. The authors also thank their colleagues Asem M. Mubarak and Muath M Rawajba from Birzeit University for their technical assistance.
Ethical Considerations
This study did not involve human participants or animals. Therefore, ethical approval was not required.
Condent to Participate
This study did not involve human participants or animals. Therefore, informed consent was not required.
Author Contributions
Conceptualization, methodology, writing-original draft preparation and study design: Saleh Sulaiman. Data curation, investigation, and formal analysis: Naji Jobran. Writing-review and editing: Mohammad Al Jabari. Supervision and project administration: Saleh Sulaiman. Analysis and Characterization: Mazen Nazal. All authors have read and approved the final manuscript and agree to be responsible for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.*
Supplemental Material
Supplemental material for this article is available online.
