Abstract
Centralized wastewater treatment plants in developing countries are often overloaded, necessitating robust decentralized alternatives. This study designed and evaluated a novel decentralized system integrating coagulation-flocculation with aluminum sulfate, a tertiary treatment unit using synthesized nano zero-valent aluminum (nZVAl) for simultaneous degradation, adsorption, and disinfection, and a sand filter. The nZVAl was characterized by XRD and SEM, confirming its composition and morphology. The optimum coagulation/flocculation conditions were observed at pH 7 and 0.5 g/L of aluminum sulfate dose. For tertiary wastewater treatment using nZVAl, the optimum operating condition was observed at pH 7.5, 0.7 g/L nZVAl dose, 40 min contact time, 200 rpm stirring rate, and 35ºC temperature. The kinetic studies indicated that the adsorption data for organic carbon (COD), biodegradable organics (BOD), nutrients (TN), and total suspended solids (TSS) were best described by the Avrami kinetic model, while phosphorus (TP) removal kinetics were best described by a pseudo-second-order model. The thermodynamic results indicated that the reaction mechanisms are exothermic for COD and TSS, and endothermic for BOD, TN, and TP. The results of ANN showed that nZVAl dose is the most effective operating conditions followed by contact time. The decentralized wastewater treatment system was tested under the optimum conditions, and the results demonstrated high efficiency, with removal rates exceeding 92% for COD, BOD, TSS, and oil and grease. High removal efficiencies were also reported for TP, TN, nickel (Ni), and copper (Cu). The treated effluent complied with all limits stipulated by the Egyptian code 501/2015, confirming its suitability for reuse in agricultural applications.
Keywords
Introduction
Developing countries face significant challenges in managing increasing amounts of wastewater, often due to population growth, rapid urbanization, and limited infrastructure (Lema, 2025; Onu et al., 2023; Qadir et al., 2010 ). Most of the centralized wastewater treatment plants in developing countries suffer from high pressure due to the large amounts of wastewater being directed to the plants, which may exceed their design capacity (Onu et al., 2023; Saadatinavaz et al., 2024). Decentralized wastewater treatment systems (DEWATS) are a solution to treat and manage wastewater on a smaller, localized scale, as opposed to centralized systems that transport wastewater to a central facility (Rahman et al., 2024; Swan et al., 2023). This approach can be used for universities or individual households. Decentralized systems offer various benefits, including the ability to tailor treatment methods to specific needs, potential cost savings, and reduced reliance on large infrastructure (Garrido-Baserba et al., 2024; Rabaey et al., 2020; Ventura et al., 2024).
Researchers have developed a decentralized system with a capacity of 0.215 m3/day that consists of a baffled septic tank integrated with an up-flow anaerobic filter (BASTAF) for treating real gray and black wastewater with high nutrients and organic content. The filter media consists of charcoal and PVC pipe cuttings. The system achieved removal efficiencies of 71.47% for biological oxygen demand (BOD) and 74.85% for chemical oxygen demand (COD) (Ventura et al. 2024). The main disadvantages of the BASTAF system are high operation and maintenance costs, as well as the high risk of sewer clogging (Ventura et al., 2024). Researchers have built a decentralized anaerobic baffled reactor (ABR) system with a capacity of 0.64 m3/day, comprising a conventional two-chamber septic tank followed by anaerobic filters. The system has achieved a BOD, COD, and total nitrogen (TN) removal efficiency of 90%, 90%, and 60%, respectively (Ventura et al. 2024). The main disadvantage of the system is the high variability in performance (Oakley et al., 2010; Rout et al., 2021). X. M. Yang et al. (2010) built a decentralized Johkasou system consisting of sedimentation, anaerobic, aeration, and disinfection units. Microorganisms were used to degrade organic contaminants. The Johkasou system has achieved a COD, TN, and TP removal efficiency of 94%, 52%, and 70%, respectively (X. M. Yang et al., 2010). The main disadvantage of the Johkasou system is that it requires regular desludging and maintenance (X. M. Yang et al., 2010). Shehabi et al. (2012) developed a community-scale DEWATS (20 m³/day capacity) with a treatment train consisting of screening, grit removal, primary sedimentation, activated sludge, secondary clarification, and UV disinfection (Shehabi et al., 2012). The system has achieved COD, BOD, TN, TP, NH4+, Nitrate (NO3-), Phosphate (PO43-), coliform removal efficiency of 95%, 99%, 86%, 88%, 99%, 83%, 94%, and 100%, respectively (Shehabi et al., 2012). Tanner et al. (2012) built a decentralized circulating fluidized bed bioreactor (CFBBR) with a capacity of 5.3 m3/day, which integrates wetlands and denitrifying bioreactors. The system has achieved COD, BOD, and TP removal efficiency of 81%, 94%, and 94%, respectively (Tanner et al., 2012). Blackett et al. (2013) have presented a decentralized passive anaerobic system with a capacity of 6.2 m3/day. The system has achieved COD removal efficiency of 54.8% (World Bank, 2014). Abou-Elela et al. (2017) has successfully constructed a novel pilot-scale passively aerated biological filter (PABF) with a capacity of 5.55 m3/day. The system has achieved COD, BOD, TN, TP, and NH4+ removal efficiency of 90%, 90%, 20%, 20%, and 20%, respectively (Abou-Elela et al., 2017). Fox and Clifford (2018) have tested a sequencing batch reactor (SBR) with a capacity of 0.9 m3/day and having treatment steps of aeration, mixing, and settling all occur within the same vessel. The system has achieved COD, TN, NH4+, and NO3- removal efficiency of >96%, 80%, 100%, and 80%, respectively (Fox & Clifford, 2018). Ranjan et al. (2019) have built a laboratory scale upflow aerobic fixed bed reactor (UAFBR) system with a capacity of 10 m3/day to treat the sewage of Vellore Institute of Technology (VIT) campus to be used for irrigation. The system consists of coagulation, anaerobic digestion, biofilm aerobic, and disinfection (using sunlight and chlorine) processes (Ranjan et al., 2019). The system has achieved COD, TN, TP, and NH4+ removal efficiency of 82%, 93%, 94.6%, and 95.7%, respectively (Ranjan et al., 2019). A decentralized membrane-less microbial fuel cell (MFC) was constructed by Feng et al. (2013). It includes three rectangular-chambered MFC bioreactors (clarifier, cathode, and anode), a composite cathode that contains iron phthalocyanine (FePc) and carbon nanoparticles, carbon cloth electrodes, and immobilized cells in the cathodic chamber. The system's primary drawbacks are its lower power density in comparison to other MFC systems and its sensitivity to high concentrations of hazardous metals like copper (Feng et al., 2013). The system has achieved COD, TN, and NH4+ removal efficiency of 92%, 82%, and 97%, respectively (Feng et al., 2013).
In this research, nano materials were used for degradation, adsorption, and disinfection of contaminants found in wastewater. Nanomaterials, particularly zero-valent metals, have been widely applied for the removal of both organic and inorganic contaminants from wastewater, owing to their high reactivity and extensive surface area. Several studies have been conducted for wastewater treatment using magnetic and nonmagnetic zero-valent metals. While zero-valent iron (nZVI) has been the most extensively documented for its reductive capabilities, zero-valent aluminum (nZVAl) offers a higher reduction potential (E°(Al³+/Al) = −1.66 V vs. SHE) compared to iron (E°(Fe²+/Fe) = −0.44 V), suggesting a potentially greater driving force for the degradation of recalcitrant contaminants. This work explores this advantage within a practical treatment train. The treatment process focused on organic matter removals such as color, poly aromatic hydrocarbons (PAHs), COD, total organic carbons (TOC), and mono aromatic compounds (BTEX) compounds by adsorption and degradation process by using nano zero-valent iron and its derivatives (Farag et al., 2018; A. S. Mahmoud et al., 2019). Nanomagnetic sorbents proved the availability for different inorganic contaminants removal (Harter, 2003; A. S. Mahmoud, Mohamed, et al., 2021; M. S. Mahmoud & Mahmoud, 2021). Mostafa et al. (2022) employed a composite of cellulose acetate polymer and nano-zero-valent iron (CA-nZVI) to achieve biological oxygen demand (BOD) removal from municipal wastewater sourced from a treatment facility in New Cairo, Egypt. Under optimized conditions—pH 7, a contact time of 30 min, a stirring rate of 200 rpm, an adsorbent dosage of 3 g/L, and an initial BOD concentration of 100 mg/L—the system attained a removal efficiency of 96.4% (Mostafa et al., 2022). A. S. Mahmoud et al. (2022) utilized alginate (Ag)-entrapped nano-zero-valent iron (nZVI) for the removal of chemical oxygen demand (COD) from aqueous solutions. The study achieved a maximum COD removal efficiency of 76% under the following optimal conditions: pH 6, a contact time of 30 min, a stirring rate of 150 rpm, an adsorbent wet dosage of 3 g/L, and an initial COD concentration of 400 mg/L. The entrapment of nZVI within the alginate biopolymer matrix served to enhance the stability and dispersibility of the nanoparticles while mitigating rapid oxidation and aggregation, thereby improving treatment efficacy (A. S. Mahmoud et al., 2022).
A. S. Mahmoud et al. (2019) investigated the adsorption of soluble organic matter from municipal wastewater using chemically synthesized nano zero-valent aluminum (nZVAl). Their findings demonstrated the efficacy of this nonmagnetic nanomaterial in adsorbing diverse soluble organic contaminants, achieving a chemical oxygen demand (COD) removal efficiency of 96%. This high removal rate was attained under optimized conditions: an initial COD concentration of 100 ± 11.8 mg/L, pH 8, a stirring rate of 100 rpm, a contact time of 10 min, and an nZVAl dosage of 0.6 g/L. The study underscores the potential of nZVAl as a highly efficient adsorbent for rapid organic pollutant removal in wastewater treatment applications (A. S. Mahmoud et al., 2019).
In this study, the nZVAl was selected as a core adsorptive and disinfection agent due to its high surface area, as well as its ability to remove a wide range of contaminants simultaneously (Nidheesh et al., 2018). Based on previous studies, nZVAl can effectively adsorb and reduce nutrients, heavy metals, and organic matter from wastewater, outperforming coagulants and conventional adsorbents due to its enhanced reactivity (A. S. Mahmoud et al., 2019; Sadek & Mostafa, 2022; Sadek et al., 2023). In addition, nZVAl also generates reactive species and destabilizes microbial cell membranes, which facilitates disinfection and improves the overall treatment performance (S. Yang et al., 2017). These advantages make nZVAl a promising material for decentralized wastewater systems where high removal efficiency must be attained within a limited footprint.
The artificial neural network (ANN) was helpful in this study due to the non-linear and complex interactions between the different operating parameters (e.g., contact time, pH, dose, stirring rate, and temperature) that govern nZVAl removal performance. Where, the non-linear dynamics between variables are often difficult to be captured by traditional statistical models. ANNs, however, can predict system performance by learning from experimental data and then identify the most significant variables (Dantas et al., 2023; Palma et al., 2025). By using ANN tool, the authors were able to reduce experimental expenses, optimize operating conditions, which facilitate scaling up of the proposed treatment system.
This study attempts to examine the efficiency of a decentralized wastewater treatment plant that has been constructed with a capacity between 80 and 100 m3/day on the campus of Badr University in Cairo, Egypt. The plant consists of three main stages: (1) coagulation/flocculation using aluminum sulfate, (2) degradation, adsorption, and disinfection using nZVAl, and (3) filtration using the sand filter. To the best of our knowledge, this is the first decentralized wastewater treatment plant that applies nanotechnology on a large scale for the treatment of municipal wastewater. The effect of pH and coagulant doses was studied by batch techniques. The effects of operating conditions were conducted to achieve the optimum operating conditions of the nano unit from pH, dose, contact time, stirring rate, and temperature. Kinetic and thermodynamic studies were performed to understand the removal mechanisms. artificial neural networks (ANNs) were conducted to generate nonlinear statistical forms including the statistics of important variables.
Material and Methods
Chemicals and Reagents
The following analytical-grade chemicals were employed without further purification: aluminum sulfate octadecahydrate (Al2(SO4)3·18H2O, extra pure, Loba Chemie), sodium borohydride (NaBH4, ⩾99%, Winlab), absolute ethanol (C2H6O, ⩾99%, Chem Lab), sodium hydroxide (NaOH, ⩾99%, Oxford Company), and sulfuric acid (H2SO4, 96%, Fisher Chemical). All solutions were prepared using deionized water (18.2 MΩ·cm resistivity).
Preparation of Nano Zerovalent Aluminum
Equation 1 describes the formation of aluminum powder after reducing it from its salt. About 13.69 g of Al2(SO4)3.18H2O powder was added into 100 mL Ethanol 50%. About 9.15 g of NaBH4 was dissolved in 1 L of 10% Ethanol.
The NaBH4 solution was transferred to a burette and added dropwise into the aluminum salt solution at a controlled rate of 1.5 mL/min under constant stirring. The formation of a aluminum precipitate was observed immediately following the addition of approximately the first 10 drops of the reducing agent. Upon complete addition, the resulting suspension was vacuum-filtered using a Buchner funnel and Whatman No. 1 filter paper. The solid product was subsequently washed three times with absolute ethanol to remove residual salts and byproducts, followed by drying under vacuum at 60 °C for 12 hr to obtain the nano zero-valent aluminum (nZVAl) powder (A. S. Mahmoud et al., 2019).
Storage and Handling of nZVAl
Due to the high reactivity and pyrophoric nature of nano zero-valent aluminum (nZVAl), which is analogous to the well-documented behavior of nano zero-valent iron (nZVI), stringent storage and handling procedures were followed to prevent passivation and oxidation before use (Stefaniuk et al., 2016). Immediately after synthesis, filtration, and drying, the nZVAl powder was stored in a vacuum desiccator under an inert atmosphere (N2) to minimize exposure to moisture and oxygen. For all batch experiments, the nZVAl was weighed in a controlled environment and transferred quickly to the reaction vessels containing the wastewater sample to initiate the reaction without significant pre-oxidation. This protocol ensured the preservation of the material's reactivity and the consistency of its performance across all experiments (A. S. Mahmoud et al., 2019).
Characterization of the Prepared nZVAl
The crystalline structure of the synthesized nZVAl nanoparticles was characterized using X-ray powder diffraction (XRD). Analysis was performed using a PANalytical X'Pert PRO MRD diffractometer (Malvern Panalytical, The Netherlands) equipped with a Cu Kα radiation source (λ = 1.5406 Å). Measurements were conducted at an operating voltage of 40 kV and a current of 30 mA. The powdered sample was packed into a stainless-steel sample holder and scanned over a 2θ range of 35° to 90° with a step size of 0.02° and a dwelling time of 1.0 s per step.
The morphological characteristics of the nZVAl nanoparticles were examined using a scanning electron microscope (SEM; Philips Quanta 250 FEG, USA) operated at an accelerating voltage of 25 kV. Samples were prepared by dispersing the nanoparticles onto a carbon tape-supported aluminum stub followed by gold sputtering to enhance conductivity. Micrographs were acquired at a magnification of 80,000× to resolve detailed surface and structural features.
Batch Studies
The batch experiments were conducted in two consecutive steps to evaluate the treatment efficiency of the proposed system:
Step 1: Coagulation/Flocculation
In the first step, aluminum sulfate (Al2(SO4)3) was used as a coagulant. A standard jar test was conducted by adding a known doses of Al2(SO4)3 to 1,000 mL of raw wastewater. Rapid mixing was performed for 3 min at 150 rpm to disperse the coagulant, followed by slow mixing for 20 min at 30 rpm to promote floc formation. The formed flocs were then allowed to settle at zero velocity until complete precipitation occurred. The supernatant obtained from this step was used as partially treated wastewater.
Step 2: Adsorption and Degradation using nZVAl
Following complete precipitation, the adsorption and degradation performance of the nZVAl was evaluated using a batch experimental approach, in which one operational parameter was varied at a time while keeping others constant. The selection of operational conditions and their respective ranges including pH, nZVAl dosage, contact time, stirring rate, and temperature was informed by established values reported in prior literature to ensure comparability and relevance to existing research (Farag et al., 2019; M. S. Mahmoud & Mahmoud, 2021). Table 1 summarizes the experimental matrix for the batch studies, detailing the ranges of operating conditions investigated: pH, nZVAl dose (g/L), contact time (min), stirring rate (rpm), and temperature (°C). In a representative experiment, a predetermined mass of 0.7 g of nZVAl was added to 1,000 mL of the pre-treated wastewater sample in an Erlenmeyer flask. The mixture was agitated at a controlled speed and temperature for a specified duration to reach equilibrium. Subsequently, the nZVAl particles were separated from the treated water via filtration using Whatman No. 2 filter paper. The filtrate was then analyzed for residual concentrations of chemical oxygen demand (COD), biological oxygen demand (BOD), total suspended solids (TSS), total nitrogen (TN), and total phosphorus (TP) in accordance with Standard Methods for the Examination of Water and Wastewater (23rd edition, Rice et al., 2017). All experiments were conducted in triplicate to ensure reproducibility, and mean values are reported (Rice & Baird, 2017).
Batch Experiments at Different Operating Parameters.

The removal efficiency for each contaminant was calculated using Equation 2:
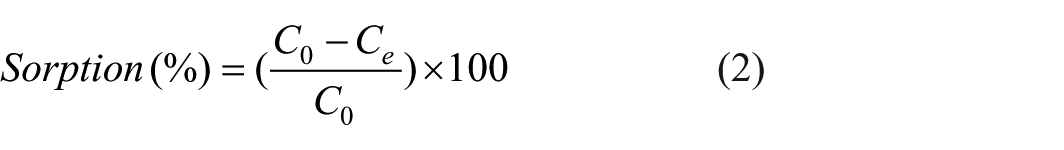
where C0 is the initial concentration (mg/L) and Ce is the equilibrium concentration in solution (mg/L) of the contaminant.
The adsorption capacity at equilibrium, representing the amount of contaminant adsorbed per unit mass of nZVAl, was determined using Equation 3:

where Qe is the equilibrium adsorption capacity (mg/g), V is the volume of aqueous solution (L), and m is the dry weight of the adsorbent (mg).
All experiments were conducted in triplicate, and the results are presented as mean values ± standard deviation. The relative standard deviation among replicates was less than 5%, confirming the high reproducibility and reliability of the experimental data.
System Preparation
A decentralized system was designed, consisting of equalization tanks, coagulation/flocculation (using a pipe flocculator), a sedimentation tank, an adsorption tank (nZVAl), and a filtration system (sand filter). The equalization tank was used for pH adjustment. The coagulation-flocculation process was designed to support fixed and variable stirring processes. Nanotechnology was recently used to adsorb and degrade a wide range of wastewater contaminants including bacteria and viruses to fulfill Egypt’s law criteria for tertiary wastewater treatment. Finally, a sand filter unit was designed to eliminate the residual suspended contaminants and the residual nanoparticles. Also, the designed wastewater treatment system supports gravity continuous flow. Figure 1 shows the scheme of the designed decentralized wastewater treatment system. The system was designed, constructed, and tested in the period from January 2023 to September 2024.

The designed decentralized wastewater treatment system.
Samples Collection
Raw wastewaters and treated water samples were collected from the influent and the effluent of the decentralized wastewater treatment plant at Badr University in Cairo Campus, Cairo, Egypt. Twelve wastewater composite samples and 12 treated water composite samples were collected. The samples were collected in 25 L in non-modified plastic bottles for testing and treatment techniques. All laboratory analyses were conducted in an ISO/IEC 17025:2017 accredited chemistry laboratory specializing in water and wastewater examination at the Housing and Building National Research Centre (HBRC). Analyses were performed in strict accordance with Standard Methods for the Examination of Water and Wastewater (23rd edition, Rice et al., 2017). This accreditation ensures that all analytical procedures—including quality control, instrument calibration, and data reporting—adhered to internationally recognized standards of precision, accuracy, and methodological rigor.
Kinetic Studies and Validation
The kinetic mechanisms for the removal of COD, BOD, TSS, TN, and TP by nZVAl particles were analyzed using nonlinear forms of several adsorption kinetic models. These included the pseudo-first-order (PFO), pseudo-second-order (PSO), Avrami fractional, Elovich, and intraparticle diffusion models. To objectively determine the best-fitting model, two error functions were employed: the sum of the squares of the errors (ERRSQ) and the Chi-square error (χ²), as defined in Equations 4 and 5, respectively (Serafin & Dziejarski, 2023; Sipos, 2021).


Where
Thermodynamic Analysis
To fundamentally characterize the adsorption interactions between the nZVAl particles and the contaminants (COD, BOD, TSS, TN, TP), key thermodynamic parameters namely the Gibbs free energy change (ΔG°), enthalpy change (ΔH°), and entropy change (ΔS°) were rigorously determined. These parameters provide critical insight into spontaneity, thermal nature, and structural changes occurring at the solid-solution interface. The thermodynamic parameters were determined by calculating the distribution coefficient, Kd (L/g), which is a fundamental measure of the adsorbent's affinity for the adsorbate. It is defined as Equations 6 to 9.
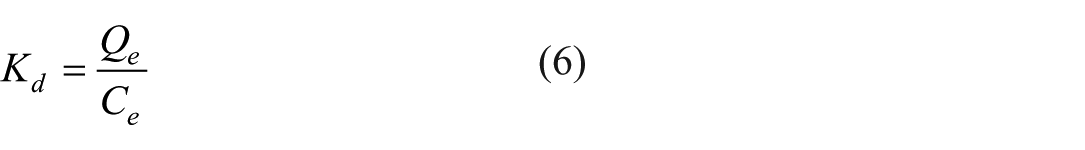
where Qe is the amount adsorbed at equilibrium (mg/g) and Ce is the equilibrium concentration in solution (mg/L). The value of Kd is related to the change in standard Gibbs free energy (∆G°) by the following equation:

By substituting the expression for ∆G° in terms of enthalpy (∆H°) and entropy (∆S°):

The two equations can be combined:


Therefore, by plotting ln(Kd) on the y-axis against 1/T on the x-axis, the slope of the resulting line is equal to −∆H°/R and the intercept is ∆S°/R. These values are then used to calculate ∆G° at different temperatures using Equation 8. This approach allows us to determine whether the adsorption process is spontaneous (∆G° <0), exothermic (∆H° <0), or endothermic (∆H° >0), and whether there is an increase or decrease in randomness at the solid-solution interface (∆S°) (Mittal et al., 2010; Sadaf et al., 2015).
Artificial Neural Network (ANN) Modeling
Two artificial neural network (ANN) models were developed using a multilayer perceptron (MLP) architecture to model the nonlinear relationships between the input operational parameters and the treatment efficiency outputs. The input variables consisted of pH, nZVAl dose (g/L), contact time (min), stirring rate (rpm), and temperature (°C). The output layer represented the removal efficiencies of chemical oxygen demand (COD), biological oxygen demand (BOD), total suspended solids (TSS), total nitrogen (TN), and total phosphorus (TP).
A single hidden layer was incorporated to capture complex nonlinear interactions within the data. The hyperbolic tangent activation function was applied in the hidden layer to introduce nonlinear transformation capabilities, while a linear activation function was used for the output layer. The network was trained using the sum of squared errors (SSE) as the loss function to quantify the deviation between predicted and experimental values.
The models were constructed and validated without exclusion of any data points to ensure robustness. Sensitivity analysis was performed to evaluate the relative and normalized importance of each input variable on the predicted removal efficiencies, providing insight into the dominant factors influencing system performance.
The structure and dataflow of the ANN model developed for predicting nZVAl-assisted wastewater treatment performance are illustrated in Figure 2.
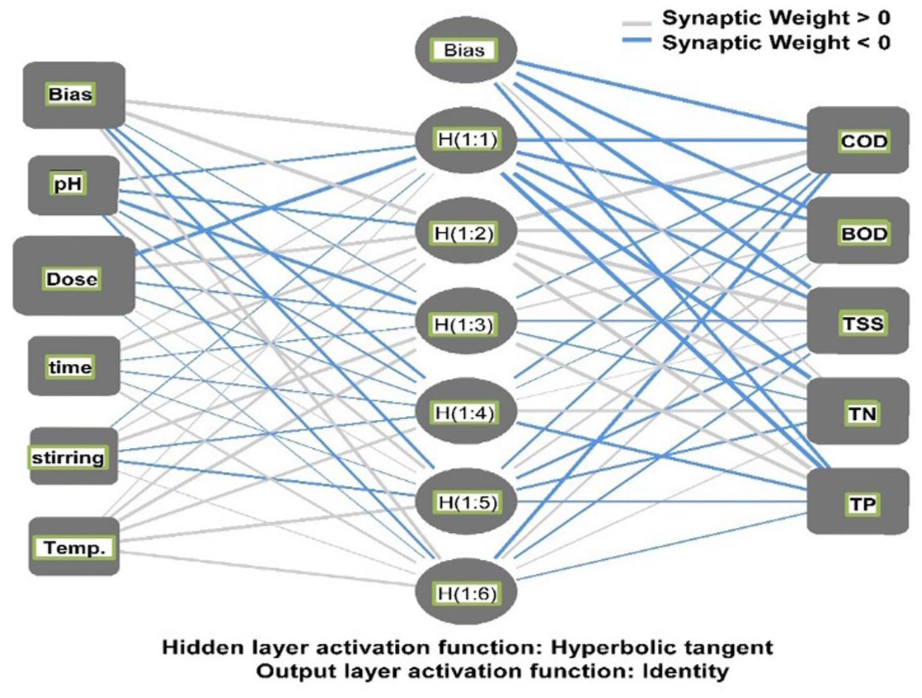
Artificial neural networks for wastewater treatment using nZVAl.
Results and Discussions
Characterization of nZVAl
Figure 3A shows the XRD pattern of nano zero-valent aluminum with a specific angle (2θ) from 35° to 90°. The XRD pattern shows five peaks at (2θ) equal 38.57°, 44.95°, 65.27°, 78.68°, and 82.92°, indicating the formation of pure referenced aluminum nanoparticles with reference number (96-151-2489). Similar findings were previously observed in studies by (A. S. Mahmoud et al., 2019). Figures 3B shows the SEM image of freshly prepared ZVAl nanoparticles. The results indicated the presence of an irregular surface structure, as well as the formation of aluminum nanoparticles with particle sizes ranging between 30 and 60 nm. The SEM image demonstrated that the prepared aluminum nanoparticles contain many pores that allow easier mass transfer and molecular diffusion inside the nanomaterial. The characterization results (XRD and SEM) confirmed that the nZVAl material synthesized for this study exhibited the same crystalline structure and morphological properties (e.g., particle size range of 30–60 nm) as previously reported and extensively characterized in our earlier works (A. S. Mahmoud et al., 2019; Sadek et al., 2023), where comprehensive analyses including XPS, BET, and EDS were presented. The focus of the current study is on the application and performance of this material within a novel multi-stage treatment system.
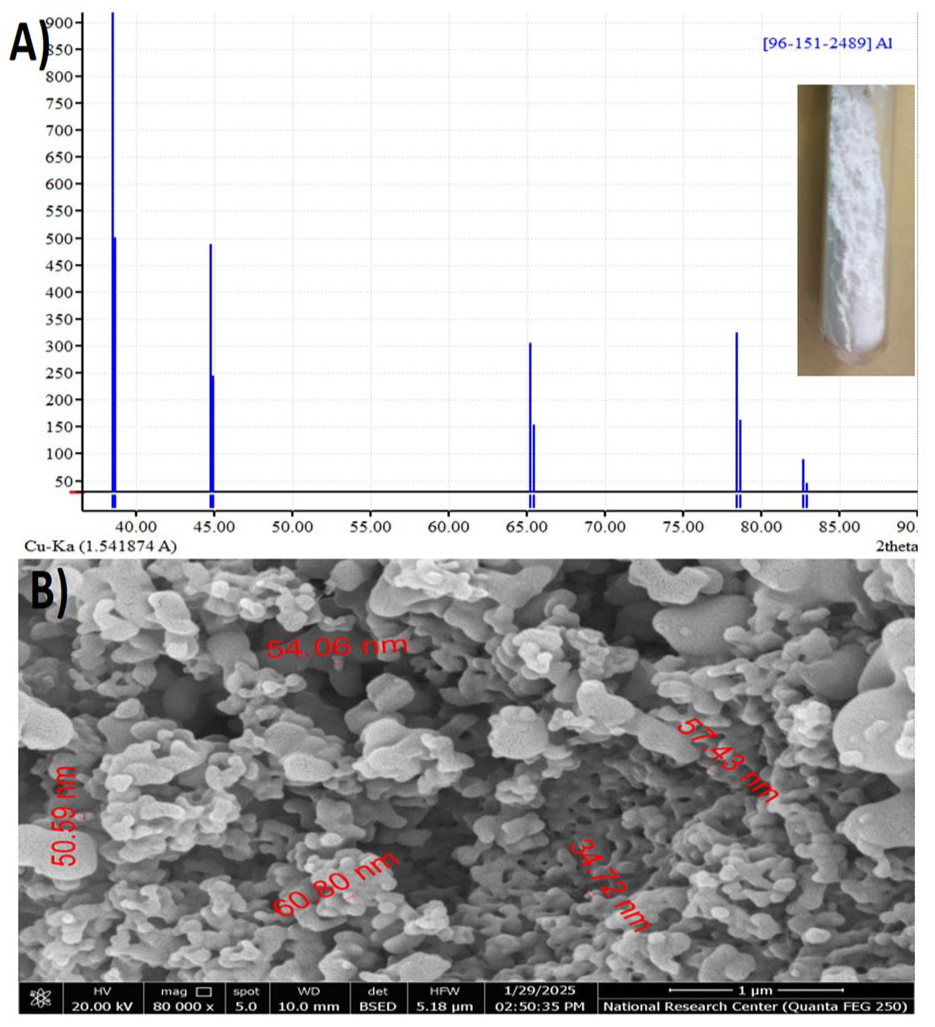
Characterization of nZVAl: (A) XRD of the freshly prepared nZVAl and (B) SEM of nZVAl.
The morphological and structural properties revealed by SEM and XRD are directly linked to the material's performance. The irregular surface structure and the presence of pores (as seen in SEM) provide a high surface area and numerous sites for the physical adsorption of contaminants. Furthermore, the confirmed presence of the zero-valent Al0 core (via XRD) is essential for the reductive degradation of pollutants, as it acts as a potent electron donor. The thin native oxide layer (Al2O3/Al(OH)3), while passivating, also provides a surface for complexation and chemisorption processes. Thus, the high removal efficiency observed in this study is a direct result of this synergistic combination of a porous adsorbent structure and a reactive metallic core.
Chemical Coagulation Using Aluminum Sulfate
Effect of pH
The effect of pH was studied at different pH ranging between 3 and 10 to optimize the suitable media for the coagulation process. The selection of suitable pH depends on two main factors, one of them the quantity and speed of flocs formation and the other the best quality of organic and inorganic removals. Figure 4A describes the variation in the removal efficiency of COD, BOD, and TSS during pH changes at constant aluminum sulfate (Al2(SO4)3) dosage of 0.5 g/L. These types of contaminants are responsible for the presence of carbon contamination in wastewater. The result indicated that the optimum pH was 7 with the removal efficiency for COD, BOD, and TSS were 65.6%, 63.6%, and 63.6%, respectively. The removal efficiency also occurred in acidic and alkaline media due to the formation of the floc but with less removal efficiency. Additionally, the high removal efficiency in the acidic media is mainly due to the high amount of TSS dissolved in that media. Finally, increasing the mass of biological growth in the alkaline media, and the non-adapted acidic and alkaline media may affect also BOD removal in that case. Although the primary metric for optimization was contaminant removal efficiency, qualitative observations indicated that the fastest sedimentation rate and most compact sludge volume occurred at the optimum pH of 7, corresponding with the highest removal efficiencies. The slowest settling rates and largest sludge volumes were observed at the pH values (pH 3 and pH 10). Figure 4B shows that the optimum pH was 7 with the removal efficiency for TN, TP, and sulfide were 58.2%, 38.3%, and 64.3%, respectively. Figure 4C shows the stability of phenol removal for different pH with 100% removal due to the presence of low phenol concentrations in wastewater (0.002 mg/L). The optimum pH was 7 with oil and cyanide removal efficiencies of 91.7% and 69.6%, respectively. Figure 4D shows that the iron removal efficiency is higher in alkaline media than in acidic media due to the solubility of some soluble iron suspension in the acidic media. Finally, the optimum zinc removal was reported in the neutral media. These results agree with the previous results for organic and inorganic matter removal using alum (Bachand et al., 2019; Georgantas & Grigoropoulou, 2005; Lin et al., 2017).

Effect of pH after Aluminum sulfate treatment on: (A) COD, BOD, and TSS, (B) TN, TP, and sulfide, (C) phenol, CN, and oil, and (D) Fe and Zn.
The observed removal efficiencies of contaminants using nZVAl can be further interpreted through the lens of aluminum Pourbaix diagrams as shown Supplemental Figure 1, which illustrates the thermodynamic stability of aluminum species as a function of pH and electrochemical potential. Under near-neutral to slightly alkaline conditions (pH 7–8), aluminum tends to form stable passive oxide layers (e.g., Al2O3 or Al(OH)3), which are critical for its role as an adsorbent and reactive material. At lower pH values (<6), aluminum may undergo dissolution, releasing Al³+ ions, which can contribute to coagulation but also reduce the available surface area and reactive sites of nZVAl particles. Conversely, at higher pH values (>9), aluminum may form soluble aluminate ions (Al(OH)4-), diminishing its effectiveness for adsorption and degradation. The optimal pH range observed in this study (pH 7.5) aligns with the region where aluminum exhibits minimal solubility and maximal surface reactivity, facilitating both adsorption and redox-mediated degradation processes. This electrochemical behavior underscores the importance of pH control in maximizing the efficacy of nZVAl-based treatment systems (S. Yang et al., 2017).
Effect of Coagulant Dose
Coagulant dosage is one of the most crucial considerations when evaluating the effectiveness of coagulants in coagulation and flocculation. Furthermore, excessive or inadequate coagulant dose would result in inefficient process performance (Getahun, Asaithambi, et al., 2024). Choosing the optimal dosage is critical for reducing coagulant costs and sludge formation while achieving maximal treatment efficiency (Getahun, Asaithambi, et al., 2024). Tests were conducted to investigate the effects of Al2(SO4)3 doses ranging from 0.1 to 0.8 g/L on the removal efficiency of all tested parameters. Figure 5 shows that the removal efficiency of all tested parameters increased with the increase in the coagulant dosage. The optimum Al2(SO4)3 dosage was reported as 0.5 g/L achieving COD, BOD, total suspended solids (TSS), total phosphorous (TP), total nitrogen (TN), iron (Fe), zinc (Zn), phenol, oil, sulfide, and cyanide (CN) removal efficiencies of 65.6%, 63.6%, 63.6%, 38.3%, 58.2%, 48.2%, 62.1%, 100%, 69.6%, 64.3%, and 91.7%, respectively. (Mostafa & Peters, 2016) have tested four different coagulants for the removal of COD, BOD, TSS, and turbidity from wastewater and the results showed similar behavior as reported in this study, where the removal efficiencies increased with the increase in the coagulants dosages. The low removal efficiency at low coagulant dosage is mainly attributed to the insufficient quantity of coagulant to destabilize the contaminants (Getahun, Asaithambi, et al., 2024). The direct relationship between the removal efficiency of all studied parameters and the coagulant dosage could be linked to the inter-particle bridging and charge neutralization mechanism caused by the polycationic character of the coagulant dosage (Getahun, Befekadu, & Alemayehu, 2024).

Effect of aluminum sulfate dose on the removal efficiency of: (A) COD, BOD, and TSS, (B) TN, TP, and sulfide, (C) phenol, CN, and oil, and (D) Fe and Zn.
Nano Treatment
Effect of pH
The effect of pH was studied in the range of 6 to 9.5 at a contact time of 40 min, a stirring rate of 200 rpm, a nZVAl dosage of 0.7 g/L, and a temperature of 35 degrees Celsius on the removal efficiency of all tested parameters. As shown in Figure 6A, the optimum pH was reported as 7.5 at COD, BOD, TSS, TN, and TP removal efficiencies of 76.2%, 77.6%, 75.9%, 34.1%, and 37.6%, respectively. The optimal performance at pH 7.5 can be directly explained by the material’s characterization. This pH is near the point of zero charge (PZC) for acid-washed ZVAl (reported range 7.2–8.47), which minimizes electrostatic repulsion and favors physical adsorption (A. S. Mahmoud et al., 2019). Furthermore, within this pH range, the native oxide layer on the nZVAl (as indicated by its composition) is stable, protecting the reactive Al0 core from rapid passivation while still allowing for controlled electron transfer for reductive degradation. At lower pH, the characterized metallic core would be consumed by rapid reaction with H+ ions, dissolving the particle. At higher pH, the formation of soluble aluminate species would compromise the adsorbent’s structure, both outcomes leading to the observed decrease in efficiency (Ahmed et al., 2024). High alkaline solutions with abundant OH− ions affect the adsorption activities of nZV metal and decrease the chemisorption of charged negative molecules due to the steric influence of negative charges (Bao et al., 2024; A. S. Mahmoud, Mostafa, & Peters, 2021; Pan et al., 2025; Saha et al., 2001). Different studies have been conducted using different sorbent materials and showed that the optimum pH ranged from 7.5 to 8, which agrees with the results of this study. A. S. Mahmoud et al. (2019) have used nZVAl for the removal of COD and the results showed that the optimum pH is 8.

The effect of nZVAl on COD, BOD, TSS, TN, and TP removal efficiencies at the operating conditions: (A) pH, (B) nZVAl dose, (C) contact time, (D) stirring rate, and (E) temperature.
Effect of nZVAl Dose
The effect of nZVAl dose was studied in the range of 6 to 9.5 g/L at an initial pH of 7.5, contact time of 40 min, stirring rate of 200 rpm, and temperature of 35°C on the removal efficiency of all tested parameters. As shown in Figure 6B, the removal efficiencies for all studied parameters were increased with the increase in the nZVAl dosage. For instance, when nZVAl dosage increased from 0.1 to 1.0 g/L, the removal efficiency increased from 21.1% to 91.1% for BOD, from 20.6% to 93.7% for COD, from 17.7% to 89.9% for TSS, from 8.3% to 55.3% for TN, and from 12.5% to 51.3% for TP. This is mostly due to an increase in the number of empty sites with increased nZVAl dose, which traps considerable amounts of molecular pollutants. Similar behavior was observed for Cu(II) ions adsorption using nZVAl, where the removal efficiency increased from 27.9% ± 1.3% to 53.2% ± 2.4% when the nZVAl dosage increased from 0.25 to 1.0 g/L (Sadek et al., 2023). Sadek & Mostafa (2022) used nZVAl for the removal of methylene blue (MB) and the results showed that increasing the nZVAl dosage from 0.1 to 1.0 g/L has led to improving the removal efficiency from 58.09% to 97.38%. It is important to note that the adsorption performance was evaluated using the pre-treated wastewater after coagulation, which had a consistent and reduced pollutant load. Therefore, to allow for comparison with other studies, the maximum observed removal capacity at the optimum dose (0.7 g/L nZVAl) can be reported. The capacities were calculated to be approximately 137.1 mg COD/g nZVAl and 84.3 mg BOD/g nZVAl. This demonstrates a high adsorption capacity relative to many other nanomaterials reported for treating complex, real wastewater matrices, underscoring the effectiveness of nZVAl as a polishing adsorbent in a multi-stage treatment.
The continuous increase in removal efficiency with higher nZVAl dose is a direct function of the material's properties. Each increment in dose provides more of the characterized reactive surface sites (from SEM) and a greater overall reductive capacity (from the Al0 core confirmed by XRD), allowing for the removal of a larger quantity of contaminants through combined adsorption and degradation.
Effect of Contact Time
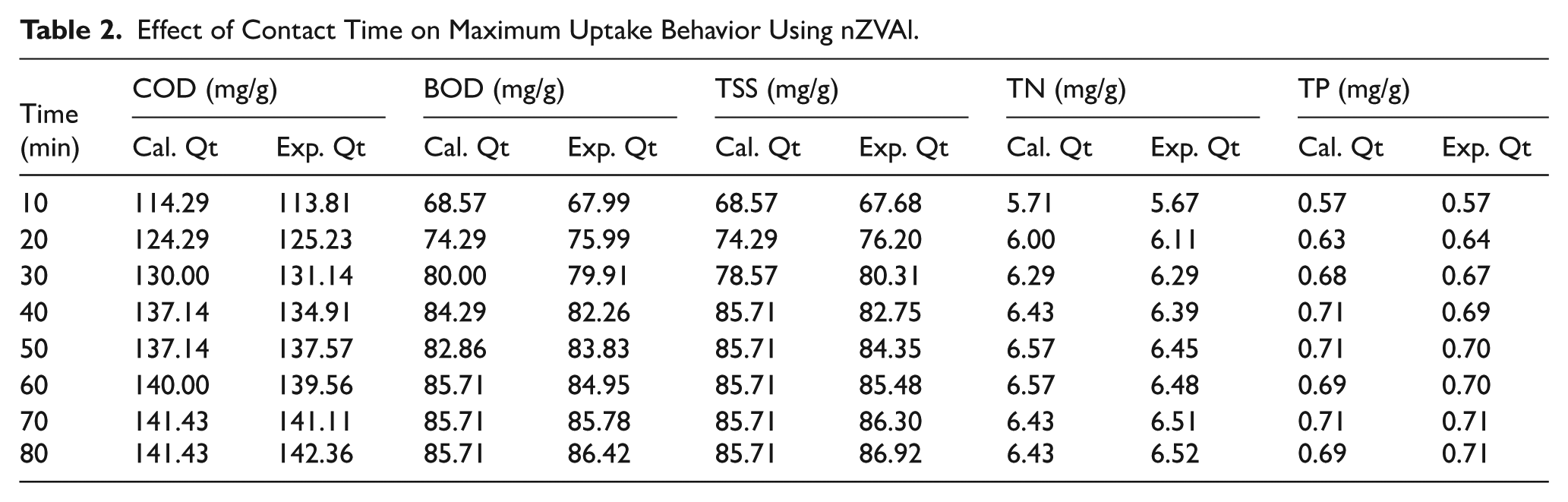
The effect of contact time was studied in the period from 10 to 80 min at an initial pH of 7.5, a nZVAl dosage of 0.7 g/L, stirring rate of 200 rpm, and temperature of 35°C on the removal efficiency of all tested parameters. The influence of equilibrated time is critical in describing the reaction mechanism between sorbed and adsorbed molecules. As shown in Figure 6C, the minimum effective time for optimal removal efficiency was around 40 min. At a contact time of 40 min, the removal efficiency of COD, BOD, TSS, TN, and TP reached 76.2%, 77.6%, 75.9%, 34.1%, and 37.6%, respectively. Table 2 shows the uptake behavior of different studied contaminants using nZVAl. The results confirm that the equilibrium time for the removal of all studied contaminants was 40 min. Mostafa et al. (2022) studied the removal of organic pollutants using nanozero-valent iron encapsulated into cellulose acetate (CA/nZVI), and the effective time for the BOD removal was 46.2 min at pH 7.3 and CA/nZVI dosage of 3 g/L; the removal percentages were nearly 80% (Mostafa et al., 2022). In general, literature findings indicated that the best period for COD and BOD removal using magnetite NPs is between 20 and 120 min, and our results are consistent with most studies.
Effect of Contact Time on Maximum Uptake Behavior Using nZVAl.
Effect of Stirring Rate
The effect of stirring rate was studied in the range from 100 to 400 rpm at an initial pH of 7.5, a nZVAl dosage of 0.7 g/L, contact time of 40 min, and temperature of 35°C on the removal efficiency of all tested parameters. As shown in Figure 6D, the optimum removal efficiencies for all studied parameters were reported at a stirring rate of 200 rpm: 76.2% for COD, 77.6% for BOD, 75.9% for TSS, 34.1% for TN, and 43% for TP. At a stirring rate of 200 rpm, there is a high chance that the contaminants come in contact with ZVAl nanoparticles and thus improved the liquid solid mass transfer. A. S. Mahmoud et al. (2020) studied the removal of COD and BOD from municipal wastewater using green synthesis nano zero valent iron (GT-nZVI), and the effective stirring rate for BOD and COD removal was 200 rpm at pH 8 and GT-nZVI dosage of 3.2 g/L; the removal percentage was 87.9% for COD and 91.3% for BOD (A. S. Mahmoud et al., 2020).
Effect of Temperature
The effect of temperature was studied in the range from 25°C to 50°C at an initial pH of 7.5, a nZVAl dosage of 0.7 g/L, contact time of 40 min, and stirring rate of 200 rpm on the removal efficiency of all tested parameters. The results obtained indicated that the most effective temperature was 35°C, as shown in Figure 6E. The removal efficiency was enhanced from 73.8% to 76.2% for COD, from 73.7% to 77.6% for BOD, from 73.4% to 75.9% for TSS, from 31.8% to 34.1% for TN, and from 31.6% to 37.6% for TP, when the temperature increased from 25°C to 35°C. This could be attributed to the reduction in the energy activation barrier at higher temperatures (Alharby et al., 2021). Other causes for the removal efficiency rise at higher temperatures include (i) the formation of new adsorption sites and (ii) an increase in the intraparticle diffusion rate of pollutants into the adsorbent (Bouhamed et al., 2012).
Comparison With Other Decentralized Systems
The proposed three-stage decentralized system (coagulation, nZVAl unit, and sand filtration) was compared with the common decentralized wastewater treatment options reported in the literature, such as sequencing batch reactors (SBRs), anaerobic baffled reactors (ABRs), membrane bioreactors (MBRs), constructed wetlands, and passively aerated biological filters. Each system has advantages and disadvantages regarding operational complexity, removal efficiency, and energy requirements (Koul et al., 2022). Table 3 includes a comparison between the proposed decentralized multi-stages system and other systems reported in the literature. Planted drying beds and constructed wetlands depend on physical, chemical, and biological processes to remove contaminants from water. The main advantages of these two approaches are energy-free and simple operation; however, these two approaches achieve moderate BOD and COD removal efficiencies (ranges from 50% to 83%), variable nutrient removal, as well as requiring large land areas (Domínguez-Solís et al., 2025; Udom et al., 2018). Trickling filters and passive biofilters are robust, operate without energy inputs, and achieve good BOD and TSS removal, however, these two approaches achieve lower pathogen and nutrient removal (Abou-Elela et al., 2017; Terry et al., 2024). MBRs can achieve high TSS and BOD removal reaching 95%, however, this approach is energy- and cost-intensive (Ventura et al., 2024). SBRs have moderate energy demands and footprint and require skilled operation, however, they can achieve high organic removal exceeding 85% (Ali et al., 2022). ABRs have the potential of biogas recovery and could achieve good COD removal, however, this system may require post-treatment (Raja et al., 2025; Yulistyorini et al., 2019).
Comparison of the Proposed Decentralized Multi-Stages System With Other Systems Reported in the Literature.

Compared to these systems, the proposed multi-stages system in this study can achieve a higher COD, BOD, and TSS removal exceeding 93%, and oil and grease removal reaching 100%. A significant reduction in TN, TP, Ni, and Cu can also be achieved. Furthermore, the treated effluent from the proposed multi-stages system can be reused in agricultural application. The proposed system also offers a high-efficiency alternative suitable for different applications where land availability is limited.
Kinetic Studies
Table 4 describes the kinetic analysis of using nZVAl for removal of COD, BOD, TSS, TN, and TP. The results indicated that COD, BOD, TSS, and TN were well described by Avrami Kinetic models with the lowest Chi error 0.065, 0.117, 0.247, and 0.009, respectively. The strong correlation with the Avrami model for COD, BOD, and TN removal suggests a complex mechanism that may involve both the adsorption of contaminants onto the nZVAl surface and their subsequent degradation or transformation through reductive reactions facilitated by the zero-valent metal core. This is consistent with the known reactivity of nZVAl, which can act as both a strong adsorbent and an electron donor. The Avrami kinetic model depicts the chemical reaction rates between adsorbed molecules and sorbed assumes that one-dimensional growth happens by solid transition from one phase to another at constant temperature (Oladoja, 2016; Pang et al., 2016). The kinetics of TP removal using nZVAl can be described by a pseudo-second order (PSO) model with the minimum Chi error of .002. The pseudo-second-order model explains a chemical reaction (chemisorption) between adsorbed molecules and nZVAl, indicating that the interaction between the adsorbate and the adsorbent is strong and involves chemical bonds (Revellame et al., 2020).
Errors and Constants of Different Kinetic Models.

Thermodynamic Studies
Both Table 5 and Figure 7 provide the ∆H and ∆S values computed from the slopes and intercepts of ln(Qe/Ce) against 1/T plot “The value of the distribution coefficient, Kd (L/g), was calculated as Kd = Qe/Ce and Thus, the linear form of the van't Hoff equation is given by Equation 9.” The negative values of ∆H for COD (−9.75 KJ/mol) and TSS (−5.30 KJ/mol) indicate that the removal process is exothermic (the system releases heat during the reaction), while the positive values of ∆H for BOD (24.32 KJ/mol), TN (3.49 KJ/mol), and TP (9.58 KJ/mol) indicate that the removal process is endothermic (the system absorbs heat during the reaction; Mustapha et al., 2019; Sahmoune, 2019). Additionally, the negative values of ∆S for COD (−20.28 J/mol/K) and TSS (−5.72 J/mol/K) indicate that there is less chaos or randomness in a system. This suggests that the system is getting more structured, and individual components are less likely to be in different states. It can happen in situations when molecules are limited, such as during physisorption, when molecules are bonded to the surface (Duodu et al., 2025; Kanwal et al., 2024; Morales et al., 2023; Sahmoune, 2019). While the positive values of ∆S for BOD (92.59 J/mol/K), TN (8.65 J/mol/K), and TP (29.26 J/mol/K) indicate that there is a rise in chaos or randomness inside a system (the solid/liquid interface), especially during contaminants adsorption onto the nZVAl surface. This indicates that as the process progresses, the system gets more chaotic and less ordered (Chiban et al., 2016; Ebisike et al., 2023; Sahmoune, 2019). The negative ∆G values for COD, BOD, and TSS demonstrated that their removal by nZVAl was feasible, and that the adsorption process may occur spontaneously. The change in ∆G indicated that pollutants were better adsorbed at higher temperatures when ion mobility on nZVAl was unrestricted (Sadek et al., 2023) (Table 6).
Thermodynamic Parameters for Conaminants Removal Using nZVAl.

ANNs for Wastewater Treatment After Using nZVAl by Using Multilayer Perceptron.


Thermodynamic relation after using nZVAl. for removal of: (A) COD, (B) BOD, (C) TSS, (D) TN, and (E) TP.
While the kinetic and thermodynamic models provide strong indications of the removal mechanisms (e.g., chemisorption for phosphorus), the complex nature of the municipal wastewater matrix makes it challenging to definitively delineate the contribution of adsorption versus degradation for the overall removal of organic content (COD/BOD).
Artificial Intelligence Neural Networks (ANNs)
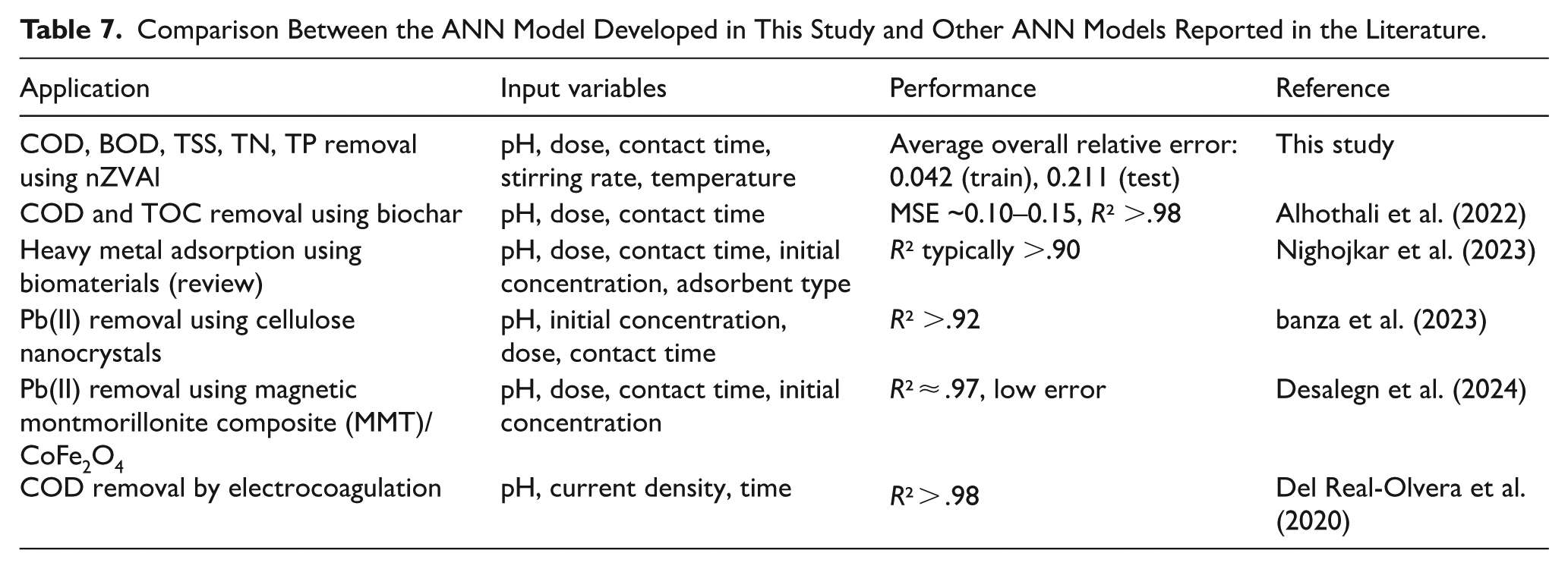
The application of artificial neural network (ANN) modeling in this study successfully captured the complex, non-linear relationships between the operational parameters and the treatment efficacy of the nZVAl process. The development of an MLP model with a 6-7-5 architecture (6 inputs, 7 hidden neurons, 5 outputs) effectively predicted the simultaneous removal efficiencies of COD, BOD, TSS, TN, and TP. The high predictive accuracy of the model, evidenced by the low average overall relative errors (0.042 for training and 0.211 for testing), confirms the capability of ANN to serve as a powerful tool for optimizing the nZVAl wastewater treatment process. The marginally higher error in the testing phase is expected and acceptable, indicating a model that generalizes well without significant overfitting to the training data. This performance is consistent with, and in many cases superior to, other ANN applications in environmental engineering, such as those for adsorption process optimization and electrocoagulation control (Alhothali et al., 2022; Del Real-Olvera et al., 2020), as detailed in Table 7.
Comparison Between the ANN Model Developed in This Study and Other ANN Models Reported in the Literature.
The sensitivity analysis yielded critically valuable insights for system design and operation. As shown in Figure 8, the overwhelming dominance of nZVAl dose (100% normalized importance) as the most influential parameter unequivocally highlights that the availability of reactive surface sites is the primary rate-limiting factor for the removal of all contaminants studied. This is a characteristic feature of adsorption-dominated processes and underscores the need for precise dosage control to maximize efficiency while minimizing material costs and sludge production. The secondary influence of contact time (25.6%) confirms that the processes of diffusion, surface reaction, and reductive degradation require sufficient time to approach completion. The significant roles of pH (23.4%) and temperature (21.2%) align perfectly with the thermodynamic and characterization results. pH governs the surface charge of the nZVAl (and thus its electrostatic interactions with pollutants) and the stability of the nanoparticles against passivation or dissolution, while temperature directly influences reaction kinetics and diffusion rates. The comparatively lower impact of stirring rate (16.5%) suggests that beyond ensuring adequate mixing to prevent settling and minimize boundary layer resistance, excessive energy input into fluid mixing yields diminishing returns.

Sensitivity analysis illustrating the relative importance and normalized importance of operational parameters on the removal efficiency of COD, BOD, TSS, TN, and TP.
These findings transcend mere statistical correlation; they provide a data-driven roadmap for optimization. For instance, operators of a future full-scale system should prioritize the precise automated dosing of nZVAl and ensure adequate hydraulic retention time in the reaction tank as the most effective levers for maintaining performance. The model's accuracy also suggests its potential use as a software sensor for real-time process control, predicting effluent quality based on easy-to-measure input parameters, thereby reducing reliance on frequent and costly laboratory analyses.
Future research should focus on expanding the ANN training dataset to include a wider range of real wastewater compositions and long-term operational data, which would further enhance the model’s robustness and predictive power for diverse scenarios. Furthermore, exploring hybrid modeling approaches that integrate ANN with mechanistic models could offer even deeper insights by combining the predictive power of machine learning with the explanatory power of physical principles.
Comparison of the Developed ANN Model With Similar Models Reported in Literature
The performance of the developed ANN model was compared with various recent studies that used ANN modeling for optimization purpose. The developed ANN model achieved low prediction errors, where the average overall relative error for training and testing was 0.042 and 0.211, respectively. Table 7 includes a comparison between the ANN model developed in this study and other ANN models reported in the literature. Alhothali et al. (2022) have applied the ANN tool to optimize COD removal from water using biochar and their results showed high accuracy of the ANN model with R² >.98 and MSE as low as 0.10 to 0.15. banza et al. (2023) have applied the ANN tool to optimize lead(II) ion (Pb(II)) removal from water using modified cellulose nanocrystals and their results showed high accuracy of the ANN model with R² >.92. Similarly, Nighojkar et al. (2023) have applied ANN models to optimize metal adsorption using various biomaterials and the results confirmed the suitability of the developed ANN model to be applied in complex adsorption processes, where the R² exceeded .90. (Desalegn et al., 2024) have achieved very high accuracy (R² ≈ .97) of the ANN model when used to optimize Pb(II) adsorption onto montmorillonite/CoFe2O4 composites. Del Real-Olvera et al. (2020) have also reported high R² value for the created ANN model exceeding .98, where the created ANN model was used to optimize COD removal from cold meat industry wastewater by applying electrocoagulation process. These comparison indicated that the performance of the developed ANN model is reliable and suitable for optimizing the propsed multi-stages treatment system using nZVAl.
Testing of the Decentralized System
Troubleshooting of the Treatment Plant
After the installation of different units of the treatment plant, the operation was started by continuous visual monitoring of the plant for troubleshooting. The first challenge was to transfer the wastewater from the manhole to the plant, but it has been observed that the transferred wastewater is not continuous and air entered the submerged pump because the manhole was not deep enough to allow continuous flow transfer. To overcome this issue, the depth of the existing manhole was increased, which allowed the pump to be submerged without blocking the way for the excess wastewater to be discharged to the main sewer pipe outside the university campus. After increasing the depth of the manhole, the wastewater transfer was continuous and no air entered the pump.
Optimization of the Plant Operation
The optimization process was conducted by adjusting the doses of NaOH and aluminum sulfate to ensure the formation of flocs. This process was conducted through pH measurement using HORIBA Water Quality Monitor and through visual observations of flocs formation. The plant was operated continuously for 8 hr until reaching the optimum doses to ensure the formation of large flocs while keeping a low concentration of aluminum in the treated effluent. A 0.5 g/L of aluminum sulfate and 0.7 g/L of nZVAl was applied to the system.
Analysis of Collected Samples
The next step was to monitor the operation of the plant by collecting 12 composite samples at different treatment stages to be analyzed in the HBRC chemical lab. The removal efficiencies were calculated, and the effluent water quality were also compared with the limits specified in the Egyptian code 501/2015 for reusing of treated wastewater in agriculture field. Table 8 and Figure 9 summarizes the analysis results for the collected samples. The results showed that the removal efficiency of total suspended solids (TSS), chemical oxygen demands (COD), biological oxygen demand (BOD5), oil and grease, ranged from 92.5% to 93.7%, 93.7% to 94.3%, 93.8% to 94.5%, 96.3% to 100%, respectively. Also, high removal efficiencies were reported for total nitrogen ranging from 52.6% to 59.9% and total phosphorus (TP) ranging from 61.4% to 98.7%. A reduction were also reported for nickel (Ni) reaching 61.1% and copper (Cu) reaching 82.6%. The results also showed that the effluent water quality did not exceed the limits specified in the Egyptian code 501/2015, which means that the treated water can be reused in agriculture application.
Analysis of Samples Collected From Different Stages of the Decentralized Wastewater Treatment Plant.


The removal efficiency of the studied parameters after each treatment process.
Limitations of the System
There are some limitations associated with the approach of applying the proposed decentralized wastewater treatment system. The first challenge is integrating it into existing urban infrastructure. The second challenge is the operational reliability of decentralized systems. Alongside removal efficiency, the volume of sludge generated is a critical operational parameter. At the optimum nZVAl dose of 0.7 g/L, the average volumetric sludge generation was quantified to be approximately 125 mL/L of wastewater treated after a 30-min settling period. This sludge consists of spent nZVAl nanoparticles (including their oxidation products, such as Al(OH)3) along with adsorbed organic and inorganic contaminants. While this represents an additional waste stream to be managed, the high removal efficiencies achieved justify the resulting volume. Future work will focus on strategies for sludge minimization, potential regeneration of the nanomaterial, and safe disposal or resource recovery pathways. The other challenge is the safe disposal of treatment products, such as sludge. Table 9 shows the main sludge treatment and disposal alternatives suitable for decentralized wastewater treatment plants, with the advantages and limitations of each alternative.
Sludge Treatment Alternatives.

Conclusions
This study investigated the efficiency of a decentralized wastewater treatment plant comprising coagulation/flocculation, nano degradation and adsorption unit, and filtration unit to be used in the treatment of real municipal wastewater. First, the lab experiments were conducted in the wastewater to obtain the optimum operating conditions for coagulation/flocculation unit and the nano unit. The results showed that the optimum coagulation/flocculation conditions were observed at pH 7 and 0.5 g/L of aluminum sulfate dose. For the nano unit, the optimum operating condition was observed at pH 7.5, 0.7 g/L nZVAl dose, 40 min contact time, 200 rpm stirring rate, and 35°C temperature. The Avrami kinetic model adequately describes the removal for COD, BOD, TSS, and TN, while the pseudo-second-order model describes the removal mechanism of TP. The thermodynamic results indicated that the reaction mechanisms are exothermic for COD and TSS, and endothermic for BOD, TN, and TP. The study also found that the developed ANN model is reliable and can be utilized to maximize the removal efficiencies of COD, BOD, TNN, TN, and TP from municipal wastewater using nZVAl under a variety of environmental conditions. Sensitivity analysis showed that nZVAl dose had considerable effects on the removal efficiency of the parameters studied. The optimum conditions obtained from the lab experiments were used to operate the decentralized wastewater treatment plant. The findings demonstrated that the suggested new multistage treatment system (coagulation/flocculation, nano unit, and filtration) can effectively treat municipal wastewater. The removal efficiencies of COD, BOD, TSS, oil and grease, TN, TP, Ni, and Cu reached 94.3%, 94.5%, 93.7%, 100%, 59.9%, 98.7%, 61.6%, and 82.6%, respectively. This result indicates that the water quality of the treated effluent did not exceed the limits specified in the Egyptian code 501/2015 and can be reused in agriculture applications. This research can be part of a decision support system regarding applying the decentralized wastewater treatment systems at the household and university levels.
Supplemental Material
sj-docx-1-asw-10.1177_11786221251392205 – Supplemental material for Innovative Decentralized Wastewater Treatment: Coupling Coagulation, nZVAl Adsorption, Sand Filtration, and ANN-Based Optimization
Supplemental material, sj-docx-1-asw-10.1177_11786221251392205 for Innovative Decentralized Wastewater Treatment: Coupling Coagulation, nZVAl Adsorption, Sand Filtration, and ANN-Based Optimization by Ahmed S. Mahmoud, Youssef Soliman, Moustafa S. Moussa, Robert W. Peters and Mohamed K. Mostafa in Air, Soil and Water Research
Supplemental Material
sj-docx-2-asw-10.1177_11786221251392205 – Supplemental material for Innovative Decentralized Wastewater Treatment: Coupling Coagulation, nZVAl Adsorption, Sand Filtration, and ANN-Based Optimization
Supplemental material, sj-docx-2-asw-10.1177_11786221251392205 for Innovative Decentralized Wastewater Treatment: Coupling Coagulation, nZVAl Adsorption, Sand Filtration, and ANN-Based Optimization by Ahmed S. Mahmoud, Youssef Soliman, Moustafa S. Moussa, Robert W. Peters and Mohamed K. Mostafa in Air, Soil and Water Research
Footnotes
Acknowledgements
The authors would like to thank Badr University in Cairo (BUC) for its support of this research. The authors would also like to thank the Academy of Scientific Research and Technology (ASRT) and the Bibliotheca Alexandrina (BA) for their continued support.
Author Contributions
MKM, ASM, YS, MSM, and RWP: research, conceptualization, methodology, validation, and data analysis. MKM, ASM, YS, MSM, and RWP: original draft writing and editing. MKM, ASM, MSM, and RWP are responsible for reviewing the first draft of the manuscript. MKM, ASM, and MSM are accountable for fieldwork and sample analysis. MKM, ASM, YS, and MSM are responsible for the design and construction of the decentralized wastewater treatment plant. MKM, ASM, MSM, and RWP: Supervision and Project Administration.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Joint Academy of Scientific Research and Technology (ASRT)/the Bibliotheca Alexandrina (BA) Research Grants [grant number 1469] awarded to the first author of this paper (Associate Professor Mohamed K. Mostafa, BUC).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The article includes all data generated or analyzed during the investigation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

