Abstract
Heavy metals are of great environmental and sanitary importance due to the toxicity they generate; therefore, a wide variety of methods for elimination in water has been studied. One of the approaches employed is bioremediation, which involves the use of biomass (microorganisms or plants), living plants (phytoremediation), or biomaterials to eliminate these elements. In this study, we investigated the technical feasibility of using the
Introduction
Many communities lack access to good quality drinking water because of the use of raw water from rivers (Calderón Núñez, 2020). Pollution of rivers usually occurs due to the release of substances derived from human activities, such as mining waste that contains heavy metals and other harmful agents for health and the natural environment (Guillén Pérez, 2020; Vergara Buitrago & Rodríguez-Aparicio, 2021). Iron is one of the common contaminants in rivers due to mining (Vergara Buitrago & Rodríguez-Aparicio, 2021), and its presence can affect the color, taste, and smell of the water (Organizacin Mundial de la Salud [OMS], 2018), as well as cause poisoning in high concentrations (Bustamante-Cristancho, 2011).
Iron ingestion causes a disease called hemochromatosis, which causes liver and heart failure, vomiting, skin damage, joint pain, and weight loss (Dey, Kotaru et al., 2022). The normal level of iron in the blood can vary depending on age, sex, and other factors. However, the level recommended by the Food and Agriculture Organization of the United Nations for irrigation water is 5 mg/L = 5 ppm. The US EPA secondary drinking water standard, MCL, is 0.3 mg/L = 0.3 ppm. (Standard Methods Committee of the American Public Health Association, American Water Works Association, and Water Environment Federation, 2018).
For the treatment of contaminated water, bioremediation has been used using plants, microorganisms, and biomaterials (Carreño-Sayago, 2015; Paredes & Ñique, 2016). In particular, to remove iron, biosorption has been used (Dey, Sreenivasulu et al., 2022).
Currently, spider web-shaped electrospun nanofibers are being fabricated to separate water from oil in wastewater treatment. (Ning et al., 2023). Natural spider webs of
The spider web has a diversity of mechanical, thermal and biomedical properties that have been widely studied (Mann et al., 2023); however, to date, there are no reports in the literature on its capabilities as a bioremediation agent for contaminated waters. Resistance studies of this type of fabric have reported values of 6308.84 kPa (Carbonell Plata, 2008), a thermogravimetric stability lower than 210°C (Xing et al., 2014), and its biocompatibility evidenced the growth of animal cells on the fabric (Bergmann et al., 2022).
This silk composed of amino acids such as glycine, allain, and sericin, which have the possibility of charging and interacting with metal ions (Römer & Scheibel, 2008). Adsorption can occur due to various mechanisms, such as electrostatic attraction, the formation of complex chemicals, and the interaction of covalent bonds between the spider web and contaminants. Currently, among the technological advances, PCL and PUR polymer membranes for water treatment, with the aim of detoxifying waters contaminated with heavy metals (Tran-Ly et al., 2020).
The iron removal can be explained through a process called biosorption, in which biological materials such as spider webs can adsorb and concentrate heavy metals or other water pollutants. Adsorption can be caused by several mechanisms, such as electrostatic attraction, the formation of chemical complexes, and the interaction of covalent bonds between the spider web and contaminants (Tran-Ly et al., 2020).
In the specific case of spider web, it has been shown that its ability to adsorb metals such as Fe is due to the presence of proteins, carbohydrates, and other organic components containing functional groups such as carboxyls, amines, and sulfhydryls. These functional groups have a high affinity for metal binding, and the spider web structure provides a porous and fibrous surface for metal adsorption (Zhou et al., 2021). In addition, the negative charge of the carboxyl and sulfhydryl groups present on the spider web attract the positively charged Fe ions. The electrostatic interaction between the functional groups and Fe ions, as well as the formation of chemical complexes between them, also contribute to the adsorption of Fe on the spider web (Pelit et al., 2011).
Specifically, the functional groups of the
We propose the analysis methodology for this research through Response Surface Methodology (RSM), which allows us to obtain optimal removal values through the effects of time (hr), iron concentration (ppm), and spider silk weight (g). There is a specific research gap in this case because the conditions for the remediation of iron in water have been evaluated using different materials, and they have not used spider webs for this purpose. Nevertheless, the results obtained demonstrate the remedial capacity of spider web and possible bioremediation mechanisms used by this process.
The objective of this study was to determine the technical feasibility of using the spider web
Materials and Methods
First, a bibliometric network was used, specifically the Science Direct database, which consisted of 622 articles, for the selection of process variables based on the keywords: Biomaterial, bioremediation, iron, and adsorption. In this study, we found that the examined articles used the variables reaction time, heavy metal concentration, and biomaterial weight more frequently in the tests. Therefore, we decided to select these variables and based on them we considered the experimental design. Subsequently, we conducted a pilot test to evaluate the biomaterial’s behavior in terms of iron adsorption. In this test, we established the following experimental conditions: a duration of 8 (hr), 0.1 g of cobweb, and three concentrations of iron (1, 2, and 3 ppm). Based on the results obtained in this test, we conclude that it is necessary to increase both the exposure time and the initial concentration. The duration of the study was approximately 12 months.
We used the following materials: Cobweb obtained from the Trichonephila clavipes species, PanReac brand iron analytical grade (High purity 97% anhydrous iron (III) chloride solution, analytical grade), and deionized water. We collect the cobwebs manually at the Cali Zoo Foundation, avoiding contact with spiders. We took extra care to minimize any disturbance to their environment and ensure their safety and well-being. In the collection, we use a 10 cm × 10 cm box and a 1.5 m stick to access the spider webs in different areas. In addition, we collect the spider web once a month so as not to affect the population of individuals.
We transport the collected spider webs to the laboratory to remove food remains, branches, and larger leaves. For cleaning, we use a Leica S6D stereoscope to eliminate any element that could affect the results. In addition, we ensured that the collection process did not alter the quality of the spider silk samples obtained, thus guaranteeing the validity and reliability of the research results.
Materials
We used an Energy Dispersive X-ray Fluorescence Spectroscope (EDX Shimadzu 7000) to obtain the readings of the initial and final iron content (ppm) in each of the water samples. When preparing ferric chloride, iron is in the Fe + 3 state and oxidized to Fe + 2. The pH of the tests was between 3.0 and 3.2. An acid pH generated by the ferric chloride reagent. The experiment was carried out in a beaker, at a volume of 40 ml, to quantify the remaining ferric iron (Fe2⁺) in a solution, we perform a chemical oxidation using a reducing agent; we use titrimetric titration with potassium dichromate. (Bosch Ariño, 1954; Szabó & Sugár, 1952) With the initial iron results, we adjusted the actual iron concentrations proposed in the experiment design (Figure 1).

Result of initial and final iron content (ppm) in each sample.
Methods
Based on the results of the pilot study, we established the experimental conditions that were determined and described in Table 1. As a response and/or dependent variable, we measured the percentage of iron removal in water and found that the selected variables (time, iron concentration, and spider silk weight) directly influenced the response variable. We employed a central composite design within the framework of response surface methodology (RSM) for forecasting and enhancing the elimination of iron from water (Y). This process considers time (hr) (X1), iron concentration (ppm) (X2), and weight of the spider web (g) (X3) as influencing factors. To maximize the percentage of iron removal, in this design, we secured eight (8) factorial points, six (6) centered points, and six (6) axial points. These 20 combinations of factor levels were executed randomly to conduct the experiment.
Response Surface Methodology Design.

According to the central composite design, we proceeded to perform various tests. We prepared samples with the corresponding iron concentrations and contact times between the solution and spider silk. Once the reaction time elapsed, we removed the spider web and filtered the sample to remove any residue. Then, we proceeded with the chemical oxidation and finally performed the reading of the samples using EDX.
Efficiency of the Removal Determination
To determine the percentage of iron removal in the contaminated water samples, through the treatment with spider web, we proceeded to perform the readings in the dispersive energy X-ray fluorescence spectroscope, located in the Optoelectronics laboratory of the Universidad Autónoma de Occidente.
To determine the iron removal percentage, Equation 1 was used, where E.R is the removal efficiency (%), C.I is the initial iron concentration (ppm), and C.F is the final iron concentration (ppm).
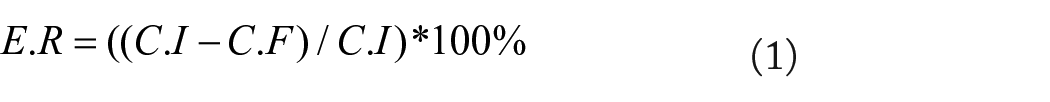
Measurement of Iron From a Spider Web
We proceeded to measure the pre-established quantities of spider web, exposed to 40 ml of each prepared iron solution. We subjected these samples to observation during the contact time established according to the experimental design. After the reaction period, we removed the spider web and proceeded to filter the resulting sample. We quantified the iron samples using the EDX technique. This method enables precise determination of iron concentrations in the samples, offering valuable insights into the interaction and adsorption of iron by the spider web.
Analysis of Response Surface Methodology (RSM)
In the experimental plan, we employed a central composite design within the RSM, which is well suited for modeling a quadratic surface commonly used in process optimization. To validate the model’s appropriateness for optimizing iron removal in water, we conducted hypothesis tests, including assessments for lack of fit, linearity, and quadratic effects. We analyzed the prediction equation using analysis of variance (ANOVA) to test the hypotheses associated with RSM. In Equation 2, shows that the second-order polynomial model optimizes the variables considered.
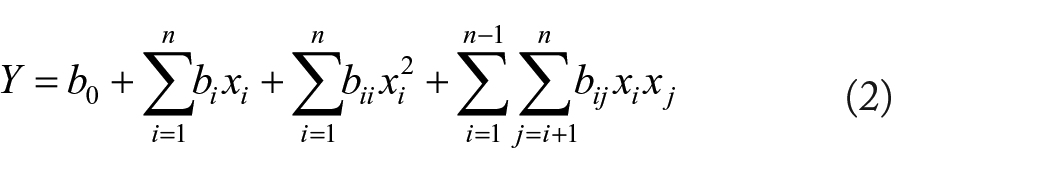
Where
Initially, we validated the assumptions about the model error ε
We obtained the coefficient of determination
For the optimization of the removal percentage, we use the predictability function method (Cornell, 1982). This approach allows the optimization of the statistical model having as its main objective to maximize the response variable removal (%). From the data of the experimental design, we obtained the optimal levels of the factors.
We performed three additional experiments in the Environmental Sciences and Optoelectronics laboratories to verify the optimal values. With these results, we calculated the relative error of the experiments and evaluated the capacity of the model to predict the optimal levels of factors (time, weight of the spider web, and iron concentration) to obtain the highest iron removal percentage. We performed the calculations using the statistical package MINITAB 19 (variance analysis, contour plots, and optimization) (Minitab Inc, 1972).
Results
Figure 2a shows the results of the averages related to the iron concentrations (ppm) and the weight of the spider web used for the removal of heavy metals in an aqueous medium. We observed that the removal efficiency was maximal when using a weight of 0.06 g and an iron concentration of 111 ppm. In Figure 2b, we present the averages of the reaction time and the weight of the spider web. A maximum value was obtained when the time was 18 hr, being this a centered point within the RSM design. In Figure 2c, we have the time and iron concentration, where the time 18 hr presents a maximum removal at an iron concentration of 111 ppm.

(a–c) Removal averages (percentage) of Fe-polluted water using a spider web.
We obtained very promising results from the iron removal test in contaminated water using spider webs, with a removal efficiency close to 80%. These results are significant because spider web is a natural and readily available material, and it can be an economical and sustainable solution for the removal of contaminants from water.
From the removal results (%) acquired in the experimentation, we obtained the regression equation, which was elaborated on the basis of the predictor variables time, real iron concentration, and web weight.
Based on these results, we found that the data fit perfectly into a regression model (RSM), which complies with the assumptions about the model error at significance levels greater than 10% (p-value). The selected model generally presented a good fit (
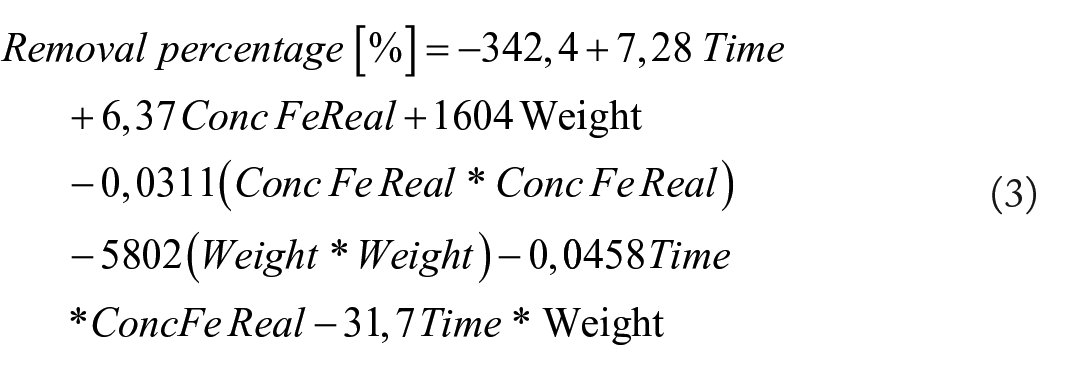
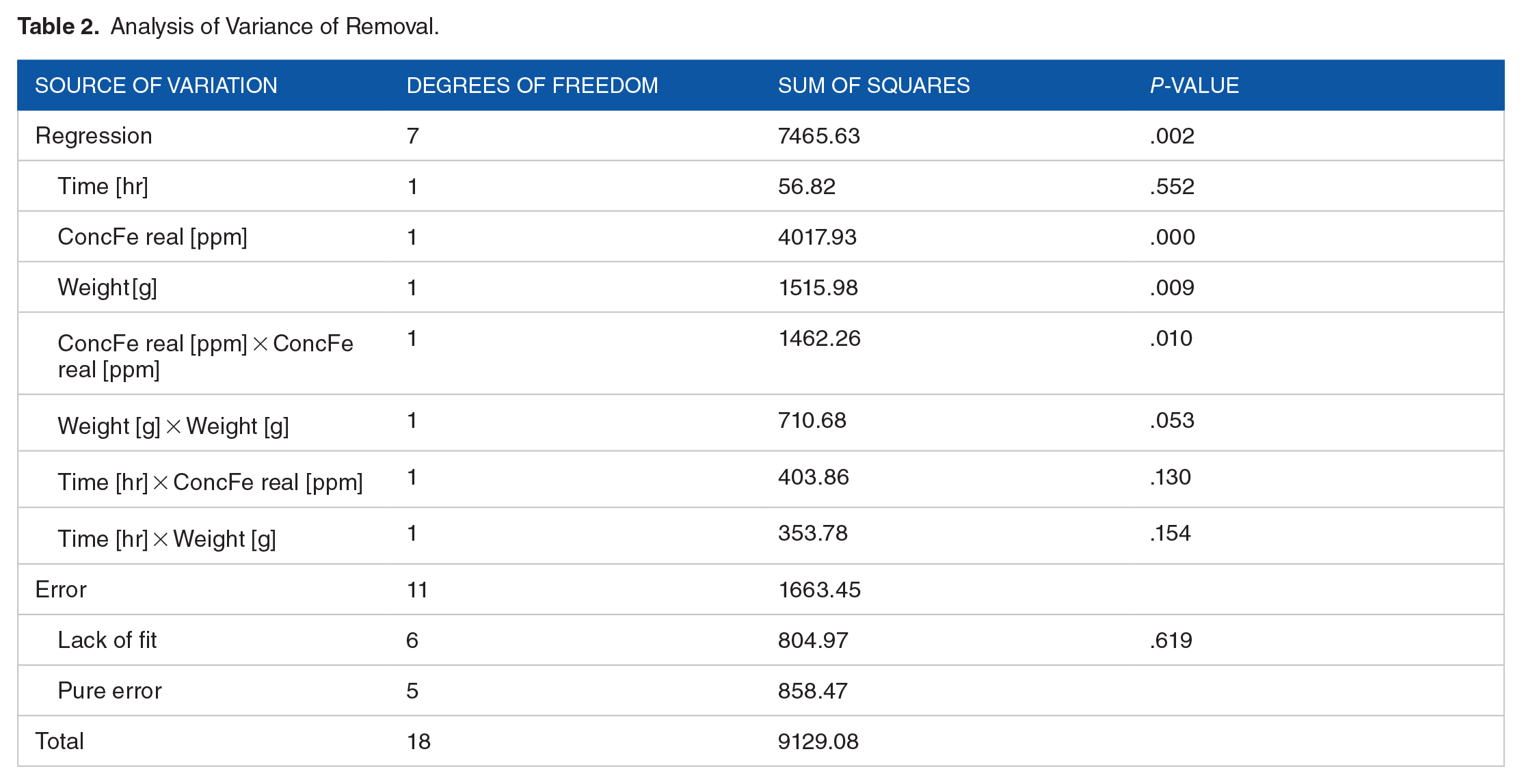
Table 2 presents the main statistical results of the regression obtained by ANOVA. The results indicate that the regression has a greater impact in the explanation of the percentage of removal compared with the random effects, (sums of squares of the regression = 7465.63 and sum of squares of the errors = 1663.45). The evaluation of the hypothesis test for lack of fit was not significant, indicating that the model is appropriate, at a significance level of 0.619 (
Analysis of Variance of Removal.
Optimization of the removal rate
Once the model with significant quadratic effects has been obtained, we proceed to optimize the removal percentage (maximize). To this end, we established the following conditions: A target value of 75% and a lower limit of 20%. The optimization presents high predictability values (D:1), in Figure 3, the
(Time: 28.09 (hr), Real Fe CONC: 81.42 (ppm), Weight: 0.062 (g) with these values, one of the best removal percentages is predicted (91.82%).
Note that the removal percentage tends to increase with time (over 20 hr).
Concerning the actual Fe concentration, it can take values between 76.02 and 88.63 that the removal is not significantly affected, taking values to predict the removal percentage between 90.46% and 89.15%.
Regarding the weight, it can take values between 0.056 and 0.075 that the removal is not significantly affected, taking values to predict the percentage of removal between 90.46% and 89.15%.
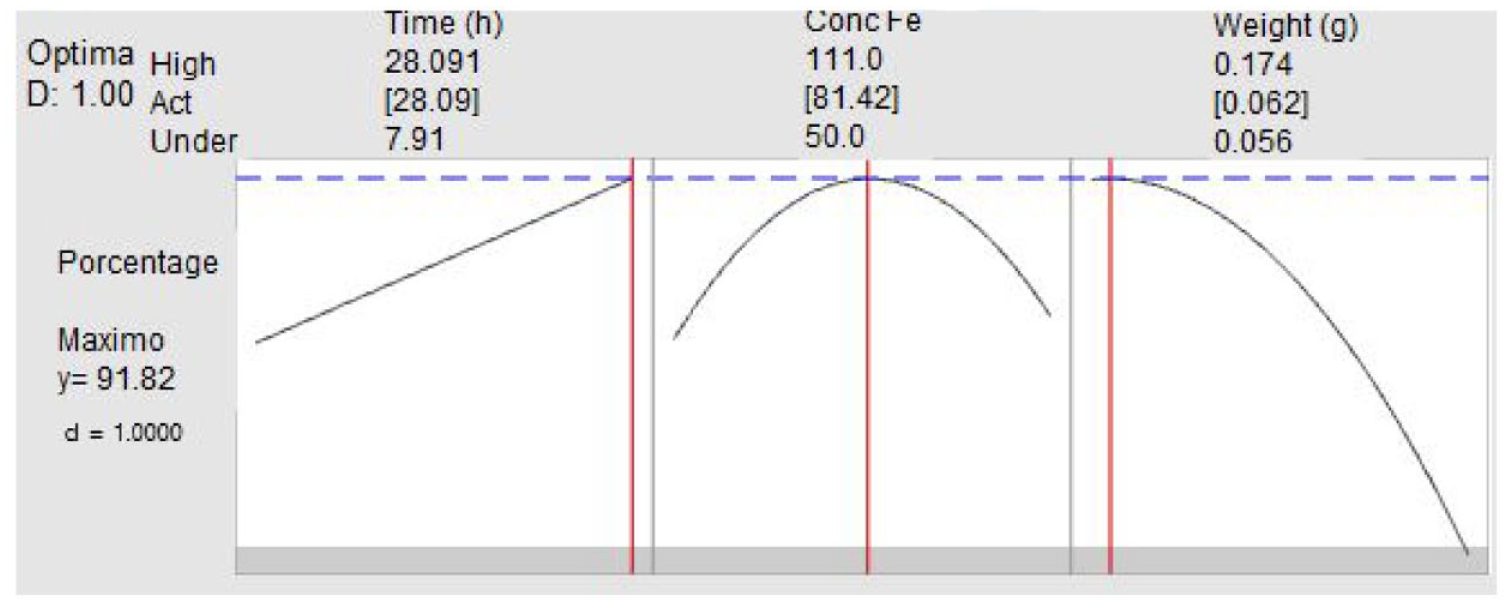
Graph of the prediction of the model with the highest percentage of Fe removal.
In general, when analyzing the results of the removal optimization (%), we observe a relationship between different variables. Regarding the weight, when it decreases or increases outside the ranges (0.056 and 0.075), the removal tends to decrease. Likewise, we found a direct relationship between time and removal, which means that as time increases, removal increases. On the other hand, when the iron concentration is outside the established ranges (76.02 and 88.63), the removal also tends to decrease. In addition, the results obtained show high predictability, indicating that it is possible to predict the result with high precision.
We present the optimization results through a contour plot depicting the removal percentage (Figure 3). The first graph here, we note that actual iron (Fe) concentrations fall within an approximate range of 66 to 98 mg/kg for extended times exceeding 20 (hr), achieving elimination percentages surpassing 80%. In the second graph, illustrating the relationship between weight and time, we observe weights below approximately 0.1 g, requiring durations exceeding 20 (hr) to achieve removal percentages exceeding 80%. In the third graph, representing the relationship between weight and actual iron (Fe) concentration, we observe weights below 0.1 g and Fe concentrations ranging between 61 and 98 ppm, resulting in removal rates exceeding 80%.
Verification of the predictions produced by the model
To ascertain the predictive capability of this model, three experiments were conducted (Table 3), demonstrating that the values forecasted by the regression equation closely align with the actual values obtained in the laboratory. We attribute this close correspondence to the good fitting observed in the modeling process, resulting in values that closely mirror those depicted in Figure 4a to c.
Summary of the Experiments and Calculation of the Relative Error of the Optimized Treatments.


(a–c) Contour plot of iron removal percentage.
Table 3 shows the relative errors of the three experiments using the optimization values established by the RSM. We obtained relative percentage error values of less than 4.34, indicating high precision in the results of test two with a relative percentage error of 0.39. This means that the results obtained are very close to the real value and that the technique used (RSM) to model the percentage of Fe removal from contaminated water.
The removal of 88% of iron water is of great importance in terms of water quality and public health. Iron is a metal that can significantly affect the taste, odor, and color of water, making it less attractive for human consumption. In addition, excessive consumption of iron in water can have negative health effects, such as gastrointestinal and cardiovascular problems. As indicated by the information from the WHO (2003), and Resolution 2115 of June 22, 2007, bottled water or water for human consumption must comply with a maximum permissible iron value of 0.3 ppm. Therefore, the removal of 88% of iron achieved in this study demonstrates that the treatment technology used can be an effective solution to ensure that drinking water meets the quality and safety requirements established by regulations. These results can have a significant impact on the protection of public health and the promotion of safer and more reliable access to drinking water (Ministerio de Vivienda y Desarrollo Territorial y Ministerio de Protección Social de Colombia, 2007).
Discussion
Previous research, such as that carried out (Seid & Gonfa, 2022; Yusuf et al., 2023), has raised that the removal of contaminants depends on the specific characteristics of the sample and that bioremediation technologies offer a promising alternative for water decontamination. The removal of iron is a critical process in the management of water and its contamination because it can affect people’s quality of life (OMS, 2018). The results obtained in this study support the efficacy of the chemical treatment used and its applicability in drinking water treatment systems, which can have a significant impact on the health and well-being of the population as well as on the protection of the environment and the sustainability of water resources (Ministerio de Vivienda y Desarrollo Territorial y Ministerio de Protección Social de Colombia, 2007).
Based on the results obtained from the RSM modeling, we can affirm that neither the time (h) should not be decreased, nor should the iron concentration (ppm) be decreased or increased outside the range of approximately 76.02 and 88.62 ppm. With respect to the weight of the spider web, values between 0.056 and 0.075 g generated optimal removal values of approximately 91%.
This study evaluated the efficiency of iron removal from contaminated water using spiderwebs as a bioremediation material. The results showed high precision, with a relative error of less than 4.34%. We achieved 88% iron removal, which is significant in terms of water quality and public health. Although this percentage, lower than that in other studies (Fito et al., 2023).
However, there is room for improvement in the iron removal process. We recommend exploring additional approaches that can enhance efficiency, such as the consideration of a “multiphase” bioremediation technique. This innovative strategy involves the implementation of two or more sequential high-capacity filters, similar to spider webs, to optimize the removal of contaminants present in the water. Thanks to this methodology, it is possible to increase the effectiveness of the treatment. This technique is relevant in compliance with drinking water quality standards (Ministerio de Vivienda y Desarrollo Territorial y Ministerio de Protección Social de Colombia, 2007), because it is possible to obtain high removal percentages of 88% in the initial phase.
In other studies related to bioremediation of water with heavy metals, using other biomaterials (carbon), we found very short process times (15, 25, and 90 min) and that the bioremediation time under the technique of this research is considerably longer. However, similar removal percentages were found in other research articles (72%, 86.49%, among others), which indicates that the removal efficiency of this work is within the average of the analyzed (Benila Smily & Sumithra, 2017; Cisneros Gómez & Laura Pezo, 2019; Fito et al., 2023).
By applying a second phase of treatment, we hope to achieve even greater removal. In addition, this technique would be beneficial for treating water contaminated with iron concentrations greater than 88.0 ppm. To perform this large-scale project, we must consider several important factors. First, we must consider the breeding of spiders because they are in charge of producing the spider web used in bioremediation. The breeding of spiders requires a specific space and habitat for their development (Foong et al., 2020); therefore, it requires a careful way of breeding and maintaining spiders in large numbers.
Another factor to consider is the amount of spider web required to perform the bioremediation process on a large scale. According to the results of this investigation, approximately 15 spider webs of 1 m diameter are required to produce 1 g of spider web. This low production density makes implementation on a large scale difficult because large numbers of spiders and spider webs must achieve the desired goals. These calculations should consider planning the scale of the project and the amount of resources needed.
Limitations of This Study
The tests in this experiment were carried out on the web of the species Trichonephila clavipes. There are other species of weaver spiders that could be used to test whether the property of adsorbing elements dissolved in water is specific to this species or if it is a property that most spider webs have. The experiment was carried out with iron and we worked with a single layer of spider web using a bioremediation technique. It would be advisable to test other heavy metals of major health and environmental importance (chromium, lead, mercury, and cadmium) as well as the “multiphase” spider web. The results obtained in a controlled laboratory environment are promising, but further research is needed to explore both its practical application, which could be limited by the real conditions of treating large volumes of water, and its large-scale application.
Conclusion
Is confirmed the efficacy of chemical treatment with spider webs for the removal of iron from contaminated water, supporting its applicability in drinking water treatment systems, with a significant impact on public health and environmental protection. The study demonstrated that it is possible to use the spider web for iron removal. We found a relative error of less than 4.34% and a removal efficiency of 88%, this percentage is lower than other studies with other types of materials, but it is significant for water quality and public health.
Based on the statistical tool used RSM, to obtain the maximum iron removal (%), mixtures with iron concentrations between approximately 76.02 to 88.62 ppm, with a weight between 0.056 and 0.075 g, should be prepared for a period of 28 (hr), the experiment was performed in a beaker, at a volume of 40 ml. Although the bioremediation time in this study is longer compared to other biomaterials that achieve very short process times (15, 25, and 90 min), the removal percentages are comparable (72%, 86.49%, etc.), indicating that the removal efficiency is within the average of other studies.
We recommend exploring additional approaches to improve efficiency, such as the “multiphase” bioremediation technique, which involves the implementation of sequential high capacity, cobweb-like filters to optimize contaminant removal and meet drinking water quality standards. Validation of the optimized iron removal for the three selected specimens corroborated that the ratios obtained through the RSM design allowed the highest removals with a predictability of one. The high removal rates obtained support the feasibility of this technique, which has important implications for environmental protection and public health. The existing relationship between the selected variables reinforces its suitability for the study of bioremediation of iron-contaminated waters.
Footnotes
Acknowledgements
The Universidad Autónoma de Occident, the Zoological Foundation of Cali, and the SEMAP-Applied Mathematics research group supported this work, and A.C. contributed to the interpretation and review of the manuscript. All authors have read and approved the final content of the manuscript and preprint of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Studies in Humans and Animals
This collection approach is respectful of the spiders and their habitat, and we adopted it to preserve the integrity of the research and minimize any negative impact on the spider population studied.
Data Availability
Data is available upon request.
