Abstract
Ulcerative giant breast cancer (UGBC) presents management challenges including uncontrolled bleeding, infection, and rapid tumor progression, which often culminate in life-threatening complications. Traditional spatially fractionated radiotherapy (SFRT) like lattice radiotherapy (LRT) offers potential for palliating bulky tumors, although its application in UGBC remains limited. We report a 51-year-old woman with a hormone receptor-positive, human epidermal growth factor receptor 2 (HER2)-negative stage IIIC UGBC (20.5 cm × 6.98 × 18.17 cm ulcerative lesion). Treatment consisted of modified lattice radiotherapy (mLRT: 15 Gy×3 fractions), followed by volumetric-modulated arc therapy (VMAT: 36 Gy/20 fractions) and systemic therapy (exemestane, capecitabine, CDK4/6 inhibitor). Rapid hemostasis was achieved within 5 days, with 39.7% tumor reduction at week 4 and progressive wound healing. At 3-month follow-up after the completion of mLRT and prior to definitive radiotherapy, the tumor had achieved a 61.8% volume reduction, with only minimal residual disease remaining. The radiation-induced skin reaction gradually alleviated with symptomatic treatment. This case demonstrates mLRT’s efficacy in achieving rapid hemostasis and significant tumor regression for chemotherapy-refusing UGBC patients. The combined approach of mLRT, VMAT, and systemic therapy provides a promising multidisciplinary strategy for symptom control and quality-of-life improvement. Further prospective studies are needed to validate these findings.
Plain Language Summary
Ulcerative giant breast cancers (UGBC) cause life-threatening bleeding, infections and severe pain, and are hard to treat in chemotherapy-intolerant patients with poor responses to standard therapies. A 51-year-old woman with a 20.5 cm × 6.98 cm × 18.17 cm stage IIIC hormone receptor-positive, HER2-negative bleeding ulcerated UGBC refused chemotherapy due to side effect concerns. She was treated with a combined regimen: modified lattice radiotherapy (mLRT, 3 high-dose fractions of 15 Gy each), subsequent volumetric-modulated arc therapy (VMAT, 36 Gy in 20 fractions), and oral systematic therapy (hormone-blocking agent, oral chemotherapy, CDK4/6 inhibitor).
Bleeding was completely controlled within 5 days; the tumor volume reduced by 24.7% at 1 week, 39.7% at 4 weeks and 61.8% at 3 months post-mLRT, with near-complete ulcer epithelialization and no cancer spread. After full combined treatment, the tumor shrank by 92.9% from baseline with a complete clinical response.
This case demonstrates mLRT’s efficacy in rapid hemostasis and tumor regression for chemotherapy-refusing UGBC patients, and the combined mLRT-VMAT-systematic therapy is a promising multidisciplinary strategy for unresectable UGBC, warranting further prospective research.
Introduction
Giant ulcerative breast cancers with persistent hemorrhage particularly present a therapeutic dilemma when conventional treatments like surgery, chemotherapy, and standard radiotherapy prove contraindicated or ineffective.1,2 Surgical resection was deemed constrained, while standard palliative radiotherapy (20-30 Gy in 5-10 fractions) can provide temporary symptom relief, but it often fails to achieve meaningful tumor regression in fungating masses. 3 Moderately escalated doses and stereotactic body radiotherapy have shown promise for smaller lesions, but their applicability to bulky tumors is limited by dose-volume constraints that endanger organs at risk (OARs).4-6 Compression dressings further complicated management by precluding invasive procedures, creating an urgent need for a rapid, non-invasive solution to control bleeding and enable subsequent therapy.
The modified lattice radiotherapy (mLRT) emerged as a feasible alternative in this setting. Since this method applies high-dose radiation to particular locations, referred to as “vertices,” it seems to satisfy the requirement for a quick tumor response and enhance patients’ quality of life quickly. By administering lesser doses to the remaining tumor volume, it simultaneously guarantees the preservation of vulnerable organs. In addition, by priming lymphocytes, activating immune cells, and causing immunological-mediated damage at locations far from the treatment field—a phenomenon called as the abscopal effect—the use of high-dose fractions triggers the immune response, which may be advantageous for patients who have spread to other areas.2,7
The aim of this report is to present a case to highlight a unique clinical scenario involving a giant ulcerative breast cancer with persistent life-threatening hemorrhage, where traditional interventions were precluded due to severe anemia and chemotherapy intolerance. Given the unmet clinical need for effective hemostatic and tumor-controlling strategies in such inoperable patients, mLRT was adopted as a salvage treatment, with the intent to evaluate its efficacy in achieving hemostasis, inducing tumor regression and enabling subsequent feasible therapeutic options. This case would underscore mLRT’s potential to address critical unmet needs in the management of giant ulcerative tumors with hemorrhagic complications, particularly in patients ineligible for standard therapies.3,8
Methods
A 51-year-old female’s breast cancer history was characterized by delayed intervention and progressive disease. In 2019, an external hospital ultrasound detected a 1-cm right breast nodule, recommending a biopsy, but the patient opted for traditional Chinese medicine (TCM) treatment instead.
On October 27, 2021, a repeat ultrasound revealed a 36 × 22 mm solid mass in the right breast (BI-RADS 4B) with a 25 × 17 mm enlarged right axillary lymph node. Despite biopsy advice, she continued TCM treatment. Tumor markers on July 28, 2022, were CEA 5.85 ng/mL and CA153 26 U/mL; an elevated FT3 (7.2 ng/mL) was noted on March 22, 2023.
By December 2023, her condition worsened. A computed tomography (CT) image on December 7 revealed a 15.26 × 15.41 × 7.38 cm (Figure 1A) ulcerated right breast cancer with axillary metastases, lung fibrotic foci, and a thyroid abnormality. The patient had no significant past medical history, and family history was unremarkable for genetic or oncologic disorders. Social history was stable with no reported psychosocial stressors. Biopsy confirmed grade 3 invasive ductal carcinoma (estrogen receptor [ER] 90%, progesterone receptor [PR] 20%, human epidermal growth factor receptor 2 [HER2]-negative, Ki-67 70%), staging cT4N3M0 (stage IIIC).

Tumor size with CT images presentation of UGBC at different times. (A) December 7, 2023. A CT scan of the right breast revealed a large ulcerative invasive ductal carcinoma with a 3-dimensional size of 15.26 × 15.41 × 7.38 cm, with central ulceration and ipsilateral axillary lymph node metastasis. (B) October 28, 2024. A follow-up CT scan demonstrated significant tumor progression to 20.5 × 6.98 × 18.17 cm with obvious effusion, active bleeding, aggravated central ulceration, and the tumor invaded the surrounding chest wall soft tissues. Imaging parameters: 16-slice CT scanner (soft tissue window, width: 600 HU, window level: 640 HU, 3 mm slice thickness, 120 kV, and 200 mAs).
On January 30, 2024, our clinic proposed taxol-based chemotherapy or aromatase inhibitor + CDK4/6 inhibitor therapy, but the patient declined, continuing TCM until October 2024. During this time, the tumor ulcerated, bled, and grew to 20.5 × 6.98 × 18.17 cm (Figure 1B), leading to severe anemia and septic shock by October 2024. Emergency management included blood transfusion, administration of ceftriaxone for infection control, and surgical debridement. Systemic therapy was initiated on October 26, 2024, consisting of exemestane (25 mg/day) and capecitabine (1250 mg/m2, 2 times a day/14 days on/21 days off). Palbociclib (75 mg/day, 21/28 days) was added on November 22, 2024. Despite systemic therapy, the tumor remained uncontrolled, with persistent bleeding and rapid progression.
Given the tumor’s massive size, irregular shape, and central ulceration with active bleeding, traditional radiotherapy approaches were deemed inadequate. Therefore, we employed mLRT, with the treatment plan designed and optimized on the Elekta Monaco 6.0 treatment planning system. This technique was chosen to achieve rapid hemostasis and local tumor control while sparing critical normal tissues such as the brachial plexus, ribs, and lungs. Tumor gross target volume (GTV) was independently contoured by 2 senior radiation oncologists on CT images (3 mm slice thickness) in the Elekta Monaco 6.0 system, with the final contour confirmed via double physician review and no inter-observer differences identified. Tumor volume was automatically calculated by the system using a voxel-based calculation method, and the maximum tumor dimensions were measured on the axial cross-section with the largest tumor area for clinical assessment of tumor regression. Serial volume measurements and target contouring were repeated at each treatment time node to dynamically evaluate changes in tumor and internal target volume (ITV) size. The mLRT lattice vertices (high-dose radiation regions) were designed with the primary principle of protecting the chest wall and skin, and the overall distribution was arranged along the tangential direction of the chest wall. Key design parameters were standardized as follows: lattice vertex diameter: 1 cm; inter-vertex spacing: 2 to 3 cm; normal tissue spacing requirement: all lattice vertices were placed at a distance of more than 1 cm from the skin, ribs, and chest wall to minimize normal tissue irradiation.
Dose Distribution Target
The tumor core at the lattice vertices was prescribed a single-fraction dose of 1500 cGy, with the peripheral tumor tissue around the vertices receiving a dose of 400 to 500 cGy;
Lattice Quantity Adjustment
The number of lattice vertices was dynamically adjusted during plan optimization and dose calculation to ensure the above dose distribution targets were achieved while maintaining strict normal tissue protection.
The first course of mLRT consisted of 15 Gy delivered in 3 fractions (total dose 45 Gy) with alternate-day irradiation (1 fraction every 2 days). Daily Cone Beam CT image guidance was used to verify tumor position, and adaptive replanning was performed before each fraction based on the latest clinical and imaging findings.
Adaptive replanning was a core component of the mLRT regimen, with a dedicated planning CT scan acquired for the patient prior to each mLRT fraction (alternate-day scanning) to guide plan adjustments. The fundamental principles for adaptive replanning were based on dynamic changes in the tumor and clinical status, with specific adjustment strategies as follows:
Tumor Morphological, Volumetric, and Bleeding Point Changes
Each fraction of 15 Gy irradiation induced clinically significant tumor regression, confirmed by direct visual observation and physical palpation immediately post-irradiation, accompanied by progressive tumor necrosis and exudation of the ulcerative lesion. These changes resulted in continuous volumetric reduction and morphological alterations of the ulcerative tumor, alongside dynamic shifts and gradual attenuation of active bleeding foci. The treatment plan was adaptively adjusted to match the newly changed tumor shape and volume, ensuring optimal coverage of the residual tumor tissue.
Bleeding Point Targeting
Active bleeding foci of the ulcerated tumor exhibited dynamic positional and intensity changes after each irradiation session, which were assessed through direct visual observation of the ulcerative surface, gentle physical palpation (avoiding iatrogenic bleeding), and quantification of daily blood loss in wound dressings. The high-dose lattice vertex regions were precisely repositioned to the newly identified active bleeding sites, with the number and dose weighting of vertices optimized accordingly to achieve accurate and targeted hemostatic radiation delivery.
Normal Tissue Protection Adaptation
The giant ulcerated tumor presented an irregular, non-spherical shape with local protrusions adjacent to the chest wall; plan adjustments were optimized to avoid irradiation of the chest wall, skin, and other normal tissues, with the lattice vertex distribution and dose weighting modified to match the altered tumor-normal tissue interface after each fraction.
The first 3 mLRT fractions were delivered on October 30, November 1, and 4, 2024, with adaptive replanning performed for each fraction based on the above principles.
The reporting of this case report conforms to the CARE (Case Report) Guidelines (EQUATOR Network) 9 (Supplementary document attached). All relevant items of the CARE Guidelines have been strictly followed to ensure the completeness and transparency of the case report.
Results
The therapeutic response demonstrated the remarkable efficacy of modified lattice radiotherapy, with this approach playing a pivotal role in achieving both rapid hemostasis and significant tumor regression. Following the initial 3-fraction lattice radiotherapy delivered on November 5, 2024, the spatially fractionated technique produced immediate clinical benefits—halting the patient’s life-threatening tumor bleeding within 5 days while simultaneously initiating rapid tumor shrinkage. In addition, the treatment response showed a characteristic pattern of progressive and sustained tumor reduction: a 24.7% volume decrease was achieved on November 8, 2024 (a week post-therapy), right after the initial mLRT course, with a further reduction to 39.7% from the baseline on December 3, 2024 (a month post-therapy). The tumor volume was reduced by 61.8% from the baseline by February 6, 2025 (4 months post-therapy). Notably, by March 18, 2025, near-complete epithelialization of the ulcerative lesion was achieved; imaging prior to the second course of conventional radical radiotherapy revealed only minimal residual tumor tissue remaining at the primary site. By March 31, 2025, the tumor volume had been reduced by 92.9% from the baseline, and a complete response (CR) was attained after the completion of the 2 sequential radiotherapy courses, with no detectable tumor tissue left, which further confirms the durable and potent antitumor effect of the modified lattice radiotherapy-based combined approach. As of the time of writing, the patient has not shown any recurrence or metastasis in clinical practice and is still undergoing endocrine therapy. She declined formal written feedback but verbally expressed appreciation for symptom relief during post-treatment visits.
The patient developed grade II radiation dermatitis (Common Terminology Criteria for Adverse Events, CTCAE v5.0, maximum grade observed during combined radiotherapy), which resolved completely within 6 weeks with topical gel and spray application. Transient numbness of the right upper limb occurred after re-irradiation, presenting with a maximum Numerical Rating Scale (NRS) score of 4; this symptom improved to an NRS score of 2 following mecobalamin administration and resolved fully by May 2025.
Figure 2 depicts the serial non-contrast CT imaging timeline of the right breast tumor throughout the entire treatment and follow-up period, while Figure 3 quantifies the dynamic changes in tumor volume with corresponding percentage reductions at key clinical time points. Figure 4 displays the treatment plan in the mLRT treatment period.
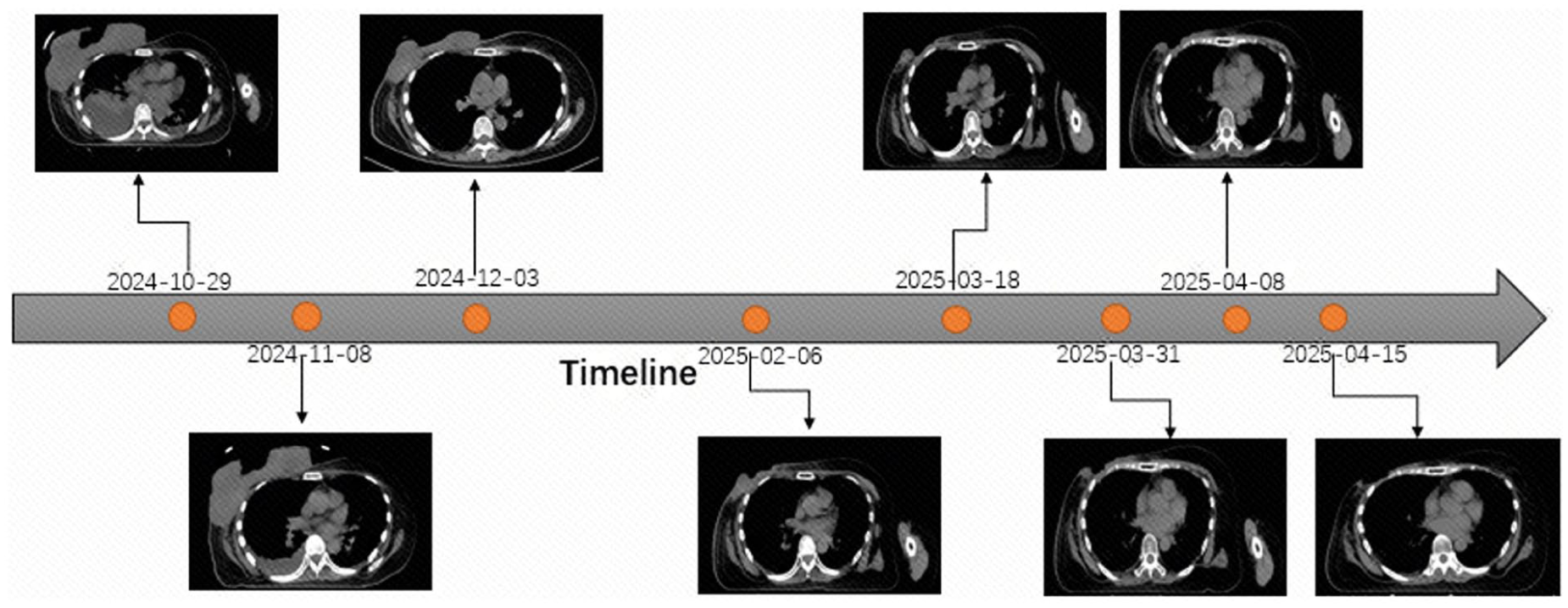
Coronal CT images of the UGBC during treatment and follow-up in chronological order. October 29, 2024: Pre-mLRT, active tumor bleeding with massive ulcerative lesion. November 8, 2024: A week post 3 fractions of mLRT (mLRT completion), complete hemostasis, and initial tumor regression (24.7% volume reduction vs pre-treatment baseline). December 3, 2024: 4 weeks post-mLRT completion, modest tumor regression (39.7% volume reduction vs pre-treatment baseline). February 6, 2025: 14 weeks post overall radiotherapy initiation, significant tumor regression (61.8% volume reduction vs pre-treatment baseline). March 18, 2025: Near-complete epithelialization of the ulcerative lesion, minimal residual tumor tissue at the primary site. March 31, 2025: 5 months post-mLRT, minimal residual disease (MRD) (92.9% volume reduction vs pre-treatment baseline). April 8, 2025: Tumor nearly completely regressed at the primary lesion. April 15, 2025: Clinically stable condition, no evidence of local recurrence or distant metastasis.

Dynamic trend of tumor volume of the UGBC from pre-treatment to 8-month follow-up. At 1 week after mLRT completion (November 8, 2024): 75.3% of the baseline volume. At 4 weeks after mLRT completion (December 3, 2024): 60.3% of the baseline volume. At 14 weeks post-baseline (February 6, 2025): 38.2% of the baseline volume. At 20 weeks post-baseline (March 18, 2025): 14.3% of the baseline volume. At 5 months post-baseline (March 31, 2025): 7.1% of the baseline volume. At 5 months and 3 days post-baseline (April 3, 2025): 7.1% of the baseline volume. At 5 months and 15 days post-baseline (April 15, 2025): 7.0% of the baseline volume.

Dose distribution of 3 sequential VMAT-based mLRT treatment plans for the UGBC (Plan 1/2/3 for the first/second/third fraction, respectively).
After the subsequent conventional radiotherapy, we analyzed the radiation doses to OARs from the combined treatment of mLRT and conventional radiotherapy. For the right lung, mLRT achieved exceptional protection with a V20 of 0.01% and a mean dose of 462.8 cGy, while combined treatment increased V20 to 20% (still below the 30% threshold) and the mean dose to 1252.1 cGy, maintaining safety. The chest wall showed minimal exposure during mLRT (V30 = 1.67%) but approached the upper limit post-conventional radiotherapy (V30 = 30.68%), necessitating close monitoring for late toxicity. Most critically, the brachial plexus exhibited concerning dose escalation: initial mLRT delivered D0.03cc = 3000 cGy (≈30 Gy) and D3cc = 1974.5 cGy (≈19.75 Gy), which surged to D0.03cc = 4759 cGy (≈47.6 Gy) and D3cc = 2885.8 cGy (≈28.86 Gy) after combined treatment, significantly exceeding single-fraction tolerance limits (typically ⩽ 26-30 Gy) and approaching/moving beyond conventional fractionation thresholds (⩽80-90 Gy EQD2). The findings underscore the importance of balancing aggressive tumor control with precise normal tissue protection, particularly for critical structures adjacent to high-dose regions.
Discussion
Rationale for the 45 Gy/3 Fractions Regimen in Modified Lattice Radiotherapy and Individualized Dose Optimization
While conventional spatially fractionated radiotherapy (SFRT) typically employs single-fraction doses of 10 Gy or 2 fractions totaling 20 Gy to achieve hemostasis in breast cancer, 10 this case employed a modified lattice radiotherapy (mLRT) regimen of 15 Gy × 3 fractions (total 45 Gy). 2 This dosing strategy was tailored to the tumor’s ultra-large ulcerated volume with life-threatening active hemorrhage and irregular morphology and guided by a stepwise approach of real-time post-fraction assessment of hemostatic response and tumor regression, rather than adherence to fixed conventional dose schemas.
The tumor in this case was exceptionally large, irregularly shaped, and featured central ulceration with active bleeding. Conventional radiotherapy with standard doses was technically straightforward, but delivering the exceptionally high ablative doses required for rapid hemostasis via this approach was unfeasible, as it would risk excessive radiation exposure and dose-limiting toxicity to adjacent critical organs. The modified lattice technique was specifically designed to address these challenges by enabling precise delivery of high doses to the tumor core while significantly sparing surrounding normal tissues.7,8,10,11
Although the biologically effective dose (BED) of this regimen exceeded 200 Gy, the spatial fractionation design ensured that the high-dose region (approximately 1500 cGy) was strictly confined to the tumor core, with surrounding normal tissues receiving substantially lower doses (< 1000 cGy in low-dose regions). This approach was further refined through an iterative, response-adapted process:
This stepwise response allowed for dynamic adjustment of the treatment plan, with the high-dose region being modified in real-time based on CT imaging and clinical observations to target residual bleeding points and tumor tissue.
This demonstrates that in cases of ultra-large, ulcerative breast cancer with active bleeding, the optimal dose is not absolute but should be determined based on a comprehensive assessment of tumor response, toxicity, and patient status. The mLRT’s ability to concentrate dose precisely while sparing normal tissues allowed for safe escalation beyond conventional SFRT doses, achieving superior therapeutic effects.11-14
The Necessity and Safety of Re-Irradiation With Conventional Fractionation After Modified Lattice Radiotherapy
Following the initial 3-fraction mLRT course, a second course of conventional fractionation radiotherapy (36 Gy/20 fractions) was administered to target residual disease. This decision was driven by the persistence of residual tumor despite significant regression after mLRT and the need for further local control to prevent regrowth and potential complications. Systemic therapy with endocrine agents (exemestane) and a CDK4/6 inhibitor (palbociclib) was continued during this period, although their full therapeutic effect typically requires 2 to 3 months.15,16
Special attention was given to OAR protection, particularly the brachial plexus, which is highly susceptible to radiation-induced neuropathy. The maximum dose to the brachial plexus was strictly limited through careful treatment planning and daily image guidance. Despite concerns about cumulative dose (the brachial plexus ultimately received approximately 45 Gy, exceeding its conventional tolerance), the patient tolerated the treatment well, with only grade II radiation dermatitis (resolved within 2 weeks) and transient right upper limb numbness (NRS score reduced from 4 to 0 within 6 months).17,18
Imaging follow-up confirmed continued tumor regression after re-irradiation, with the ulcerated lesion shrinking to near-complete epithelialization by March 18, 2025. This suggests that carefully planned re-irradiation with conventional fractionation can be safely administered after mLRT in selected cases, provided that OAR constraints are meticulously observed and treatment response is closely monitored. The case highlights the importance of individualized OAR protection strategies, particularly in patients with large tumors requiring multiple courses of radiotherapy.1,19,20
Synergistic Effects of Systemic Therapy and Radiotherapy in Disease Management
Systemic therapy played a critical role in suppressing systemic disease progression throughout the treatment course. Although endocrine therapy (exemestane) and CDK4/6 inhibitors (palbociclib) have a slower onset of action (typically 2-3 months for significant effects), their combination with radiotherapy provided a synergistic benefit21,22:
The mLRT achieved rapid local control and hemostasis within 5 days, stabilizing the patient’s critical condition and preventing life-threatening complications such as hemorrhagic shock and sepsis.
This immediate tumor control created a therapeutic window for systemic therapy to exert its effects, preventing distant metastasis and managing micrometastatic disease.8,10,22,23
Follow-up imaging showed no signs of recurrence or metastasis, confirming the effectiveness of this combined approach in achieving both short-term local control and long-term disease management. 1
The choice of a relatively mild endocrine regimen (exemestane + capecitabine) was tailored to the patient’s advanced disease stage, poor general condition, and treatment tolerance. This underscores the importance of individualizing systemic therapy based on patient factors, balancing efficacy with toxicity to optimize outcomes in frail or heavily pretreated patients.
The Role of Interval Irradiation and Adaptive Planning in Enhancing Safety and Efficacy
A key feature of this treatment strategy was the use of interval irradiation—spacing out the fractions of mLRT and re-irradiation to allow for tumor response assessment, normal tissue repair, and adaptive replanning. This approach offered several advantages.
Clinical Monitoring
The intervals allowed close observation of tumor regression and hemostasis, enabling timely adjustments to the treatment plan based on observed response.
Normal Tissue Recovery
Intervals permitted partial recovery of irradiated normal tissues, potentially reducing late toxicity. 24
Adaptive Optimization
Imaging (CT/magnetic resonance imaging [MRI]) performed during intervals guided modifications to the treatment plan, ensuring continued tumor coverage while minimizing exposure to normal structures. Given the high doses used in mLRT, this adaptive strategy was particularly important for maintaining safety and efficacy throughout the course of therapy.
This “adaptive radiotherapy” approach, facilitated by frequent imaging and flexible planning, represents a best practice for managing complex cases where standard rigid protocols may be suboptimal. 25
Evidence of Systemic Antitumor Effects (“Abscopal Effect”)
An intriguing observation in this case was the evidence of a systemic antitumor effect, commonly referred to as the “abscopal effect,” following mLRT. Three months after treatment initiation, the patient exhibited regression not only in the primary breast tumor but also in contralateral axillary lymph nodes and resolution of pleural effusion—a distant site not directly targeted by radiation.26,27 While the exact mechanisms remain under investigation, this phenomenon is hypothesized to result from the release of tumor antigens and activation of systemic immune responses triggered by localized high-dose radiation. 15
This observation aligns with emerging evidence that SFRT, particularly when combined with immunotherapy, can enhance systemic antitumor immunity. Although the patient did not receive concurrent immunotherapy in this case, the spontaneous abscopal response suggests that mLRT may have immunomodulatory effects that warrant further exploration.8,28,29
Transformative Potential of Modified Lattice Radiotherapy in Advanced Breast Cancer Management
This case demonstrates that mLRT can safely and effectively achieve rapid hemostasis and significant tumor regression in patients with ultra-large, ulcerative, and previously untreatable breast cancer. By converting a previously inoperable and life-threatening condition into one amenable to further palliative or potentially curative interventions, mLRT expands the therapeutic options for patients who have exhausted conventional approaches.1,3,30
Moreover, the successful integration of mLRT with systemic therapy and supportive care highlights its potential to buy critical time for systemic treatments to take effect, ultimately improving patient outcomes. The ability to deliver high doses precisely while sparing critical structures also opens avenues for combining mLRT with other emerging therapies (eg, immunotherapy, targeted agents) in future studies.
Conclusion
The mLRT achieves rapid hemostasis and marked tumor regression in a chemotherapy-refusing patient with stage IIIC ulcerative giant breast cancer, with complete bleeding control within 5 days and a significant tumor volume reduction till CR at follow-up. Combined with conventional radiotherapy and systemic therapy, mLRT safely balances aggressive local tumor control and normal tissue protection, even for ultra-large ulcerated lesions. This multimodal strategy creates a therapeutic window for systemic therapy to exert efficacy and elicits potential abscopal effects. The mLRT represents a promising palliative and salvage approach for unresectable ulcerative breast cancer with life-threatening hemorrhage, expanding therapeutic options for patients ineligible for standard treatments.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234261441378 – Supplemental material for Rapid Hemostasis and Significant Tumor Regression in Ulcerative Breast Cancer Via Combined Modified Lattice Radiotherapy and Systemic Therapy: A Case Report
Supplemental material, sj-docx-1-bcb-10.1177_11782234261441378 for Rapid Hemostasis and Significant Tumor Regression in Ulcerative Breast Cancer Via Combined Modified Lattice Radiotherapy and Systemic Therapy: A Case Report by Weiwei Sang, Yanyan Shi, Bin Wang and Lihua Zhang in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
The authors would like to express sincere gratitude to the medical staff of the Department of Radiation Oncology, Jiahui International Cancer Center, Shanghai, for their dedicated efforts in patient care, treatment implementation, and data collection throughout the study. The authors also appreciate the patient’s willingness to participate in this case report and share her clinical information for academic advancement. In addition, the authors thank the Ethics Committee of Shanghai Jiahui International Hospital for their guidance on research ethics and compliance. Finally, the authors acknowledge the professional English editing service provided by Dr Volker R. Rudat, with expertise in medical oncology, which enhanced the clarity and academic rigor of the manuscript.
Ethical Considerations
This study involves the retrospective analysis of a single patient’s clinical data and treatment outcomes, which was reviewed and approved by the Ethics Committee of Shanghai Jiahui International Hospital.
Consent to Participate
The patient was fully informed of the purpose of the study and provided written informed consent for the use of her de-identified clinical information, treatment records, and imaging data in this research.
Consent for Publication
Written informed consent was obtained from the patient prior to the initiation of all therapeutic interventions (including modified lattice radiotherapy, volumetric-modulated arc therapy, and systemic therapy). The patient was fully informed of the nature, risks, benefits, and alternative treatment options of the proposed multimodal therapy, as well as the purpose of this case report and the use of her de-identified clinical data, imaging findings, treatment records, and photographic materials for research and publication purposes. The patient provided explicit written consent for the collection, analysis, and presentation of her de-identified clinical information in this manuscript and for its subsequent publication in a peer-reviewed journal. A signed copy of the informed consent form is retained in the institutional medical record archive of the Department of Radiation Oncology, Shanghai Jiahui International Cancer Center, and is available for review by the journal editorial office upon formal request.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The de-identified clinical data (eg, radiotherapy parameters, laboratory test results, follow-up records) and imaging data supporting the findings of this case report are stored in the electronic medical record system of Shanghai Jiahui International Hospital. Due to privacy restrictions and compliance with the Health Insurance Portability and Accountability Act (HIPAA) and local medical privacy regulations, the raw data cannot be publicly shared. However, reasonable requests for access to the de-identified data may be submitted to the corresponding author (Lihua Zhang,
Supplemental Material
Supplemental material for this article is available online.
AI Tool Disclosure
The authors used Grammarly (Version 1.0) solely for language refinement, including grammar correction, punctuation adjustment, and improvement of sentence fluency in the manuscript. No artificial intelligence (AI) tools (including but not limited to large language models, data analysis software with AI functions, or specialized scientific research AI tools) were used to generate, modify, or analyze any scientific data, clinical results, imaging findings, radiotherapy planning parameters, or statistical outcomes reported in this study. All core scientific content and research data are derived from actual clinical practice and manual analysis by the authors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
