Abstract
Background:
Adenomyoepithelioma (AME) of the breast is a rare biphasic neoplasm characterized by proliferation of epithelial and myoepithelial cells. Although most AMEs demonstrate indolent behavior, malignant transformation has been reported, and diagnostic uncertainty often complicates clinical management. Given the rarity of AME, most data are limited to case reports and small series, leaving optimal management undefined.
Objectives:
To describe the clinical presentation, imaging characteristics, histopathologic features, and outcomes of patients with breast AME.
Design:
Retrospective single-institution case series.
Methods:
We retrospectively reviewed 15 patients diagnosed with breast AME between 2010 and 2023. Demographic, clinical, imaging, core needle biopsy (CNB) findings, surgical pathology, and outcomes data were analyzed.
Results:
The median age at diagnosis was 56 years. More than half of the patients (53.3%) were asymptomatic at presentation. Mammography most frequently demonstrated discrete ovoid masses (61.5%), and ultrasound most often showed hypoechoic lesions (66.7%) with lobulated margins (50%). Core needle biopsy identified benign AME in 38.4% of cases, while 61.5% yielded indeterminate findings with AME included in the differential. Five patients underwent CNB alone without surgical excision. Of the 9 patients who underwent both CNB and excision, 6 (66.7%) had AME on final pathology, whereas 3 were upgraded (2 invasive carcinoma, 1 atypical ductal hyperplasia). Of the 3 cases with cytologic atypia on CNB, 1 was upgraded to invasive carcinoma. Over a median follow-up of 10 months, no recurrences, metastases, or breast cancer-related deaths were observed.
Conclusions:
Breast AME generally follows a benign clinical course, but diagnostic challenges exist due to histologic heterogeneity. Core needle biopsy features such as cytologic atypia may indicate higher malignancy risk. These findings highlight the ongoing dilemma of when observation is sufficient versus when surgical excision is warranted. Our results add to the limited body of evidence and align with prior reports, underscoring the need for larger, multicenter studies with longer follow-up to clarify long-term outcomes and optimal management strategies.
Introduction
Adenomyoepithelioma (AME) is a rare breast neoplasm characterized by biphasic proliferation of ductal epithelial and myoepithelial cells. 1 Although most AMEs behave in a benign manner, they carry a recognized risk of local recurrence, malignant transformation, and, in rare cases, distant metastasis.2 -4 Clinically, patients often present with a painless, palpable breast mass, though a substantial proportion are asymptomatic and diagnosed incidentally on screening mammography or other imaging modalities. 5
Accurate diagnosis remains challenging. Imaging findings are nonspecific and histologic heterogeneity on core needle biopsy (CNB) can obscure the diagnosis. Immunohistochemistry is often required to confirm the biphasic cellular composition.6 -9 Given these diagnostic limitations, CNB findings may under- or overestimate malignant potential, complicating treatment decisions.
Management guidelines for AME remain undefined. Surgical excision with close surveillance is generally recommended, largely due to the malignant potential reported in case studies, including metastases to the lungs, thyroid, bone, and brain.10,11 Some authors advocate for routine surgical excision of all AMEs to mitigate the risk of underdiagnosis and malignant transformation, while others propose a more conservative approach in cases with benign histology, particularly for small, asymptomatic lesions. This lack of consensus underscores the clinical uncertainty faced by both patients and providers.8,11 Additionally, existing literature is limited to case reports and small case series, restricting the ability to generalize outcomes and inform evidence-based strategies.
The present study aims to characterize the clinical, radiologic, and histopathologic features of breast AME in a single-institution cohort. By contributing additional series-level data, we seek to expand the current understanding of AME, highlight diagnostic challenges, and provide insights to help guide management decisions and improve long-term patient outcomes. The reporting of this study conforms to the PROCESS guidelines. 12
Methods
We conducted a retrospective chart review of patients treated within the Novant Health system from 2016 to 2022. Inclusion criteria included female patients ⩾ 18 years-old, with a diagnosis of, or suspicion for, AME based on initial CNB or final surgical pathology. Exclusion criteria included patients who underwent surgical treatment outside of the Novant Health system or cases in which AME was not included in the pathologic differential diagnosis.
Data were extracted from the electronic medical record (EMR). Patients were identified via EMR query. Clinical variables collected included demographic, presenting symptoms, imaging findings, histopathology, treatment, and outcomes. All pathology slides were reviewed by an in-house pathologist and underwent a second internal review in accordance with institutional protocol. When diagnostic uncertainty persisted, cases were referred for external pathology review to confirm the diagnosis. Outcomes measured included recurrence, metastasis, and survival. Descriptive statistics were used to summarize the demographic and clinical variables. Frequencies and percentages were computed for any categorical data. No inferential statistical tests were conducted. All statistical analyses were performed using Microsoft Excel (version 16.74). 13 No scientific data has been generated or modified using AI. This case series has been reported in line with the PROCESS guidelines. 12
All patient information was de-identified and informed consent was not required. Patient data was not shared with third parties. This study received ethical approval from the Novant Health Institutional Review Board (IRB Protocol ID: 22-2118), ensuring compliance with ethical standards and appropriate research procedures.
Results
A total of 16 patients were identified, with 1 patient excluded after pathology review findings were inconsistent with true AME. The final cohort comprised 15 patients with a median age at diagnosis of 56 years (range, 40-89) and 60% were postmenopausal. Regarding clinical presentation, 8 patients (53.3%) were asymptomatic at diagnosis, with lesions detected on screening mammogram. The remaining 7 patients were symptomatic with a palpable breast mass, pain, or both (Table 1).
Patient characteristics.

Tumor size ranged from 1.2 to 36 mm (mean 14.3 mm). Among tumors 20 mm or larger, 75% were symptomatic, compared with 36.4% of tumors smaller than 20 mm. Most lesions (86.7%) were detected via mammography, while 2 were identified incidentally on MRI or CT. Of the 11 patients who underwent both mammogram and ultrasound during the diagnostic process, 8 (72.7%) demonstrated discrete, ovoid masses visible on both modalities. Additional mammogram findings included asymmetry with calcifications in 2 patients (15.4%), asymmetry without calcifications in 2 (15.4%), and calcifications alone in 2 (15.4%). Of the 12 patients who underwent ultrasound, 11 (91.2%) demonstrated hypoechoic masses, with internal vascularity in 33.3%, and lobulated margins in 50.0% (Table 2). Representative mammographic and ultrasonographic images from a diagnosed case of AME are shown in Figures 1 and 2.
Clinical presentation and imaging findings.

US: ultrasound, MRI: magnetic resonance imaging, CT: computed tomography.
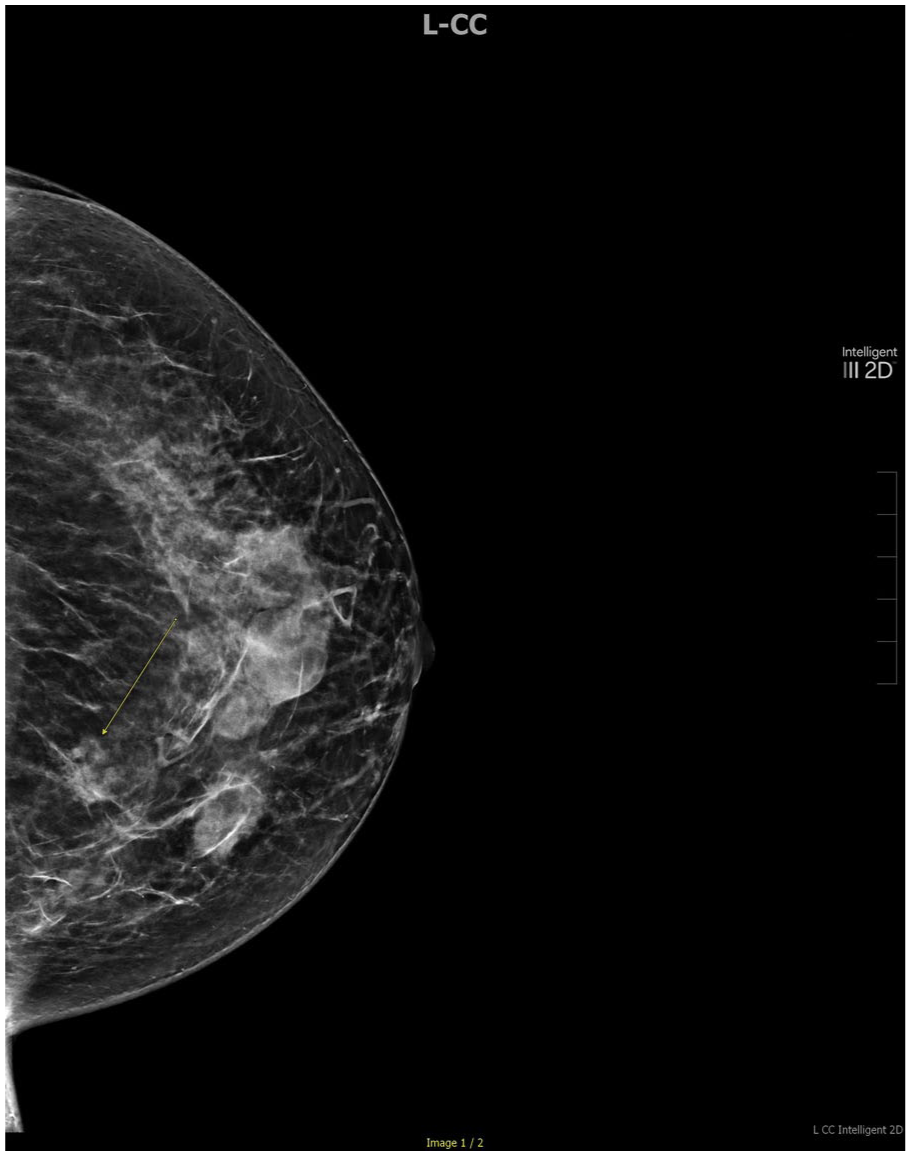
Left breast mammogram demonstrates an asymmetric mass (yellow arrow) in a patient with multiple stable well-circumscribed cysts in the left upper medial breast without associated calcifications.
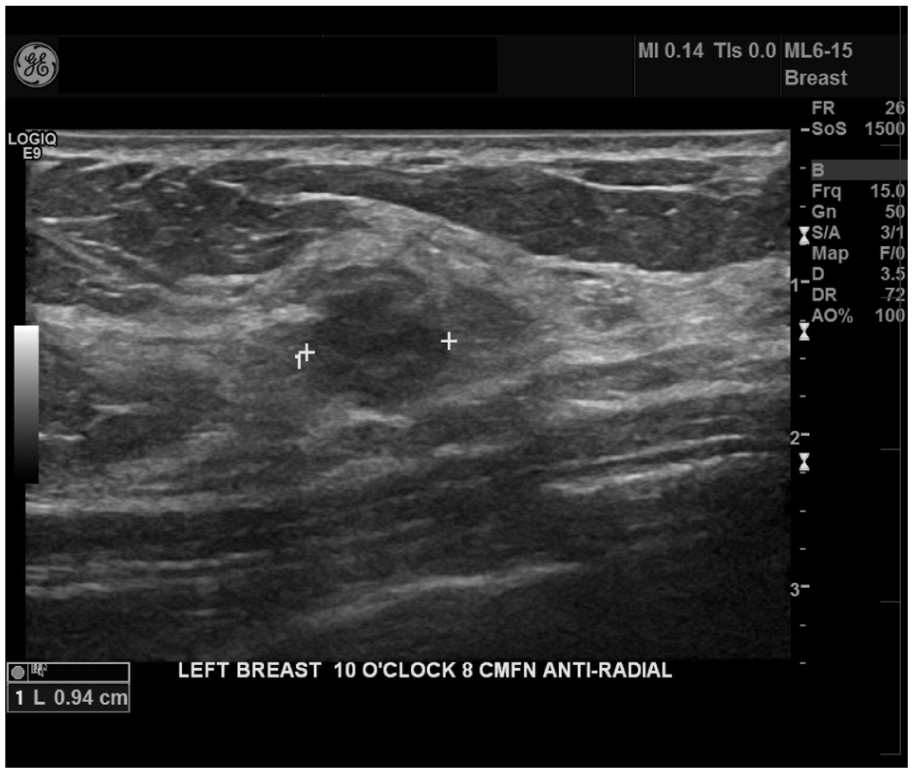
Left breast ultrasound confirms a 13 mm hypoechoic, lobulated mass in the upper inner left breast.
Following imaging, 13 patients underwent CNB, while 2 required surgical biopsy due to lesion location. Histological findings on CNB included spindle-shaped epithelial or myoepithelial cell configuration (61.5%), prominent papillary component (46.2%), and sclerosis/fibrosis (38.5%). Cellular atypia was observed in 3 patients (15.4%) and calcifications were noted in 1 (7.69%) (Table 3). Representative histologic and immunohistochemical images from a diagnosed case are shown in Figure 3A to C.
Management and histologic findings.

CNB: core needle biopsy, AME: adenomyoepithelioma, ADH: atypical ductal hyperplasia.

(A) Excisional biopsy specimen showing a multilobulated, circumscribed cellular proliferation (H&E, 40×). (B) Biphasic cell population comprised of compressed, cytologically bland tubules with a prominent myoepithelial component in the background (H&E, 200×). (C) Well-formed tubules lined by conspicuous myoepithelial cells (H&E, 200×).
CNB rendered a diagnosis of benign AME in 5 patients (38.4%), whereas 8 (61.5%) yielded indeterminate results with AME included in the differential diagnosis. Among the 5 patients with a CNB diagnosis of benign AME, 3 elected for observation, while 2 proceeded with surgical excision which confirmed a definitive diagnosis of AME. Overall, 10 patients (66.7%) underwent surgical excision and 5 (33.3%) were managed with observation alone.
Final pathology following excision confirmed AME in 7 of 10 patients (70%), while 2 were upgraded to invasive carcinoma and 1 to atypical ductal hyperplasia (ADH). All 3 upgraded patients were symptomatic at diagnosis, presenting with a palpable mass or tenderness, and mean lesion size (15.3 mm) was slightly larger than the overall cohort average (14.3 mm). On imaging, while all appeared as round, circumscribed masses on mammography, ultrasound findings varied: 1 was hypoechoic with lobulated margins and internal vascularity, 1 was hypoechoic without vascularity, and 1 was an oval, hypoechoic mass with ductal extension and calcifications. On CNB, all 3 upgraded patients had papillary architecture; 2 also showed cellular atypia, including 1 with increased mitotic activity. Notably, all 3 cases with cellular atypia on CNB ultimately upgraded on final surgical pathology.
The median follow-up duration was 10 months (range, 0-68). Three patients were lost to follow-up. No recurrences or metastases were observed during the study period. One patient died of advanced age, but no deaths were attributable to AME or breast cancer.
Discussion
Breast AMEs are rare tumors characterized by biphasic proliferation of epithelial and myoepithelial cells. They are generally considered benign and indolent, although rare cases of malignant transformation or local recurrence have been reported.2,3,14,15 In our cohort, no recurrences, metastases, or breast cancer-related deaths occurred during follow-up, supporting prior reports of its typically favorable clinical course. However, the relatively short median follow-up interval in this study (10 months) limits our ability to fully assess long-term outcomes, particularly the risk of late recurrence. Based on limited data, AME has been shown to recur primarily between 8 months and 5 years following resection.16,17 Recurrences of AME have been shown to be either benign or malignant AME. One literature review from 2017 by Logie et al 18 demonstrated a median time to recurrence of 2.3 years. Longer observation periods are therefore necessary to more definitely characterize the natural history of AME.
Clinically, AMEs are most often reported as solitary, palpable masses in middle-aged to older women.11,19 Our cohort reflected a similar age distribution, but more than half of patients (53.3%) were asymptomatic at diagnosis. Only 3 patients (20%) presented with a painless, solitary breast mass. There was also variation found in tumor size which played a role in symptom presentation. We found that tumors ⩾ 20 mm were more likely to be symptomatic, with 75% presenting as palpable masses (with or without tenderness), compared with 36.4% of tumors < 20 mm. This finding aligns with previous observations that larger tumors are more likely to produce symptoms, whereas smaller lesions are frequently detected incidentally.7,20
Imaging findings for AME are generally nonspecific. Mammography typically revealed round, oval, or lobulated masses with smooth or occasionally indistinct margins and rare calcifications, while ultrasound often showed solid, hypoechoic lesions with variable vascularity.11,19 Our findings are largely consistent with these descriptions. Among the 11 patients who underwent both mammography and ultrasound, 8 (72.7%) had discrete, ovoid masses visible on both modalities. The next most common mammographic finding was an asymmetry with or without associated calcifications in 4 patients (30.8%). Previous studies have reported rare calcifications in AME; in one case series of 13 cases, only a single patient demonstrated calcification, while the remainder presented as nonspecific masses.8,21 In contrast, 30.7% of our cohort demonstrated calcifications. 22 This discrepancy may be partially explained by the presence of 2 cases that were ultimately upgraded to invasive carcinomas on final pathology.
On ultrasound, lesions were predominantly hypoechoic (66.7%), often had lobulated margins (50%), and one-third exhibited internal vascularity. These findings are comparable to previously published data demonstrating that AMEs often appear as well defined, hypoechoic masses with lobulation or irregular margins. 19 Furthermore, the presence of internal vascularity on Doppler in one-third of our cases has also been described as a useful, though nonspecific, feature suggesting solid, proliferative lesions.11,19 Overall, the imaging characteristics of AMEs are variable and can overlap with those of malignant breast lesions, underscoring the need for histopathologic confirmation to achieve definitive diagnosis.11,23,24
Histologically, AMEs demonstrate a biphasic proliferation of myoepithelial and epithelial cells, typically arranged in a well-circumscribed, often lobulated or tubular architecture.2,15 Morphologic heterogeneity is common, with variation in the distribution of glandular and myoepithelial cells, lobulation, the presence of spindle-shaped myoepithelial cells, papillary architecture, tubular patterning, and associated fibrosis.3,14,15 The histologic diversity complicates a definitive diagnosis using CNB alone, and adequate sampling and careful pathologic review is essential to avoid missing focal malignant features.3,6
Although most AMEs are benign, prior studies have identified cytologic atypia, increased mitotic activity, and infiltrative growth as markers of atypical AME or malignant potential.4,6,19,25 Our findings similarly suggest that cytologic atypia on CNB may raise suspicion for malignancy on final pathology. Of the 15 included patients, 3 were found to have cytologic atypia on CNB; including one with both cytologic and cellular atypia and was upgraded on final pathology to invasive breast cancer, while the other 2 patients had a benign AME and an AME with atypia. Papillary architecture on CNB was present in all 3 patients whose final pathology was upgraded, though it was also observed in 3 cases confirmed as AME, highlighting variability in its predictive value. Notably, all 3 upgraded cases were symptomatic at presentation, with a palpable mass or tenderness, suggesting a possible correlation between clinical presentation and risk of malignancy.
Our findings add to the ongoing debate regarding the need for routine surgical excision of all AMEs diagnosed on CNB. In our cohort, CNB established a diagnosis of benign AME in 5 patients, 3 of which opted for observation and 2 underwent surgical excision. Surgical pathology for those 2 patients confirmed a definitive diagnosis of benign AME. The remaining 8 who underwent CNB had indeterminate results that included AME in the differential; surgical pathology confirmed AME in 5, but 2 were upgraded to invasive carcinoma and 1 to ADH. While CNB plays a critical role in the initial evaluation, pathologic heterogeneity may obscure malignant components and lead to underdiagnosis.3,14,19 Historically, complete surgical excision has been recommended for all AMEs diagnosed on CNB, given the potential for malignant transformation or local recurrence, especially in the presence of atypical features.4,26,27 More recent studies, however, advocate for individualized management guided by multidisciplinary review and radiologic-pathologic concordance, reserving surgery for cases with atypia, suspicious imaging, or diagnostic uncertainty.19,26- 28
In our study, no recurrences, metastases, or breast cancer-related deaths were observed during the follow-up period. Importantly, all patients with a more definitive CNB diagnosis of benign AME who underwent surgical excision were confirmed as AME on final pathology, while upgrades occurred only in the indeterminate group. These findings support the possibility that select patients with heterogenous AME features on CNB may be managed with observation. However, indeterminate CNB results or concerning histologic features such as cytologic atypia, increased mitotic activity, infiltrative growth, or papillary architecture, based on current limited data, should still be considered for excision to rule out malignancy. Ultimately, our results support the potential for a more nuanced approach to management while underscoring the need for larger studies to define which patients may safely avoid surgery.
This study is limited by its retrospective design, small cohort size, rarity of the lesion, and relatively short follow-up period, which may limit our ability to assess long-term risks. In addition, this was a single-institution series, which may limit generalizability to broader patient populations. Diagnostic variability is another limitation as pathologic interpretation of AME can be challenging and CNB findings may not fully capture tumor heterogeneity. Selection bias is also possible, as patients referred for surgery may represent a population with more indeterminate or suspicious features.16,17 Further multi-institutional, prospective studies with standardized criteria and extended follow-up are needed to better define long-term outcomes and optimal management strategies.
Conclusions
Adenomyoepithelioma is generally regarded as a benign neoplasm with a low risk of malignancy, a finding supported by our cohort. In this study, we found no evidence of disease progression, metastasis, or death related to AME. Among patients with a more definitive diagnosis of AME on CNB, no upgrades were identified on final pathology. These results highlight important clinical implications: while surgical resection is the most recognized treatment approach, observation alone may be a reasonable option in carefully selected patients, particularly when CNB demonstrates typical heterogenous AME features. Incorporating radiologic-pathologic concordance may help reduce overtreatment in this rare entity. Future research with larger cohorts and longer follow-up is essential to refine criteria for conservative management and better define long-term outcomes.
Footnotes
Acknowledgements
Not applicable.
Ethical Considerations
This study received ethical approval from the Novant Health IRB (approval #22-2118).
Consent to Participate
All patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Consent for Publication
Not applicable.
Author Contributions
Lyndsay Cooper: Conceptualization, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing.
Saheli Parekh: Conceptualization, Data curation, Investigation, Writing – review & editing.
Korene Gbozah: Conceptualization, Writing – review & editing.
Patrick Wilson: Data curation, Writing – review & editing.
Sean Kisch: Investigation, Writing – review & editing.
Dipika Misra: Conceptualization, Writing – review & editing.
Kristina Shaffer: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
