Abstract
Background:
Human epidermal growth factor receptor 2 (HER2) is an important predictive and prognostic biomarker in breast cancer. Immunohistochemistry (IHC) is the preferred initial test due to its cost-effectiveness and simplicity. While fluorescence in situ hybridization (FISH) is the traditional gold standard test for HER2 gene amplification, DISH has emerged as an accepted alternative that allows evaluation under a standard light microscope.
Objectives:
To evaluate agreement between HER2 IHC (2+/3+) and DISH in node-positive primary breast cancers and compare findings with published IHC and FISH data.
Design:
Retrospective single-center cohort study using nationwide referral specimens.
Methods:
Cases of pathologically confirmed lymph node metastasized invasive breast carcinoma with HER2 IHC scores of 2+ and 3+ were retrieved. Interpretation of HER2 IHC was performed using the 2023 ASCO/CAP guideline. HER2 DISH was conducted and evaluated by the HER2/CEP17 signal ratio on primary tumors first, and on metastasized lymph nodes in cases of persistent technical failure.
Results:
Among 1,307 breast cancers, DISH detected HER2 amplification in 933 cases, including 92% (760) of IHC 3+ cases and 36% (173) of IHC 2+ cases. Seven cases with persistent technical failure on primary tumors were resolved when switching to lymph node specimens. Comparison with the meta-analysis data of IHC and FISH showed no significant differences, indicating that DISH is a reliable alternative to FISH.
Conclusion:
Our study demonstrates a high concordant rate between HER2 IHC and DISH in the IHC 3+ group and a low positive rate in the IHC 2+ group. We found no significant difference in the positive rates of HER2 IHC to DISH when compared with prior data of IHC to FISH, reaffirming the use of HER2 DISH as an effective and more accessible alternative to FISH in HER2 2+/3+ breast cancer.
Introduction
The human epidermal growth factor receptor 2 (HER2) gene, locating on band q21 of chromosome 17, encodes a type of tyrosine kinase receptor. Approximately 15% to 20% of breast cancers exhibit HER2 gene amplification and/or protein overexpression, and this HER2 status is both prognostic and predictive of response to HER2-targeted therapy.1 -4
The 2023 American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guideline recommends utilizing immunohistochemistry (IHC), bright-field in situ hybridization, or fluorescent in situ hybridization (FISH) as detection tests. The new guideline reaffirms the use of HER2 targeted therapy in patients with HER2 IHC 3+ and IHC 2+ with amplified in situ hybridization (ISH) while acknowledging the benefits of newer generation HER2 combined drugs in HER2-low patients. 3
Due to its cost-effectiveness and simplicity in performing and interpreting, many laboratories prefer using IHC as a screening test and perform ISH only if the IHC result is 2+ or 3+. For ISH tests, FISH is still considered a gold standard test for HER2 amplification. However, FISH has several limitations when compared with bright-field dual-color in situ hybridization (DISH).5,6 Several studies have demonstrated very high to almost perfect agreement on HER2 amplification between FISH and DISH, suggesting that DISH can be used as a more accessible alternative to FISH.7 -21
While there are extensive studies on the concordance of HER2 IHC and FISH, the existing literature on direct comparison between IHC and DISH is still lacking. Therefore, the objectives of our study are to provide additional insights into the agreement rate between equivocal (2+) and positive (3+) HER2 IHC and DISH in the context of lymph node metastasized breast cancer, and to compare the results with the existing data on the association between HER2 IHC and FISH.
Methods
Sample
All cases of lymph node metastasized invasive breast carcinoma with HER2 IHC scores of 2+ and 3+ in King Chulalongkorn Memorial Hospital (KCMH) and the Biobridge project, which received specimens from 86 hospitals across Thailand from 2015 to 2021, were included. As per the project protocol, all samples had appropriate pre-analytical factors, pathologically documented lymph node metastasis, and validated IHC assays were used to evaluate HER2. The formalin-fixed paraffin-embedded (FFPE) tissue blocks, H&E slides, and the initial HER2 IHC slides were retrieved. Two pathologists, KI and AP, independently reviewed the HER2 IHC slides according to the 2023 ASCO/CAP guidelines. Any discrepancies were resolved through joint review and consensus. If any problems interfered with slide interpretation, re-staining would be performed in KCMH, and the slides would be reevaluated. Twenty-three cases were reclassified to negative HER2 IHC, thus excluded, while 18 cases were reclassified from 3+ to 2+. In addition, 9 cases were excluded due to inadequate remaining invasive tumors. Ultimately, a total of 1,307 cases remained in the study, and HER2 DISH was then performed. Of these, 826 cases had a score of 3+, and 481 cases had a score of 2+.
IHC
The FFPE tissue blocks were sectioned into 3-μm-thick sections. Anti-HER2/Neu (4b5) antibody (Ventana Medical Systems, Inc, Tucson, Arizona; catalog number 05278368001) was applied as per the manufacturer’s protocol, using the automated Benchmark Ultra system (Ventana Medical Systems, Inc, Tucson, Arizona).
The interpretation of repeated HER2 IHC slides was done in the same way as the IHC slides received from the Biobridge project.
DISH
The INFORM HER2 Dual ISH probe cocktail (Ventana Medical Systems, Inc, Tucson, Arizona; catalog number 760-6072) was performed as per the manufacturer’s protocol on 4-μm-thick tissue sections, using the automated Benchmark Ultra system (Ventana Medical Systems, Inc, Tucson, Arizona). Quality assessment for HER2 and Centromere Enumeration Probe 17 (CEP17) signals in both internal control and tumor cells, as well as background staining, was conducted. If any interference was detected, repeat staining with proper adjustment was done. If the issue persisted, we switched to performing DISH on the lymph node specimen.
The interpretation of HER2 DISH slides was performed independently by 2 pathologists (KI and AP) according to the manufacturer’s validated protocol, using light microscopes with 40× and 60× objective lenses, and their consensus was used. The HER2 and CEP17 signals were enumerated in 20 invasive tumor cell nuclei. If the HER2/CEP17 ratio was within the range of 1.8 to 2.2, an additional 20 nuclei would be counted. The final HER2/CEP17 ratio of equal to or greater than 2.0 was regarded as positive or amplified, while the HER2/CEP17 ratio of less than 2.0 was interpreted as negative or nonamplified.
Statistical Analysis
The data were statistically analyzed using SPSS version 28 (IBM Corp., Armonk, New York). Metrics calculated were the concordant rate of positive IHC and DISH tests, the positive rate of HER2 DISH in the equivocal (2+) IHC group, 95% confidence intervals (CIs) for the concordant rate and positive rate, and Fisher’s exact test to compare the significance of associations between concordant rate and positive rate of IHC and DISH to IHC and FISH.
Result
Concordance of HER2 IHC and DISH
Patient age and DISH metrics, stratified by HER2 IHC score, are provided in Table 1. Of the 1,307 cases, 933 samples showed HER2 amplification by DISH. HER2 amplifications were detected in 92% of IHC 3+ cases (95% CI = 90.2%-93.9%) and 36% of IHC 2+ cases (95% CI = 31.7%-40.3%). The Fisher’s exact test demonstrated a nonrandom association in the data (P < .001). The result is summarized in Table 2, and a representative case with tumor heterogeneity is shown in Figure 1.
Patient age and HER2/CEP17 signals stratified by IHC score.
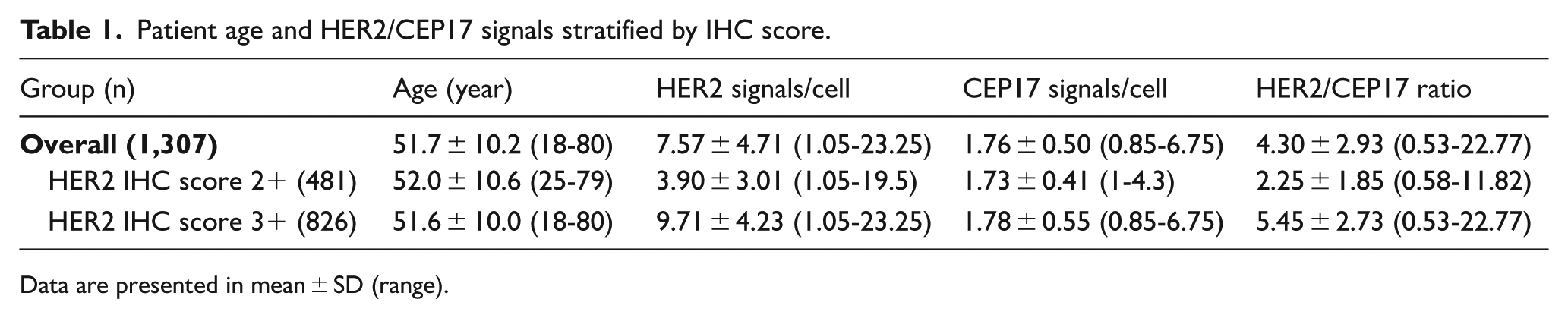
Data are presented in mean ± SD (range).
Result of HER2 IHC and DISH.

P-value < .001.

Tumor heterogeneity in HER2 gene amplification and expression. (A) Hematoxylin and eosin stain showing invasive breast carcinoma of no special type. (B) HER2 IHC revealing a positive 3+ result on the left and a negative 1+ result on the right. (C) HER2 DISH with red CEP17 signals and black HER2 signals displaying positivity in area corresponding to the HER2 IHC positive region. (D) HER2 DISH with red CEP17 signals and black HER2 signals showing negativity in area corresponding to the HER2 IHC negative region.
Persistent technical failure of DISH occurred in 7 primary-tumor samples (0.5%); repeat testing on matched lymph node specimens resolved all cases, as illustrated in Figure 2.

Persistent DISH failure in the primary tumor resolved by testing a metastasized lymph node. (A) Hematoxylin and eosin section of the breast showing poorly preserved carcinoma. (B) HER2 DISH on the primary tumor displaying only faint red CEP17 signals despite manufacturer-recommended troubleshooting to enhance black HER2 signals. (C–D) HER2 DISH on a metastatic lymph node from the same patient, demonstrating clear HER2 amplification.
Comparison Between IHC to DISH and IHC to FISH
Comparison of our IHC and DISH data to the data of IHC and FISH from the meta-analysis 22 was performed. The Fisher’s exact test indicated no statistically significant differences in either HER2 IHC score 2+ and 3+ groups. The comparative data are shown in Table 3.
Comparison of HER2 IHC and DISH to IHC and FISH.

Discussion
Our study aimed to evaluate the concordance between HER2 IHC and DISH in lymph node metastasized breast cancer, providing a comparative perspective against the well-established concordance between HER2 IHC and FISH. The findings could contribute to the ongoing discourse on optimizing HER2 testing methodologies to enhance diagnostic accuracy and therapeutic outcomes.
The results demonstrate a very high concordance between HER2 DISH and IHC in the IHC 3+ group and a low positive rate of HER2 IHC 2+ when compared with HER2 DISH. These data align well with the current recommendations on HER2 testing, which emphasize the reliability of IHC as a primary screening method, followed by ISH confirmation in equivocal cases. 3 Although there are minor discrepancies between our findings and the results of some prior studies, which are presented in Table 4, we believe these slight differences do not affect the testing recommendation.
Prior studies comparing HER2 IHC 2+ and 3+ to DISH in breast cancer.
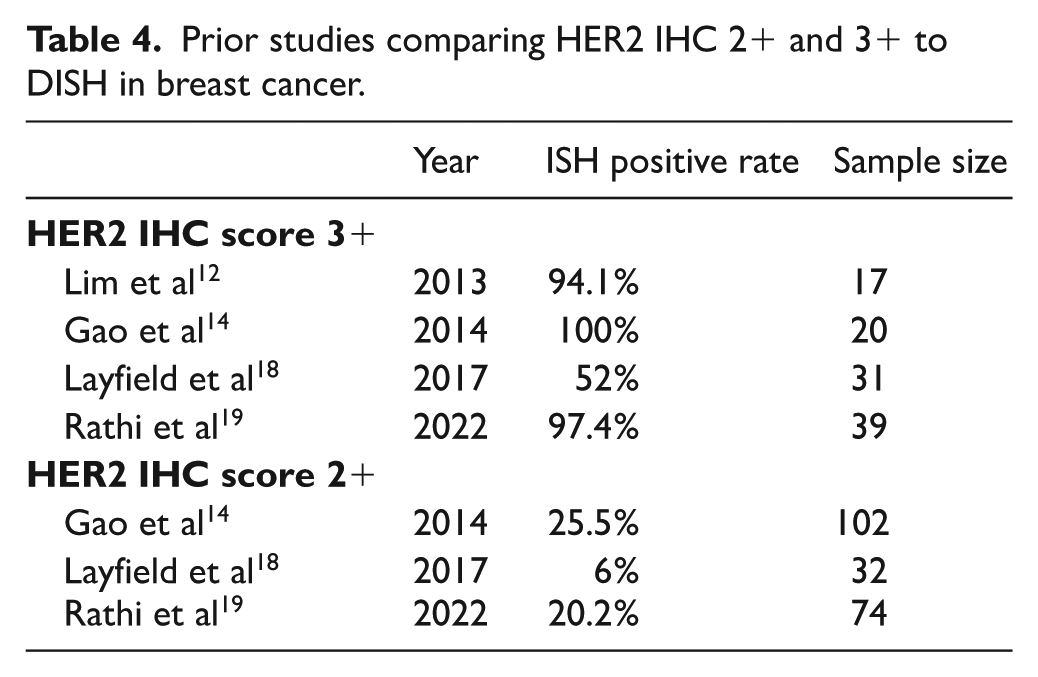
Prior studies have reported varying failure rates for performing HER2 DISH, ranging from 2.8% to 11% with different definitions from initial to persistent failure.14,15,17,20 As our laboratory technicians routinely assessed the slide quality and tried to correct some issues before sending the slides to pathologists, we did not record the initial failure rate. However, we observed an extremely low rate (0.5%) of persistent technical failure when performing DISH in the tissue blocks of primary breast tumors. All such cases could be corrected by shifting to perform DISH in metastasized lymph nodes instead. Previous HER2 FISH and bright-field ISH studies demonstrated high concordance between primary breast carcinomas and matched axillary lymph node metastases, supporting the use of lymph node tissue as a valid alternative when primary tumor material is unavailable or inadequate.23,24
The main advantages of FISH over DISH is that FISH provides clearer signals and lower technical failure rate. 15 However, DISH excels in several other aspects. First, DISH uses chromogenic signals, which can be viewed using a routine light microscope, while FISH requires a significantly more expensive fluorescent microscope and a darkroom setup. This not only increases costs but also makes FISH less practical for many small laboratories. Second, fluorescent signals fade much faster than chromogenic signals, restricting the ability to review the slide. Third, HER2 DISH offers a quicker turnaround time, as it requires less time for staining and interpretation. Fourth, DISH allows for better morphological assessment, making it easier to identify invasive tumor cells.6,21 Finally, DISH slides can be easily scanned using standard whole slide imaging sets, which opens up the possibility for digital pathology in telepathology consultations or for use with artificial intelligence assistance.25,26
The strength of this study lies in its large sample size. The 1,307-sample size is the largest study on this topic in the current English literature, especially when considering the problematic 2+ IHC score group. In addition, this study included samples from multiple hospitals across Thailand. While this subjected the study to increased heterogeneity in pre-analytic factors, the strong agreement in the IHC 3+ group validates its applicability in wider settings.
Limitations
One limitation of this study is the lack of the gold standard FISH test. Nonetheless, as previously discussed, prior investigations show almost perfect to excellent agreement between DISH and FISH tests.7 -21 We also find no significant difference when comparing our result to the result of IHC and FISH, thereby justifying the use of DISH as a substitute for FISH. Another limitation of this study is the lack of cases with IHC scores of 0 and 1+. This limits the ability to precisely calculate some statistical values such as sensitivity, specificity, overall concordance and Cohen’s kappa agreement.
Conclusion
Our study adds valuable evidence to the body of literature supporting the use of DISH as an effective alternative to FISH for confirming HER2 status in breast cancer, especially in resource-limited settings. The concordance rate of the IHC score 3+ group is 92%, and the positive rate of the IHC score 2+ group is 36%. The data reveal an expected correlation between HER2 IHC and DISH in lymph node metastasized breast carcinomas, comparable with prior studies between HER2 IHC and FISH in breast cancers.
Footnotes
Acknowledgements
The Biobridge Project Thailand was funded by Roche Thailand Ltd. to encourage the utilization of HER2 DISH.
ORCID iDs
Author contributions
Kroonpong Iampenkhae: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Writing—original draft, Writing—review & editing.
Adiluck Pisutpunya: Data curation, Formal analysis, Investigation, Methodology, Writing—review & editing.
Chawinthorn Vuthithammee: Data curation.
Piyakawin Sodsoon: Data curation.
Sompon Apornvirat: Formal analysis, Writing—original draft, Writing—review & editing.
Ethical considerations
This retrospective study was approved by the Institutional Review Board of the Faculty of Medicine, Chulalongkorn University, reference number 938/64.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no external funding. The publication fee was partially supported by the author’s university.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The first author was involved with the project and obtained permission to use the data, but did not receive any incentive or obligation to publish the result in an academic journal.
The remaining authors declare no conflict of interest.
Data availability statement
The data sets used and/or analyzed during this study are available from the corresponding author upon reasonable request.
